Abstract
An 8-year-old, male, mongrel dog developed severe cough and anorexia and died within 3 months. Autopsy revealed an invasive grayish-white mass in the right kidney and multiple nodules in the lungs, thoracic wall, and spleen. Histologically, the renal mass and the other nodules were mainly composed of papillotubular structures lined by oval-to-polygonal pleomorphic cells. The cells were reactive with DBA, PNA, and UEA-1 lectins and positive for vimentin but negative for CD10 and high molecular weight cytokeratin. Because of its histological, histochemical, and immunohistochemical similarities with human collecting duct carcinoma (CDC), a diagnosis of renal collecting duct carcinoma with pulmonary, thoracic, and splenic metastases was established. To our knowledge, this is the first case report of CDC in animals.
In dogs, primary renal neoplasia is uncommon; its prevalence is estimated to be 0.3–1.5% of all canine neoplasms. 2, 10, 11, 14 Generally, such neoplasms arise in middle-aged dogs without breed predilection. The most common clinical signs of canine renal tumors are anorexia, lethargy, weight loss, hematuria, and proteinuria. 4, 18 The most common histologic type of renal neoplasm is renal cell carcinoma (RCC). Although RCCs in dogs have been classified previously into several subtypes, based on their histologic (papillary, tubular, and solid type) and cytologic (chromophobic, eosinophilic, and clear cell type) patterns, the differences in the biologic behaviors of these subtypes remain to be elucidated. 10
Human RCCs are classified as clear cell, papillary, chromophobe, and collecting duct carcinoma (CDC) based on histopathologic and molecular biologic properties. Oncocytoma, a benign neoplasm derived from the collecting duct, is also described. Among renal tumors, CDC is the most aggressive. 8 Here, we describe a canine renal neoplasm with morphologic, histochemical, and immunohistochemical characteristics similar to those of human CDC.
An 8-year-old, male, mongrel dog was referred to the veterinary hospital with the complaint of persistent cough and anorexia. The main findings were the presence of multiple nodules in the lungs on thoracic radiography and elevated serum alkaline phosphatase level (1,439 mU/ml). There were no signs of urinary tract disease, and urinalysis was not performed. A primary pulmonary neoplasm was suspected, and a combination chemotherapy with filacul, adriamycin, cisplatin, and prednisolone was instituted. Despite intensive care, the dog progressively lost weight, developed anemia, and finally died of respiratory failure 87 days following initial presentation.
Necropsy, performed 5 hours after death, revealed a grayish-white mass (2.5 × 2.3 × 2.0 cm) in the right kidney (Fig. 1). On cut section, the mass invaded the renal parenchyma from the medulla to the cortex in a radiating manner. Multiple nodules were also noted in the lungs (Fig. 2), thoracic wall, and spleen; the pulmonary parenchyma was almost replaced by the nodules. The right cardiac ventricle was dilated, presumably due to altered pulmonary hemodynamics. Based on macroscopic findings, the presumptive diagnosis was primary renal malignancy with multiple metastases in the lungs, thoracic wall, and spleen.
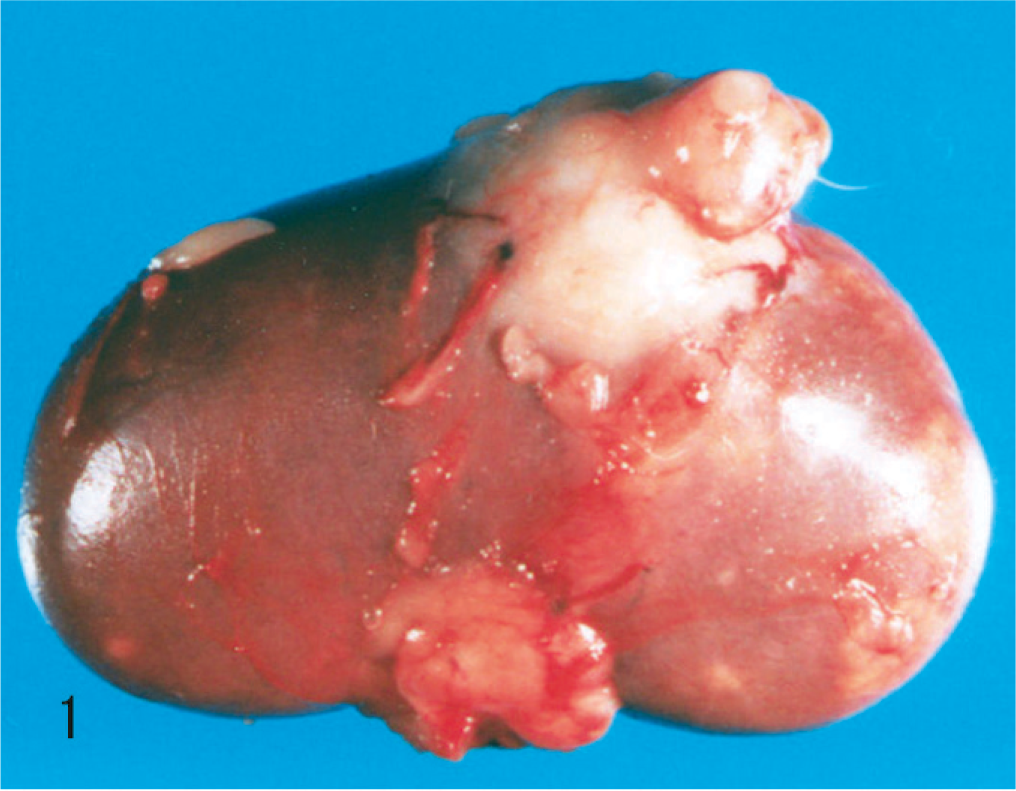
Kidney; dog. Renal collecting duct carcinoma. A grayish-white mass bulges from the capsular surface of the right kidney.
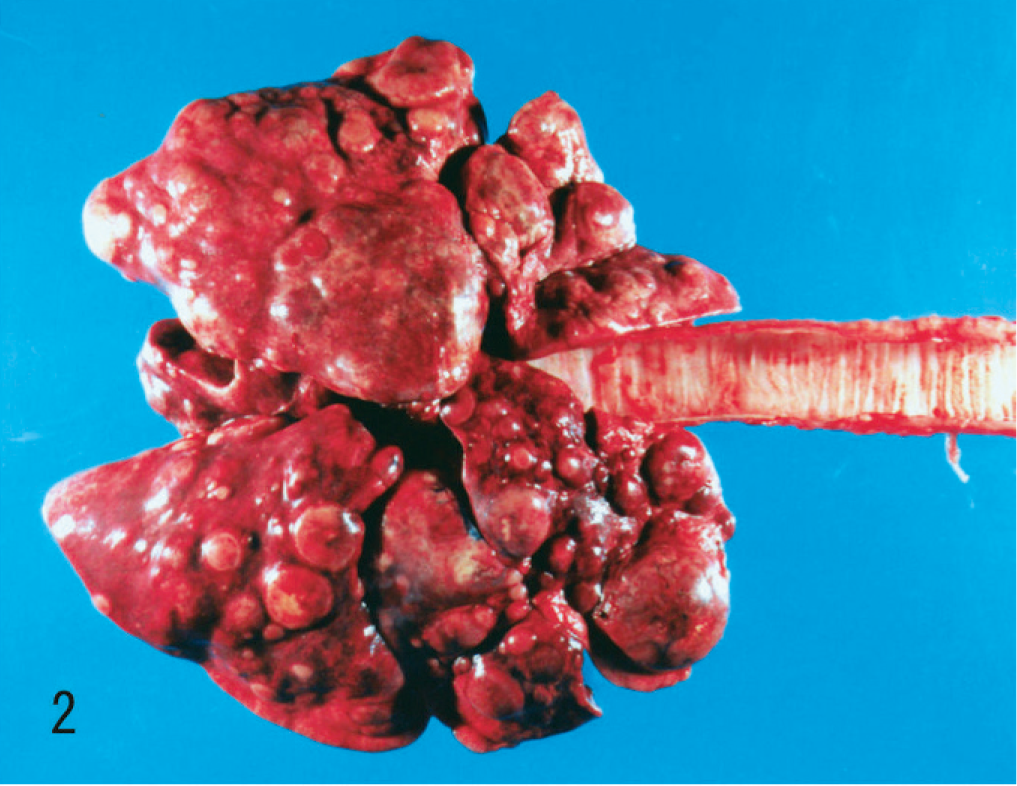
Lungs; dog. Metastatic renal collecting duct carcinoma appears as multiple nodules scattered through both lungs.
Selected tissues were immediately fixed in 10% neutral-buffered formalin, embedded in paraffin, and cut into 4-µm-thick sections for histologic examination. The tissue sections were stained with HE and Verhoeff van Gieson. The nuclear grade of the neoplastic cells was determined according to Fuhrman's 4-grade system for human renal tumors. 7 For phenotypic characterization of the neoplastic cells, lectin histochemistry and immunohistochemistry were performed by using serial paraffin sections.
For lectin histochemistry, the tissue sections were incubated with biotinylated lectins at room temperature for 3 hours. The lectins used were Dolichos biflorus agglutinin (DBA, 1 : 50; Seikagakukogyo, Tokyo, Japan), peanuts agglutinin (PNA, 1 : 50; Seikagakukogyo), and Ulex europaeus agglutinin-1 (UEA-1, 1 : 50; Seikagakukogyo). The adsorbed lectins were detected by incubation with peroxidase-conjugated streptavidin, followed by a 3, 3′-diaminobenthidine (DAB) reaction.
For immunohistochemistry, the following primary mouse monoclonal antibodies were used: anti-CD10 (1 : 50; Novocastra, Newcastle, UK), high molecular weight cytokeratin (HMW-CK, 1 : 50; Dako, Carpinteria, CA), and antivimentin (1 : 100; Dako). The labeled antigens were detected using the streptavidin-biotin (SAB) method with the HistoFine kit (Nichirei, Tokyo, Japan), followed by a DAB reaction. Samples from the contralateral kidney were processed in an identical manner and used to compare the reactivity of neoplastic cells with nonneoplastic renal tissue.
The renal mass observed at necropsy had an invasive and radiating growth pattern from the inner medulla to the outer cortex, with necrosis and hemorrhage (Fig. 3). Histologically, the neoplasm was composed of papillotubular structures lined by simple or pseudostratified epithelium. The tumor was associated with desmoplastic stroma with few granulocytes. Neoplastic cells varied in size, were oval to polygonal, and had pleomorphic nuclei with prominent nucleoli and scant eosinophilic cytoplasm (Fig. 4). Numerous mitotic figures were observed. Nuclear features corresponded to a grade-4 neoplasm, according to Fuhrman's system. 7 Neoplastic cells disrupted the renal capsule, invaded the perirenal fat, and were observed within the lumen of veins (Fig. 5). Renal pelvic urothelium did not have any neoplastic change (Fig. 6), and there was no evidence of pyelonephritis. The nodules found in the lungs, thoracic wall, and spleen were histologically similar to the renal proliferation. No metastases were detected in the brain.
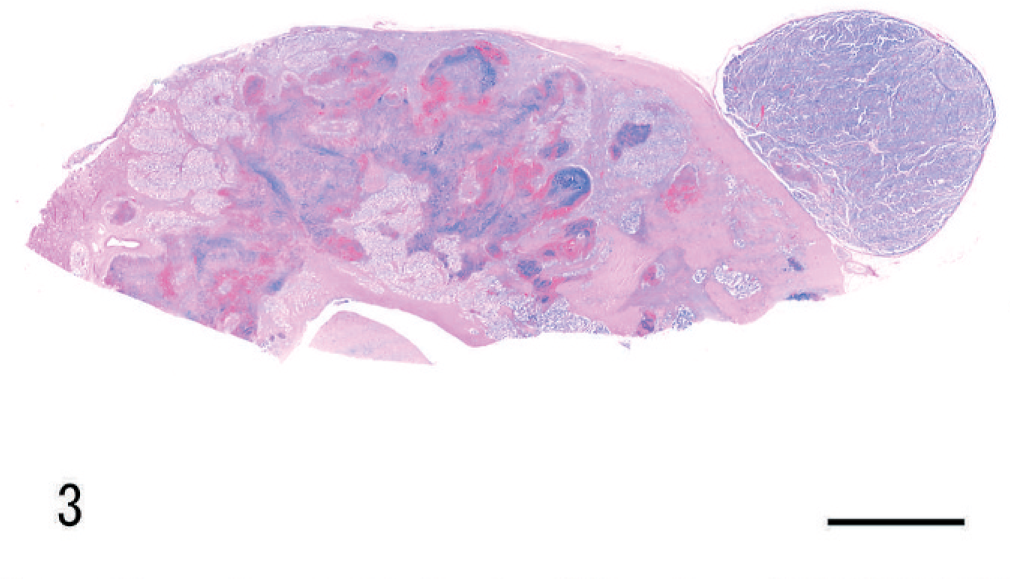
Kidney; dog. Renal collecting duct carcinoma. Invasion of the pelvis and medulla. Note the necrosis and hemorrhage. HE. Bar = 0.5 cm.
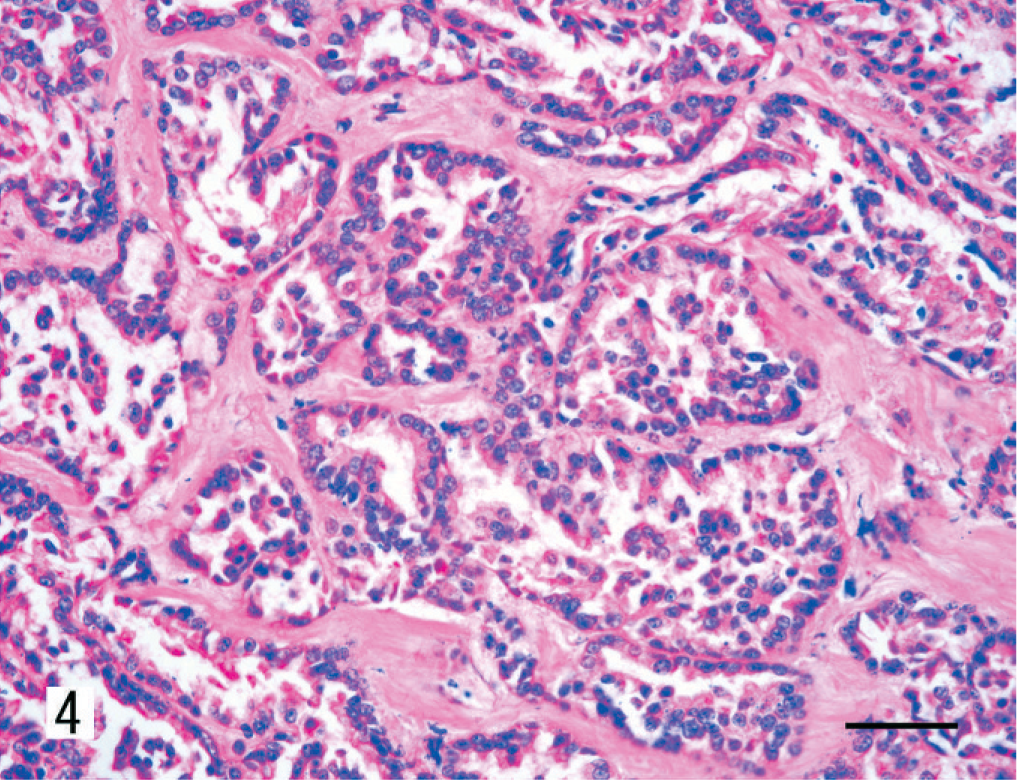
Kidney; dog. Renal collecting duct carcinoma. The tumor cells vary in size and shape, from an oval to a polygonal shape, and display a high degree of nuclear atypia and an eosinophilic cytoplasm. HE. Bar = 25 µm.
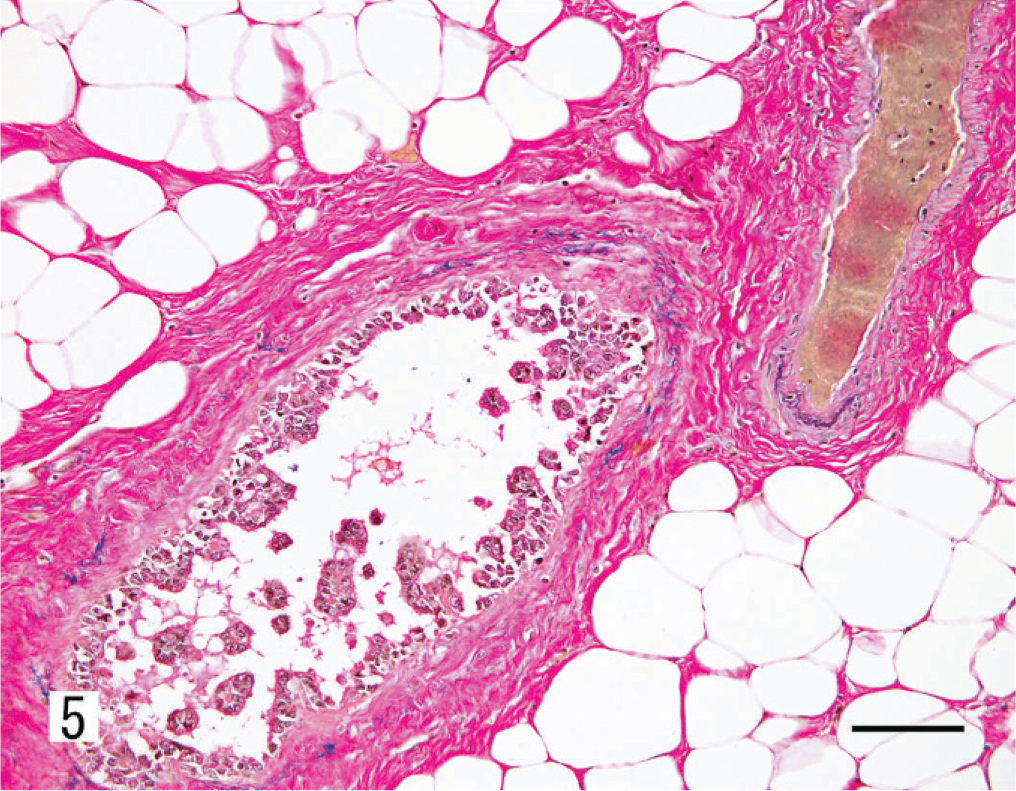
Kidney; dog. Renal collecting duct carcinoma. Invasion of blood vessels by the neoplastic cells. Verhoeff van Gieson. Bar = 50 µm.
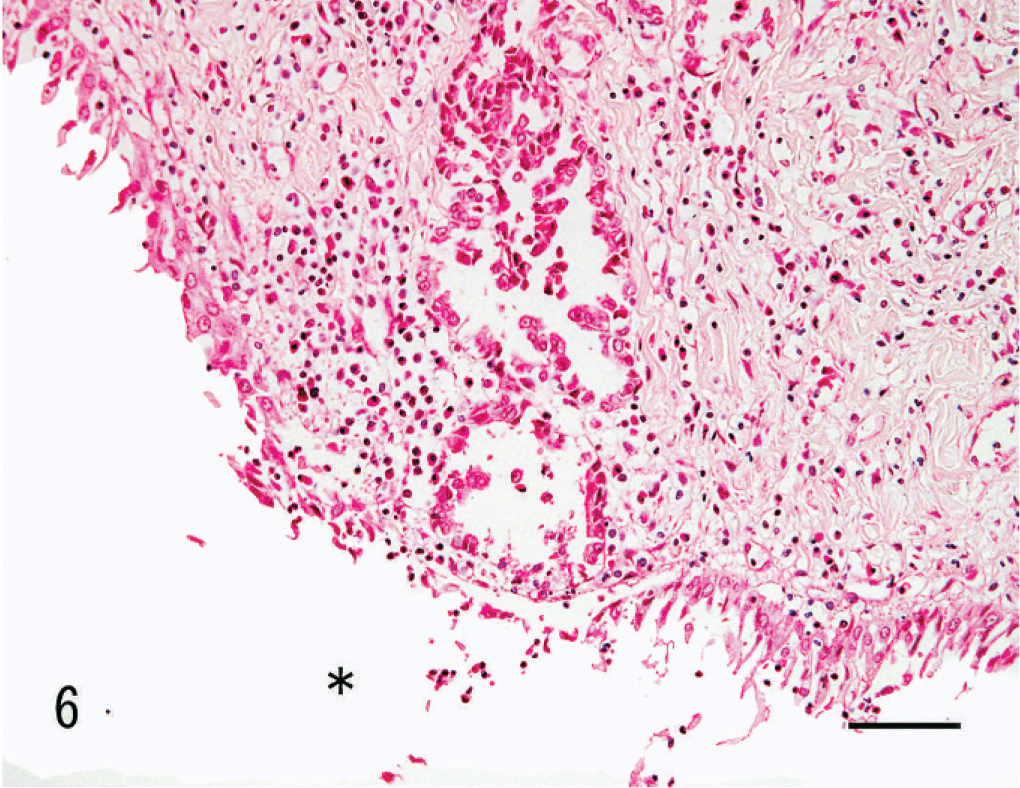
Kidney; dog. Renal collecting duct carcinoma. Neoplastic cells have replaced the epithelial lining of the collecting duct at the opening to the pelvis without involvement of the pelvic mucosa. The asterisk indicates the pelvic cavity. HE. Bar = 40 µm.
The lectin histochemical and immunohistochemical features of the renal neoplasm and nonneoplastic renal tissue are listed in Table 1. The proximal tubules were CD10-positive. Reactivity with DBA, PNA, and UEA-1, and immunopositivity for HMW-CK were evident in the distal tubules, collecting tubules, and collecting duct. Notably, DBA was reactive only in the collecting duct and in the portion of the collecting tubule that was continuous with the collecting duct (Fig. 7). The glomerular epithelium and vascular endothelium were both positive for vimentin. Neoplastic cells were reactive with DBA (Fig. 8), PNA, and UEA-1, and were also positive for vimentin. The metastatic neoplastic cells in the lung had the same patterns of reactivity as the renal neoplasm (Fig. 9).
Results of histochemical and immunohistochemical analysis of a canine renal CDC.
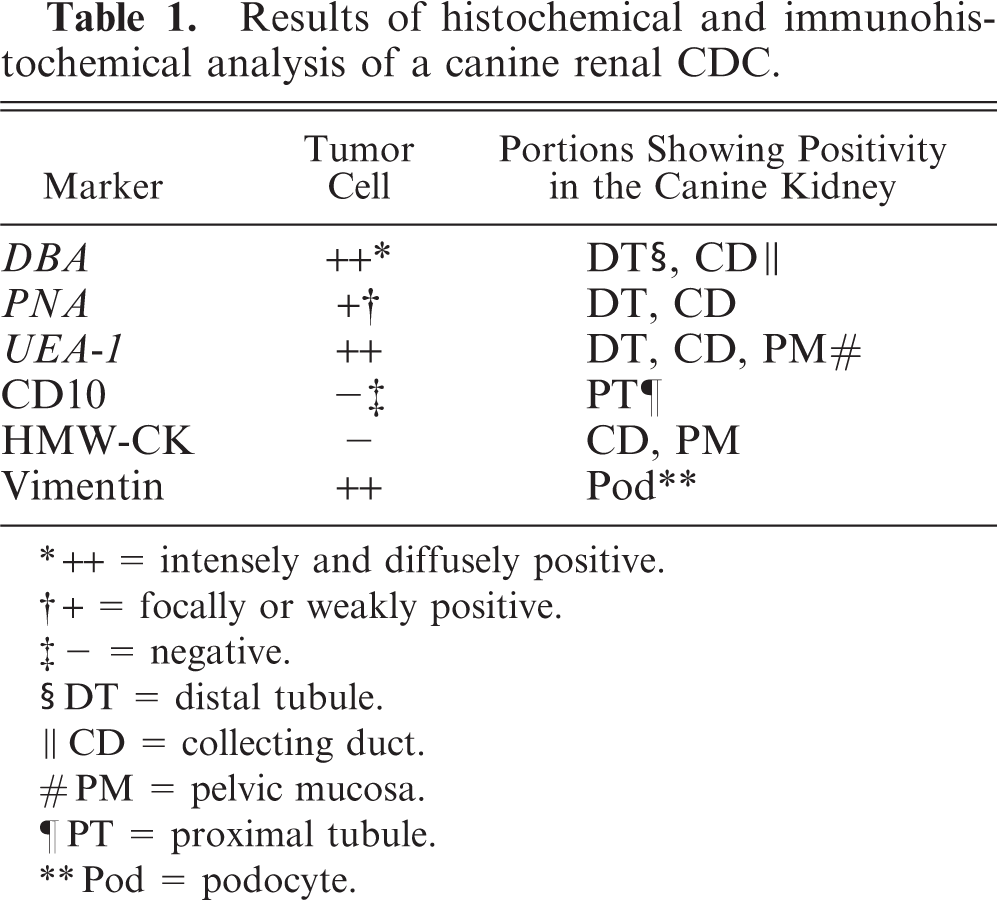
++ = intensely and diffusely positive.
+ = focally or weakly positive.
- = negative.
DT = distal tubule.
CD = collecting duct.
PM = pelvic mucosa.
PT = proximal tubule.
Pod = podocyte.
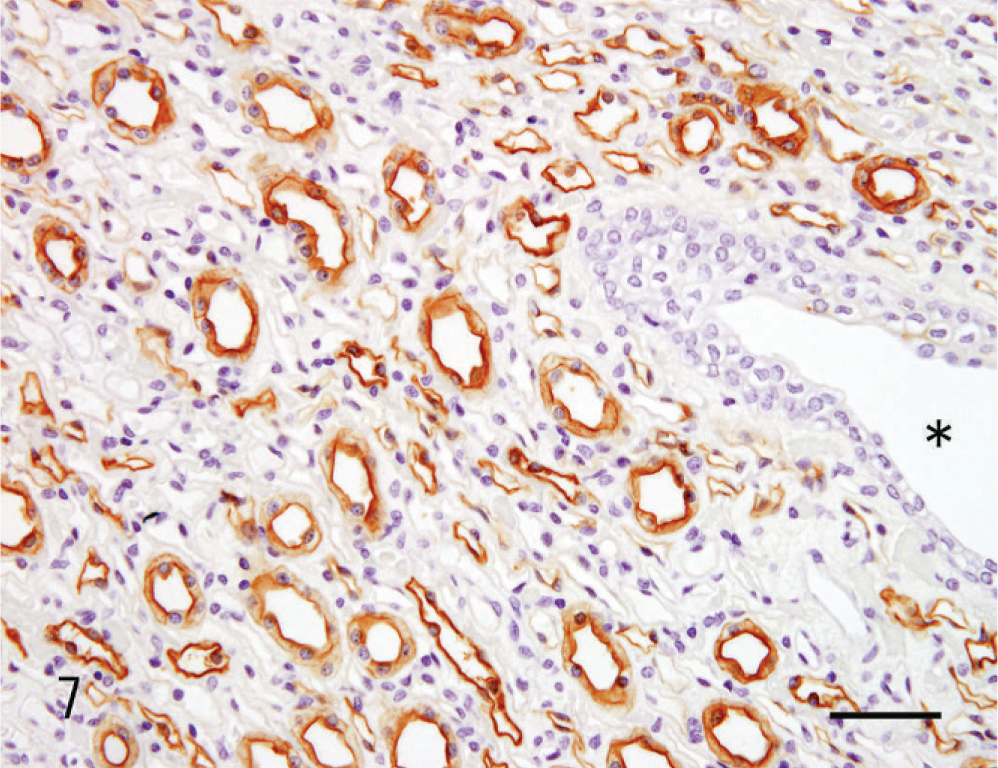
Kidney; dog. Renal collecting duct carcinoma. In normal kidney tissue, DBA is reactive only with the collecting duct epithelia and not with pelvic urothelium. The asterisk indicates the pelvic cavity. Bar = 25 µm.
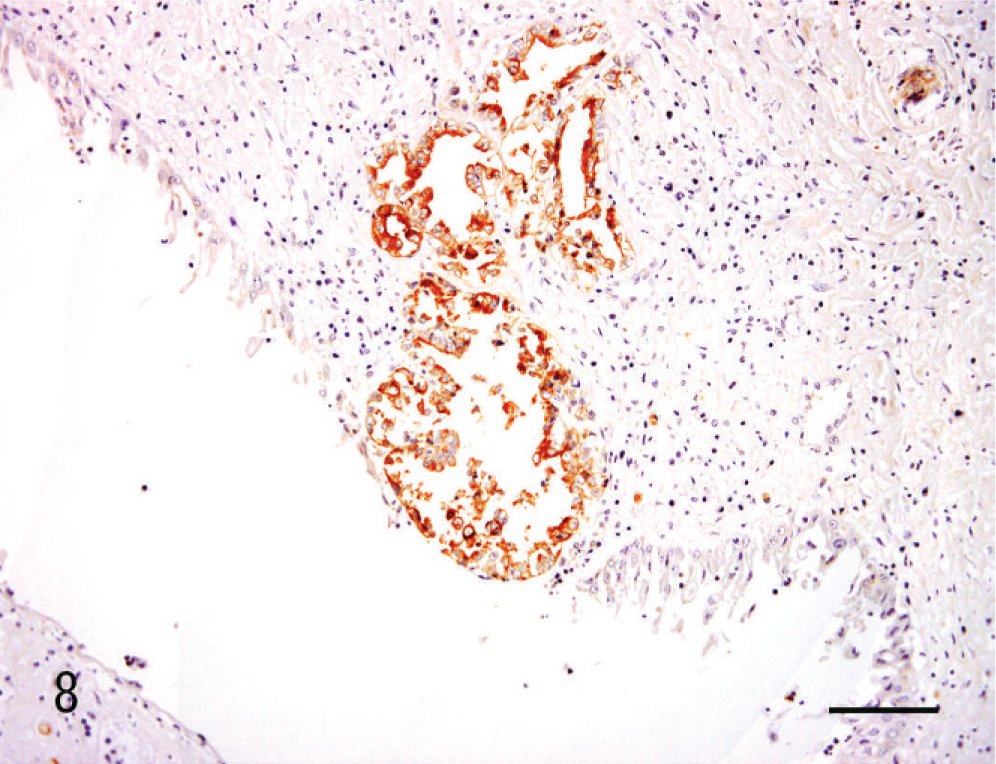
Kidney; dog. Renal collecting duct carcinoma. DBA is reactive with canine renal CDC tumor cells. Bar = 40 µm.
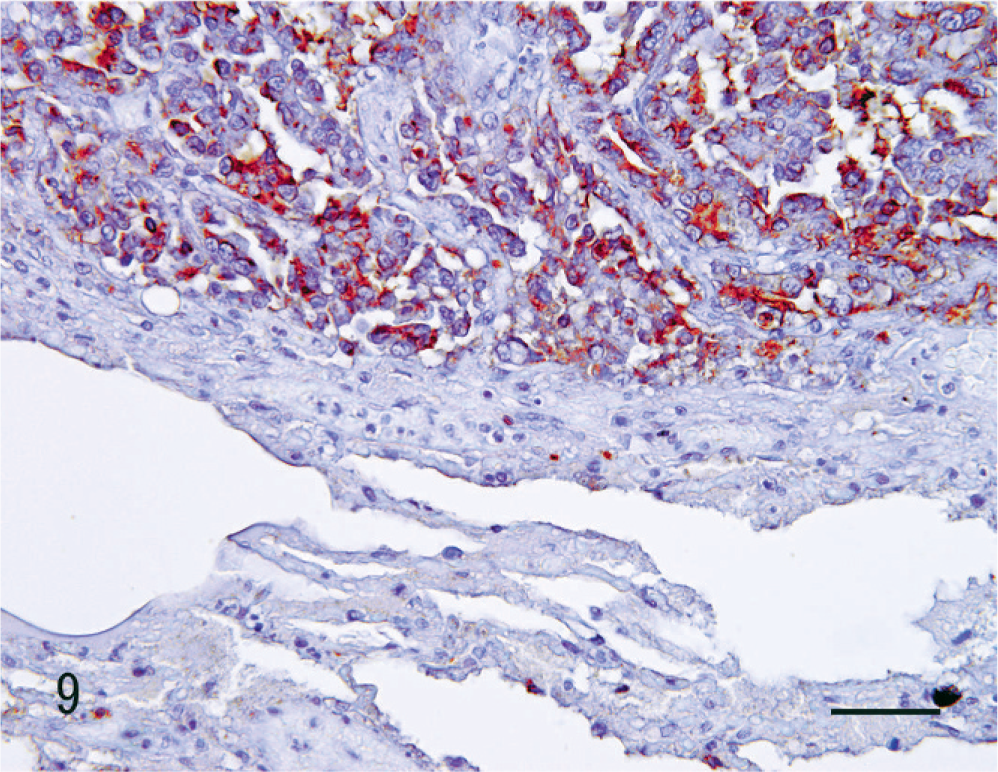
Kidney; dog. Renal collecting duct carcinoma. DBA is reactive with tumor cells in the pulmonary metastasis. SAB method, counterstained with hematoxylin. Bar = 25 µm.
Human renal epithelial neoplasms are classified according to their histopathologic and molecular biologic characteristics. 18 The majority of the human RCCs are of the clear cell type, which is derived from the proximal tubular epithelium. In contrast, chromophobe RCCs, CDCs, and oncocytomas are derived from the collecting duct. 13, 18 Human CDC was first reported by Mancilla-Jimenez et al. 9 as “a papillary carcinoma associated with adjacent dysplastic collecting ducts.” Subsequently, this tumor type was thought to be derived from the collecting duct epithelia close to the pelvic mucosa. However, the diagnostic criteria for CDC are still controversial, even though the World Health Organization (WHO) has proposed several criteria for the diagnosis of these lesions (Table 2). 8, 13, 18
Assessment of the current canine renal CDC case in relation to the WHO diagnostic criteria for human CDC.

NA = Not applicable.
The canine tumor was of large size.
Previously, clear cell RCC, oncocytoma, urothelial carcinoma, and squamous cell carcinoma have been reported in dogs, 5, 6, 10, 12 but no cases of CDC. To our knowledge, we present the first report of a canine renal tumor that is morphologically similar to human CDC. As shown in Table 2, this case met most of the major and minor criteria required for a diagnosis of CDC. 18 With respect to the location criteria, Minor Criterion 1 (central location) was applied instead of Major Criterion 1 (location in a medullary pyramid) due to the large size of the tumor in this case. Notably, tumor cells were found to have replaced the epithelial lining of the collecting duct opening to the pelvic region (Fig. 6), which was the most convincing evidence that this tumor was indeed derived from the collecting duct. There was no evidence of urothelial carcinoma in the pelvic mucosa.
Human CDC lesions can be associated with atypical epithelium in the neighboring collecting duct, which is considered a precancerous lesion. 8, 13, 18 In our canine case, such lesions were not observed. Reactivity to UEA-1 and immunopositivity for HMW-CK are included as WHO criteria for diagnosing human CDC. 18 Neoplastic cells in human CDC retain lectin histochemical characteristics and immunohistochemical properties like those of the distal nephron, particularly of the collecting duct. 8, 13, 15, 16, 18 The lectin histochemical reactivities analyzed in the contralateral kidney (nonneoplastic) tissues of dogs differed from those of the human kidney. In dogs, UEA-1 is strongly reactive with the distal tubule rather than the collecting duct epithelia, and DBA reacted only with the collecting duct epithelia and that part of the collecting tubule adjacent to the collecting duct. Hence, DBA would be a more beneficial marker than UEA-1 in the diagnosis of canine CDC. Therefore, our immunohistochemical data for the current canine CDC case were consistent with those of human CDC.
Immunoreactivity with HMW-CK has been included among the WHO diagnostic criteria for human CDC. However, positivity for HMW-CK is not necessarily observed in these tumors. 15, 16 Interestingly, our canine case did not express HMW-CK. Human high-grade epithelial malignancies are frequently found to be positive for vimentin, a mesenchymal cell marker, but not for cytokeratin, an epithelial cell marker. 3 This might be a result of epithelial-mesenchymal transition. The gross and histopathologic features of our current animal case therefore met the established diagnostic criteria for human CDC. Consequently, we conclude that this canine tumor should be diagnosed as a CDC.
Clinically, hematuria is the most common symptom of canine renal tumors and has been noted in 32% of the cases. 4 Although hematuria, flank pain, and flank mass are also classical characteristics of human RCC, 4 none of these features was evident in our current case. Hence, the clinical diagnosis of primary renal malignancy was not established.
The direct cause of death of the dog was dyspnea due to multiple metastases that had replaced the pulmonary parenchyma. The right atrium and ventricle of the heart were compressed by these metastases, although the right ventricle was dilated due to increased pulmonary vascular resistance. Given that the median survival of canine renal carcinoma has been reported to be 16 months (range 0–59 months), 4 the clinical course of this case was short (87 days); this may reflect the severity of the pulmonary metastases.
In humans, each type of renal tumor has different biologic behaviors and properties, and thus requires a specific therapeutic regimen. 1 Many cases of RCC have been successfully treated by surgical resection in the early stages of the disease; this has been facilitated by advances in radiologic imaging. However, CDCs have an unfavorable prognosis due to their high frequency of postoperative recurrence and distant metastases. 19 At present, an effective therapy has not been established for human CDC. 11 The impact of adjuvant chemotherapy upon the survival in canine renal tumors has not been established. 4 In this case, the anemia and hemorrhagic diathesis may have been secondary to the chemotherapeutics.
In conclusion, we report a case of renal CDC in a dog that satisfies most of the accepted diagnostic criteria for human CDC. 17, 18 To our knowledge, this is the first report of CDC in animals. A precise diagnosis and classification of renal tumors is important for prognostic purposes and for the future establishment of effective treatments.
