Abstract
Exocrine pancreatic carcinoma is a particularly malignant neoplasm of the dog. Clinical and pathologic findings of an unusual variant of exocrine pancreatic neoplasia termed hyalinizing pancreatic adenocarcinoma were evaluated in 6 dogs. On microscopic examination, neoplasms were composed of tubules and acini of epithelial cells, with bright eosinophilic granular apical cytoplasm. Tubular lumina and tumor stroma contained abundant hyaline material that resembled amyloid. The hyaline material was not congophilic, and tumor cells and hyaline material were immunohistochemically negative for amyloid A, immunoglobulin light chains (κ and λ), amylin (islet amyloid polypeptide), laminin, and α1-antitrypsin. Two patients survived longer than 15 months after diagnosis; one of these dogs was untreated and had grossly evident metastasis at the time of diagnosis. The deaths of the other 4 dogs occurred as a result of poor recovery after partial pancreatectomy or in association with other concurrent life-threatening conditions. Two dogs were diagnosed with panniculitis, a condition rarely associated with pancreatic disease. Further evaluation is needed to determine the composition and biologic significance of intratumor hyaline material. Studies that associate exocrine pancreatic carcinoma grade and histologic subtype with prognostic outcomes in the dog are warranted such that appropriate therapy can be elected.
Introduction
Pancreatic neoplasia is uncommon in dogs and occurs less frequently in cats and rarely in other domestic animals. 11, 19, 24 Most pancreatic neoplasms of the dog are of epithelial origin, and neoplasms derived from exocrine epithelium predominate. 11, 21, 24 In the dog, exocrine pancreatic neoplasia is usually malignant. Transcoelomic metastasis, metastasis to the liver or regional lymph nodes, extensive contiguous growth, or infiltration usually occur before the onset of clinical signs. 2, 11
Exocrine pancreatic cancer may be derived from ductal or acinar epithelium. In humans, clinically significant subclassifications of exocrine pancreatic neoplasia have been established according to cell type of origin and an assortment of diverse histologic morphologic features. 3 In domestic animals, the histologic features of exocrine pancreatic neoplasia are highly variable among and within neoplasms. 10, 11 Common histologic patterns comprise tubules, acini, or solid sheets, and tumor stroma ranges from delicate to scirrhous. Cells may be cuboidal, polygonal, or columnar, and have varied polarity and cytoplasmic granulation. Although histologic subclassifications exist for pancreatic tumors of domestic animals, 10 correlations with histogenesis or biologic behavior have not been clarified for the dog.
A distinct variant of exocrine pancreatic carcinoma was diagnosed in 6 dogs by the Colorado State University Veterinary Diagnostic Laboratory between 2003 and 2005. These neoplasms were termed hyalinizing pancreatic adenocarcinoma because of the accumulation of glassy, homogeneous, eosinophilic, extracellular material within tumor stroma and tubular lumina. Each was composed of well-granulated epithelial cells, with mild cellular atypia, suggestive of low-grade malignancy. Because this histologic subtype of exocrine pancreatic carcinoma had not been described, the prognosis for affected dogs was unknown at the time of diagnosis. The objectives of the present case series were to describe these neoplasms, to biochemically characterize the nature of the hyaline tumor matrix, and to contrast clinical findings from this series with those of typical exocrine pancreatic carcinoma of the dog.
Methods
Diagnostic materials and patient records that pertained to 6 dogs with hyalinizing pancreatic adenocarcinoma were reviewed. Sections 3–5 µm in thickness were prepared from formalin-fixed paraffin-embedded pancreatic tumor samples from each case. Sections from each tumor were stained with hematoxylin and eosin (HE); periodic acid–Schiff (PAS), with and without diastase; mucicarmine; Masson's trichrome; and Congo red.
Immunohistochemistry (IHC) was performed on formalin-fixed paraffin-embedded sections of pancreatic neoplasms from all dogs of the study population to characterize the biochemical nature of the intratumor hyaline material. Specifically, IHC was conducted to detect the presence of amyloid A (AA), amylin (islet amyloid polypeptide), λ and κ immunoglobulin light chains, laminin, and α1-antitrypsin. Paraffin embedded tissues from pancreatic tumors were sectioned at 5 μm, placed onto Probe-On Plus microscope slides (Fisher Scientific, Pittsburgh, PA), and routinely deparaffinized and rehydrated. Endogenous peroxidase was quenched with 3% hydrogen peroxide for 15 minutes, and sections were rinsed in 0.05M Tris-buffered saline solution (TBS/Tween 20). Nonspecific binding was blocked with normal goat serum (10% in TBS/Tween 20) for 15 minutes. The antigen retrieval method, primary antibody dilution, incubation time, and antibody source are listed in Table 1. Tissues were then incubated at 23°C with primary antibody at the appropriate dilution and time, and then rinsed. Next, sections were incubated with the linking reagent (EnVision+ System, goat anti-rabbit IgG/HRP-dextran polymer conjugate, Dako North America, Inc., Carpinteria, CA), rinsed 5 minutes in TBS/Tween 20, and color developed with 3-amino-9-ethylcarbazole reagent (ready-to-use, Dako North America) for 5–15 minutes, as appropriate. Sections were then rinsed in running tap water for 5 minutes and counterstained with Mayer's hematoxylin (Sigma-Aldrich, St. Louis, MO) for 5 minutes. The negative controls were processed by the above-described procedure, with the primary antibody omitted. Positive controls for the primary antiserum are listed in Table 1. IHC staining for laminin (Dako North America) was performed on a Dako Autostainer, as previously described, by using a labeled streptavidin-biotin-peroxidase complex system to visualize the immune reactions. 25
Primary antibodies for IHC.
Results
Clinical findings
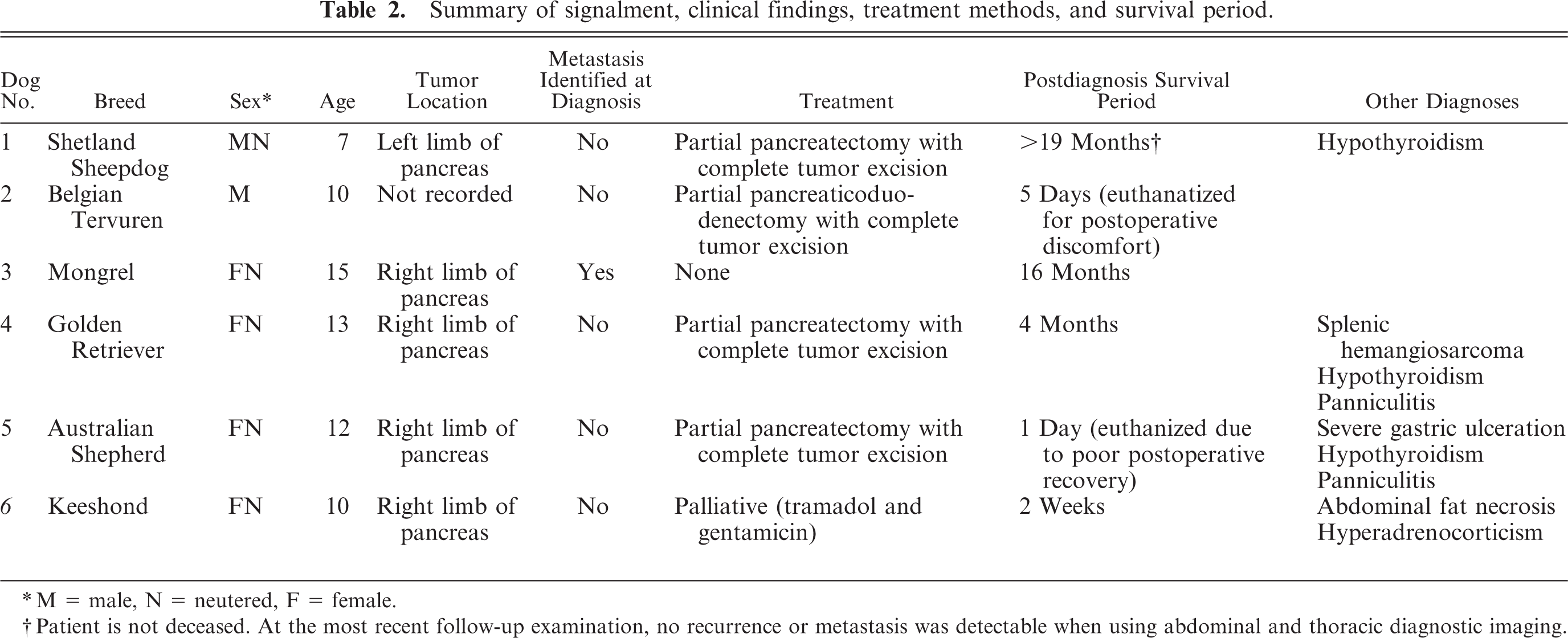
Signalment, clinical findings, treatment methods, and survival periods for all dogs are listed in Table 2.
Summary of signalment, clinical findings, treatment methods, and survival period.
M = male, N = neutered, F = female.
Patient is not deceased. At the most recent follow-up examination, no recurrence or metastasis was detectable when using abdominal and thoracic diagnostic imaging.
Sixteen months later, the dog became anorexic and unable to stand. A CBC revealed mild neutrophilia (13,752 cells/µl; RI, 2,060–10,600 cells/µl) and monocytosis (1,146 cells/µl; RI, 0–840 cells/µl). A serum biochemistry panel demonstrated azotemia (urea nitrogen, 202 mg/dl [RI, 6–25 mg/dl]; increased creatinine, 9.2 mg/dl [RI, 0.5–1.5 mg/dl]; phosphorus, 20.3 mg/dl [RI, 2.5–6.0 mg/dl]), and lipase (4,197 U/liter; RI, 77–695 U/liter), hypoalbuminemia (2.5 g/dl; RI, 2.7–4.4 g/dl), and hypercholesterolemia (350 mg/dL; RI, 92–324 mg/dl). The dog was euthanatized. A postmortem examination was not performed.
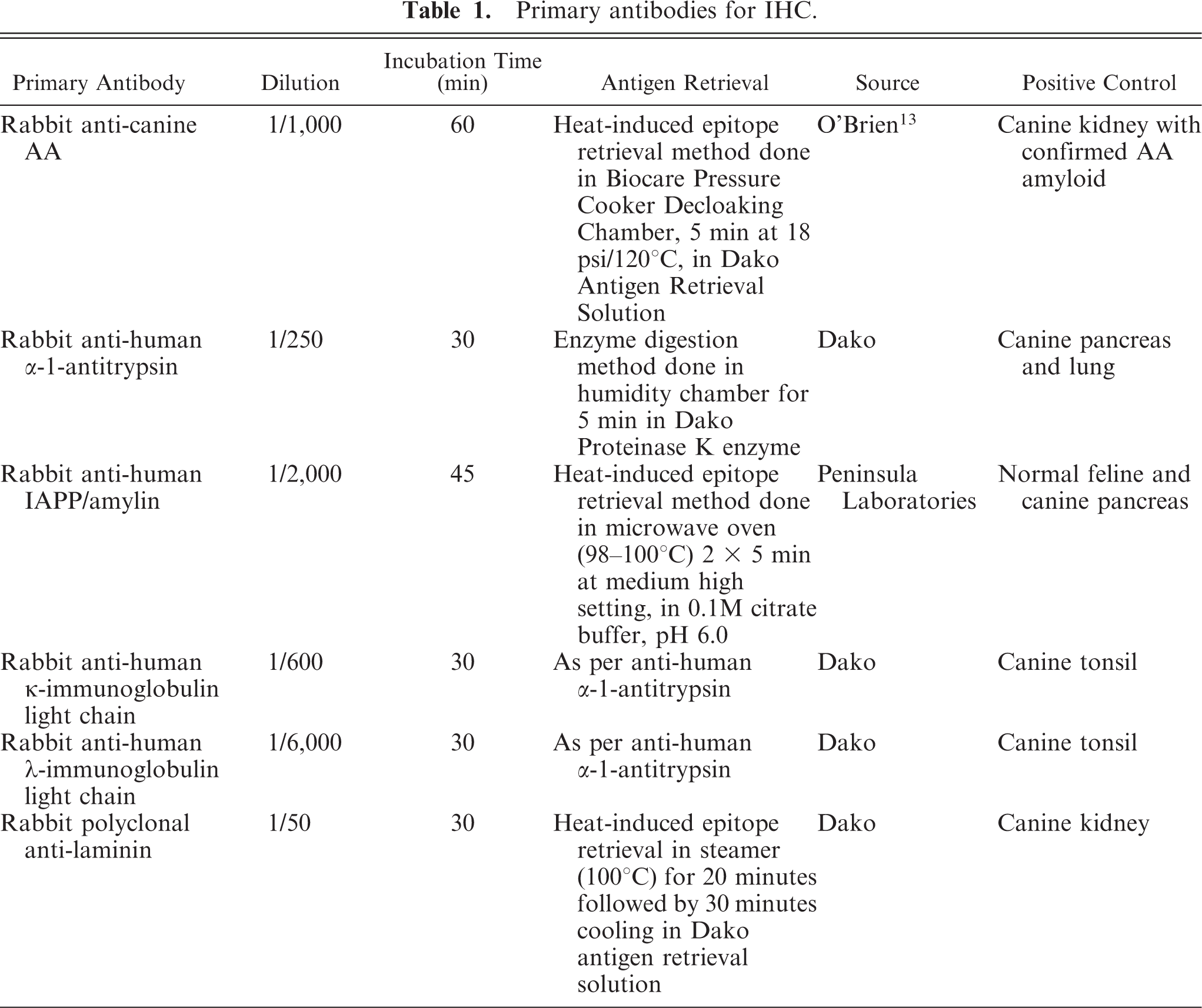
Four days after presentation, the dog developed anorexia, vomiting, and epistaxis. An additional area of subcutaneous swelling had developed along the right lateral thorax. A CBC revealed hypochromic, normocytic, nonregenerative anemia (hct, 24.5% [RI, 37–55%]; hemoglobin, 9.2 g/dl [RI, 12–18 g/dl]; mean corpuscular volume (MCV), 65.3 fl [RI, 60–77 fl]; reticulocyte percentage, 0.5%); neutrophilia (46,080 cells/µl; RI, 3,000–12,000 cells/µl); and monocytosis (3,940 cells/µl; RI, 300–2,000 cells/µl). A serum biochemistry profile included hypoalbuminemia (1.9 g/dl; RI, 2.2–3.9 g/dl) and elevated ALP (556 U/liter; RI, 23–212 U/liter), amylase (2,429 U/liter; RI, 0–480 U/liter), lipase (4,506 U/liter; RI, 60–645 U/liter), and cholesterol (359 mg/dl; RI, 110–320 mg/dl). An ultrasound showed an echogenic mass near the right limb of the pancreas, hepatomegaly, and bilateral enlargement of the adrenal glands (both 7.8–8.1-mm wide). Ultrasound-guided needle biopsy specimens were collected from the pancreas and caudal mammary subcutaneous tissue. Cytology of an impression smear of the pancreatic mass indicated that the mass was composed of columnar cells, which sometimes formed acinar structures, consistent with pancreatic epithelial cells (Fig. 1). Cytology was interpreted as normal pancreatic tissue; however, because the cytologic sample was believed to represent a mass lesion, pancreatic neoplasia could not be ruled out. Per histology, the pancreatic biopsy specimen was diagnosed as a nodular pancreatic hyperplasia. Histopathologic examination of the subcutaneous tissue biopsy specimen of the caudal mammary area revealed chronic-active panniculitis.
Pancreatic mass; dog No. 6. Columnar epithelial cells from an acinus-like structure within a basophilic proteinaceous vacuolated background with scattered erythrocytes. Note granular apical cytoplasm suggestive of pancreatic origin. Some cells have large or multiple nucleoli. Wright-Giemsa. Bar = 15 µm.
After a poor response to tramadol and intravenous gentamicin therapy, the dog was euthanatized 2 weeks after initial presentation. At postmortem, the pancreas was diffusely enlarged, firm, and white-pink. Several nodular masses, 2–4 cm in diameter, extended from the pancreas into the adjacent mesentery. On cut surface, thick, opaque, yellow-green fluid oozed from cavitated areas within the masses. Firm, raised, iridescent areas, consistent with fat necrosis, were present throughout the mesentery and omentum. Subcutaneous fat of the dorsum and caudal mammary area was swollen, soft, and oozed oily yellow-tinged fluid on the cut surface.
Histopathology
Histopathologic characteristics of hyalinizing pancreatic adenocarcinoma were similar in all pancreatic tumors of the study population. The presence of lakes and globular aggregates of eosinophilic hyaline material that resembled amyloid, which expanded the fine fibrovascular stroma between tumor cells and was present within tubular structures, was a distinctive feature of all tumors (Fig. 2). Tubular and tubulocystic architectural patterns predominated but alternated with sparse solid areas where cells were organized in acini, dilated acini, nests, and trabeculae. Tumor cells were cuboidal, polygonal, or columnar, and had abundant eosinophilic and granular apical cytoplasm, suggestive of zymogen granules. Nuclei were basal, round, and hyperchromatic, with large nucleoli and frequent mitotic figures. Additional features of cellular atypia included mild anisocytosis and nuclear pleomorphism. Stromal accumulations of hyaline material contained entrapped nests or cords of shrunken, atrophic neoplastic cells, or clear spaces where cells had presumably been lost. Intratubular hyaline material was more deeply eosinophilic than interstitial hyaline material, and contained a few neutrophils, macrophages, eosinophils, and necrotic cellular debris. Tumors were discontinuously bordered by fibrous connective tissue, suggestive of collapsed pancreatic interstitium. Infiltrative nests of neoplastic epithelial cells extended into the fibrous connective tissue. The hyaline material and cytoplasmic granules did not stain with mucicarmine or PAS. With Masson's trichrome, hyaline material stained blue-gray (Fig. 3). Hyaline material was neither congophilic nor birefringent (Fig. 4).
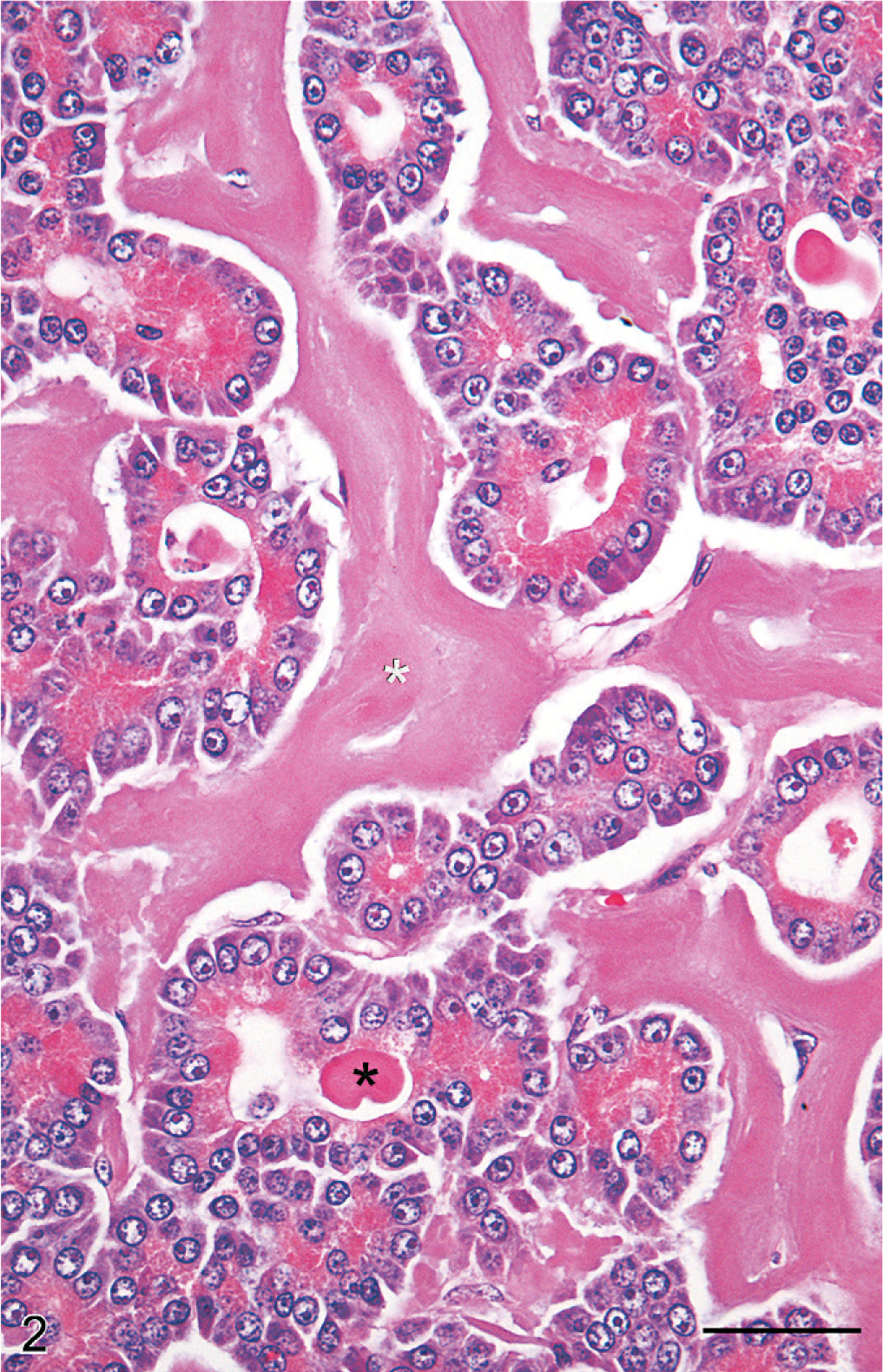
Pancreatic mass; dog No. 6. Neoplastic epithelial cells form tubular structures that are surrounded and separated by abundant hyaline material (white asterisk). Similar hyaline material is within tubular lumina (black asterisk). Note prominent zymogen granules within neoplastic epithelial cells. HE. Bar = 50 µm.
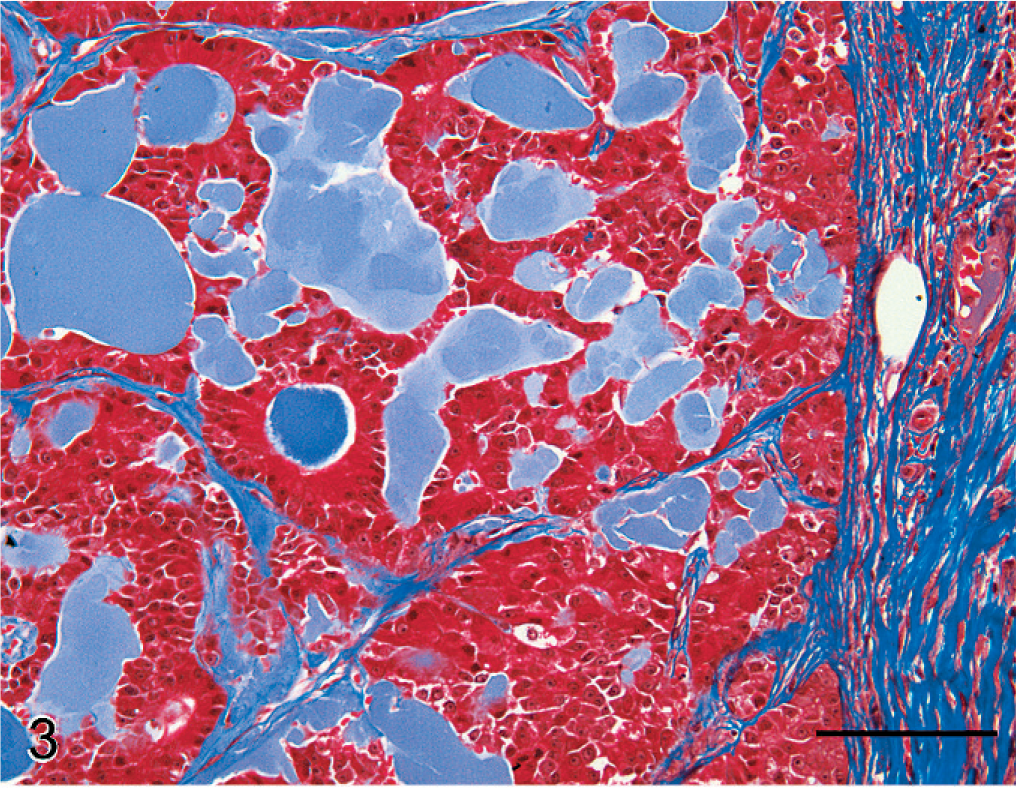
Pancreatic mass; dog No. 3. Hyaline material stains blue-gray in contrast to collagen-rich connective tissue which stains dark blue. Masson's trichrome. Bar = 100 µm.
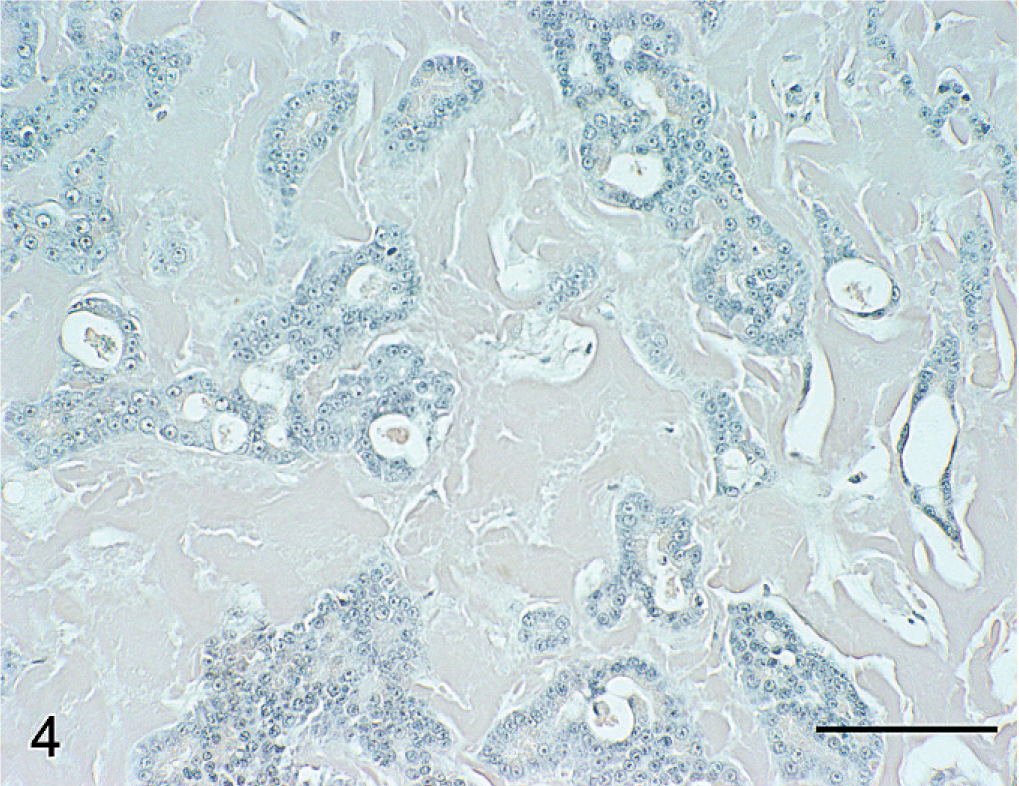
Pancreatic mass; dog No. 6. Tumor matrix fails to stain for amyloid. Congo red. Bar = 100 µm.
IHC
When sections of tumor from all dogs were examined with immunohistochemistry for canine AA, amylin, lambda light chains and kappa light chains, laminin, and α1-antitrypsin, no immunoreactivity was associated with the cells or hyaline material, which comprised the tumors (Fig. 5). Antigen-specific immunostaining was present in all positive-control tissues evaluated.
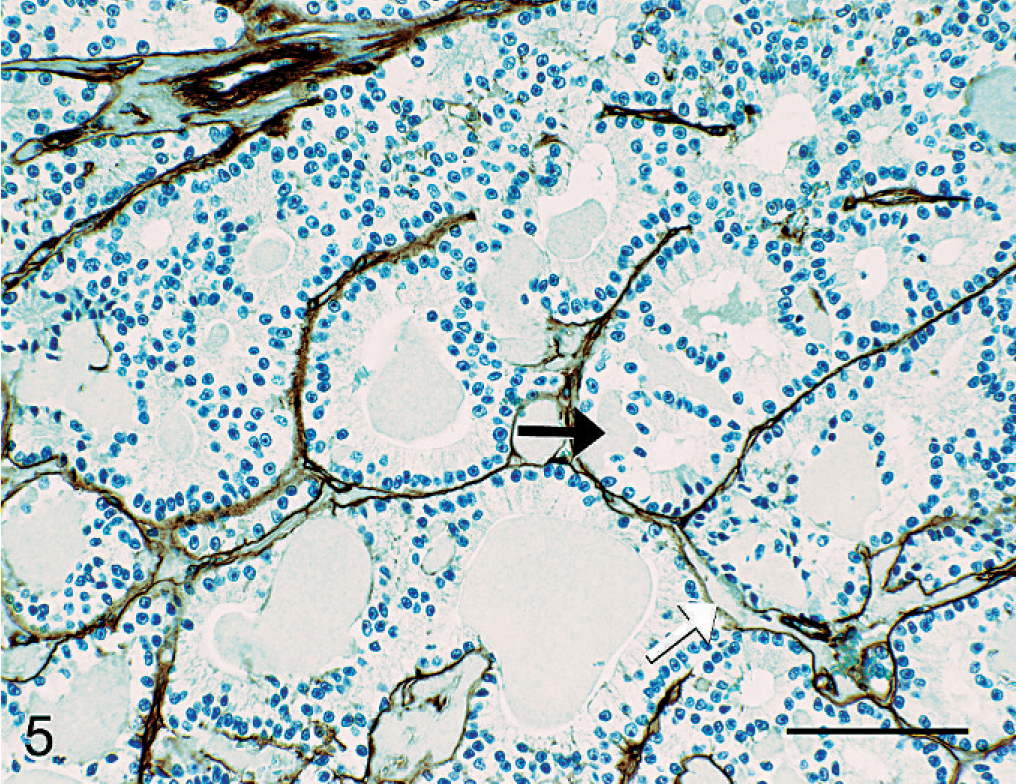
Pancreatic mass; dog No. 3. Laminin immunolabeling defines vessel walls and basement membranes of tubules formed by neoplastic epithelium. No laminin immunolabeling is identified within hyaline tumor matrix. Note the accumulation of hyaline material between basement membranes of tubules (white arrow), and between basement membrane of tubules and basal aspect of neoplastic cells (black arrow). LSAB2 HRP, DAB, Mayer's hematoxylin counterstain, Bar = 100 µm.
Discussion
The present study describes a unique variant of exocrine pancreatic carcinoma that was identified in 6 mature-to-elderly mid-sized breed dogs. The neoplasm usually involved the right limb of the pancreas and was grossly evident as a solitary mass. For most dogs of the study population, the discovery of a pancreatic mass was an incidental finding. Substantially elevated serum lipase and amylase levels were common among affected dogs.
On microscopic examination, hyalinizing pancreatic adenocarcinoma is distinguished from conventional exocrine pancreatic carcinoma by the presence of accumulated extracellular hyaline material. Hyalinizing pancreatic adenocarcinoma does not seem to conform to the current World Health Organization classification for exocrine pancreatic neoplasia of domestic animals. 10 Neoplastic cells were often columnar and arranged into tubules, consistent with ductal adenocarcinoma. However, the presence of numerous cytoplasmic zymogen-like granules is also consistent with acinar-cell carcinoma. In all studied neoplasms, histologic features suggested low-grade malignancy. The histologic pattern included consistent tubular organization. Furthermore, the cells were uniformly well differentiated, having distinctly granulated cytoplasm. However, some features of malignancy were apparent, including the absence of an intact tumor capsule, mild cellular atypia, frequent mitotic figures, and invasion of marginal stroma.
Canine hyalinizing pancreatic adenocarcinoma appears to be biologically less aggressive than conventional exocrine pancreatic carcinoma of the dog. Although the median survival time for exocrine pancreatic carcinoma of the dog has not been reported, this neoplasm tends to have metastasized and/or extensively invaded adjacent tissues at the time of diagnosis. 24 Metastasis at the time of diagnosis was only identified in 1 dog of the study population (dog No. 3); however, this dog lived 16 months after diagnosis. Two patients of the study population were euthanatized shortly after a partial pancreatectomy (dog Nos. 2 and 5); however, this is not surprising, because, given the critical location of pancreatic tumors, postoperative mortality of dogs with exocrine pancreatic cancer is high. 24 Dogs of the study population without postoperative complications or concurrent life-threatening disease survived longer than expected for dogs with exocrine pancreatic cancer and within a range reasonably anticipated for elderly dogs. Cell-to-matrix interactions play a major role in tumor growth and invasion. 15 Therefore, it is possible that certain stromal matrix accumulations may mechanically or biochemically impede malignancy. Alternatively, the impression of reduced malignancy in the present case series may largely be a reflection of the degree of differentiation of tumor cells. Further study is needed to identify associations between certain prognostic factors (including histologic subclassification or the presence of cytologic features of malignancy) and the survival period of dogs diagnosed with exocrine pancreatic carcinoma.
Two dogs (dog Nos. 5 and 6) of the study population were diagnosed with panniculitis. Panniculitis associated with pancreatic neoplasia has been reported in humans, 5 dogs, 4 and cats. 8 The pathogenesis of “pancreatic panniculitis” is unknown; however, it has been speculated that it may be the result of lipolytic pancreatic enzymes reaching systemic circulation 18 or the result of a deficiency in serum enzyme inhibitors, including α1-antitrypsin or α2-macroglobulin. 12, 23 In all species, “pancreatic panniculitis” is an uncommon sequel to pancreatic disease with a poor prognosis. 4, 5, 8 Therefore, it is surprising that 2 of 6 dogs (33%) of the study population had panniculitis. It is possible that in one or both of these dogs, panniculitis was an incidental condition unrelated to pancreatic disease. Further study is required to determine whether this condition truly is more prevalent in dogs with hyalinizing pancreatic adenocarcinoma than in dogs with other pancreatic diseases.
The origin of the intratumor hyaline material is unknown. Because the material that accumulated within the lumina of tubules of neoplastic epithelial cells was similar to that which accumulated in the tumor stroma, it is possible that the material represents a secretory product. However, the biochemical nature of the hyaline material within pancreatic tumors of the study population remains undetermined. When stained with HE, the material appeared amyloid-like because of the hyaline and homogeneous staining pattern, entrapment of cells, atrophy of entrapped cells, displacement of normal architecture, and lack of significant inflammatory response. Several neoplasms of animals and humans accumulate amyloid, including thyroid medullary carcinoma, pancreatic islet–cell neoplasms, and immunoglobulin light-chain producing immunocyte dyscrasias. 1 However, the matrix of canine hyalinizing pancreatic adenocarcinoma is probably not amyloid, because it was not congophilic, and it lacked green birefringence with polarized light. 7 Furthermore, immunoreactivity for potential amyloid constituents, including amyloid A, amylin, and immunoglobulin light chains, was not demonstrated.
Several human neoplasms accumulate hyaline material that is not amyloid. Electron microscopy and immunohistochemistry for laminin and collagen indicate that extracellular hyaline material in clear-cell carcinoma of the ovary consists of accumulated basement membrane. 16 Stromal deposition of hyaline material that consists of basement membrane has also been reported to occur in synovial sarcoma and hyalinizing trabecular thyroid adenoma. 6, 14 However, IHC failed to identify laminin in the hyaline material of the present tumors, which suggests that basement membrane is not a major component. The hyaline material that accumulates in hyalinizing clear-cell carcinoma of the salivary gland is composed mostly of collagen I and fibronectin. 9, 17 However, collagen does not seem to be a primary component of the hyaline material within the present tumors; such connective tissue stains dark blue with Masson's trichrome, in contrast to the blue-gray staining of the hyaline material in the studied tumors. 22 Clear-cell ductal pancreatic adenocarcinoma accumulates hyaline globules within glandular lumina formed by neoplastic cells. 20 This material is immunoreactive for α1-antitrypsin and is strongly PAS positive. However, the hyaline material within the present tumors did not react with PAS, and IHC failed to identify accumulated α1-antitrypsin. Additional histochemical, IHC, and ultrastructural study may help elucidate the biochemical composition, source, and biological significance of the hyaline material.
The present report documents 6 cases of a seemingly rare variant of exocrine pancreatic carcinoma of dogs. Clinicians and pathologists should be aware that dogs with this neoplasm may have a longer postdiagnosis survival period than for other exocrine pancreatic carcinomas, especially when other conditions with poor prognoses are lacking. Given the risks of pancreatic surgery, it is unclear whether surgical excision is justifiable for low-grade pancreatic cancer. Research is needed to delineate associations between histologic classifications and grades of pancreatic cancer, including the variant described herein, with outcomes in the dog.
