Abstract
This report describes the morphologic, ultrastructural, and immunophenotypic features of a nodular ganglioneuroblastoma in the jejunum of a 13-month-old Holstein-Friesian heifer. On histologic examination, the mass was composed of clusters of neuroblasts and isolated ganglionic neurons in abundant neurophilic matrix that was surrounded by scanty Schwannian stroma. On ultrastructure examination, the large ganglionic neuron-like cells had unmyelinated neurites. Most ganglionic neuron-like tumor cells expressed neurofilament, neuron-specific enolase, chromogranin A, and S-100, whereas the Schwann-cell-like stromal cells expressed S-100 and vimentin. Both brain-derived neurotrophic factor (BDNF) and tropomyosin-related kinase-B (Trk-B) were expressed in ganglionic neuron-like tumor cells, which suggested the activation or reactivation of an embryonic autocrine BDNF/Trk-B pathway that could have prolonged cell survival and promoted differentiation with neurite formation.
Keywords
Neuroblastic tumors of the peripheral nervous system are believed to originate from a neural crest stem cell, which explains the spectrum of morphologic variants from benign ganglioneuromas to malignant undifferentiated neuroblastomas. 5, 18 The current World Health Organization histologic classification of the tumors of the nervous system of domestic animals differentiates peripheral neuroblastomas from ganglioneuromas. 7 In contrast to the simple system used in veterinary pathology, human neuroblastic tumors are subdivided into 4 categories with subtypes based on specific morphologic criteria. 16 The categories are as follows: neuroblastoma (Schwannian stroma-poor subtype), nodular ganglioneuroblastoma (composite Schwannian stroma-rich/stroma-dominant, and stroma-poor subtypes), intermixed ganglioneuroblastoma (Schwannian stroma-rich subtype), and ganglioneuroma (Schwannian stroma-dominant subtype). 16 Most peripheral neuroblastic tumors occur in cranial and spinal nerve ganglia; adrenal medulla; or retropleural, retroperitoneal, and mediastinal sites in humans, domestic animals, and laboratory animals. 7, 8, 16, 17 This report describes histologic, immunohistochemical, and ultrastructural features of a bovine jejunal nodular ganglioneuroblastoma in a Holstein-Friesian heifer.
A 13-month-old Holstein Friesian heifer was recumbent, with severely reduced sensitivity of the hind legs, an atonic tail, and reduced anal reflex. A caudal physeal fracture of the third lumbar vertebra with ventral dislocation of the epiphysis was detected by radiography.
Because of the poor prognosis, the animal was euthanatized and submitted for necropsy. Tissue samples were fixed in 10% neutral buffered formalin, processed routinely in paraffin wax, sectioned at 4-μm thickness, and stained with hematoxylin and eosin. For transmission electron microscopy, tissue samples were post-fixed in 5% glutaraldehyde followed by 1% osmium tetroxide, then embedded in epoxy resin. Ultrathin sections were contrasted with uranyl acetate and lead citrate and then were analyzed with a transmission electron microscope (EM10C, Zeiss, Oberkochen, Germany). Immunohistochemistry of serial sections was performed with the avidin-biotin-peroxidase complex method (ABC) (Elite PK6100; Vector Laboratories, Burlingame, CA, USA) by using monoclonal antibodies for vimentin (1 : 100), neurofilament (NF) (1 : 200), neuron-specific enolase (NSE) (1 : 100), synaptophysin (1 : 100), pancytokeratin (1 : 100) (all from DAKO Diagnostika, Hamburg, Germany), and chromogranin A (1 : 50; Novocastra Laboratories Ltd, Newcastle upon Tyne, UK). Polyclonal antibodies were used for glial fibrillary acidic protein (GFAP) (1 : 2000; DAKO), S-100 protein (1 : 800; Sigma-Aldrich Chemie, Taufkirchen, Germany), myelin basic protein (MBP) (1 : 800; Chemicon Europe, Hofheim/Taunus, Germany), brain-derived neurotrophic factor (BDNF) (1 : 50), and tropomyosin-related kinase-B (Trk-B) (1 : 50) (both from Santa Cruz Biotechnology, Heidelberg, Germany). The specificity of the immunohistochemical reactions was confirmed by the use of normal bovine cerebrum, cerebellum, spinal cord, and peripheral nerve. As a negative control, serial sections were treated with Balb/c mouse ascites fluid or nonimmune rabbit serum, instead of the primary monoclonal or polyclonal antibodies, respectively.
At necropsy, the heifer was in good nutritional condition and weighed 311 kg. There was an acute complete transversal fracture of the caudal epiphyseal plate of the third lumbar vertebral body, which was interpreted as the cause of the aforementioned neurologic deficits. A well-demarcated multinodular mass with a diameter of approximately 0.5–3.0 cm and a smooth surface partially replaced the submucosa, muscularis, and serosa of the jejunum and adjacent mesentery, approximately 3 m from the pylorus (Fig. 1). The mass comprised soft white nodules with scattered foci of necrosis and calcification separated by gray stroma. This infiltrative mass caused circumferential thickening of the jejunal wall, which led to partial luminal occlusion. The tunica muscularis of the jejunum oral to the mass was mildly to moderately thickened, but there was no prestenotic impaction. There also was a high ventricular septal defect (2.5 cm in diameter) in the heart, which was accompanied by mild ventricular dilatation. Because of the lack of signs of cardiac insufficiency, the septal defect was interpreted as an incidental finding.
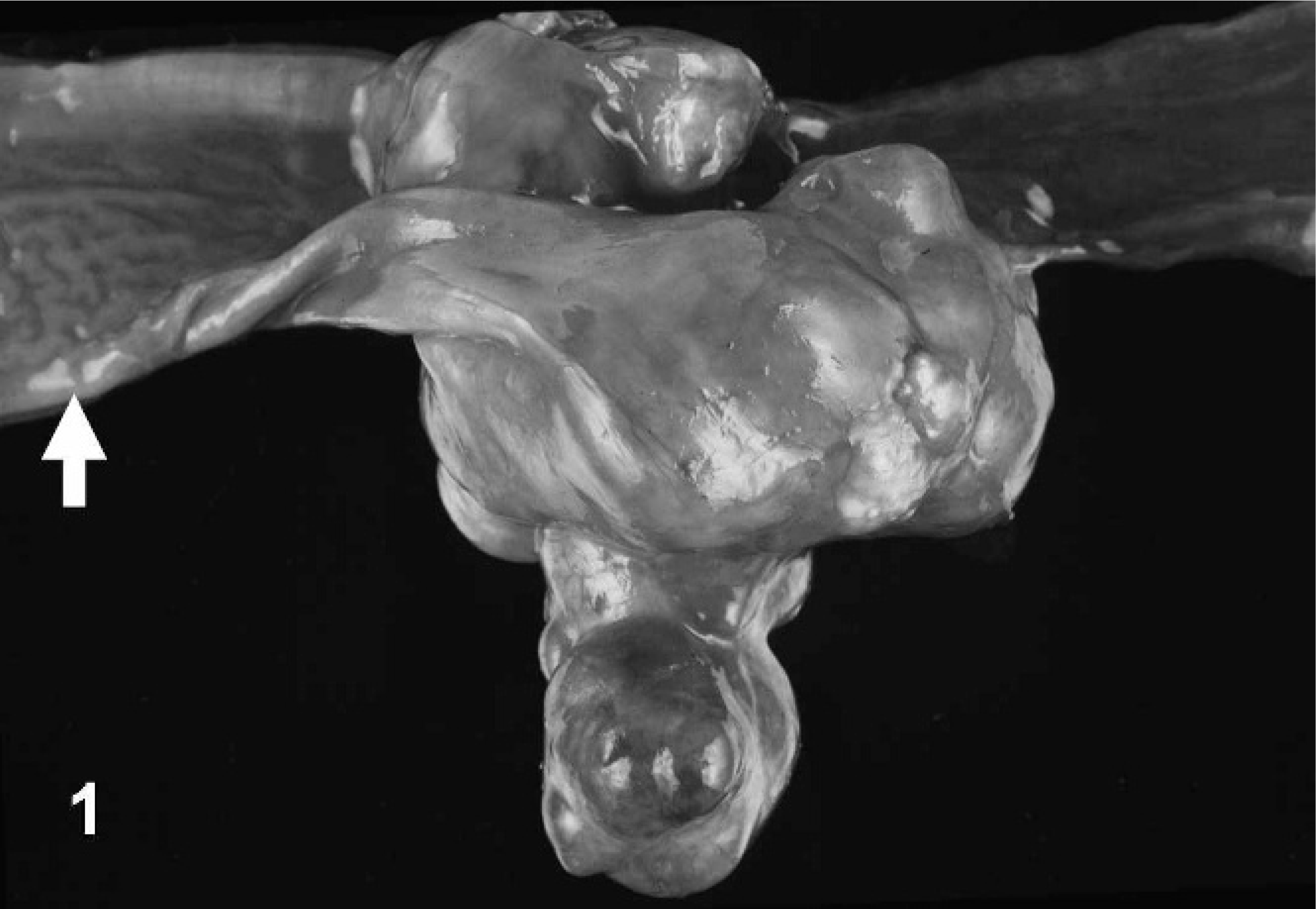
Jejunum; bovine. Nodular ganglioneuroblastoma with partial jejunal obturation and thickening of the tunica muscularis oral to the mass (arrow).
On histologic examination, the jejunal mass was a multinodular tumor with poorly and densely cellular areas separated by thin stroma-dominant regions. The mass occupied the submucosa, with infiltration into the tunica muscularis and serosa. The densely cellular areas were composed of irregular sheets and nests of round-to-polygonal cells (Fig. 2). These cells had a moderate amount of finely granular eosinophilic cytoplasm and oval to bizarrely shaped nuclei, with coarsely clumped chromatin and one or two prominent nucleoli. A few multinucleated tumor cells were present. The hypocellular areas were composed of tissue that resembled neuropil, with individual or small nests of large polygonal cells, separated by thin strands of spindloid stromal cells (Fig. 3). The variably sized (10–40 μm) polygonal cells that resembled ganglionic neurons contained large, round, pale euchromatic nuclei with a single prominent nucleolus, abundant eosinophilic cytoplasm, and coarsely granular basophilic cytoplasmic components that resembled Nissl substance. A few cells were binucleated. The perinodular, stroma-dominant fascicles were predominantly composed of spindloid cells that resembled Schwann cells. These cells contained scanty eosinophilic cytoplasm and elliptical nuclei with indistinct nucleoli (Fig. 4). These Schwann-cell–dominant areas also contained numerous parallel axon-like structures and a few well-differentiated ganglionic neuron-like cells. Areas of hemorrhage, necrosis, and calcification were scattered throughout the tumor. The mitotic index varied greatly, from 0 in well-differentiated areas to 3 mitotic figures per high power field (0.0625 mm2) in regions with poorly differentiated cells.
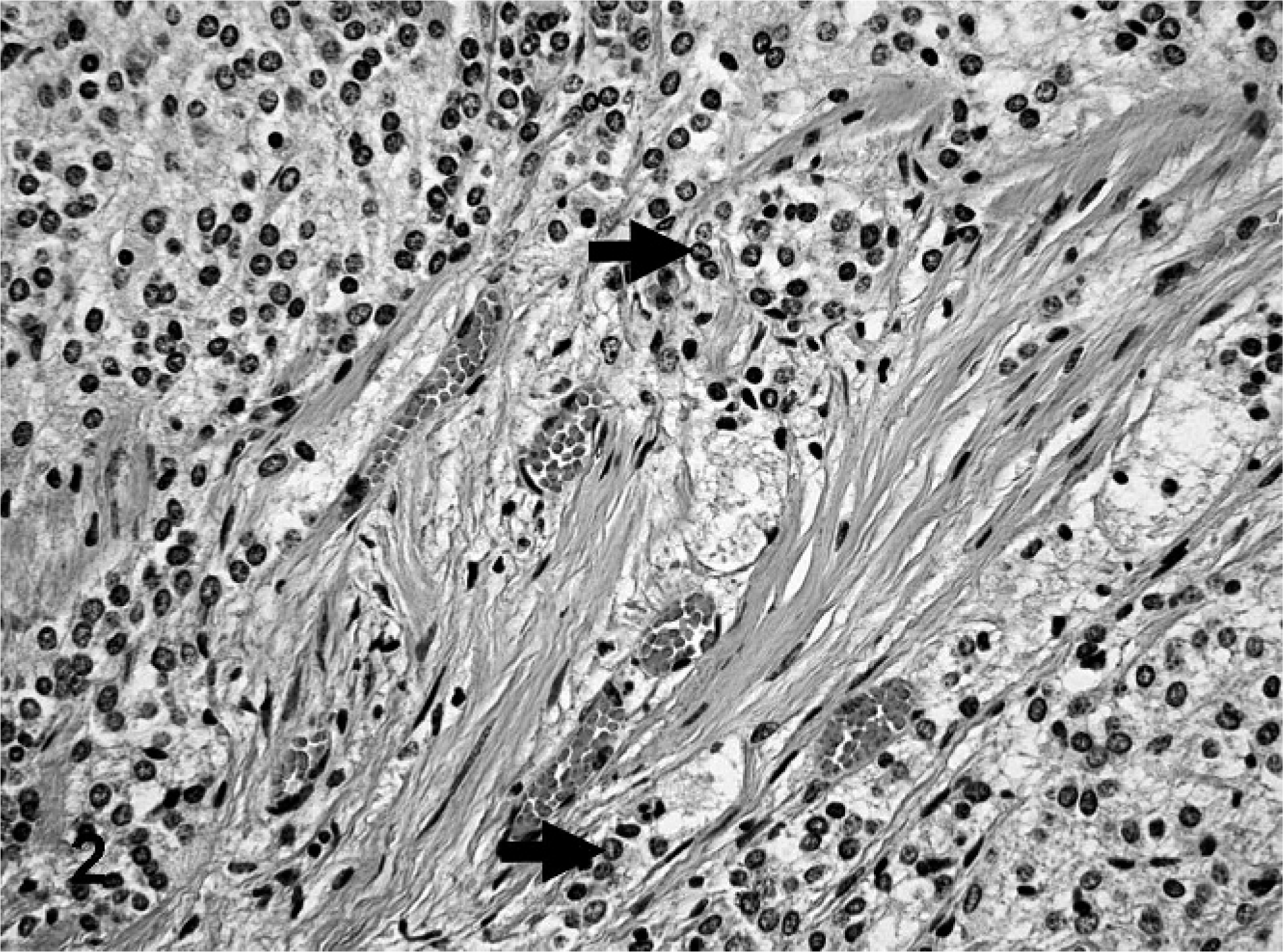
Jejunum; bovine. Poorly differentiated neuroblastic tumor cells (arrows) infiltrating the tunica muscularis. HE.
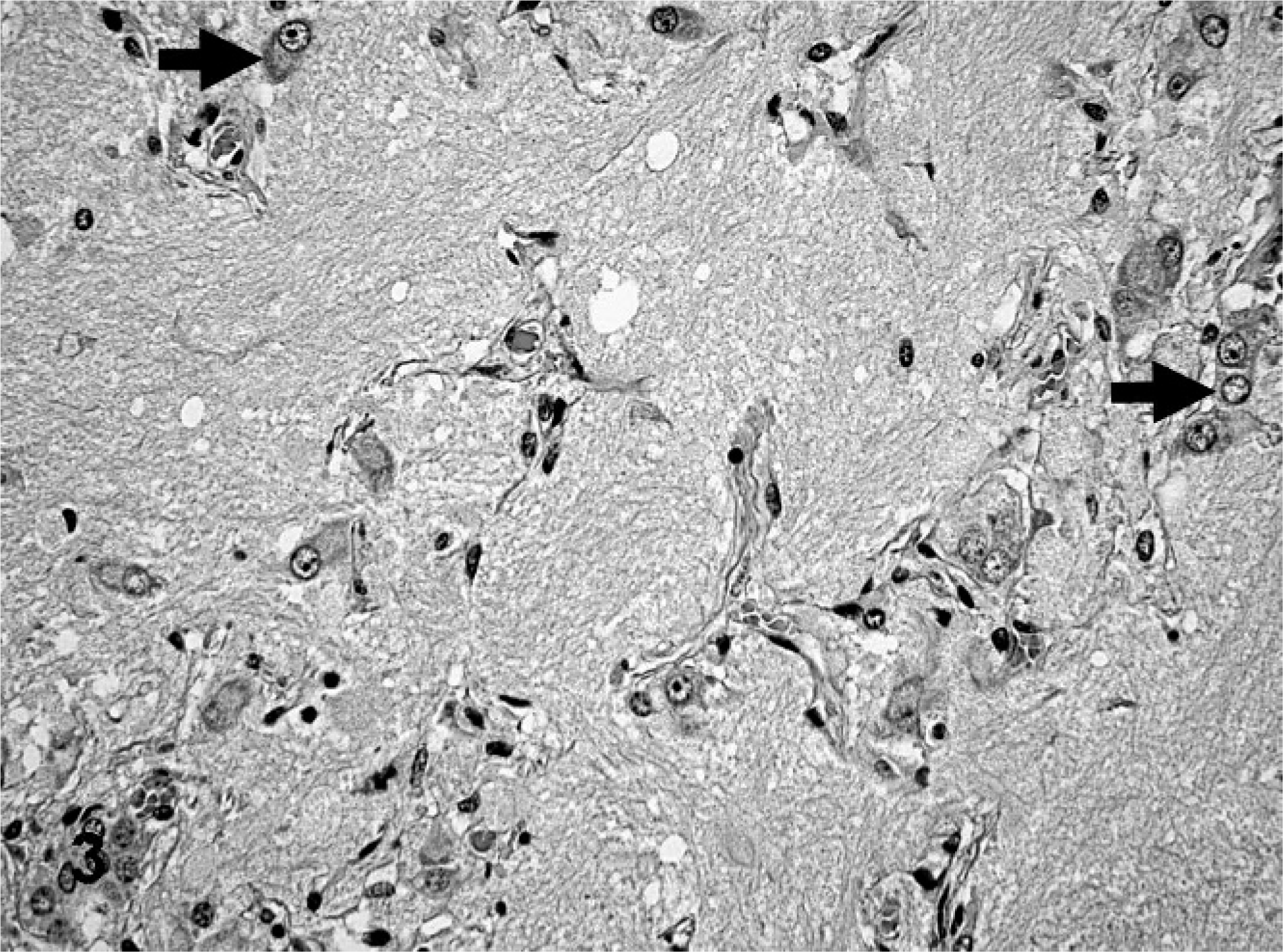
Jejunum; bovine. Individual or small clusters of maturing ganglionic neuron-like tumor cells (arrows) in abundant neurophilic matrix. HE.
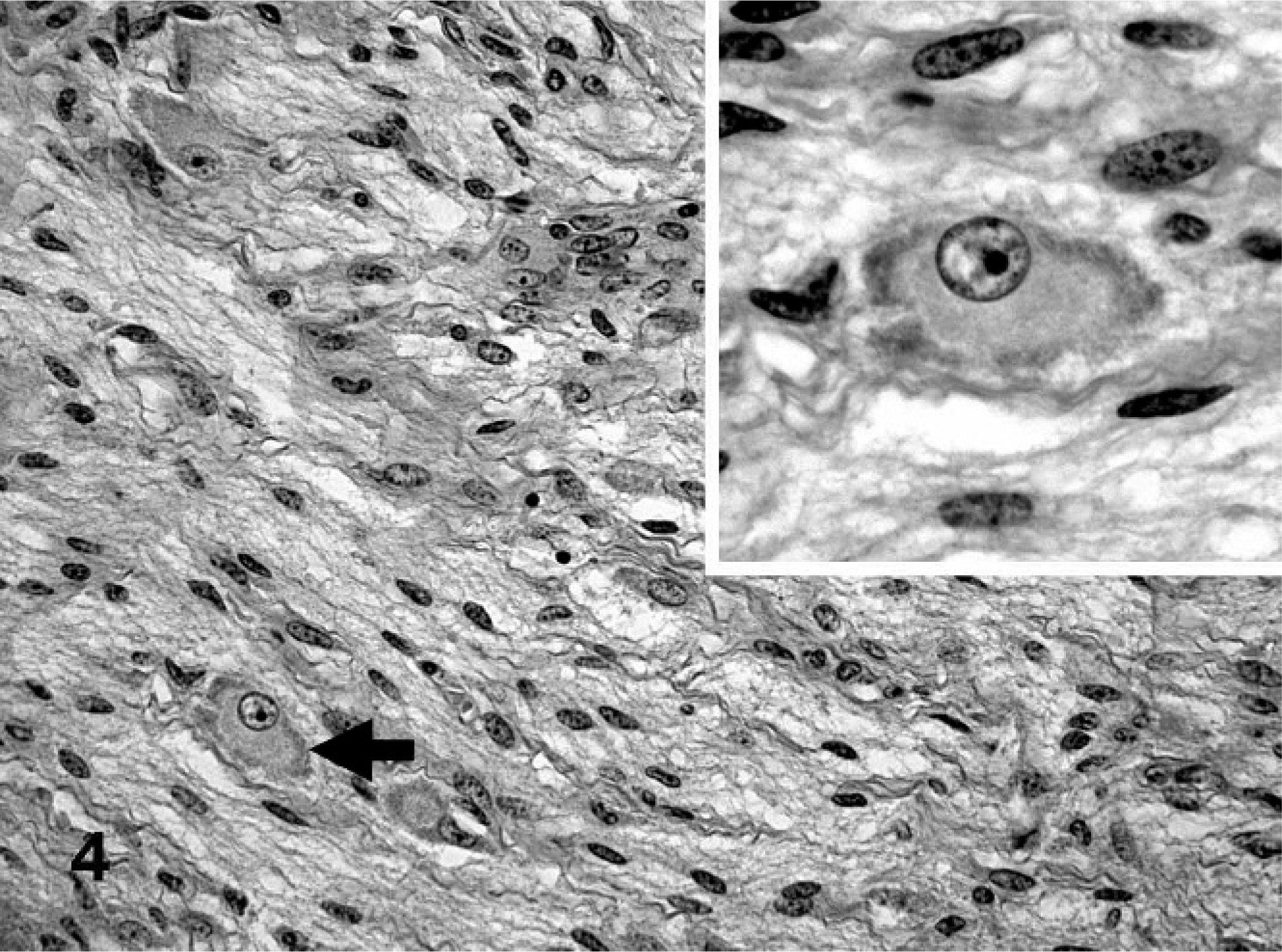
Jejunum; bovine. Schwannian stroma-dominant tumor area with few maturing ganglionic neuron-like tumor cells (arrow). Inset: Higher magnification. Maturing ganglionic neuron-like tumor cell with a large round pale nucleus, prominent nucleolus, and abundant cytoplasm with peripherally localized basophilic granulated Nissl substance. HE.
On ultrastructural examination, most neuroblastic cells had an undifferentiated phenotype. In contrast, the larger tumor cells that resembled ganglionic neurons contained abundant mitochondria, stacks of rough endoplasmic reticulum, lysosomes, and membrane-limited neurosecretory granules with an electron-dense core. The ganglionic neuron-like cells were embedded in a matrix that resembled neuropil and was mainly composed of vesicular neurites with neurofilaments, numerous dense-core neurosecretory granules, and mitochondria (Fig. 5). In general, no myelinated neuritic processes were identified.
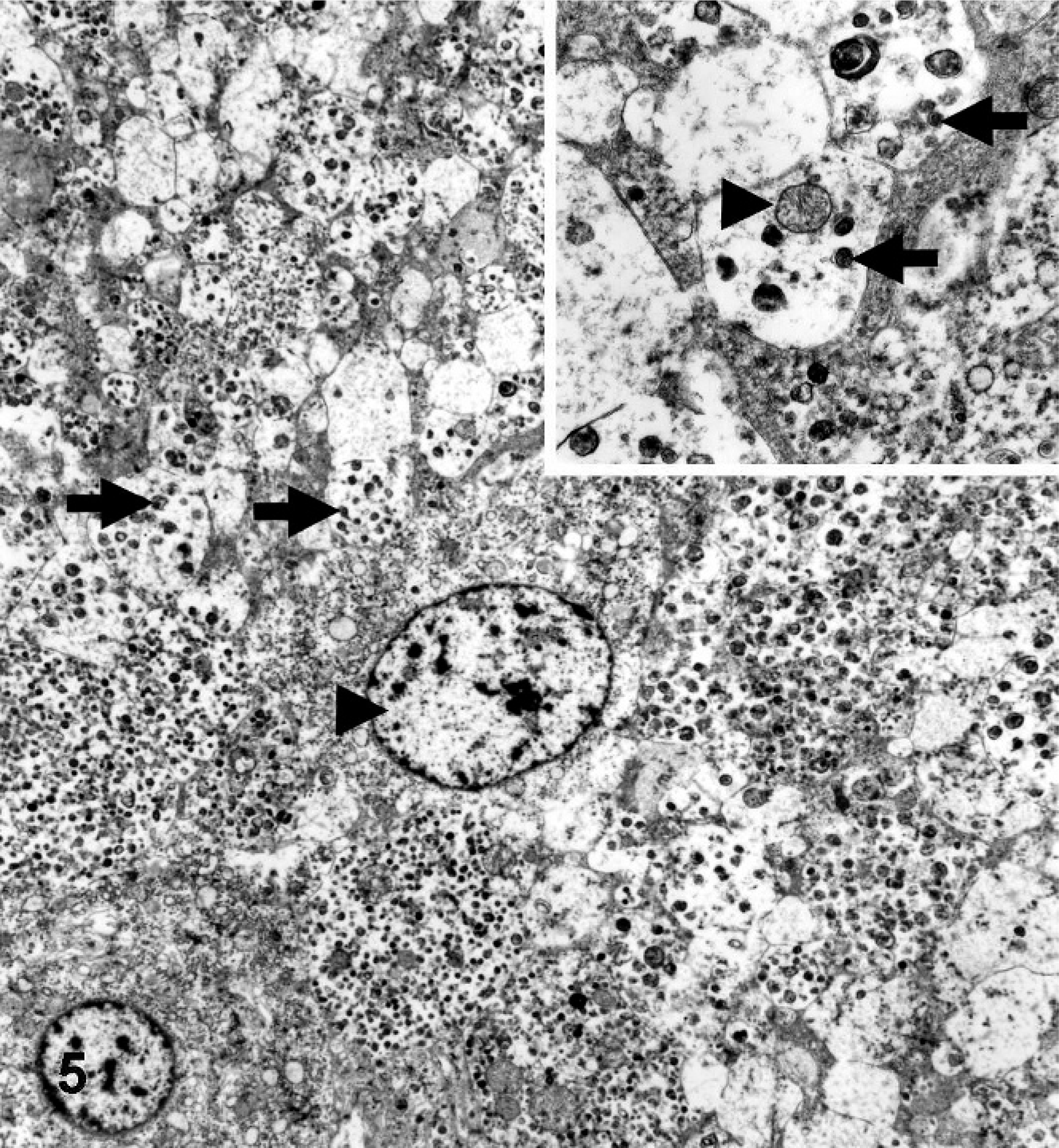
Jejunum; bovine. Ganglionic neuron-like tumor cell with a large pale euchromatic nucleus (arrowhead) in neurophilic matrix of neuritic processes containing neurofilaments and dense-core neurosecretory granules (arrows). Inset: Higher magnification. Neuritic processes contain neurofilaments, mitochondria (arrowhead) and dense-core neurosecretory granules (arrows). Electron microscopy.
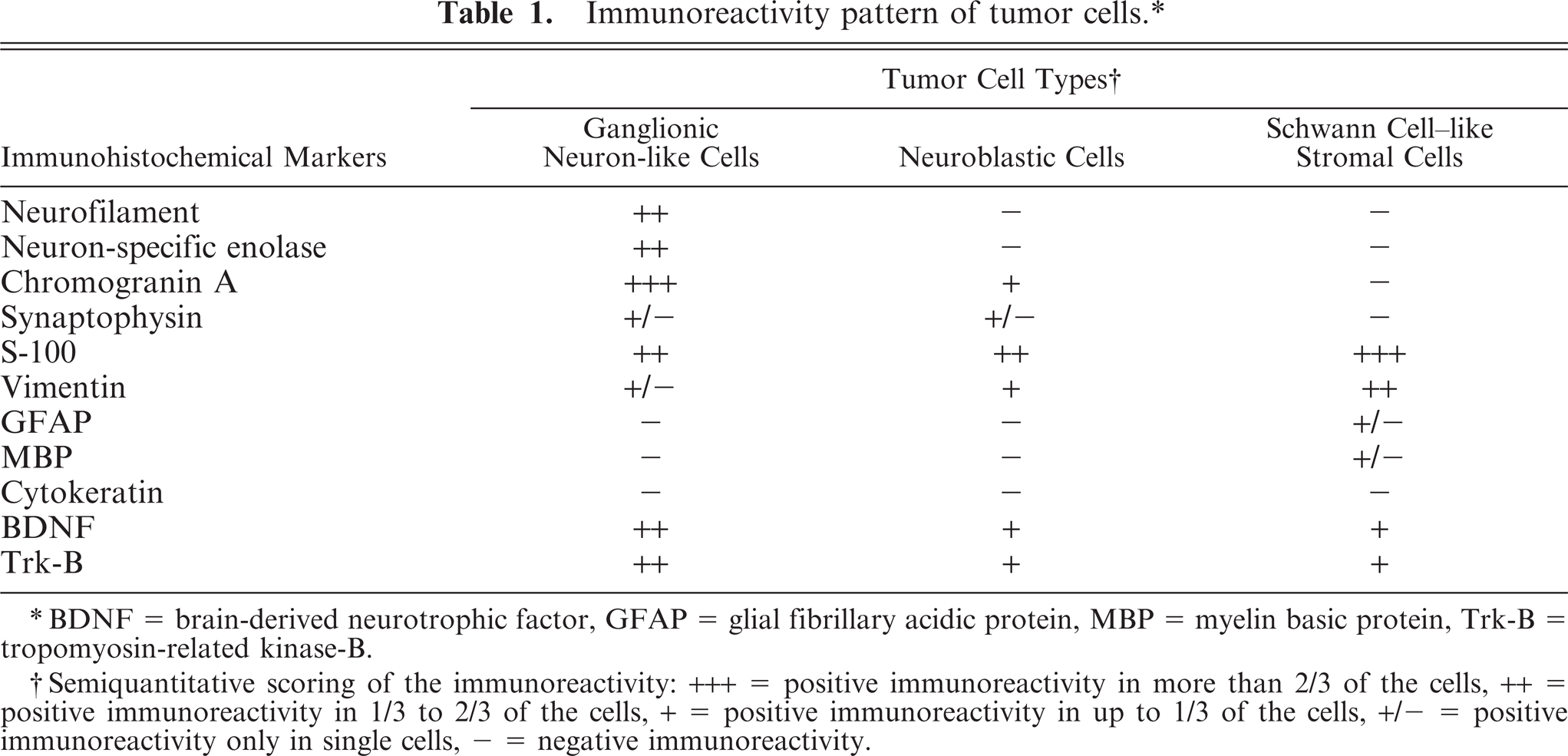
The results of a semiquantitative evaluation of the immunoreactivity of the tumor cells are summarized in Table 1. Neurofilament immunoreactivity was particularly intense in perikarya of many of the ganglionic neuron-like cells and neurite-like cellular processes but was not detected in neuroblastic cells or cells that resembled Schwann cells. Immunoreactivity for NSE was weaker than that for NF and was restricted to the perikarya of ganglionic neuron-like cells. Immunoreactivity for NF and NSE clearly distinguished the ganglionic neuron-like cells from the poorly differentiated neuroblastic cells. At least moderate S-100 immunoreactivity was observed in nearly all Schwann cell–like cells. In addition, a weak-to-moderate S-100 expression was detected in the soma of many neuroblastic and ganglionic neuron-like tumor cells. Chromogranin A had a granular cytoplasmic immunoreactivity pattern in nearly all ganglionic neuron-like cells, with a few disseminated granules in the neurophilic matrix (Fig. 6). Only individual GFAP- or MBP-expressing cells were found throughout the neoplasm. Most tumor cells expressed vimentin; none expressed cytokeratin. Intense cytoplasmic BDNF immunoreactivity (Fig. 7) and less intense Trk-B immunoreactivity (Fig. 8) were detected in the ganglionic neuron-like tumor cells, especially in the poorly cellular, neuropil-rich areas of the tumor.
Immunoreactivity pattern of tumor cells.∗
BDNF = brain-derived neurotrophic factor, GFAP = glial fibrillary acidic protein, MBP = myelin basic protein, Trk-B = tropomyosin-related kinase-B.
Semiquantitative scoring of the immunoreactivity: +++ = positive immunoreactivity in more than 2/3 of the cells, ++ = positive immunoreactivity in 1/3 to 2/3 of the cells, + = positive immunoreactivity in up to 1/3 of the cells, +/- = positive immunoreactivity only in single cells, - = negative immunoreactivity.
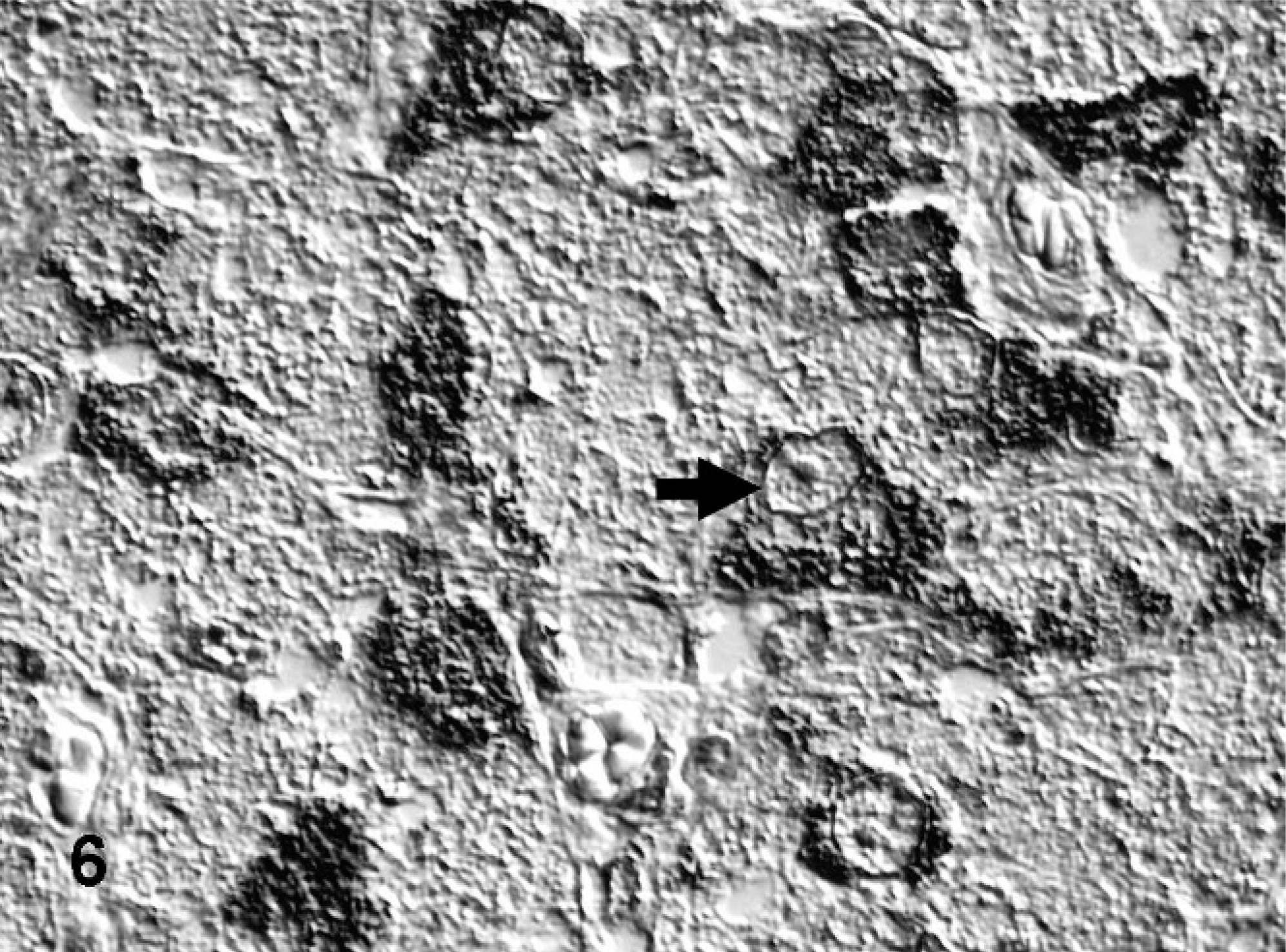
Jejunum; bovine. Granular cytoplasmic immunoreactivity for chromogranin A is present in nearly all ganglionic neuron-like cells (arrow) with few disseminated granules in the neurophilic matrix. ABC method, hematoxylin counterstain, Nomarski differential interference contrast.
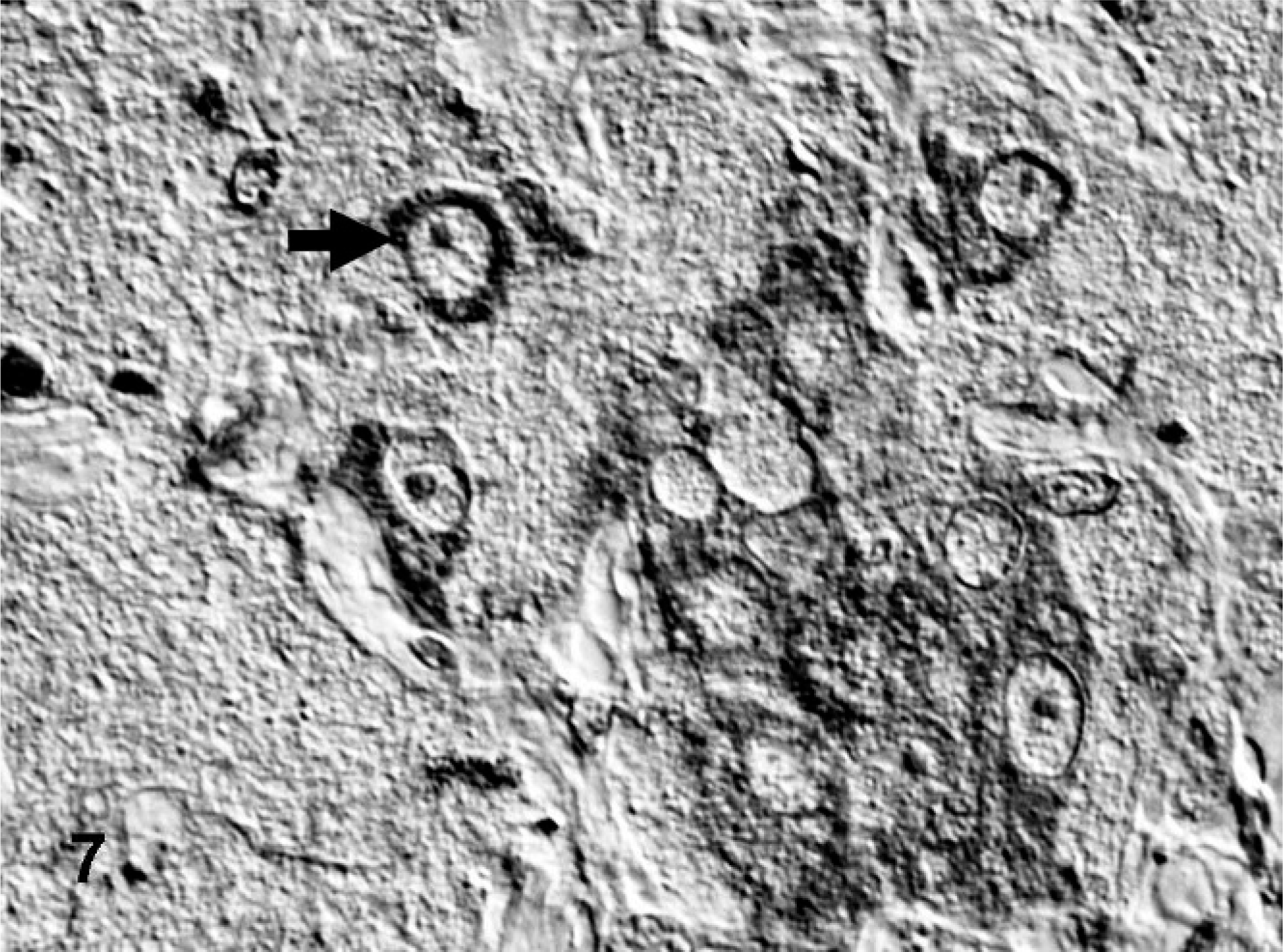
Jejunum; bovine. Immunoreactivity of BDNF in the cytoplasm of most ganglionic neuron-like tumor cells (arrow). ABC method, hematoxylin counterstain, Nomarski differential interference contrast.
Jejunum; bovine. Immunoreactivity of tropomyosin-related kinase-B (Trk-B) in the cytoplasm of most ganglionic neuron-like tumor cells (arrow). ABC method, hematoxylin counterstain, Nomarski differential interference contrast.
To confirm the neuronal specificity of the polyclonal anti-BDNF and anti-Trk-B antibodies, we analyzed normal bovine nervous system tissue and observed an immunoreactivity pattern that was comparable with the murine in-situ hybridization data of the Allen brain atlas (http://www.brain-map.org). 10 Cytoplasmic BDNF immunoreactivity was detected in many neurons of the cerebral cortex and hippocampus, in a few cerebellar Purkinje cells, and in many central and peripheral axons, whereas glial cells were negative. Trk-B immunoreactivity was weaker and more disseminated in neurons of the normal brain. In addition, a moderate ependymal Trk-B immunoreactivity was detected.
Paraparesis in this heifer was attributed to lumbar vertebral fracture. The jejunal nodular ganglioneuroblastoma was an unrelated incidental finding at necropsy. The tumor was composed of nodules of undifferentiated and differentiated neuroblastic cells with abundant tissue that resembled neuropil and perinodular Schwannian stroma–rich tissue. Ganglioneuroblastomas represent an intermediate type in the spectrum of neuroblastic tumors that range from the undifferentiated neuroblastoma to the benign ganglioneuroma. 5, 18 According to Shimada et al., 17 the term ganglioneuroblastoma should be restricted to those tumors with a mature Schwannian stromal component, individual mature and/or maturing ganglion cells, and a neuroblastic component. The term “ganglion cell” or “ganglion-like cell” includes only those further differentiated neuronal tumor cells that resemble normal peripheral ganglionic neurons. 16, 17 The neuroblastic component may be present in distinct macroscopic nodules (nodular ganglioneuroblastoma), as in this tumor, or as multiple microscopic foci (intermixed ganglioneuroblastoma). 16, 17 The neuroblastic cells of nodular ganglioneuroblastomas are conceptually regarded as the consequence of development of one or multiple aggressive or malignant clones, either because of newly acquired genetic aberrations or the persistence of two or more genetically and biologically different cell types. 16, 17 The maturation of peripheral neuroblastic tumors is usually accompanied by the appearance of Schwann cells. Although in vitro studies demonstrate that neuroblastoma cell lines can differentiate into neuronal, Schwannian, and melanocytic cells, depending on the culture conditions, 21 the origin of the intratumoral Schwann cells is still debated. 1, 12 Schwann cells are assumed to produce antiproliferative and differentiation-inducing factors. 9 Therefore, the presence of Schwann cells and stroma in neuroblastic tumors is regarded as a positive prognostic indicator in human oncology. 16, 17
The main ultrastructural features that helped to identify the present tumor as a differentiating neuroblastic tumor included neurofilament-containing processes and dense-core neurosecretory granules. 5, 11, 15 As described by Misugi et al., 11 an increasing number of neurosecretory granules, proliferation of rough endoplasmic reticulum and Golgi complexes, and an overgrowth of nonmyelinated neurites are associated with the progressive differentiation from neuroblastoma to ganglioneuroma.
The observed immunoreactivity pattern is consistent with the findings of Omi et al., 14 who described more pronounced NF and NSE expression in bovine peripheral nervous system tumors with predominantly neuronal cells and a higher S-100, and vimentin expression in those tumors with an abundant stromal component. The expression of vimentin and S-100 but not of NF by the undifferentiated neuroblastic cells in this jejunal ganglioneuroblastoma is concordant with the findings in a case of bovine spinal neuroblastoma. 19 The expression of chromogranin A mainly in the maturing ganglionic neuron-like tumor cells is validated by the ultrastructural demonstration of dense-core neurosecretory granules. 6, 20
The presented case is the first description of BDNF and Trk-B expression in a peripheral neuroblastic tumor of domestic animals. Investigations that use human cell lines and tumor tissue demonstrate that neuroblastoma cells express little or no neurotrophin-3 (NT) and nerve growth factor (NGF), whereas BDNF and NT-5 were expressed at high levels. 3, 13 A potential autocrine BDNF stimulation, which would induce tyrosine phosphorylation of Trk-B, appears to promote cell survival, differentiation, and neurite outgrowth in neuroblastoma cell lines. 3, 13 An autocrine BDNF/Trk-B loop is activated in developing neurons of the embryonic central and peripheral nervous system, 4 which supports the survival of dopaminergic neurons. 2 The finding of BDNF and Trk-B expression in this bovine nodular ganglioneuroblastoma could support the described autocrine BDNF/Trk-B loop as a common pathway in mammalian neuroblastic tumorigenesis, which leads to prolonged tumor-cell survival and neurophilic differentiation.
Footnotes
Acknowledgements
We thank Brigitte Behrens, Bettina Buck, Petra Grünig, and Kerstin Rohn for excellent technical assistance, and Frances Sherwood-Brock for critical review of the manuscript.
