Abstract
Eleven rhesus monkeys developed multifocal erythematous and a vesicular rash. Most recovered spontaneously, but a 21–year-old female became moribund and was euthanized. Findings were of vesicular dermatitis and widespread multifocal hemorrhagic necrosis of the lungs and other viscera, with intralesional intranuclear inclusions. Simian varicella virus was identified as the cause by polymerase chain reaction analysis and serologic testing.
History, Clinical Findings, and Gross Lesions
From August 2001 through July 2002, 11 indoor housed rhesus monkeys of both sexes developed a multifocal erythematous rash that affected the inguinal areas, abdomen, axillae, and face. Individual lesions were roughly circular and up to about 1 cm in diameter, and some contained small vesicles (Fig. 1). Most affected monkeys were juveniles, 2 to 3 years old, but one (monkey No. 1) was a 21-year-old female. Some of the younger animals had undergone organ transplantation procedures, which included immunosuppression; the others had received no experimental manipulation. Except for monkey No. 1, none of the animals was clinically ill, and the skin lesions resolved within 3 weeks. Monkey No. 1 was reported inappetent one day and lethargic with nasal hemorrhage the next. She was euthanized, and a postmortem was done. In addition to the skin lesions, the lungs were purple and heavy, and exuded small amounts of serosanguineous fluid from the cut surface. There were petechiae in the mucosa of the tonsils, stomach, and intestine.

Papulovesicular dermatitis; monkey No. 3.
Differential Diagnoses
The primary differential diagnoses for vesicular dermatitis, with or without disseminated disease in macaques are simian varicella virus (SVV; Cercopithecine herpesvirus 9) 6 and B virus (BV; Cercopithecine herpesvirus 1). 4 A rash and pneumonia also occur in simian measles. 7
Microscopic Findings
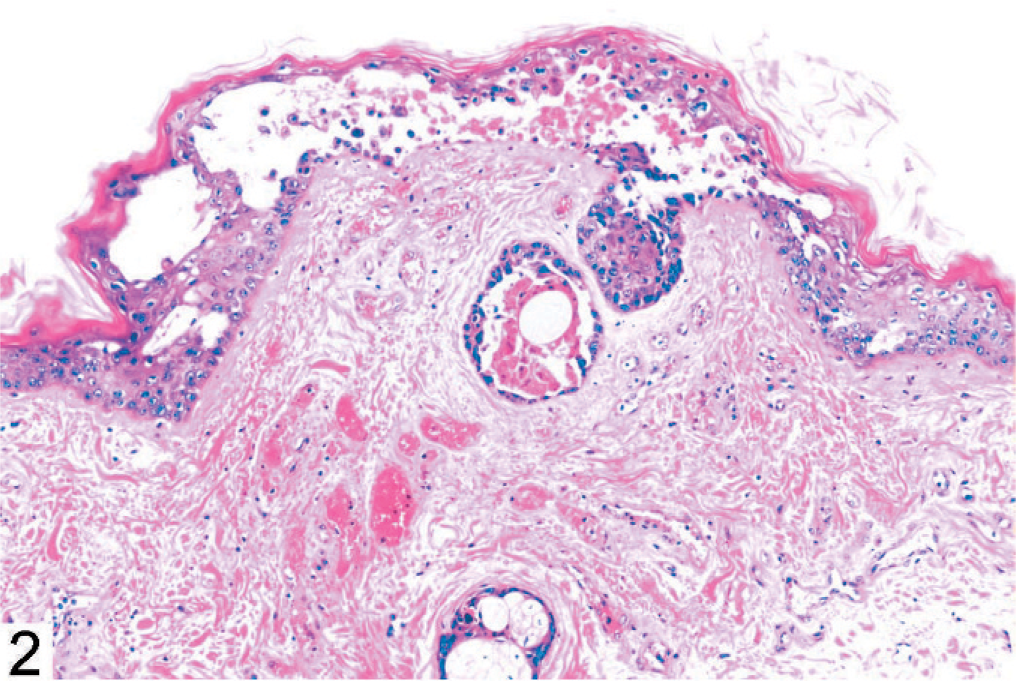
The skin lesions in monkey No. 1 were characterized by degeneration, necrosis, and separation of the cells of the basal, granular, and spinose layers of the epidermis, and dermal edema (Figs. 2 and 3). Eosinophilic intranuclear inclusions were prominent, and there were a few small syncytia in the affected epidermis (Fig. 3, arrow). Other findings included severe multifocal necrotizing and hemorrhagic pneumonia (Fig. 4), splenitis, conjunctivitis, pharyngitis, tonsillitis, glossitis, adrenalitis, and hepatitis with intranuclear inclusions similar to those in the epidermis.
Vesicular dermatitis; monkey No. 1. HE.
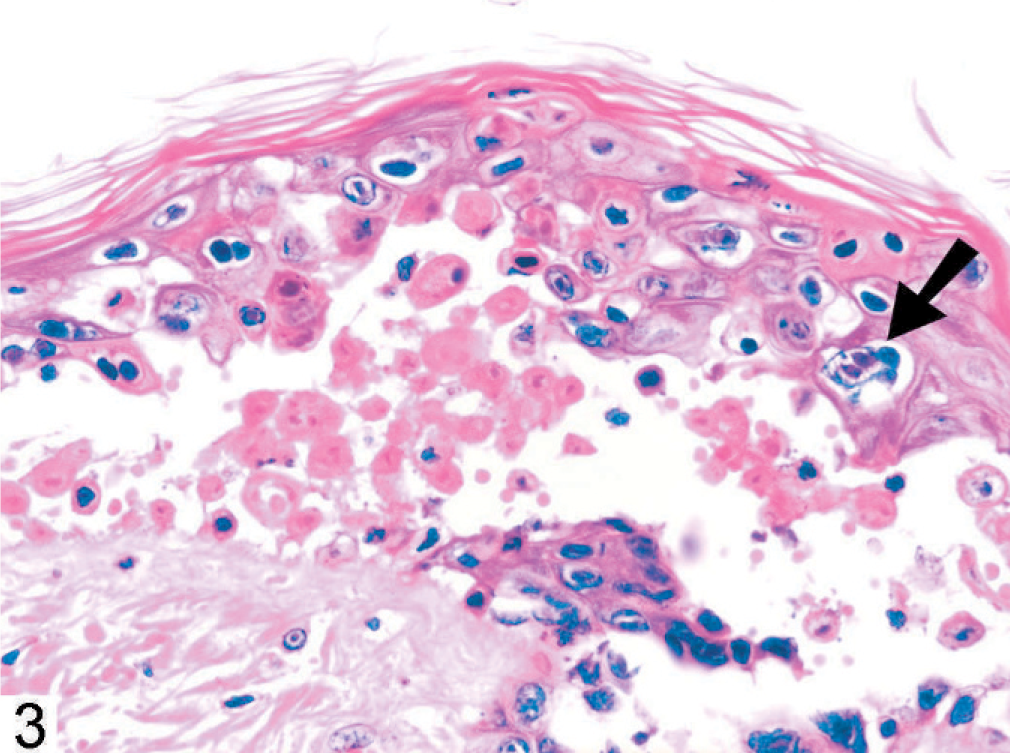
Vesicular dermatitis with intranuclear inclusions and syncytia (arrow); monkey No. 1. HE.
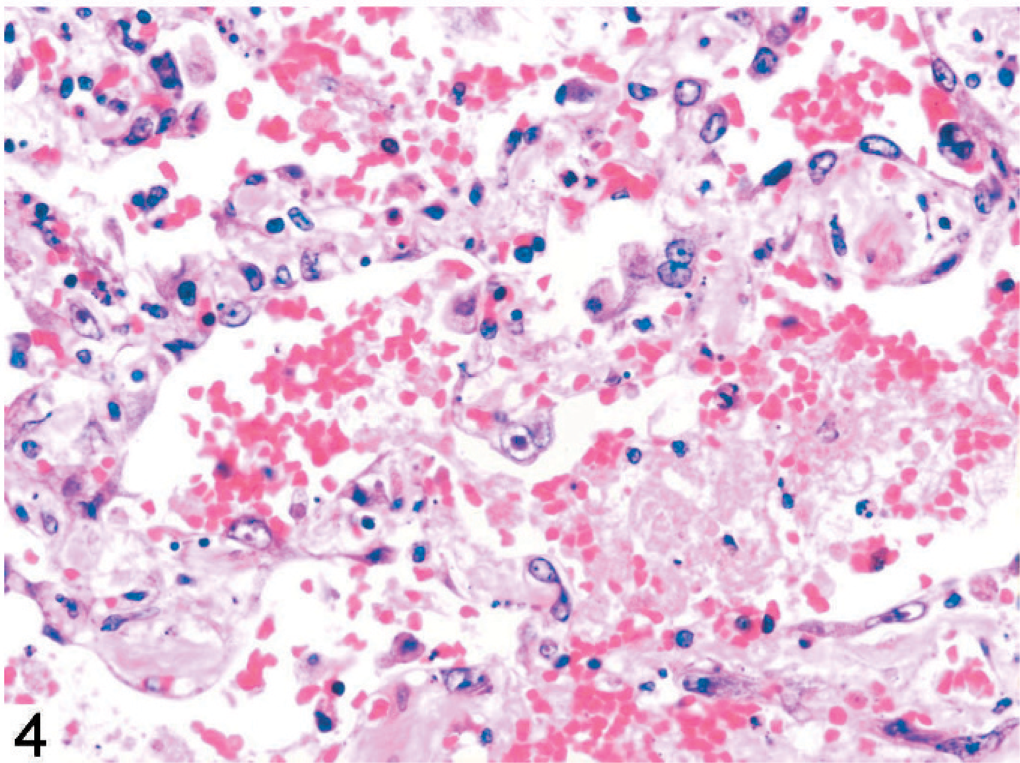
Necrotizing and hemorrhagic pneumonia with intranuclear inclusions; monkey No. 1. HE.
Laboratory Findings
Vero monkey kidney cells were used to propagate SVV (strain Δ provided by Dr. W. Gray, University of Arkansas, Little Rock, AR) and Herpesvirus papio 2 (HVP2; Cercopithecine herpesvirus 16; to test for BV) and prepare viral antigens for enzyme-linked immunosorbent assay (ELISA) and deoxyribonucleic acid (DNA) for SVV polymerase chain reaction (PCR).
DNA was isolated from formalin-fixed lung and spleen with a DNeasy Tissue Kit (QIAGEN, Valencia, CA) and from Vero cells with DNAzol reagent (Molecular Research Center, Cincinnati, OH), according to the manufacturer's instructions. PCR for BV and human herpes simplex virus (HSV1 and HSV2) was done as previously described 2 with rTth polymerase (Applied Biosystems, Foster City, CA) and approximately 100 ng of template DNA. Two primer sets were designed from the SVV glycoprotein B gene (GenBank accession no. AF275348). Primers 300 (5′-CGCAGACAGAGTTCCCATACCAGTATCTGAG-3′) and 302 (5′-GCTGTCATAAGGGATATATGATCGAGCATCGAC-3′) amplified a 300–base pair (bp) fragment, primers 301 (5′-ACCAGAGGATATATGGAACCCACCACTAGG-3′) and 303 (5′-CCTTTCGAGCTTCATCTTTAACGCAGTGAC-3′) amplified a 260-bp product, and primers 300 and 303 amplified a 700-bp product. After an initial 3-minute denaturation at 94°C, amplification was carried out by 35 cycles of 96°C-30 seconds/62°C-30 seconds/72°C-1 minute, followed by 72°C for 5 minutes. PCR products were purified with Wizard PCR Preps (Promega, Madison, WI), and both strands directly sequenced to confirm their derivation from SVV.
HVP2/BV and SVV ELISAs were done as previously described, 10 and a commercial kit was used for measles virus antigen (Sigma Chemical Co., St. Louis, MO). All ELISAs used biotinylated anti-human immunoglobulin (Ig) G followed by avidin-biotinylated horseradish peroxidase (Vector Laboratories, Burlingame, CA) to detect bound macaque IgG. Sera were initially screened at a dilution of 1 : 100. Positive sera were titered by testing serial twofold dilutions. Paired serum samples from 5 affected macaques for which preoutbreak samples were available, including monkey No. 1, were tested. All results for HPV2/BV and measles virus were negative. SVV results for monkey No. 1 also were negative. Two other monkeys (Nos. 2 and 3) had preoutbreak titers for SVV that were considered positive (400 and 3,200, respectively) and had modest increases after infection (titers 1,600 and 6,400, respectively). The remaining 2 monkeys (Nos. 4 and 5) had little or no preoutbreak SVV antibody (titers <100) and seroconverted strongly (titers 3,200 and 12,800, respectively).
DNA samples from the lung and spleen of monkey No. 1 were tested by PCR. Results of nested PCR for detection of BV and HSV were negative. PCR with 3 sets of SVV primers yielded products of the expected size for 2 of the primer sets (Fig. 5). DNA sequencing of the products confirmed that they were SVV. Alignment of the 700-bp sequence derived from lung and spleen of monkey No. 1 with that of the reference SVV sequence indicated that the lung and spleen sequences were identical to each other and 98% homologous with the reference sequence. Of the 12 nucleotide substitutions, one was located near the 3′ end of primer 302, which explained the failure to amplify products from tissue samples with this primer.
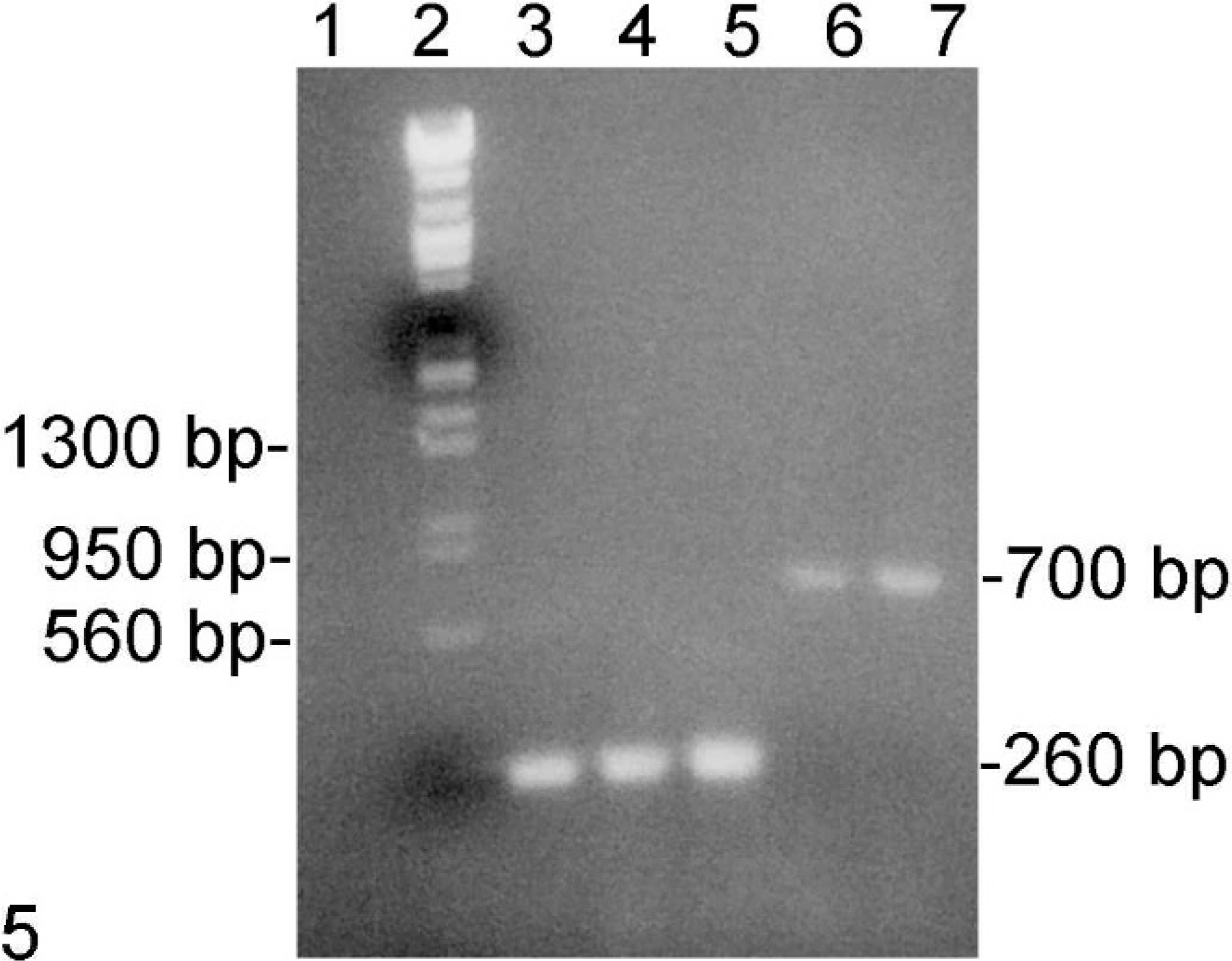
PCR results; monkey No. 1. Lane 1, negative; 2, marker; 3, SVV; 4 and 6, lung; 5 and 7, spleen; 1–5, primers 301 and 303; 6 and 7, primers 300 and 303. Ethidium bromide.
Discussion
Simian varicella and BV disease are cause for serious concern: SVV because of the potential for epizootics with significant mortality and BV because of the potential for zoonotic human disease. Simian varicella is caused by an alpha-herpesvirus related to the human varicella-zoster virus and is transmitted primarily by aerosol. 6 The disease has occurred spontaneously in captive nonhuman primates of several Old World species, including rhesus and other macaques, African green monkeys, and patas monkeys, as major and minor epizootics and as individual cases. Severity varies widely, from mild self-limiting disease to fatality. Patas monkeys seem to be most susceptible to fatal disease, but cynomolgus macaques also can be severely affected. The lesions described above are typical of those previously reported. It is not clear why monkey No. 1 was the only animal severely affected or why the outbreak was limited to 11 cases among up to 75 monkeys potentially exposed. The affected monkeys were housed in the same facility in several rooms on one floor, and all were used by the same research group. There was no apparent pattern in cage or room location among affected monkeys. The source of the outbreak also is not known. The younger monkeys had been imported from 3 commercial sources within the year preceding the outbreak but were not tested for SVV infection by the vendors or during importation or quarantine. Because 2 monkeys had clear serologic evidence of preoutbreak infection and because latent SVV infection can be reactivated in ganglionic neurons, it is likely that exposure of susceptible monkeys resulted from shedding of virus by one or more monkeys with a reactivated latent infection, possibly as a result of immunosuppressive treatment.
BV occurs naturally in macaques and is an alpha-herpesvirus related to HSV1 and HSV2. 4 It is a rare cause of disseminated multifocal necrotizing disease in its natural hosts and in patas, colobus, and DeBrazza's monkeys. 1, 3– 5, 8, 9 Its major significance, however, is its potential to cause encephalitis in humans, a rare occurrence but having a 70% case fatality rate if untreated.
Measles can affect nonhuman primates of many different species and is characterized by multifocal parakeratotic dermatitis, proliferative pneumonia with extensive formation of syncytia that contain eosinophilic intracytoplasmic and intranuclear inclusions, and syncytia in lymphoid tissues. 8 It was once common in laboratory primates but now is rarely encountered.
Footnotes
Acknowledgement
This work was partially supported by NIH grant RR07849 to RE.
