Abstract
Allergic dermatoses are common in people and domestic animals. Resultant lesions are routinely biopsied and submitted for histological examination to confirm a diagnosis or rule out diseases with overlapping or atypical clinical features. Diagnostic pathologists and clinicians are often faced with the difficult task of determining whether an allergic reaction pattern is present on both the microscopic and macroscopic levels and correlating histopathologic findings with clinical and historical data to achieve a precise clinical diagnosis. The bulk of the current veterinary literature on allergic dermatoses focuses on atopic dermatitis in dogs, distantly followed by cats, horses, and other animals. The objectives of this review are to demonstrate the key histopathologic and clinical diagnostic features of the various allergy-mediated reaction patterns, and to provide diagnosticians with a practical guide for clinicopathological correlations. Current concepts in the pathophysiology of immediate hypersensitivity reactions, with a focus on atopic dermatitis, are discussed. Points of potential histopathologic overlap between the “classic” allergic reaction pattern and less common inflammatory, predominately eosinophilic, conditions that may mimic this pattern will be discussed with the goal of highlighting the critical need for collaboration between pathologists and clinicians in furthering patient care.
The Coombs and Gell classification scheme breaks hypersensitivity reactions into 4 categories. 18 Allergic diseases are generally considered to be type I hypersensitivity reactions (both immediate and late phase) to foreign substance involving immunoglobulin (Ig) E, and less frequently type IV (delayed, cell-mediated) reactions. Allergic skin disease includes atopic dermatitis, cutaneous adverse food reaction (CAFR; food allergy), hypersensitivities to ectoparasites, contact hypersensitivity, and some adverse drug reactions (ADRs), although ADRs may fall under any of the 4 Coombs and Gell subtypes. Allergic hypersensitivity reactions may affect other organ systems and mucosal surfaces with separate, though potentially concurrent, disease manifestations such as allergic bronchitis (asthma), allergic rhinitis, allergic conjunctivitis, inflammatory bowel syndromes, oral eosinophilic granuloma, and anaphylaxis.
Allergic dermatitis is common in companion animal veterinary practice, with pruritus being the most frequent presenting complaint. 41 Although the problem is commonplace, the pathogenesis of allergic dermatitis is incompletely understood. Diagnosis and effective treatment of allergic dermatitis are challenging due to varied clinical presentations, varied responses to therapy, and presenting clinical signs that can mimic other cutaneous diseases. 109 Diagnostic pathologists may be equally challenged when asked to diagnose or rule out allergic skin disease as the histopathologic findings are not always straightforward, clinical information that is critical to a diagnosis may be lacking, and other disease processes can have histopathologic overlap. The main task of the diagnostician in this scenario is not to diagnose allergic skin disease, but to rule out other conditions that have overlapping clinical phenotypes and to assess the tissue for histopathologic features that support or refute the clinical diagnosis. As such, it behooves the pathologist to understand the known pathophysiology of allergic skin disease in domestic animal species and common histopathologic and clinical correlates.
Most of the veterinary literature on allergic dermatoses is focused on atopic dermatitis of companion animals, primarily dogs, with fewer publications available on cats, distantly followed by horses, livestock, and nontraditional pets. Canine atopic dermatitis (cAD) has been thoroughly reviewed by a prior task force of the American College of Veterinary Dermatology (2001), the International Task Force on Canine Atopic Dermatitis (2003–2011), and currently the International Committee on Allergic Disease of Animals (since 2013).33,34,59,76,90
Eosinophils are not resident leukocytes in the skin.81,91 Eosinophilic dermatitis may lead a diagnostician to consider a hypersensitivity response or parasitism. Despite the commonality of allergic skin disease, other dermatoses with eosinophilic infiltrates should be carefully ruled out. For example, varying severities of perivascular eosinophilic dermatitis can be encountered in biopsy samples of pemphigus foliaceus, canine acute eosinophilic dermatitis and edema (CAEDE), zinc-responsive dermatosis of the Siberian husky and Nordic breed dogs, sterile pustular erythroderma of miniature schnauzers, and in samples of furunculosis (where bare hair shafts and keratin may provoke eosinophil chemotaxis).8,31,107 Falling back on allergic skin disease as a probable diagnosis with simply the presence of an eosinophilic dermatitis is a pitfall that should be avoided if other clinical or histopathologic features are not supportive.
Before diving in, practical considerations to optimize the diagnostic utility of a cutaneous biopsy sample need to be addressed. An open line of communication between the clinician and diagnostician is essential. A concise, pertinent, and accurate clinical history are needed, including signalment, responses to prior therapy, current ectoparasite control measures, lesion descriptions and distribution, and clinical differential diagnoses. If glucocorticoid therapy has been utilized, this should be known and a “wash-out” period avoiding glucocorticoids for 3 weeks is recommended, when possible, due to the effects of glucocorticoids on leukocytes, particularly eosinophils, which may alter the histopathologic interpretation. A longer wash-out period may be needed if reposital steroids are used.52,66 Surgical scrub and clipping should be avoided so as not to impart artifacts to the epidermis and to preserve the diagnostic value of pustules and crusts. Multiple punch biopsy samples (preferably two to four, 6 to 8 mm diameter samples) are recommended for most cases as diagnostic information may vary between sites due to the natural evolution of lesions, secondary surface trauma and microbial infections.
Classic Cutaneous Hypersensitivity Reactions
Canine Atopic Dermatitis
Atopic dermatitis is a genetically predisposed inflammatory and pruritic skin disease with characteristic clinical features associated with elevated allergen-specific IgE antibodies, most commonly directed against environmental allergens.33,38 Though clinical signs of cAD are characteristic, they can vary with pruritus (the unpleasant sensation of itch) being the most consistent finding. Canine atopic dermatitis is common, chronic, and progressive, generally with a juvenile to early adult age of onset of 6 months to 3 years.25,103 Strong breed predilections for the development of cAD support a genetic basis, with golden retrievers, West Highland white terriers, German shepherd dogs, cocker spaniels, and French bulldogs commonly reported, though predilections also appear to have geographic variations.6,50 Based on genomic studies, the predisposing genotypes may be breed-specific.83,84,94,97,104,119
To date, there is no test available to establish a diagnosis of cAD and histopathologic findings are highly variable. Canine atopic dermatitis is generally a diagnosis of exclusion that requires first ruling out other common and treatable causes of pruritus in young dogs (eg, ectoparasites and superficial bacterial and fungal infections) and then ruling out other causes of allergic skin disease (eg, ectoparasite hypersensitivity and adverse food reaction). The diagnosis of “atopic-like disease” has been made previously if elevated IgE levels are not detected following exposure to an allergen and the dog has signs otherwise consistent with cAD, although it is not entirely clear whether this represents a separate disease or a failure of allergen-specific IgE detection.33,38
Canine atopic dermatitis offers a spontaneous large animal model for study of AD in people. There is significant overlap in the lesion distribution, immunologic profile, prevalence, and therapeutic considerations. The progression differs as people develop AD at an early age and often have a reduction in the severity of disease in adulthood. In people, the atopic state may progress to involve other allergic conditions such as allergic bronchitis and allergic rhino-conjunctivitis/sinusitis (“hay fever”). This progression, which is termed the “atopic march,” is not recognized with cAD.6,41,58,98
Clinical diagnosis
The clinical diagnosis of cAD follows exclusion of other causes of pruritus and a response to anti-inflammatory therapy, and may be supported by a demonstrated reaction to a particular allergen via intradermal testing or serum-based allergen testing. Most of the clinical lesions recognized with cAD are secondary to self-trauma and may be complicated by microbial infection (predominately Staphylococcus pseudintermedius or Malassezia spp.). Biopsy is not commonly recommended by dermatologists to support a diagnosis of cAD, but a biopsy may be procured to rule out other dermatoses if lesions are atypical or refractory to standard therapies. A detailed set of clinical diagnostic criteria for cAD have been developed and modified over the years as an aid to practitioners and for standardizing clinical trial participants.25,79,88,118 Presently, the use of the criteria of Favrot is employed in an attempt to differentiate cAD (including non-food-induced and food-induced cases) from other non-cAD conditions. 25
The common lesions of cAD include erythema, alopecia, and lichenification of haired skin in the perioral, periocular, ventral and pedal regions (Fig. 1a–h), and flexural surfaces (Fig. 1a–c, g). Pedal lesions may also include nodular interdigital cysts/furunculosis (Fig. 1e). Dogs may have a history of chronic or repeated episodes of otitis externa, which has been reported as the initial presenting complaint in 43% of dogs with cAD. 25 There are common variations to the distribution of lesions that are somewhat breed-dependent. These phenotypes have been reviewed by Wilhelm et al. 115 A prospective study by Picco et al 87 showed that cAD and CAFRs/food allergy (see below) are indistinguishable based on clinical presentation. Therefore, practitioners may use the term cAD to cover both food-induced and non-food-induced cAD cases. It is not clear whether food-induced cAD shares the same pathogenesis as other environmental allergens or if it is a manifestation of CAFR without concurrent gastrointestinal signs. Based on current studies, the engrained dogma that lesion distributions vary between cAD and CAFRs (ie, that food allergic dogs frequently present with otitis and perianal dermatitis or “ears and rears”) is no longer supported.25,87
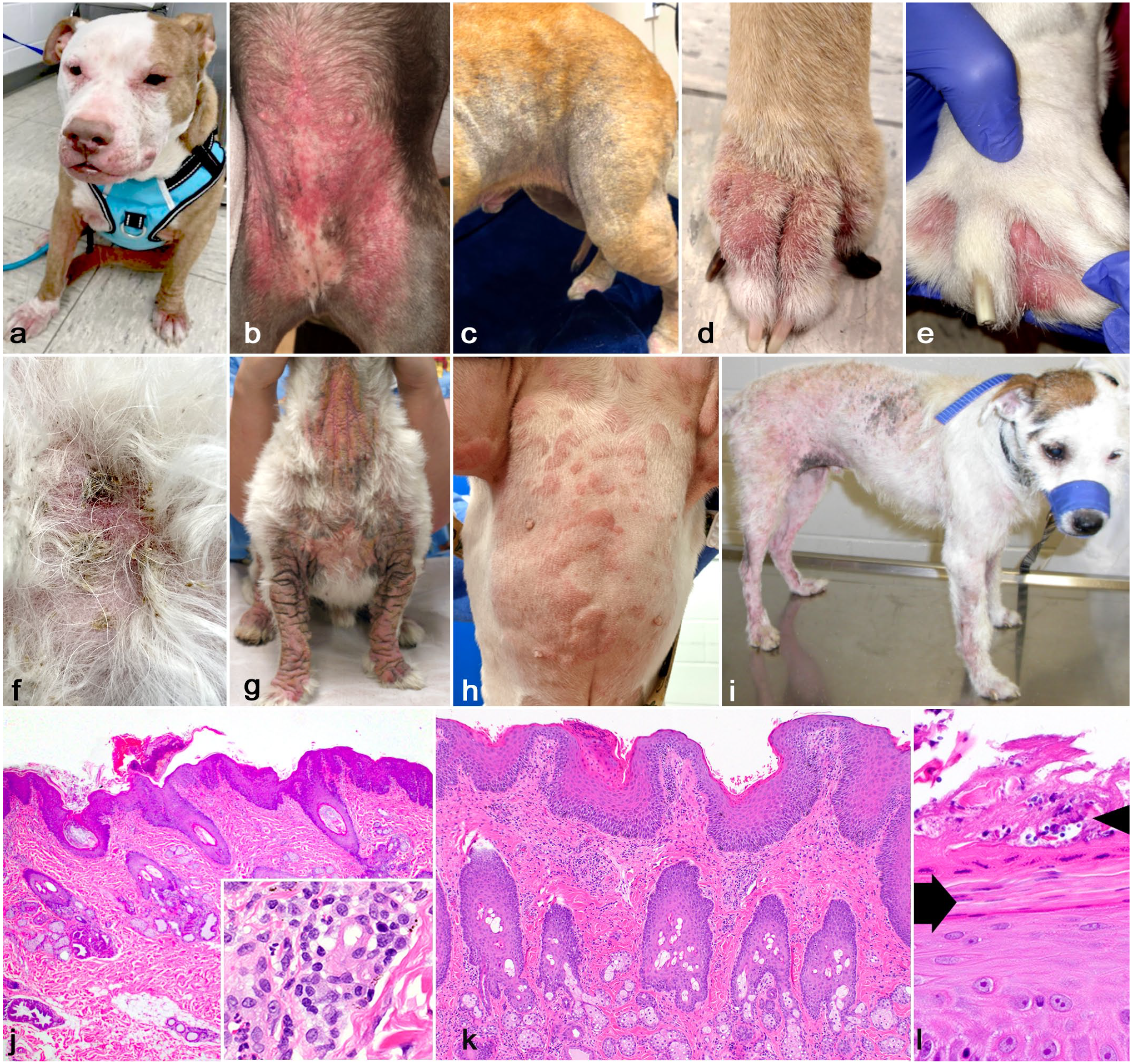
Cutaneous hypersensitivity reactions. (a) Canine atopic dermatitis (cAD), American pit bull terrier, dog. Severe periocular, perioral, ventral, and dorsal pedal alopecia and erythema. There is thickening from lichenification and edema of the lips, muzzle, and axillary skin. (b) cAD, ventrum, mixed breed, dog. Coalescing macular and regionally extensive ventral abdominal and inguinal erythema during a flare state. (c) cAD, flank and inguinum, shepherd-mixed breed, dog. Regional alopecia, lichenification, and hyperpigmentation predominately along a flexural surface. (d) cAD, paw, American pit bull terrier, dog. Dorsal pedal erythema, lichenification, and alopecia. (e) cAD, paw, bulldog. Interdigital erythema with nodules corresponding to chronic hyperplastic dermatitis, infundibular hyperkeratosis, and furunculosis. (f) cAD and pyoderma; ventrum, mixed breed dog. Patchy alopecia with erythema, crust, and follicular casts. Aerobic culture isolated Staphylococcus pseudintermedius with cocci identified cytologically. Recurrent bacterial infections are common with cAD (photo courtesy of Dr Peter Canning). (g) Malassezia dermatitis and cAD, West Highland white terrier, dog. Formerly termed epidermodysplasia of West Highland white terriers or “Westie Armadillo Syndrome.” There is severe ventrally oriented to generalized alopecia, lichenification, and hyperpigmentation. (h) Urticaria; ventrum; French bulldog. Multifocal and coalescing erythematous wheals may be seen in flare states of allergic skin disease. (i) Flea allergy dermatitis, Jack Russell terrier, dog. Alopecia, erythema, and multifocal hyperpigmentation. Note that head and neck are spared as lesions are largely attributed to biting/chewing in addition to scratching. (j) Histopathologic findings in cAD; The common histopathologic finding is a mixed perivascular dermatitis including lymphocytes, plasma cells, eosinophils, and occasional neutrophils. Inflammation may be of varying severity in cAD and lesions are nonspecific. The stratum corneum is compact with a surface crust and the epidermis is hyperplastic. There is generalized sebaceous gland hyperplasia. The inset shows a mild perivascular infiltrate of lymphocytes, plasma cells, and eosinophils with moderate spongiosis. Hematoxylin and eosin (HE). (k, l) Histopathologic findings in Malassezia dermatitis and cAD, West Highland white terrier, dog. There is severe epidermal hyperplasia with superficial perivascular dermatitis. There is generalized sebaceous gland hyperplasia. HE. (l) Higher magnification of the surface shows parakeratotic hyperkeratosis (arrow) and compact orthokeratosis with crust containing few cocci and surface yeast with a morphology of Malassezia spp. (arrowhead). HE.
Clinical differential diagnoses for cAD may include other allergic skin diseases such as CAFR, ectoparasitism, Malassezia dermatitis (MD; Fig. 1g), ADR, and pemphigus foliaceus. In senior dogs, epitheliotropic cutaneous lymphoma may mimic the inflammatory lesions and pruritus that are typical of cAD, and a biopsy may be indicated.
Histopathologic findings
The primary, consistent histopathologic lesion in cAD is superficial perivascular dermatitis (Fig. 1j, k). In terms of achieving a final diagnosis, this is the least helpful of the dermatopathologic patterns. The degree of inflammation in cAD can be minimal. As the clinical lesions of cAD are secondary to surface trauma and microbial infection, the histopathologic changes encountered in a biopsy, especially in chronic refractory cases, are no different. Epidermal hyperplasia, surface-oriented inflammation, edema, superficial fibroplasia, and pustules or folliculitis associated with secondary microbial infection are often the predominant findings. The histopathologic changes only allow for exclusion of other differentials and to support the clinician’s observations. The pathologist must correlate their findings with the clinical distribution of lesions. Clinical photos or body maps, in addition to lesion descriptions, may be useful.
Canine atopic dermatitis is associated with perivascular mononuclear dermatitis with varying degrees of eosinophilic and neutrophilic infiltrates. 7 Early in the course of disease or in nonlesional atopic skin, the degree of inflammation can be minimal. As lesional skin is generally biopsied, the degree of inflammation may range from mild to severe, and other chronic histologic changes are often prominent, including epidermal hyperplasia and acanthosis, spongiosis, sebaceous gland hyperplasia, and variable apocrine gland ectasia (Fig. 1j, k). Of note, sebaceous gland hyperplasia in wire-haired fox terriers has been associated with proliferation of Demodex injai and may not represent allergy (Supplementary Fig. S1).40,70,82 This lesion has also been seen by the authors in other terrier dogs. As an extension of the epidermis, follicular walls may be acanthotic and spongiotic (Fig. 1j, k). In the face of chronic inflammation, atrophy of the inferior follicle is common and telogen phase may predominate. Close attention should be paid to the stratum corneum to look for evidence of surface trauma. These changes include compact orthokeratotic hyperkeratosis and foci of parakeratotic hyperkeratosis, presumably from rapid keratinocyte turnover and failure of complete maturation (Fig. 1j, m). Small eosinophilic intraepidermal pustules may be identified. 81 Eosinophilic intracorneal and subcorneal pustules may also be encountered with ectoparasitism, 68 and ectoparasitism (e.g., sarcoptic mange) can easily be misdiagnosed as allergic dermatitis in the absence of surface mites (Fig. 2a). If larger eosinophilic pustules are present, scrutiny for evidence of acantholysis, including step sectioning, is recommended to rule out pemphigus foliaceus (Fig. 2c).
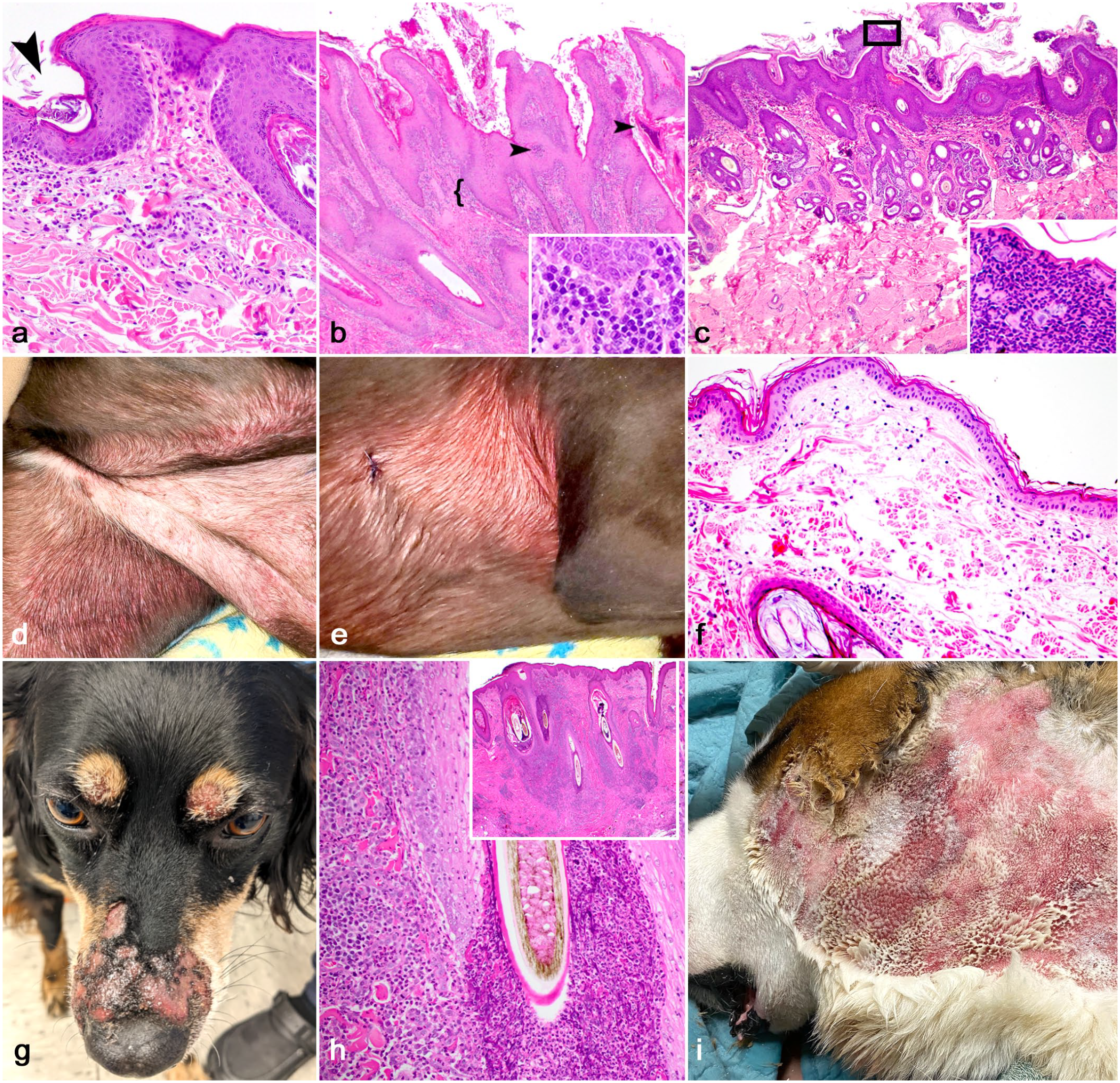
Adverse reactions to treatment of allergy and other canine dermatoses to consider. (a) Sarcoptic mange, haired skin, dog. There is moderate, superficial eosinophilic inflammation with epidermal hyperplasia. A fragment of a sarcoptic mite is captured burrowing in the superficial epidermis (arrowhead). The histopathologic findings in the absence of the mite overlap extensively with chronic allergic skin disease. The clinical history in this case included intense generalized pruritus. Hematoxylin and eosin (HE). (b) Lymphoplasmacytic psoriasiform lichenoid dermatosis, haired skin, dog. This lesion is often associated with chronic cyclosporine therapy (or other calcineurin inhibitors) and is characterized by discrete, irregular epidermal hyperplasia with hyperkeratosis (orthokeratotic and parakeratotic with colonies of bacterial cocci), small epidermal neutrophilic and eosinophilic pustules and crusts (arrow heads), and a lichenoid band (lacking an interface reaction) predominated by lymphocytes and plasma cells (bracket and higher magnification inset). HE. (c) Pemphigus foliaceus, haired skin, dog. Pemphigus foliaceus is a pustular acantholytic dermatitis. If surface crusts or pustules are lacking, the hyperplastic surface-oriented inflammation may contain scattered neutrophils and eosinophils with a similar histopathologic pattern of allergic dermatitis. Acantholytic cells are demonstrated in the higher magnification inset. HE. (d, e) Canine acute eosinophilic dermatitis and edema (CAEDE), dog. Diffuse ventral erythroderma and edema with coalescing red papules and macules (photo courtesy of Dr Christine Cain). (f) CAEDE, pattern 1, dog. The dermis is expanded by edema and ectatic blood vessels with a minimal perivascular to interstitial infiltrate containing eosinophils. Note the lack of epidermal hyperplasia as the clinical progression is generally acute and marked. The degree of inflammation may be especially mild considering the degree of clinical erythroderma (d, e). HE. (g) Eosinophilic furunculosis of the face, Australian shepherd dog. Ulcerated and crusted nodules and pustules overlying the nasal bridge and dorsal periocular skin. (h) Eosinophilic furunculosis of the face, muzzle, mixed breed, dog. Severe luminal folliculitis with outer root sheath rupture and eosinophilic dermatitis. The epidermis is acanthotic and spongiotic. The lesion is putatively associated with arthropod bite hypersensitivity. The lower magnification inset demonstrates an adnexal-oriented nodular to diffuse pattern of inflammation predominated by eosinophils. HE. (i) Acute moist/pyotraumatic dermatitis, face, 1-year-old St. Bernard dog. Severe periauricular erythema, erosions, and ulcers with purulent exudate. These lesions are intensely pruritic and historically are associated with flea allergy dermatitis and/or canine atopic dermatitis (photo courtesy of Dr Elise Enners).
Early in cAD, or during flare states, an eosinophilic inflammatory infiltrate may be present, although this is frequently a minor component of the inflammation described in published studies.78,117 As the cytokine profile shifts from a Th2- to a Th1-weighted immune response, the inflammation is more commonly lymphocytic and plasmacytic.89,100 Perivascular mast cells may be more numerous and reactive with nuclear prominence. 113 Superficial vascular ectasia may be slight or prominent, occasionally with marginated granulocytes, imparting the erythema seen grossly. A biopsy of nonlesional skin in dogs with cAD may reveal minimal lymphocytic and eosinophilic inflammation without epidermal or adnexal hyperplasia or histopathologic changes may be absent. 78
Pathogenesis of cAD
Categorization as a type 1 (late phase) hypersensitivity reaction oversimplifies the complexity of cAD, whose pathogenesis involves intertwining alterations in the epidermal barrier and dysregulation of immune responses to allergens and commensal microorganisms. An inciting cause has not been pinpointed in cAD as the barrier, immune system, cutaneous microenvironment, and microbes continuously alter one another with multilayered interactions, although genetic predispositions are proposed.
The epidermal barrier
Primary epidermal barrier dysfunction is likely an early contributor to development cAD. Prior studies have focused on the epidermis as a mechanical barrier, and defects in the epidermis allow for allergen penetration and interaction with an altered immune system.10,86 In the process of cornification, filaggrin (FLG, filament aggregating protein) binds keratin tonofilaments to facilitate cell compaction, and its breakdown products (hygroscopic amino acids) are moisturizing and acidifying. 92 In humans, homozygous FLG deletions or loss of function defects causes ichthyosis vulgaris, which is associated with AD in up to 50% of cases. 86 People with heterozygous FLG mutations are also predisposed to AD. 10 It should be noted that many atopic individuals do not have an FLG mutation. In cAD, altered expression of FLG has been proposed based on reduced immunolabeling of FLG in keratinocytes in atopic compared with normal beagles. 60 Chervet et al 17 also demonstrated reduced immunofluorescent labeling of the C-terminus of FLG in corneocytes compared with the N-terminus, supporting a loss of function mutation in cAD. Aside from a group of Labrador retrievers from the United Kingdom, a significant association with specific FLG mutations and cAD has not been made. 120 Altered lipid composition and organization of the stratum corneum are documented in cAD, including reduction in ceramides (a major constituent of extracellular lipids).45,73,101 Keratinocytes also contribute to and respond to the inflammatory environment of cAD via lymphocyte stimulation and act as facultative antigen-presenting cells with varied antimicrobial peptide production (see Supplemental Material).
The immune response
Investigations into the immune response of cAD have recently revolutionized treatment strategies in veterinary dermatology with the small molecule inhibitor oclacitinib (Apoquel; Zoetis, Parsippany, NJ, USA) and the advent of the monoclonal antibody lokivetimab (Cytopoint, Zoetis), which targets interleukin (IL)-31. Oclacitinib is a member of the Janus kinase/signal transducers and activators of transcription (JAK/STAT) family. Oclacitinib is a nonselective JAK inhibitor, but it predominately inhibits JAK1 and the function of JAK1-dependent cytokines (IL-2, IL-4, IL-6, IL-13, and IL-31). 29 Lokivetimab is a caninized anti-canine IL-31 monoclonal antibody which neutralizes IL-31 and prevents it from binding to dorsal root ganglia of pruritogenic peripheral nerve endings. 27 Calcineurin inhibitors (eg, cyclosporin) are also used commonly in an attempt to avoid adverse effects of chronic glucocorticoid therapy and inhibit T-cell proliferation via inhibition of IL-2 production. 61 Atopica (Elanco, Greenfield, IN, USA) is a modified cyclosporine labeled for refractory AD in dogs and cats. Understanding the complex cutaneous immune environment gives context to these therapies and may provide avenues for future therapeutic success.
Inflammation in AD is predominately lymphocytic, and in cAD increased numbers of T cells are present in lesional and nonlesional skin.81,102 Evidence supports that Th-2 weighted immune responses are involved in the development/sensitization and perpetuation of cAD, but with disease progression the immune response appears to shift to a mixed Th-1/Th-2 or Th-1 predominance.80,89,100 This may coincide with overlapping microbial infection. Cutaneous mRNA profiling in cAD has demonstrated increased IL-4 (which stimulates IgE production) and IL-5 in lesional skin. 75 Others have failed to detect differences in the levels of IL-4 in cAD and healthy skin, although this may speak to disease chronicity and shifting away from a Th-2 predominant cytokine profile.57,81,100 IL-31 is produced by activated Th2 cells, mast cells, dendritic cells, eosinophils, and basophils and is a potent inducer of pruritus in dogs; hence, the target of lokivetimab and oclacitinib in breaking the itch-scratch cycle.15,65 Other Th subsets are more recently highlighted as contributory to the cytokine profile in cAD, including Th9 (IL-9 promotes mast cell growth, cytokine production, and regulatory T-cell stimulation) and Th22 (IL-22 plays a role in epithelial proliferation and differentiation, and antimicrobial peptide production), and upregulation of both IL-9 and IL-22 has been demonstrated in dogs. 80
Dermal mast cells can degranulate following IgE cross linking as well as with IgE-independent pathways.35,20 Studies have reported varied results on whether or not mast cell numbers in the skin of cAD are increased, decreased, or similar compared with normal dogs.36,113,117 It is the authors’ impression that they are more numerous or prominent in lesional skin of cAD. Mast cell degranulation allows for the release of histamine, leukotrienes, prostaglandins, proteases, and cytokines that promote vasodilation, eotaxis, and the Th2 immune response.56,113
The presence of an eosinophilic infiltrate in canine skin, even if mild, should prompt an evaluation for or a suspicion of a hypersensitivity response (not solely type I). The degree of eosinophilic infiltration in studies evaluating the skin of dogs with cAD is highly variable.77,81,89,117 Circulating eosinophilia and neutrophilia have been reported to be significantly higher in dogs with cAD compared with normal dogs. 15 Neutrophilic infiltrates are also variable, and less contributory toward a diagnosis as they may be a reflection of a superimposed infection. Basophils are rarely reported in association with cAD, though are more frequently implicated in the cutaneous allergic immune response in human and mouse studies. 67 This may be due to lack of detection in routine histology and a lack of a validated immunohistochemical assay for canine basophils.
Microbial interactions
The cutaneous microenvironment contains a complex ecosystem of commensal bacteria and fungi, including potential pathogens. Abnormal shifts in the cutaneous microflora (dysbiosis) are documented in both AD and cAD. In dogs with cAD, reduced bacterial diversity in lesional and nonlesional atopic skin and increased proportions of Staphylococcus spp. have been shown with both culture-dependent and culture-independent methods.9,16,54,93 S. pseudintermedius, followed by Staphylococcus schleiferi, both of which may be identified as commensals, are the most frequently detected pathogens in canine bacterial dermatitis and folliculitis (Fig. 1f), whereas Staphylococcus aureus is most associated with AD in people.9,14,26,37,54 Lesional skin in cAD may harbor more bacteria, particularly Staphylococcus, compared with normal skin. 37 Whether this is due to an altered keratinocyte barrier, changes in the chemical microenvironment, or decreased antimicrobial peptide production is unclear. Increased colonization may lead to pruritus and contribute to clinical signs of cAD. 26 Lesions of bacterial dermatitis and folliculitis may manifest clinically as “moth-eaten” alopecia, papules, pustules, crusts, and collarettes (Fig. 1f). The bacterial infection and the subsequent inflammatory response may mask or exacerbate the histopathologic changes, including the superficial perivascular dermatitis and epidermal hyperplasia seen in cAD.
Dermatitis associated with Malassezia overgrowth (Malassezia dermatitis, MD) is typically pruritic, erythematous, and may exacerbate lesions and discomfort in cAD. Secondary lesions of MD include excoriation, lichenification, hyperpigmentation, and intertrigo (Fig. 1g). Paronychia (inflammation of the claw bed), often with red-brown discoloration of the claw and hair, and ceruminous otitis are commonly identified with MD. 32 To further complicate the matter, type I hypersensitivity reactions to Malassezia pachydermatis extracts have been reported in atopic dogs. Malassezia may not only act as an opportunistic pathogen but also as an allergen. 69 Hyperplastic dermatosis of West Highland white terriers is a rare disease, formerly referred to as epidermal dysplasia of West Highland white terriers or colloquially as “armadillo Westie syndrome” (Fig. 1g). Based on the prevalence of MD in this condition, and prior case reports of concurrent cAD, it is likely that this represents either a severe allergic response with secondary MD or an allergic response to Malassezia itself.23,72 Histopathologic findings of MD in dogs overlap those with cAD and may include patchy parakeratotic hyperkeratosis, irregular epidermal hyperplasia, edema, and lymphocyte exocytosis (Fig. 1k, l) T-cell-predominant superficial perivascular dermatitis is generally present. Yeast is inconsistently present histologically. 64
CAFRs and Food Allergy/Sensitivity
CAFRs in dogs encompass cutaneous lesions secondary to food allergy and food intolerance. Food intolerance is due to an alternate pathogenesis with maldigestive/malabsorptive enteropathies, toxicities, or idiosyncratic causes of cutaneous, enteric, or other signs. Food allergies (also known as food sensitivities) are caused by an amplified immune response to a dietary antigen. Clinical manifestations of CAFR with an allergic phenotype (food allergy) include pruritus or urticaria with or without gastrointestinal signs. Strictly speaking, to date most cases of food allergy reported in the veterinary literature fall under the broader umbrella of CAFR as immunologic profiling of reported cases has not been routinely performed. 48 Cutaneous lesions of food allergy are clinically indistinguishable from cAD and dermatologists/clinicians may refer to this as food-induced cAD when there is a lack of gastrointestinal signs, as discussed above. 87 The gold standard for diagnosis of a CAFR (including food allergy) is an elimination diet trial (including novel or hydrolyzed protein diets) followed by food-item provocation. Serum food-specific IgE and IgG elevations in dogs demonstrate low repeatability and poor accuracy in documentation of food allergy and intradermal testing is also unreliable for a diagnosis. 71 Histopathology of the gastrointestinal tract is nondiscriminatory for differentiating CAFR or food allergy from other causes of chronic enteritis/inflammatory bowel disease.1,110 The recommendations for the duration of an elimination diet trial vary, but an elimination trial of 8 weeks may provide upward of 90% sensitivity for a diagnosis.95,105
Other diagnostic assays have been investigated for CAFRs. Lymphocyte proliferation assays have been reported to have good accuracy (80%), with 100% positive and 50% negative predictability in the diagnosis of CAFR, but these tests are currently only offered in research facilities due to rapid processing requirements.21,46,47,53 Patch testing (with controlled surface application of a potential allergen and evaluation of a reaction) was found to be accurate and negative predictability was excellent, though positive predictability is low. There was unsatisfactory accuracy of diagnosis with endoscopic evaluation, fecal food-specific IgE, and hair and saliva testing.4,51,71,105
No sex predispositions for CAFR are recognized.87,108 Breed predilections are infrequently reported for CAFR. Picco et al 87 reported that German shepherd dogs, West Highland white terriers, Boxers, Rhodesian ridgebacks, and Pugs were overrepresented, though many of these breeds are also predisposed to cAD. As the clinical history and the outcomes of treatment are not always discerning between CAFR and non-food-induced cAD, the cutaneous histopathologic findings reported to date for either disease state (as described above) should be viewed in the same light and both should be maintained as differential diagnoses by the pathologist along with ectoparasitism.
Flea Allergic Dermatitis
Flea allergic dermatitis (FAD; aka flea bite dermatitis, flea bite hypersensitivity) is common and most frequently associated with the bite of Ctenocephalides felis. It is encountered in geographical climes that are favorable to the flea and may be nonseasonal. 11 Dogs may be infested by fleas with little to no clinical signs and flea infestation is not synonymous with FAD. 74 The onset of FAD is generally between 1 and 6 years of age. Clinical lesions are typically distributed along the caudal dorsum, hind legs, and tail base, and chewing may be seen more commonly compared with scratching, licking, and rubbing (Fig. 1i). Alopecia and papular dermatitis may develop. In German shepherd dogs and bulldog-type breeds, fibropruritic nodules may be encountered. Historically, FAD has been associated with pyotraumatic dermatitis or “hot spots” (Fig. 2i), but the relationship is not exactly clear and concurrent cAD is possible. FAD is thought to involve both immediate and late phase type I hypersensitivities. Low-molecular-weight haptens in flea saliva and numerous allergens have been identified by studying IgE reactivity of sera.3,35
Histopathologic changes in known flea bite sites include edema and perivascular dermatitis composed of mast cells, eosinophils, and mononuclear cells with intraepidermal eosinophilic pustules. These findings are variable and histologic lesions in the skin are nonspecific.111,116 With flea bites or intradermal testing of flea antigen, erythematous wheals develop within 1 minute and resolve in 2 to 4 hours. 30 At 4 hours, eosinophils predominate (the late phase reaction) with smaller numbers of neutrophils. Chronic lesions are not fully documented in the literature, but superimposed lesions of self-trauma are seen. Further complicating the clinical picture, dogs with cAD may be predisposed to development of FAD, and FAD may be a complicating factor in cAD. Ectoparasitism by mites (e.g., Sarcoptes scabiei, Cheyletiella spp.) may have similar histopathologic findings (Fig. 2a). Again, the diagnostician is left with a reaction pattern supportive of underlying allergic skin disease and/or ectoparasitism, and a final diagnosis is left to the clinician after documentation of disease progression, clinical examination, and response to treatment.
Allergic Contact Dermatitis
Allergic contact dermatitis (ACD) is generally considered uncommon to rare, but the incidence may be geographically dependent as it is more frequently reported in tropical and subtropical regions. 74 A definitive diagnosis of ACD is difficult, as it requires restriction and provocation testing and patch testing with the allergen. ACD is more frequently reported in people, with some hypothesizing that the canine haircoat may offer some degree of protection. In addition, ACD in people is most commonly caused by haptens (eg, heavy metals, industrial chemicals, cosmetics) that dogs do not contact. Lesion distribution of ACD is generally along sparsely haired regions of the ventrum, face, ears, scrotum, perineum, and ventral paws or at sites of application of the sensitizing or irritating agent. 42 Differential diagnoses include cAD, ectoparasitism, CAFR, ADRs, infection, and irritant contact dermatitis. ACD and cAD may occur concurrently, which can confound the diagnosis, and historical reports of ACD in dogs should be scrutinized.
ACD is an exaggerated antigen-specific immune response (historically considered a type IV hypersensitivity, but potentially with a component of a type I hypersensitivity) to percutaneous penetration of allergen (haptens), requiring prior exposure/sensitization. 42 This is delineated from irritant contact dermatitis (ICD), which represents a nonspecific inflammatory reaction to an irritating or caustic surface agent, for which prior exposure is not required (though irritants may act as allergens in allergic individuals) for which prior exposure is not required. To date, the pathogenesis of ACD in dogs has not been established, but parallels can be drawn from investigations in people and rodent models. Reported allergens include metals, topical medications and their vehicles (eg, neomycin and propylene glycol), shampoos and topical dermatological products, plastics, cement, wood, and plants (potentially contributing to the geographically variable incidence) including the families Commenlacae (eg, “wandering jew” or “wandering dude,” Tradescantia fluminensis and spiderwort, Tradescantia virginiana) and Amarlidaceae (Hippeastrum), and Trachelospermum asiaticum (Asian jasmine).42,74
Histopathologic findings in ACD are nonspecific and inconsistent, and a biopsy sample may be nondiagnostic with a pattern of superficial perivascular to lichenoid dermatitis, potentially with a predominance of neutrophils or eosinophils, and variable spongiosis and vesicle formation. 31 Robust studies on histopathologic findings of ACD in domestic animals are lacking. There may be histopathologic overlap with ICD and ACD, although ICD may impart a more direct cytotoxic effect on keratinocytes with subsequent inflammation. Therefore, careful attention should be paid to epidermal changes, including confluent parakeratosis with variable hyperplasia and spongiosis, compact orthokeratotic hyperkeratosis (ichthyosiform irritant reaction), or necrosis (as a chemical/caustic insult).42,112 If there is time for the epidermis to begin to repair following exposure, a band of diffuse parakeratosis in the stratum corneum subtended by a layer of basketweave orthokeratin may be suggestive of a surface irritant.
Urticaria and Angioedema
Urticaria (multifocal and coalescing erythematous wheals) and angioedema (Fig. 1h) occur secondary to mast cell degranulation and histamine release, and unlike other erythematous dermatoses lesions blanch under applied pressure (diascopy).74,122 These lesions may be encountered with allergic flares, but nonimmunologic mechanisms can also cause mast cell degranulation. Causes include adverse food or drug reactions, arthropod envenomation, excessive heat or cold, and dermatographism.74,96 The diagnostician is tasked with ruling out other causes for vasodilatation and edema such as vasculitis, lymphangitis, infectious disease processes, and neoplasia. 74 Urticaria/angioedema may manifest histopathologically as edema with separation of collagen bundles and vascular dilation with minimal inflammation. With chronicity, there may be interstitial fibrosis and interstitial histiocytes engulfing edema fluid. If there is a basophilic tint to the edema (ie, myxedema or mucin), cutaneous mucinosis of shar-pei dogs or hypothyroidism may be considered as histopathologic differentials, but the clinical progression and signalment should be telling.22,121
Adverse Reactions to Treatment of Allergy
ADRs to medicaments used to treat chronic hypersensitivities occur and the resultant skin lesions may be biopsied. In these cases, the history and clinical courses may otherwise be supportive of chronic allergic skin disease. The histologic pattern may differ from chronic hyperplastic superficial perivascular dermatitis, as above. Examples of ADRs secondary to the treatment of chronic hypersensitivities include calcinosis cutis, cutaneous and adnexal atrophy with chronic glucocorticoid therapy, and adverse reaction to topical sprays containing betamethasone. 99 Chronic use of cyclosporine or other calcineurin inhibitors has been associated with the development of gingival hyperplasia and lymphoplasmacytic lichenoid psoriasiform dermatosis (Fig. 2b).24,114 More recently, opportunistic infections by Burkholderia cepacia complex have been reported in dogs treated with cyclosporine and were associated with severe neutrophilic and pyogranulomatous furunculosis, with potential for progression to sepsis.2,13 Other systemic and cutaneous signs associated with cyclosporine therapy are rarely reported.24,85 An increased incidence of cutaneous histiocytomas has been reported with oclacitinib therapy versus cyclosporine therapy, 39 but a larger retrospective cohort study showed no increased prevalence of skin masses with oclacitinib therapy versus age-/breed-matched control dogs that were oclacitinib-naïve. 55
Other Canine Dermatoses to Consider
With the commonality of allergic skin disease, the nonspecific histopathologic findings in many cases, and the potential for a disconnect between the clinician and the diagnostician in terms of history, sample acquisition, or communication, there are other entities that the diagnostician should keep in mind to avoid a misdiagnosis or misinterpretation. If the dermal inflammatory infiltrate contains a significant eosinophilic component, other features in the biopsy samples or history may aid in arriving at a correct or more specific diagnosis. In some instances, there may be a history and clinical lesions supportive of chronic allergic skin disease, but the biopsy submitted to the diagnostician may indicate another disease. A few examples are included below.
Pemphigus Foliaceus
Pemphigus foliaceus is the most common cutaneous autoimmune disease in dogs with autoantibodies targeting desmocolin 1 of the desmosome and subsequent acantholysis. 5 Pemphigus foliaceus is characterized histopathologically by broad pustules with nondegenerate granulocytes and acantholytic keratinocytes that span multiple adnexal units. Both neutrophils and eosinophils may be present in the dermis and pustules, and the significance of the eosinophilic versus neutrophilic infiltrate is not known. 107 If pustules and crusts are not captured, the resultant histopathologic picture is that of a chronic hyperplastic dermatitis, which is also seen in allergy and pyoderma (Fig. 2c, d). Anecdotally, cases of pemphigus may have a higher incidence of chronic allergic dermatitis, but whether this is a co-morbidity, trigger, or a true association is not yet determined. ADRs may have histologic lesions similar to those observed in pemphigus foliaceus.
Canine Acute Eosinophilic Dermatitis and Edema
CAEDE (formerly Wells-like syndrome) is an uncommon syndrome in dogs with an acute presentation of erythroderma and edema, and in most cases is associated with gastrointestinal signs preceding or concurrent with skin lesions.44,62 Skin lesions may manifest as generalized violaceous erythroderma (Fig. 2d, e), plaques, red targetoid macules, or patches. While most dogs fully recover, systemic signs may be severe and potentially fatal leading to euthanasia or death (multiorgan failure). Peripheral eosinophilia may be encountered along with hypoproteinemia and hypocholesterolemia, which are attributed to vascular leakage with marked peripheral edema.12,62,63 Holm et al 44 first reported on this condition in a case series of dogs, drawing clinical comparisons with Wells syndrome in people. The gastrointestinal signs are not attributed to a specific gastrointestinal disease and are not universally present.12,62 ADR has also been investigated as a putative cause retrospectively, and many cases have a recent history of treatment with drugs directed at enteric disturbances. However, not all cases have positive drug associations when subjected to a scoring rubric.8,63 Histopathologic patterns associated with CAEDE, which were reported by Cain et al, 12 encompass a spectrum of severity. Inflammatory patterns range from mild superficial inflammation with vascular ectasia (pattern 1), moderate mid-dermal to superficial dermal inflammation and edema (pattern 2), to severe and diffuse eosinophilic infiltrates in the dermis with edema, variable epidermal vesiculation, and the formation of collagen flame figures (pattern 3). 44 The histopathologic pattern can overlap with AD, especially in patterns 1 and 2, but key discriminating features include abrupt onset, often with gastrointestinal signs, lack of histologic features of chronicity (epidermal, adnexal hyperplasia), and dramatic erythema with relatively sparse inflammation in pattern 1 (Fig. 2f). Distinguishing eosinophils from neutrophils in CAEDE can be difficult due to eosinophil degranulation, but may be aided by a Luna stain. An attempt to distinguish CAEDE from sterile neutrophilic dermatosis should be made. 8 Sterile pustular erythroderma of miniature schnauzers may also contain an eosinophilic infiltrate and can have overlapping histopathologic features of CAEDE (pattern 3) and sterile neutrophilic dermatosis.
Eosinophilic Furunculosis of the Face
Eosinophilic furunculosis of the face is an uncommon skin disease in dogs that is clinically distinctive and is characterized by an eruption of papules, pustules, and ulcerative plaques on the dorsal muzzle/nasal bridge (Fig. 2g). The periocular region, pinna, and rarely other foci of haired skin are involved.19,28,106 No breed predispositions are reported, though anecdotally younger to middle-age large breed dogs may be overrepresented. There have been suggestions that bites or stings be an inciting cause with a subsequent hypersensitivity reaction. 31 The signalment, rostral muzzle location, and marked eosinophilic inflammation are the compelling factors that support the hypothesis that this is due to an insect bite hypersensitivity reaction (eg, Hymenoptera bites/stings).
Typical histopathologic findings are severe eosinophilic folliculitis and furunculosis with necrosis and disruption of the follicular outer root sheath and a massive dermal infiltrate consisting of eosinophils and fewer neutrophils (Fig. 2h). The attendant inflammation may become granulomatous over time. Lesions typically respond rapidly to systemic glucocorticoid therapy.
Zinc-Responsive Dermatosis
Eosinophils can be identified as a superficial perivascular infiltrate in some cases of zinc-responsive dermatosis, which are seen most frequently in the Siberian husky and other Nordic breed dogs. The lesions are variably pruritic. It is not clear whether these are dogs with concurrent allergic skin disease or whether this infiltrate resolves with zinc supplementation. Although parakeratosis may be encountered from surface trauma and increased keratinocyte turnover, in zinc-responsive dermatosis the parakeratosis is generally more diffuse with involvement of the follicular ostium. In the authors’ experience, clinical histories are not typically supportive of concurrent allergic skin disease.
Lesions of Surface Trauma
Localized lesions secondary to chronic or intense pruritus with or without superficial or deep bacterial infection may be biopsied, including fibropruritic nodules, acral lick dermatitis/granuloma, and pyotraumatic/acute moist dermatitis. Fibropruritic nodules in dogs are exophytic, restricted to the superficial dermis, and are composed of dense collagen, reactive fibroblasts, and a perivascular inflammatory infiltrate. These nodules are grossly distinctive, but may be biopsied to rule out infectious or neoplastic disease processes. The epidermis is markedly hyperplastic with exaggerated rete and may be compact from repetitive surface trauma. The inflammatory infiltrate is often mixed with a combination of lymphocytes, plasma cells, and neutrophils with few eosinophils. 31 Fibropruritic nodules are most frequently encountered on the dorsal lumbosacral region and caudal thighs in association with chronic FAD. German shepherd and bulldog-like breeds may be predisposed. There may be histopathologic overlap with other forms of chronic trauma and dermatitis (eg, acral lick dermatitis/granuloma). Acral lick dermatitis/granulomas tend to occur on the distal limb and are resultant of chronic repetitive surface trauma. Pruritus, secondary to allergic skin disease, may be an inciting factor in some cases. The histopathological lesions may be like that described above, though frequent foci of furunculosis and subsequent granulomatous dermatitis are encountered.
Pyotraumatic dermatitis (colloquially known as “hot spots”) are focal ulcers or plaques that are frequently intensely pruritic (Fig. 2i). These lesions may present acutely and frequently require glucocorticoid therapy to quell the inflammation. There has historically been an association with these lesions and FAD or cAD as the inciting cause of the intense “itch-scratch cycle.” Lesions most frequently occur on the cheek, lateral thigh, neck, and lower back. Histopathological findings include ulcers with superficial neutrophilic inflammation with pustules, folliculitis, and furunculosis variably present. These lesions may or may not have a significant eosinophilic infiltrate. 43
Other clinical lesions (reviewed by Morris et al) that may be seen in association hypersensitivity reactions include mucocutaneous pyoderma (MCP), lip fold pyoderma, and intertrigo (bacterial and/or yeast overgrowth within skin folds or due to superficial friction, which results in barrier disruption). 49
Conclusion
Allergic dermatitis is common in dogs, though challenging to definitively diagnose and treat. Lesions are frequently complicated by self-trauma and microbial infection. Careful consideration of the clinical history of these cases allows for optimal interpretation of biopsy samples and provides a useful clinical aid. As surface-oriented eosinophilic dermatitis may have a similar histopathologic presentation across many diseases, maintaining a broad list of differential diagnoses and scrutinizing nuances in the biopsies and clinical history is essential. The diagnostic pathologist may not be able to provide a definitive diagnosis in many cases of allergy, but they still provide a critical function. The importance of working relationships and strong communication with clinical colleagues to achieve a final diagnosis cannot be overstated.
Supplemental Material
sj-pdf-1-vet-10.1177_03009858231189298 – Supplemental material for A review of cutaneous hypersensitivity reactions in dogs: A diagnostician’s guide to allergy
Supplemental material, sj-pdf-1-vet-10.1177_03009858231189298 for A review of cutaneous hypersensitivity reactions in dogs: A diagnostician’s guide to allergy by Charles W. Bradley, Elizabeth A. Mauldin and Daniel O. Morris in Veterinary Pathology
Footnotes
Declaration of Conflicting Interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: D.O.M. provides consultation services and has received research support from Stallergenes Greer Laboratories, North Carolina, USA.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
