Abstract
Exercise-induced pulmonary hemorrhage (EIPH) is common in horses following intense exertion, occurring in up to 75% of racing Thoroughbreds and Standardbreds. In spite of this, the pathogenesis of EIPH is poorly understood. In 7 racing Thoroughbred horses with EIPH, 6 sections were collected from the left and right lung, representing the cranial, middle, and caudal region of the dorsal and ventral lung (84 sites total). Grossly, both right and left lungs had numerous dark brown to blue-black foci along the caudodorsal visceral pleura. Tissue sections were stained with hematoxylineosin, Masson's trichrome, and Prussian blue. Verhoeff Van Gieson and immunohistochemistry for α-smooth muscle actin were used to assess the pulmonary vasculature. Histologic scores (HS = 0–3) were assigned to each region/slide for the presence and severity of 5 findings: interstitial fibrosis, hemosiderin accumulation, pleural/interlobular septal thickness, arterial and venous wall thickness, and evidence of angiogenesis (maximum cumulative HS = 15). Thirty-nine of the 84 (46%) sections were histologically normal (HS = 0); 33/84 (39%) were mildly to moderately affected, with small amounts of hemosiderin and fibrosis (HS = 1–9) while 12/84 (14%), primarily from the dorsocaudal lung, had severe vascular remodeling, fibrosis, and hemosiderin accumulation (HS = 10–15). In the latter, veno-occlusive remodeling of the intralobular veins colocalized with hemosiderosis, fibrosis, hypertrophy of vessels within the pleura, and interlobular septa and bronchial neovascularization. We propose that regional veno-occlusive remodeling, especially within the caudodorsal lung fields, contributes to the pathogenesis of EIPH, with the venous remodeling leading to regional vascular congestion and hemorrhage, hemosiderin accumulation, fibrosis, and bronchial angiogenesis.
Introduction
Exercise-induced pulmonary hemorrhage (EIPH) is common in horses following bouts of intense exertion; it is a particular problem in racing Thoroughbreds and Standardbreds, and occurs in up to 75% of these animals. 2, 8, 23 The hemorrhage is usually detected endoscopically after racing, although in severe cases it may manifest as epistaxis.
Because EIPH does not usually result in the death of the animal, and is not an impediment to the use of the horse after the racing career is over, there have been limited studies describing the histopathology of this disease. The most extensive investigation of the gross and histologic features of EIPH was performed 20 years ago. 17, 20 These investigators described the clinical features of 26 EIPH-affected horses and the pathology of 19. 16 The gross lesions of EIPH include bilaterally symmetrical dark discoloration of the pleura of dorsocaudal lung, which is firmer than normal lung. 17 Histologically, the primary findings were scattered bronchiolitis, hemosiderophage accumulation, and fibrosis. 20 In addition, the authors used latex vascular perfusion to identify foci of bronchial arterial neovascularization, and suggested that EIPH may arise from the bronchial circulation. 18 More recently, Oikawa analyzed the lungs of 13 Thoroughbreds. The gross lesions of EIPH were similar to those described in the earlier study, and the histologic lesions were described as distortion of bronchioles, and alveolar epithelization. 22
In spite of the frequency of EIPH in the horse population, there is a poor understanding of the pathogenesis of the disease. Many of the factors suggested to contribute to EIPH have been proposed following examination of the histology and ultrastructure of the lungs from EIPH-affected horses; hypothesized mechanisms include capillary stress failure, pulmonary fibrosis, and antecedent small airway disease. 21, 27 Currently, the most accepted hypothesis for EIPH pathogenesis is exercise-induced pulmonary hypertension, resulting in alveolar capillary stress failure. 7, 27 However, this hypothesis does not account for the lesions of EIPH, i.e., fibrosis and bronchial circulation proliferation.
The present study was performed to further evaluate the pulmonary pathology of EIPH in adult racing Thoroughbreds. We characterized the presence, distribution, severity, and features of lesions from 12 lung regions from 7 horses that were removed from racing because of repeated epistaxis during or following racing. Our histologic findings confirm the presence of fibrosis, hemosiderin, and bronchial angiogenesis. In addition we identify, heretofore unreported, pulmonary vascular changes. Specifically, we documented morphologic evidence of pulmonary veno-occlusive remodeling that bears striking similarity to pulmonary veno-occlusive disease in humans. Based upon these findings, we suggest that regional veno-occlusive remodeling, especially within the caudodorsal lung fields, contributes to the pathogenesis of EIPH.
Materials and Methods
Selection criteria for EIPH horses
All of the horses included in the study were racing Thoroughbreds from the Singapore Turf Club, registered with the Malaysian Racing Association. A detailed log of the frequency of bleeding following racing was not available, but it is known that each of the horses was taken out of racing following at least 1 episode of epistaxis during racing.
Postmortem tissue collection
Following euthanasia of the animals, the entire cardiopulmonary system was removed from the thoracic cavity, and the lungs were photographed prior to sampling. Six sections were collected from both the right and left lung representing the cranial, middle, and caudal region of the dorsal and ventral lung (D1-3; V1-3) for a total of 84 sections from the 7 horses (Fig. 1). The tissues were fixed in 10% neutral buffered formalin and shipped to Michigan State University for processing and evaluation.
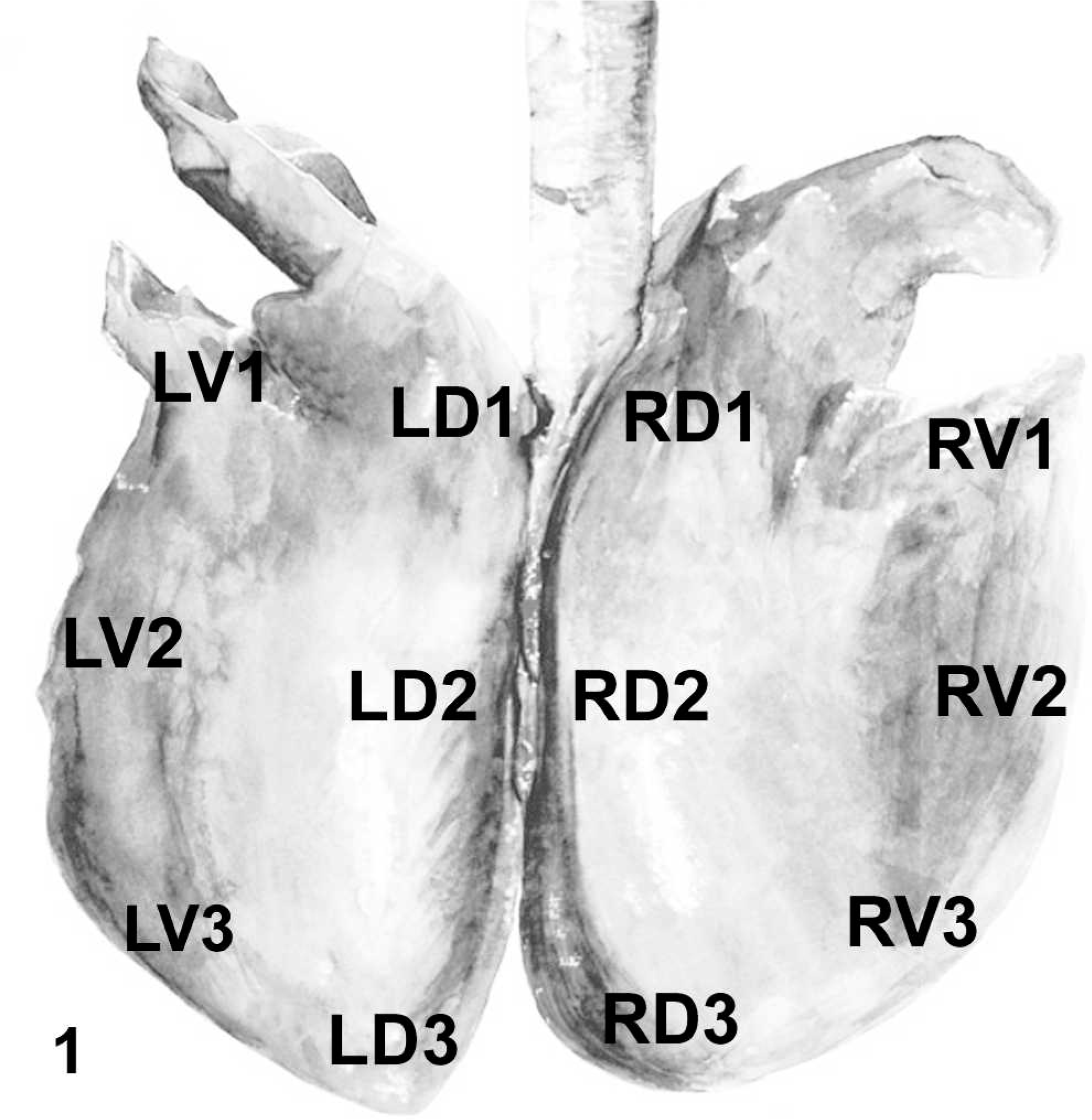
Equine lung diagram; pictorial depiction of the 12 sites sampled from EIPH horses, representing left (L), right (R), dorsal (D), and ventral (V) lung fields.
Histopathology
The sections were processed and embedded in paraffin. Six-μm sections were placed on glass slides and routinely stained using hematoxylin eosin, Masson's trichrome, Prussian blue, and Verhoeff-Van Gieson. To assess the amount and distribution of collagen, slides were stained with picrosirius red, a collagen-specific stain, following previously published methods.
24
All of the slides were evaluated without knowledge of sampling location using brightfield microscopy, except for the picrosirius red–stained slides, which were evaluated using polarized light. Each of the slides was assigned a histopathologic score based upon 5 criteria: interstitial fibrosis, amount of hemosiderin, pleural/interlobular septal thickness, evidence of neovascularization, and vascular (arterial and venous) wall thickness. The scoring for each of the 5 lesions ranged from 0 (not present) to 3 (severe); a cumulative histopathology score was calculated for each slide (maximum possible = 15). The slides were placed into 3 categories based upon histological score: 0 = histologically normal; 1–9 = mild to moderate lung changes; 10
Immunohistochemistry
α-SMA immunohistochemistry was used to assess the thickness and location of the vascular tunica media vis-à-vis the elastic lamina and adventitia; it was also used to detect foci of arteriolar neovascularization. Six-μm tissue sections were deparaffinized in xylene and rehydrated in a graded series of ethanol before being incubated overnight at 4°C with a 1 : 100 dilution of mouse monoclonal antibody against α-smooth muscle actin (Dako Corporation; Carpentaria, CA). This was followed by incubation with avidin–biotin conjugated horseradish peroxidase as per manufacturer's instructions (Vector Laboratories; Burlingame, CA) and diaminobenzidine (Sigma Chemical; St. Louis, MO).
Statistical analysis
The histopathologic score of the left and right lung were compared using a Student's t-test. Because the mean HS of the right and left lung did not differ significantly, these values were combined for further analysis. The histopathologic scores of the 6 lung regions were compared by use of a repeated measure ANOVA. When P < 0.05, means were compared with the Student Newman–Keuls test.
Results
Signalment
The 7 horses were all male-castrate Thoroughbred racehorses; the average age of the horses, when available, was 6 years (range 5–7). The interval between the last reported episode of EIPH and euthanasia averaged 97 days.
Gross pathology
Gross lesions were restricted to the lungs and similar to those described in previous reports. 17, 22 Both right and left lungs of all of the animals had numerous dark brown to blue–black foci along the caudodorsal visceral pleura of both right and left lungs. Examination of the formalin-fixed tissues after sectioning for histopathology revealed fine thickening of interlobular septa, and small discrete (1–2 mm) foci of dark brown–black lung parenchyma (Fig. 2); these changes were only detected in those lung regions with the highest histopathology score (see below).
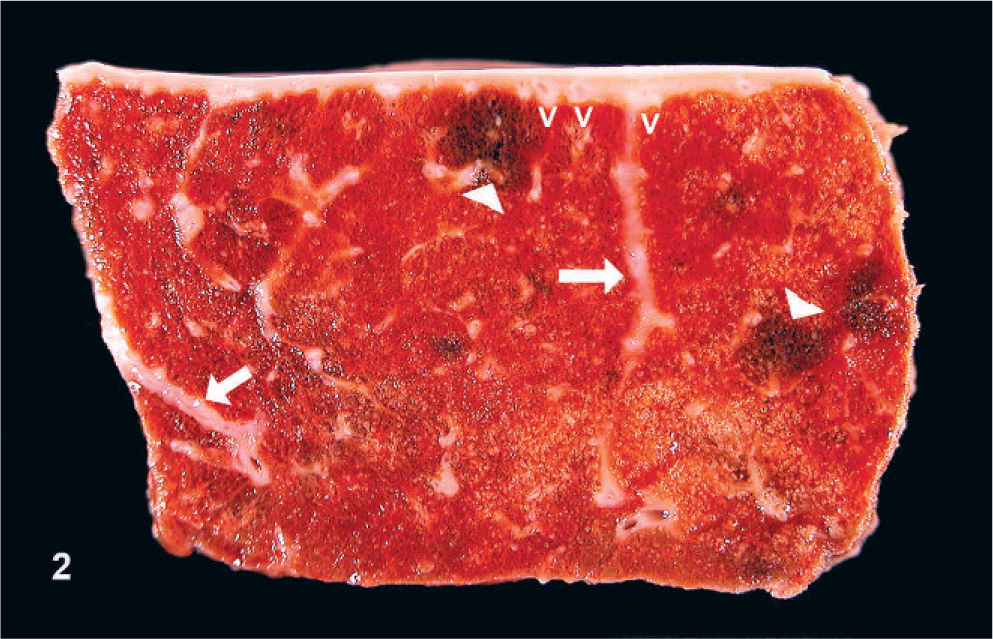
Equine lung; horse No. 4. Formalin-fixed lung from high HS region of EIPH lung. The interlobular septa are thickened (arrows), and there are small foci of parenchymal discoloration (arrowheads). Hypertrophied vessels (V) are visible within the pleura.
Histopathologic scoring (HS)
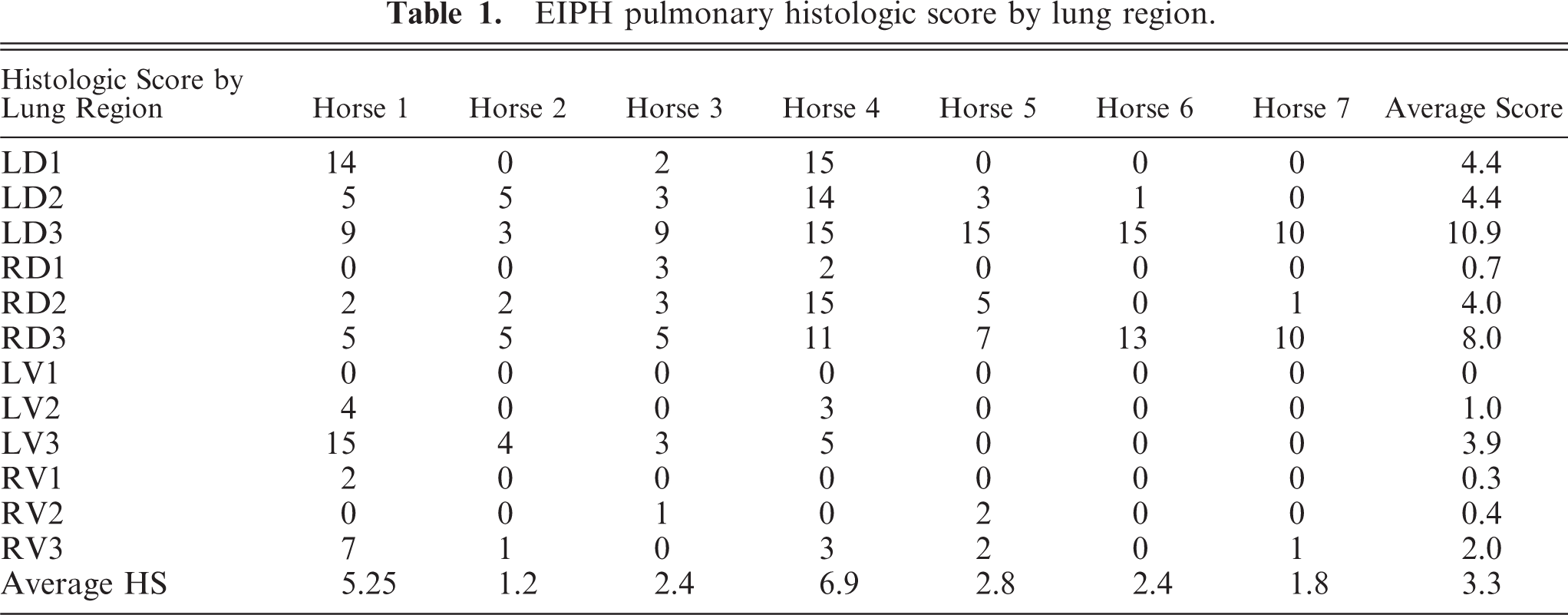
The HS of the individual sections of lung ranged from 0 to 15 (Table 1). Thirty-nine/eighty-four (46%) were histologically normal (cumulative score = 0); 33/84 (39%) had HS between 1 and 9 and 12/84 (14%) had HS between 10 and 15. The mean HS of the right and left lung did not differ significantly, and were combined for further analysis. The HS of region D3 was significantly higher than any other region (Fig. 3). Furthermore, the HS of region D2 was significantly higher than regions V1 and V2.
EIPH pulmonary histologic score by lung region.
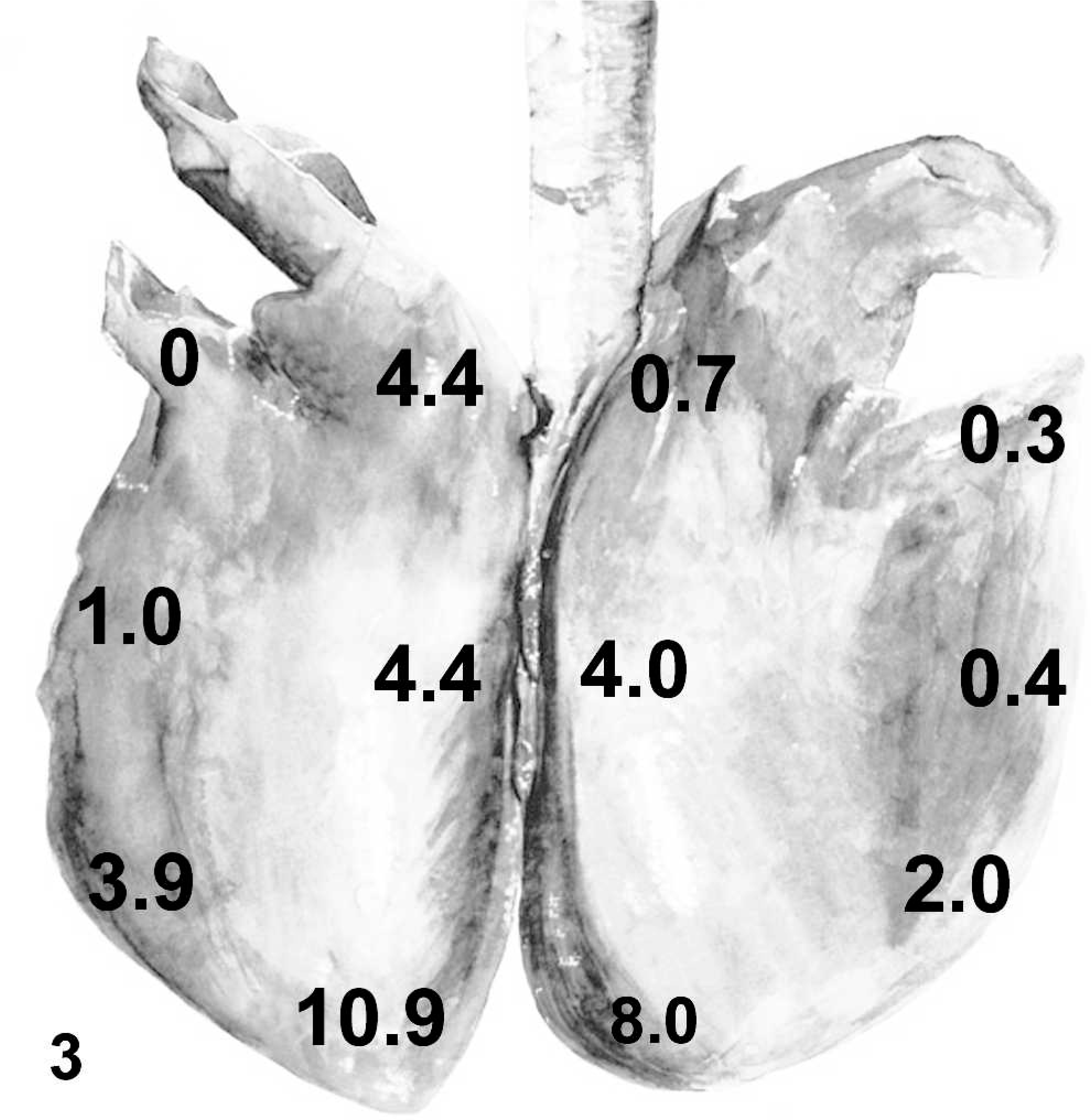
Equine lung diagram; average HS from the 12 lung sites sampled in the EIPH horses.
Histopathology
Lung regions with a HS = 0 were histologically normal (Fig. 4). In HS 1–9 sections, the primary findings consisted of mild to moderate hemosiderin accumulation and fibrosis. The hemosiderin was mostly within the cytoplasm of macrophages (hemosiderophages). Small numbers of hemosiderophages were present within the lumen of bronchioles, with the remaining primarily found associated with the bronchovascular bundle, around intralobular veins, along the septa, and beneath the pleura. The fibrosis was mild, and primarily around small numbers of intralobular veins and bronchovascular bundles, and to a lesser extent within the interlobular septa.
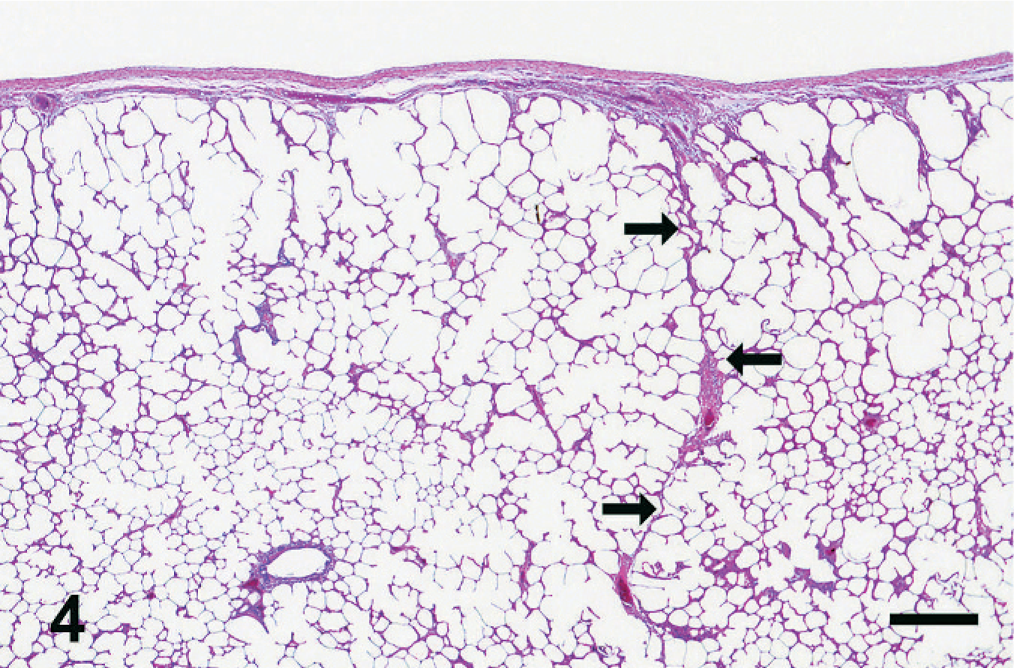
Equine lung; horse No. 4. HS = 0 region of equine lung. The interlobular septum is thin and delicate (arrows), and there is no evidence of hemosiderin or fibrosis. HE. Bar = 500 μm.
The spectrum of histologic changes in HS 10–15 consisted of fibrosis, hemosiderin accumulation, and vascular remodeling. This constellation of changes was consistently distributed at 4 major sites within the lung: alveolar interstitium, perivascular/peribronchiolar, subpleural, and associated with the interlobular septa (Fig. 5).
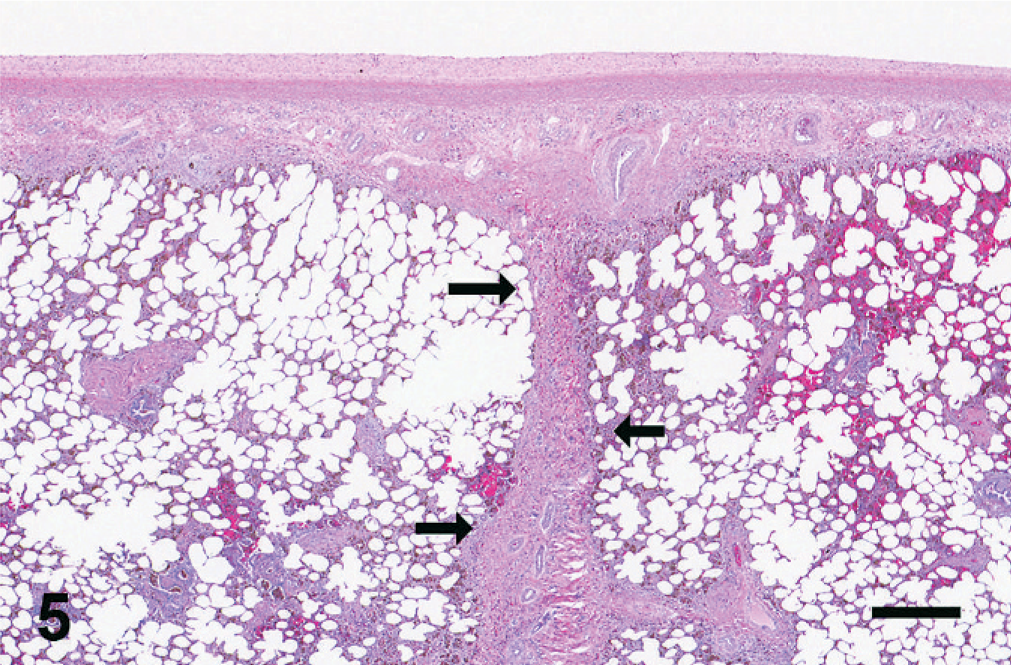
Equine lung; horse No. 4. HS = 15 region of EIPH lung (histologic appearance of lung in Fig. 2). The interlobular septum (arrows) and pleura are markedly thickened and fibrotic. Fibrosis is also present around the bronchiolovascular bundles and within the interstitium. HE. Bar = 500 μm.
Hemosiderin accumulation was abundant in HS 10–15 sections and was especially evident in Prussian blue–stained sections (Fig. 6). Cell-associated hemosiderin was found within the cytoplasm of macrophages, and hemosiderin was often free within the tissue; the hemosiderin colocalized with the fibrosis. Small foci of mineralization were noted within the collagen bundles of the interlobular septa and within the collagen of the bronchiolovascular bundles (Fig. 7). Prussian blue also detected iron within the mineralized collagen in the septa and the bronchiolovascular bundles.
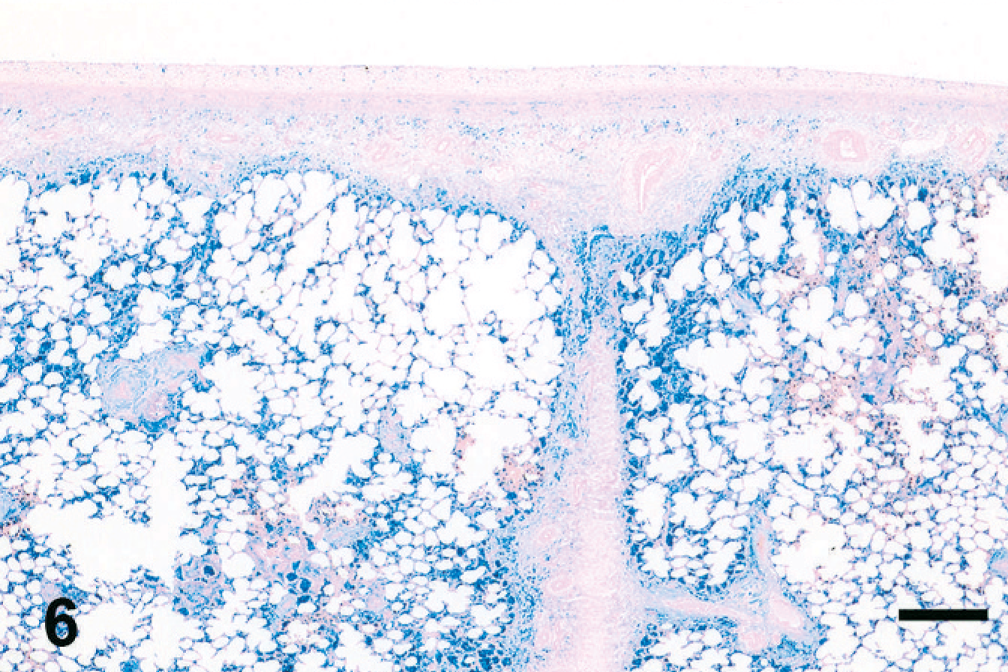
Equine lung; horse No. 4. Histochemical detection of iron in HS = 15 region of EIPH lung (same region as Figs. 2, 5). Iron heavily accumulates in regions of fibrosis, particularly adjacent to the thickened septum, beneath the pleura, and around airways and vessels, but is also present within alveolar interstitium. Prussian blue. Bar = 500 μm.
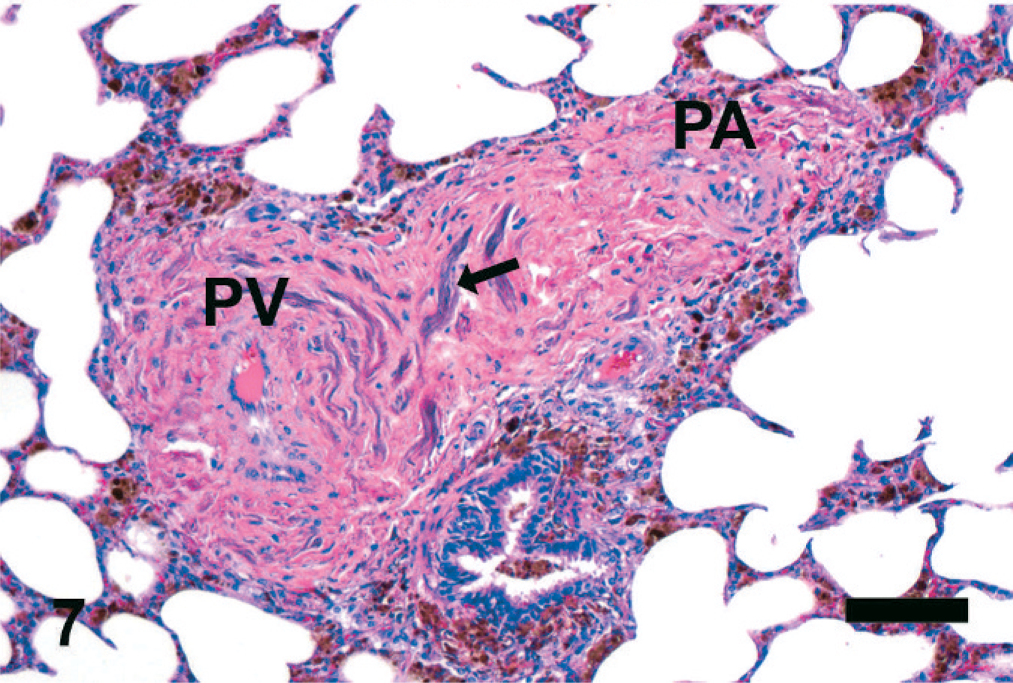
Equine lung; horse No. 4. Fibrosis and mineralization of collagen in bronchiolovascular bundle. The collagen deposition surrounds the pulmonary vein (PV) and pulmonary artery (PA). Mineralization of collagen can be found in these areas of fibrosis (arrow). HE. Bar = 50 μm.
Picrosirius red revealed the increases and distribution changes of collagen in affected versus unaffected lung. Collagen distribution in the unaffected lung was localized to the pleura, thin interlobular septa, and around airways and the vasculature; focal aggregates of collagen were also found at alveolar branch points (Fig. 8). The fibrosis at all locations consisted of well-organized bundles of mature collagen, which stained intensely with Masson's trichrome, and illuminated brightly under polarized light in the picrosirius red–stained tissues (Figs. 9, 10). The interlobular septa and subpleural lung were expanded by collagen, which often blended with adjacent foci of alveolar interstitial fibrosis (Figs. 9, 10), and dense collagen was found surrounding the intralobular veins (Fig. 10).
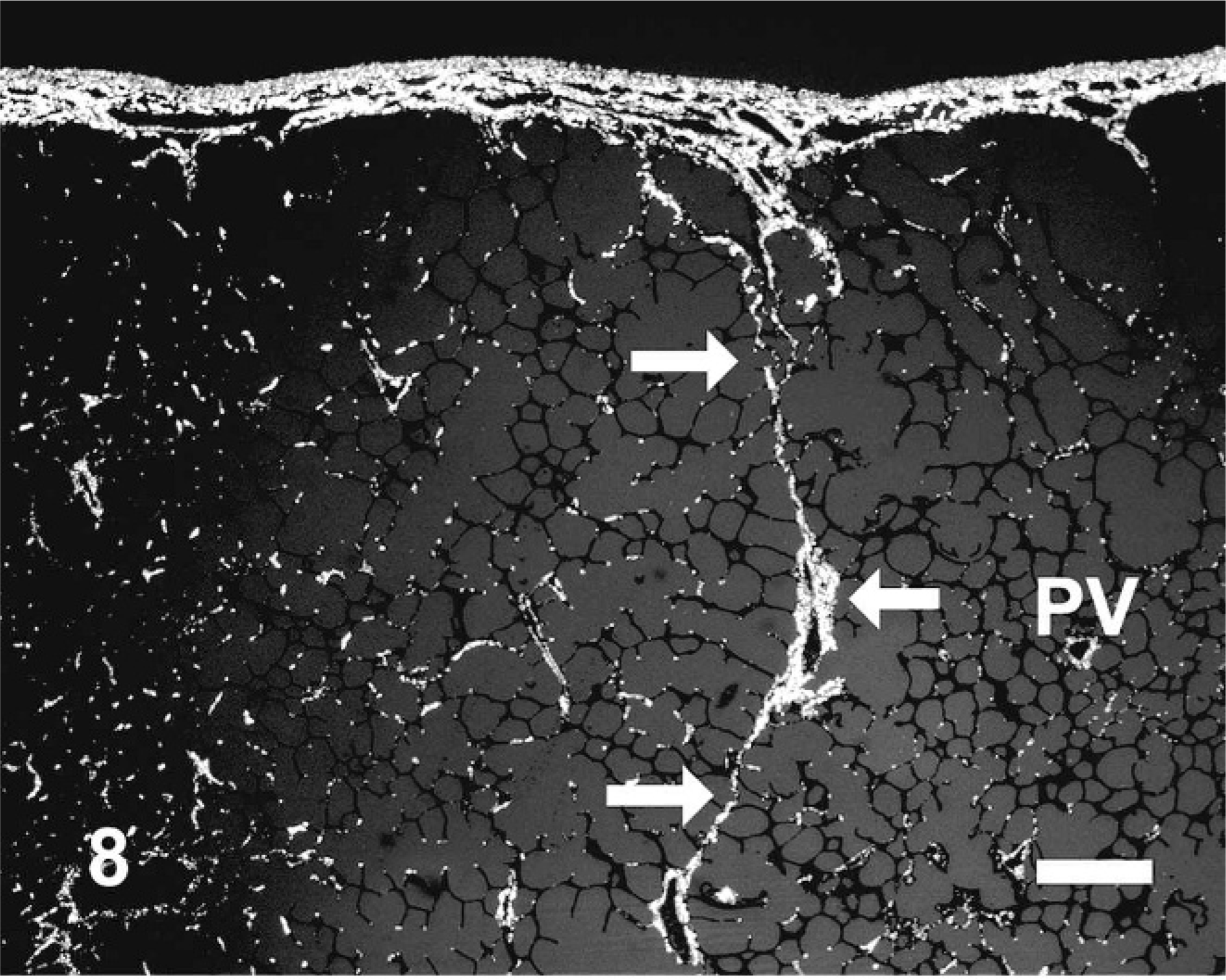
Equine lung; horse No. 4. Histochemical detection of collagen using picrosirius red and polarized light microscopy in HS = 0 region of equine lung. Collagen is mostly restricted to the pleura and interlobular septum (arrows). Note the small amount of collagen surrounding a normal intra-lobular pulmonary vein (PV). Picrosirius red, polarized light. Bar = 50 μm.
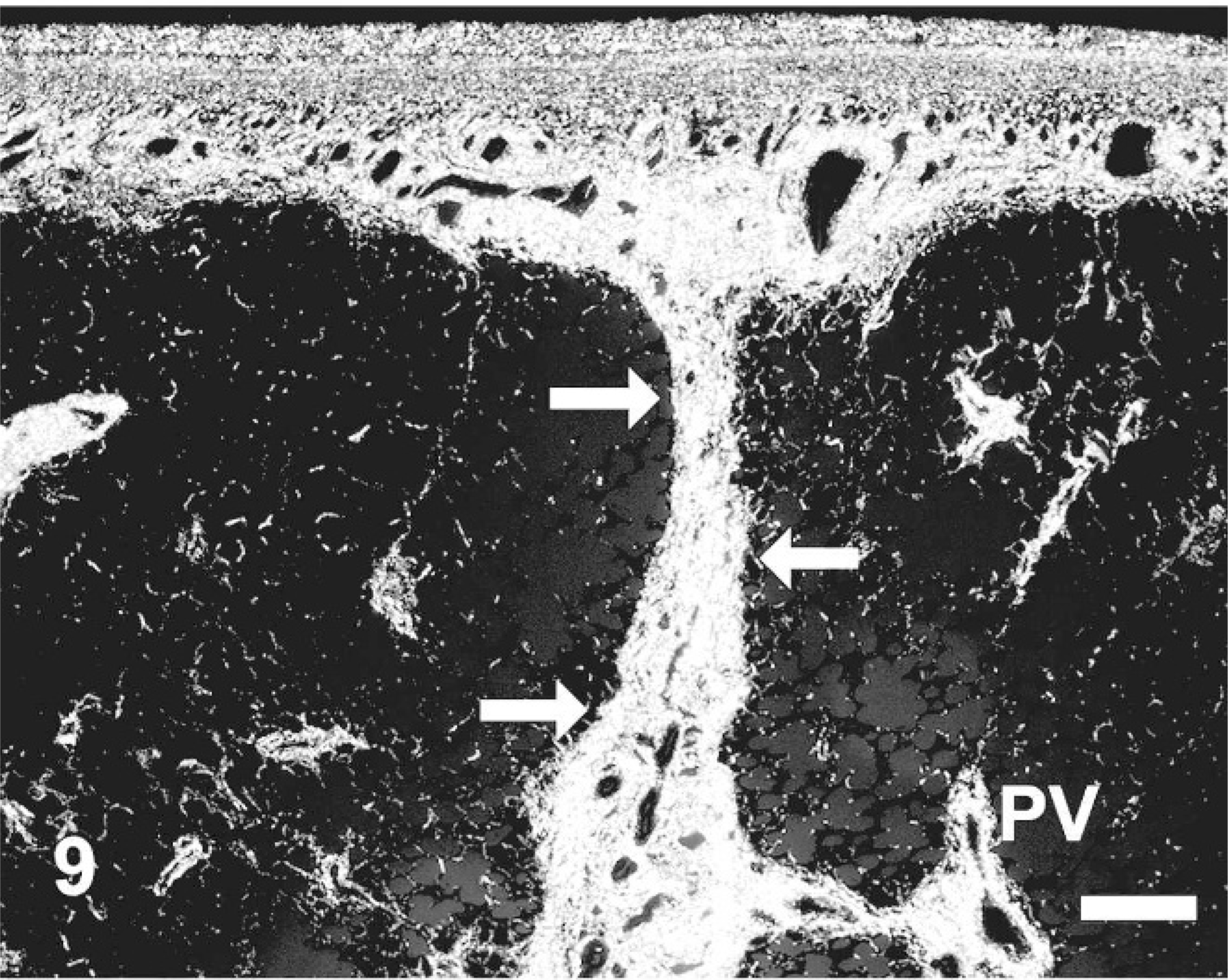
Equine lung; horse No. 4. Histochemical detection of collagen using picrosirius red and polarized light microscopy in HS = 15 region of EIPH lung. Increases in collagen are evident within the pleura and interlobular septum (arrows), as well as around a pulmonary vein (PV). Picrosirius red, polarized light. Bar = 50 μm.
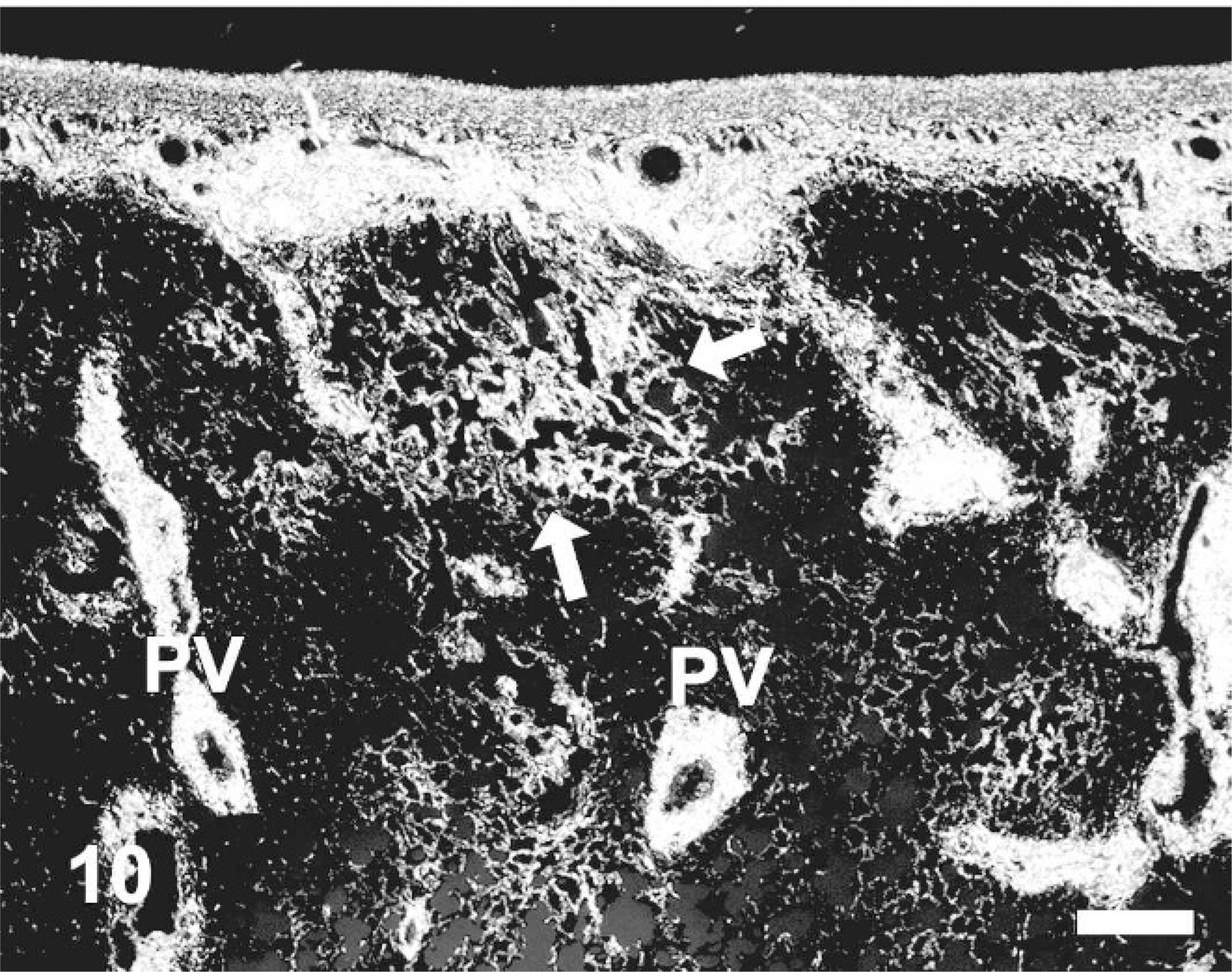
Equine lung; horse No. 4. Histochemical detection of collagen using picrosirius red and polarized light microscopy in HS = 15 region of EIPH lung. Increases in collagen are evident within alveolar interstium (arrows) and around intralobular veins (PV). Picrosirius red, polarized light. Bar = 50 μm.
Vascular histopathology
The vascular histology within the HS = 0 and HS = 1–9 categories was normal; a brief review of important micro-anatomic features follows. Large- and medium-sized branches of the bronchial artery could be found within the normal equine pleura; these vessels had a well-developed tunica media and a thin internal elastic lamina (Fig. 11). Pulmonary arteries and veins were distinguished by location and histologic features; pulmonary arteries followed the conducting airways, had a well-developed tunica media, a roughly equal wall thickness-to-lumen diameter, and an internal and external lamina (Fig. 12). Branches of pulmonary veins were found along the conducting airways, as intralobular branches within the pulmonary parenchyma, and within the interlobular septa and pleura. Pulmonary veins were thin-walled, with less smooth muscle than arteries, had a low wall thickness-to-lumen ratio and a single elastic lamina separating the media from the adventitia; intralobular veins also had small numbers of elastic fibers present within the adventitia (Fig. 13).
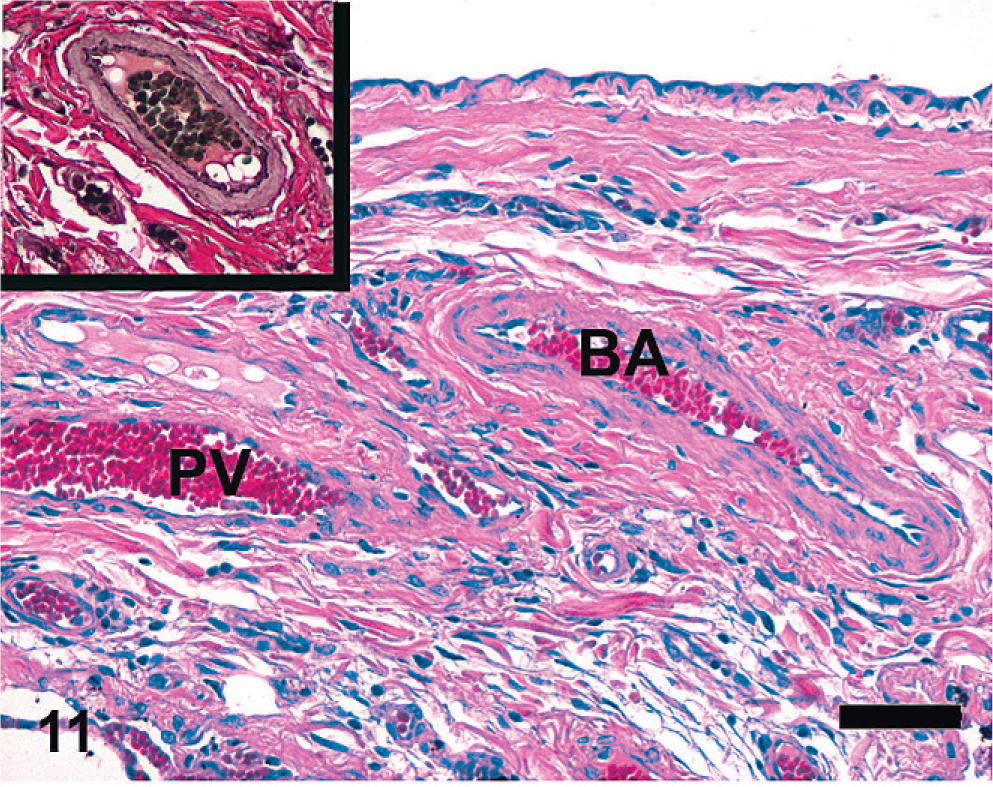
Equine lung; horse No. 1. Microscopic anatomy of pleural vasculature, HS = 0. Branches of the bronchial artery (BA) are present within the pleura and interlobular septa in horses; these arteries have well-developed internal elastic lamina (inset). Thin-walled branches of pulmonary vein (PV) are also present. HE. Bar = 50 μm.
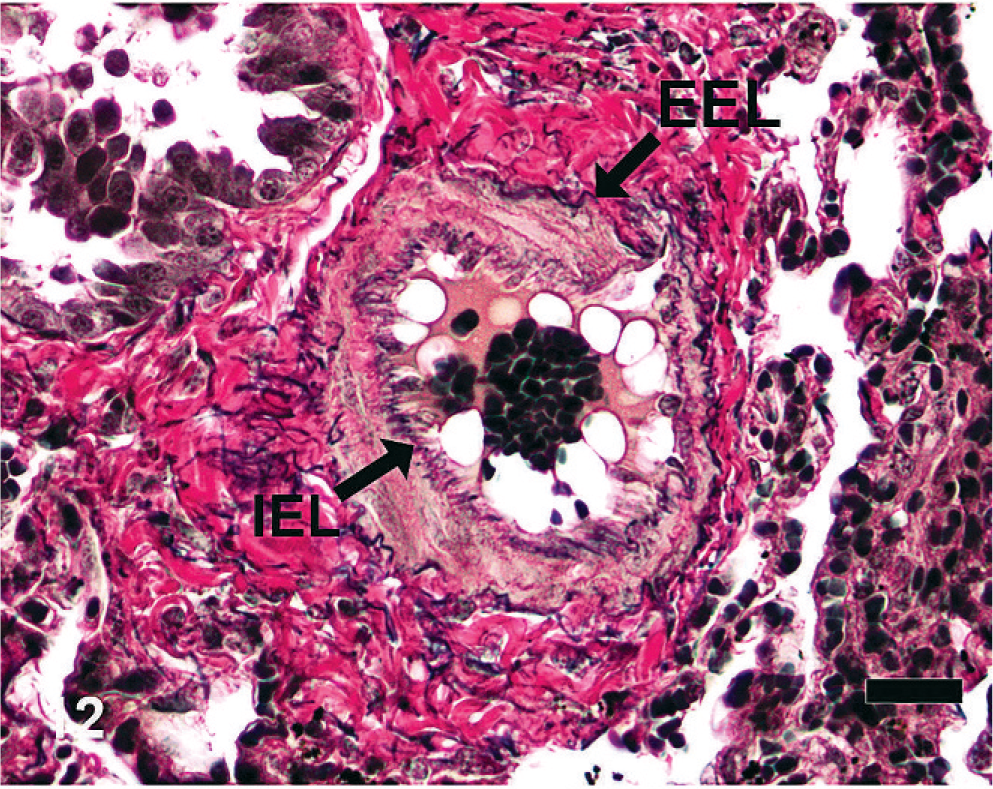
Equine lung; horse No. 1. Microscopic anatomy of pulmonary vasculature, HS = 0. Branches of the pulmonary artery run alongside the conducting airways and have well-developed internal (IEL) and external (EEL) elastic lamina. Verhoeff-Van Gieson. Bar = 20 μm.
In HS 10–15, there was extensive vascular remodeling involving the venous and arterial systems. There were numerous thick-walled vessels within the pleura and interlobular septa (Fig. 14). These vessels had marked medial smooth muscle hypertrophy and mild to moderate intimal hyperplasia. The elastic lamina in these vessels was inconsistent; unlike arteries, most of these vessels lacked organized elastic lamina, while other vascular profiles had a small internal elastic lamina, and fewer possessed both an internal and external elastic lamina as seen in bronchial arterial branches (Fig. 15). Throughout the affected regions, most intralobular veins were surrounded, and obscured, by a prominent collar of mature collagen (Fig. 16); in Van Gieson–stained sections, the increased extracellular matrix was found within the adventitia, and interposed between the elastic lamina and the lumen, often resulting in a markedly reduced lumen of the affected veins (Fig. 17). There was a marked increase in bronchial arteriolar profiles within the fibrotic alveolar interstitium, pleura/subpleura, and within the thickened interlobular septa (see below).
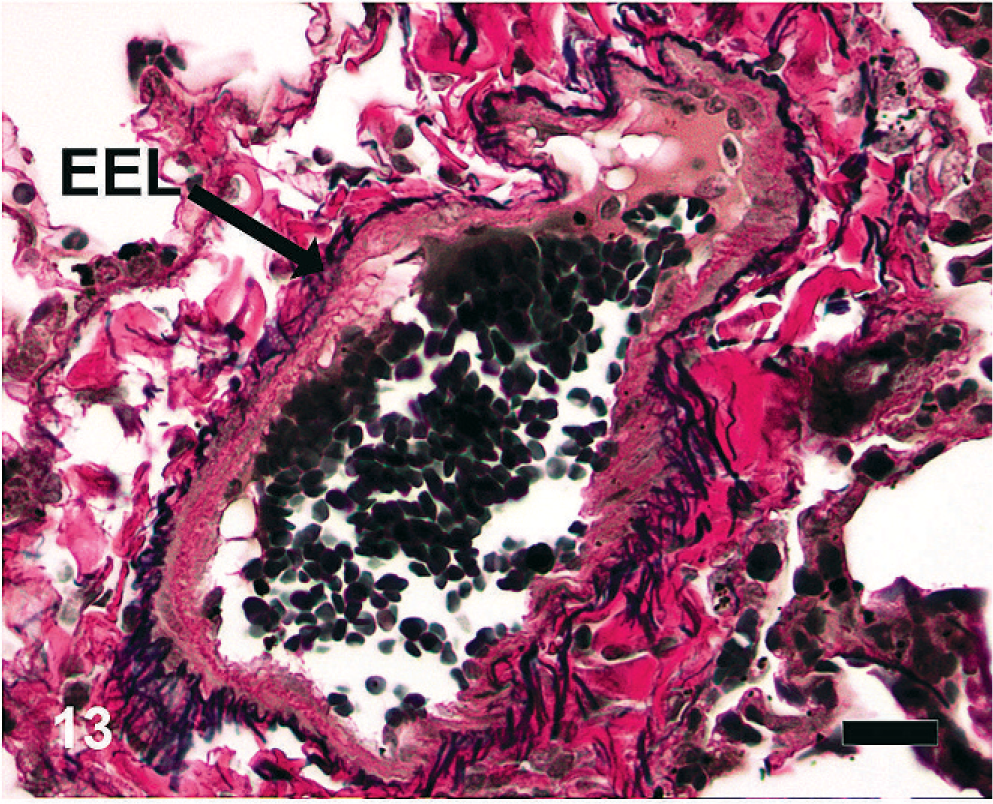
Equine lung; horse No. 1. Microscopic anatomy of pulmonary vasculature, HS = 0. Intralobular branches of the pulmonary veins are found within the alveolar parenchyma. The vessels have an external elastic lamina (EEL) between the adventitia and tunica media. Note the adventitial collagen and elastin fibers. Verhoeff-Van Gieson. Bar = 20 μm.
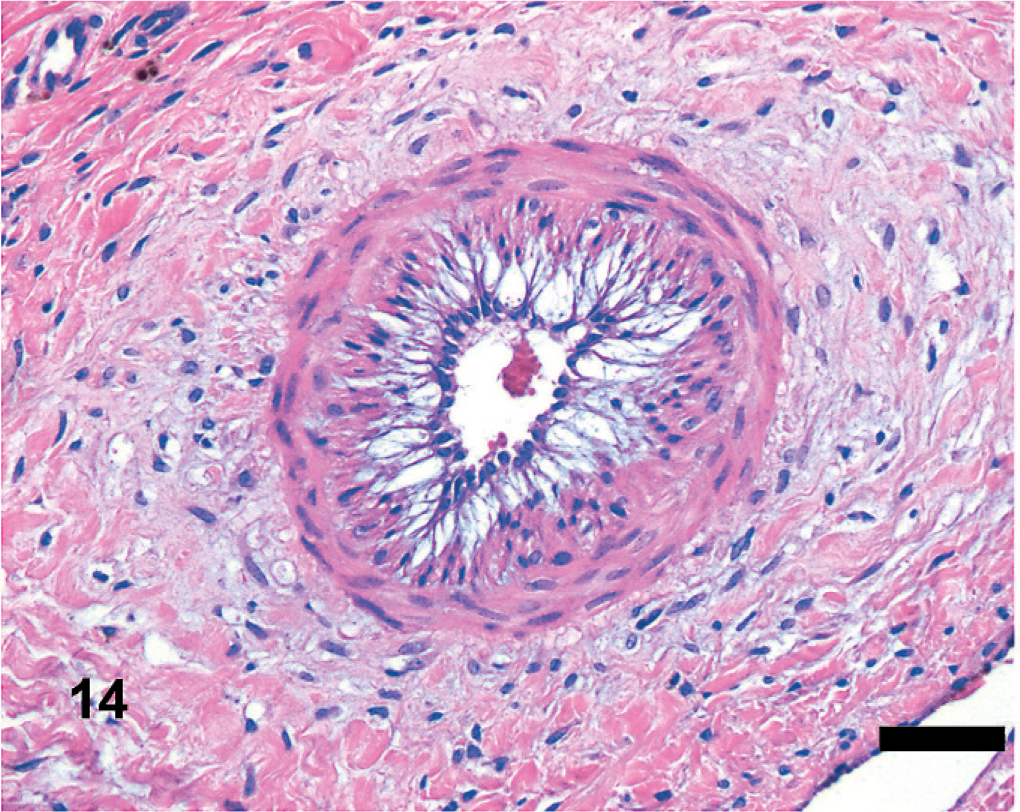
Equine lung; horse No. 1. Microscopic anatomy of pleural vasculature, HS = 15. Hypertrophied vessels are present within the pleura and interlobular septa. The tunica media is thickened and there is intimal hyperplasia. HE. Bar = 50 μm.
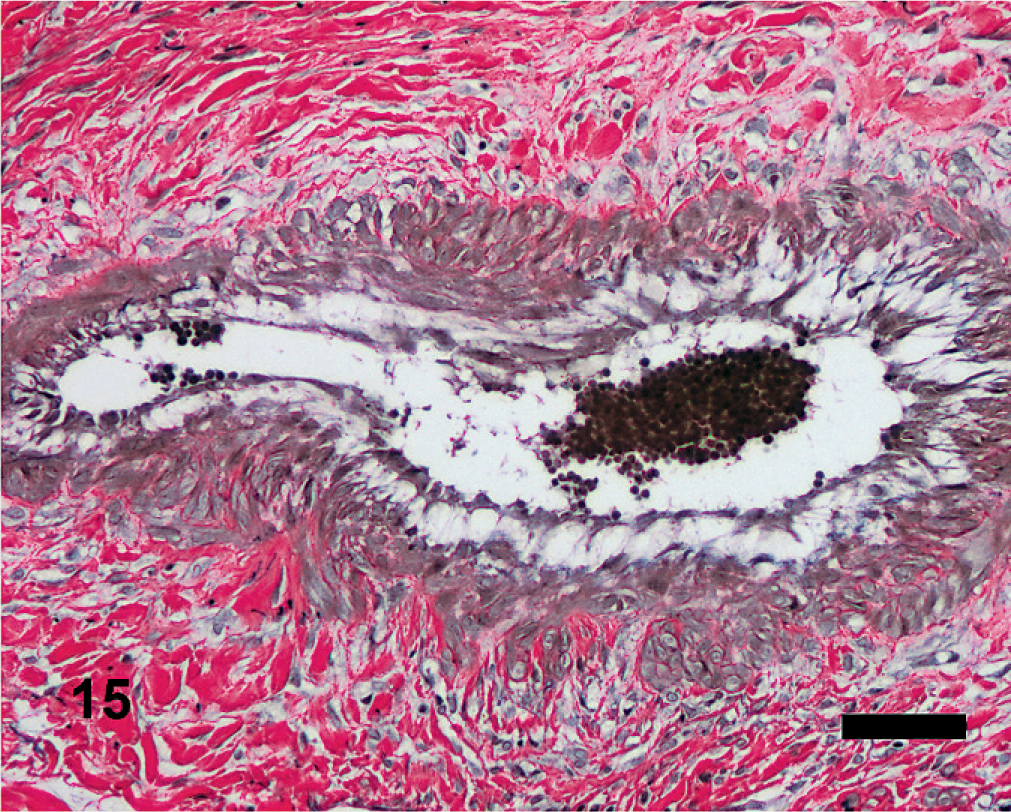
Equine lung; horse No. 1. Microscopic anatomy of pleural vasculature, HS = 15. The hypertrophied vessels often lack well-developed internal or external elastic lamina. Verhoeff-Van Gieson. Bar = 50 μm.
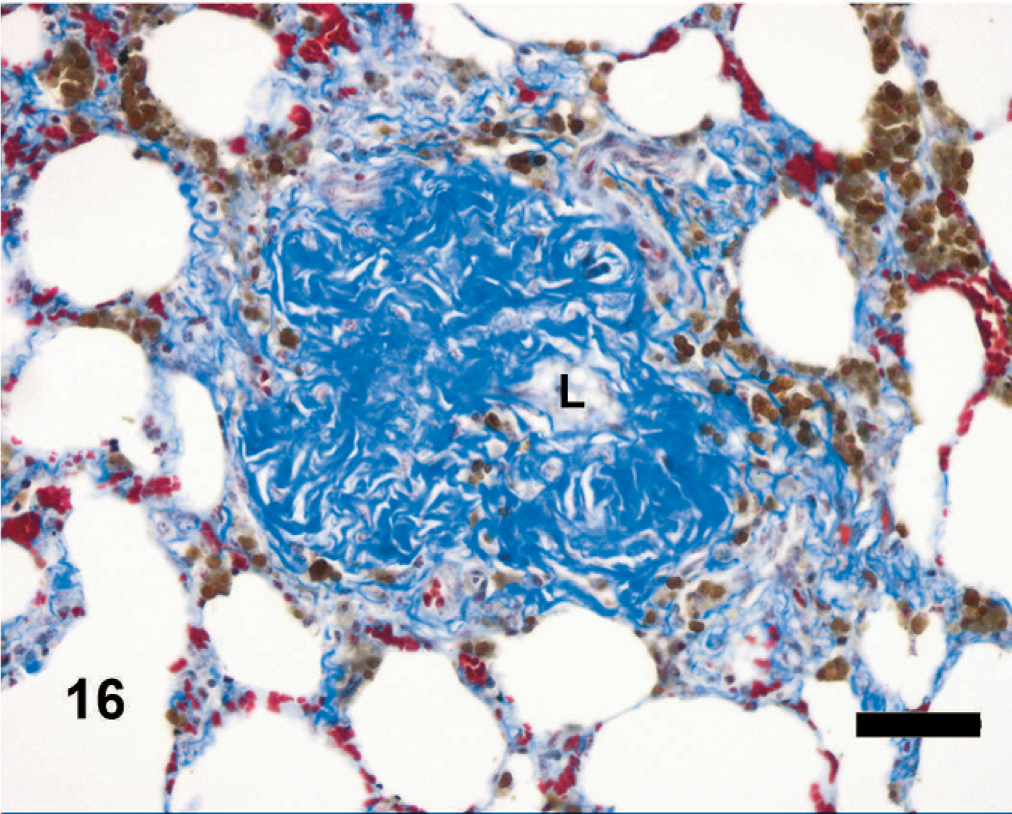
Equine lung; horse No. 1. Microscopic anatomy of pulmonary vasculature, HS = 15. Veno-occlusion of intralobular veins. The fibrosis surrounds and partly occludes an intra-lobular pulmonary vein. The collagen fibers form a dense collar around the vessel, the vessel wall is obscured, and the lumen (L) is reduced. Note the abundant hemosiderin within the surrounding alveoli. Masson's trichrome. Bar = 50 μm.
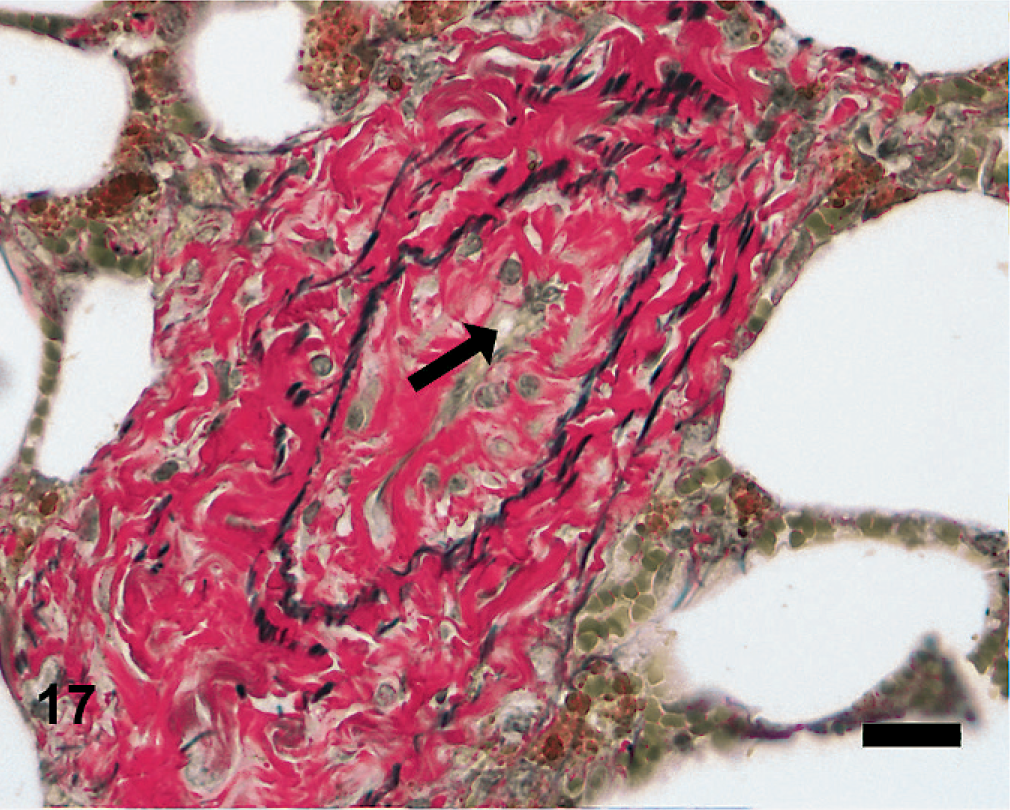
Equine lung; horse No. 1. Microscopic anatomy of pulmonary vasculature, HS = 15. Veno-occlusion of intralobular veins. The fibrosis expands the adventitia and is interposed between the elastic lamina and vein lumen. The lumen of the vessel (arrow) is severely reduced. Verhoeff-Van Gieson. Bar = 50 μm.
Smooth muscle actin immunohistochemistry
In HS = 0 and HS = 1–9 sites, all branches of the pulmonary and bronchial arterial systems had a tunica media of normal thickness, while the media of the pulmonary veins was much thinner, and was located internal to the external elastic lamina (Fig. 18). As noted above, in the sections with HS 10–15, there was a marked increase in bronchial arteriolar profiles within the alveolar interstitium, the pleura/subpleura, and interlobular septa (Fig. 19). α-SMA labeled the markedly increased thickness of the media of the muscular vessels within the pleura and interlobular septa (Fig. 19). The media of the intralobular veins was not appreciably thickened, was internal to the thickened adventitia noted above, and adjacent to the compressed lumen (Fig. 19).
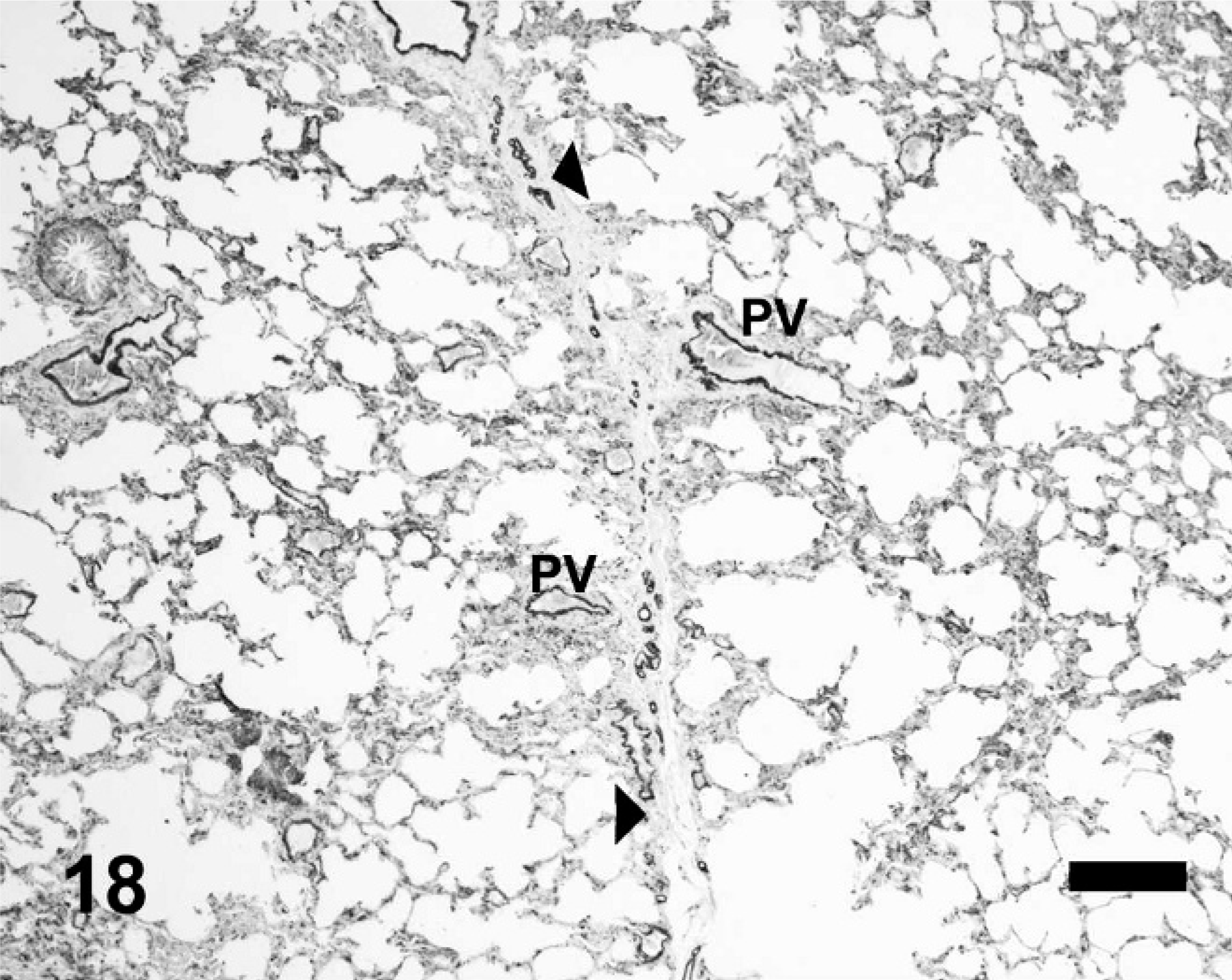
Equine lung; horse No. 1. α-Smooth muscle actin immunohistochemistry, HS = 0. Smooth muscle actin is evident within the media of vessels within the interlobular septum (arrowheads), surrounding bronchioles, and within the wall of large blood vessels. PV = pulmonary veins. Diaminobenzidine with no counterstain. Bar = 200 μm.
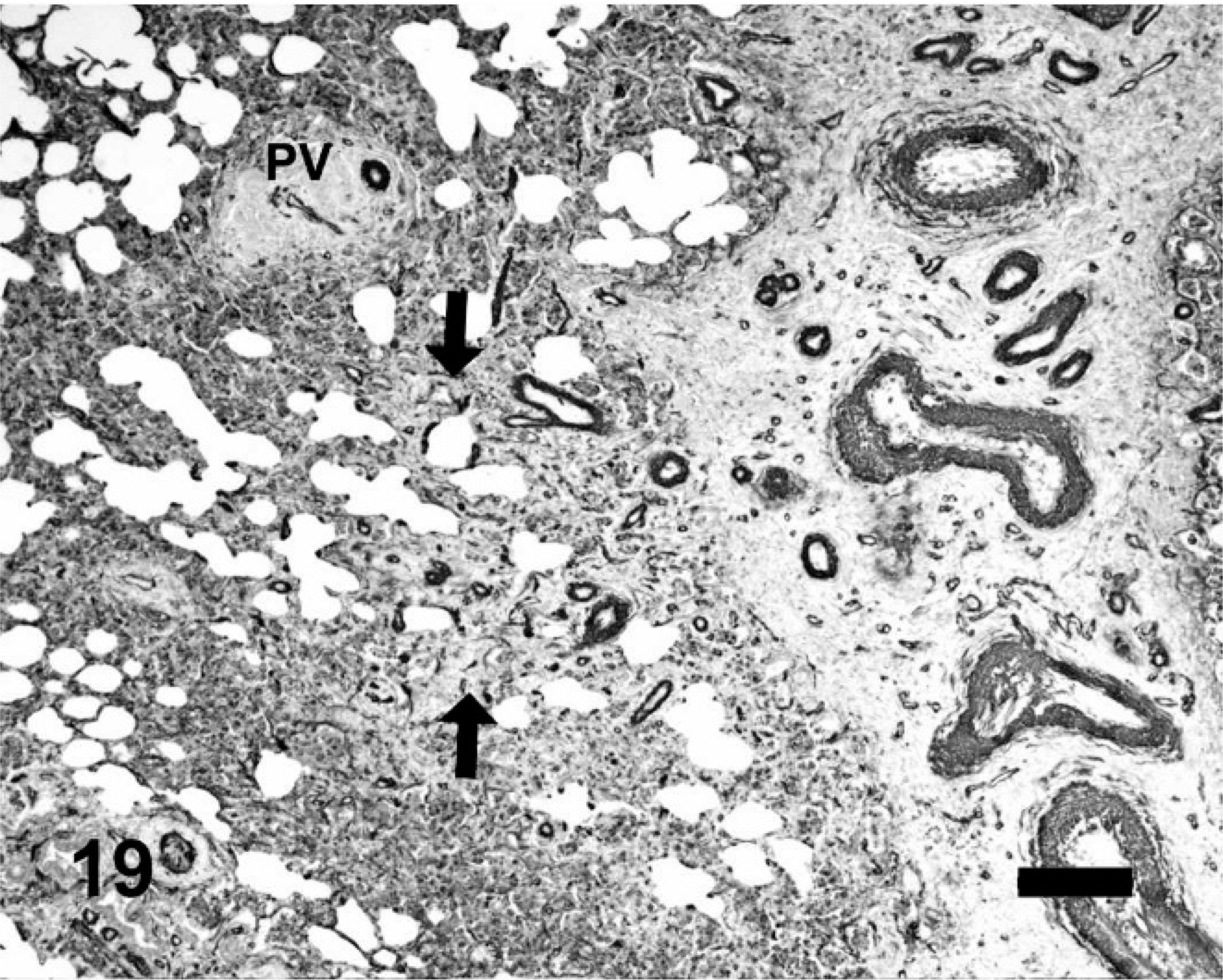
Equine lung; horse No. 4. α-Smooth muscle actin immunohistochemistry, HS = 15. Smooth muscle actin is present within the hypertrophied tunica media of the vessels within the interlobular septum. Small muscular arterioles are present within foci of neovascularization (between arrows). In the occluded intralobular veins (PV), the tunica media is surrounded by the collar of collagen, and adjacent to the reduced vein lumen. PV = pulmonary veins. Diaminobenzidine without counterstain. Bar = 200 μm.
Discussion
Although previous studies using electron microscopy, histopathology, and latex vascular perfusion techniques have implicated both the pulmonary capillary bed and the bronchial circulation as the source of bleeding in EIPH, 18, 27 there is no unifying hypothesis that explains the contributions of the 2 circulations to the histopathology of the disease. The present study is the first report of regional veno-occlusive remodeling in EIPH. The veno-occlusion is colocalized with extensive fibrosis and hemosiderin accumulation within primarily the caudodorsal lung fields. While we have demonstrated an association between veno-occlusive remodeling in the lungs and EIPH, it remains to be determined if the veno-occlusion is causative for EIPH or is a consequence of chronic bleeding within the lung. A review of the literature failed to find documentation of incidental vein-specific collagen accumulation in the lung or other organs experiencing hemorrhage or tissue hemosiderosis. We think it is more likely that the venous pathology/occlusion plays a major part in the initiation of EIPH.
The veno-occlusive remodeling has not previously been reported in association with EIPH. This process affected primarily the intralobular veins within the high HS regions of lung and was absent from the lower HS categories. The hallmark of the lesion was extensive deposition of collagen in the wall of the vein, resulting in a visibly reduced vein lumen. Previous papers have described arterial wall hypertrophy and increased connective tissue around small arterioles, especially within the most severely affected regions of the lung, but not changes to the pulmonary veins. 20, 22 It is possible, however, that remodeled pulmonary veins could be misidentified as pulmonary arteries; indeed a VVG-stained vessel identified as an artery in the O'Callaghan study is morphologically similar to the veno-occlusion found in the current study (see O'Callaghan et al., Fig. 11). 20 In interpreting the venous pathology, it is important to remember that the tissues in this study were not fixed via vascular perfusion at a constant pressure. Because of this, it is possible that the apparent decrease of the vein lumen represents postmortem vascular wall collapse because of collagen accumulation, rather than intravital vein narrowing. The use of VVG to highlight the location of the elastic lamina relative to the collagen deposition and lumen suggests that there is a true decrease in lumen diameter, rather than an inward collapse of the vessel wall.
The spectrum of histologic findings within the high HS regions of the EIPH lung, especially the anatomic colocalization of veno-occlusive remodeling, fibrosis, and hemosiderin, is very similar to pulmonary veno-occlusive disease (PVOD) in humans. In people, the latter is an uncommon cause of pulmonary hypertension and is occasionally associated with hemoptysis. 10, 11 The histologic features of long-standing cases of PVOD are pulmonary venous sclerosis, interstitial and septal fibrosis, marked hemosiderin accumulation, and proliferation of the bronchial circulation. 25, 26 As in EIPH, foci of collagen mineralization and siderosis are also features of chronic PVOD. 26 The accumulation of large amounts of hemosiderin in the lungs is known to be associated with interstitial fibrosis as has been described in cases of idiopathic pulmonary hemosiderosis in humans. 3, 15
As has been reported by others, 17, 20, 22 EIPH lesions were most common in the caudodorsal lung fields. In lung regions with the highest HS, the major lesions, i.e., veno-occlusive remodeling, hemosiderin, and fibrosis always occurred together; we did not detect significant hemosiderin and fibrosis without veno-occlusive remodeling. The data from our study suggest that the bleeding in EIPH horses largely derives from regions with vascular remodeling, hemosiderin accumulation, and fibrosis primarily within the dorsocaudal lung field. The descriptions of the histopathology of EIPH in the studies of O'Callaghan et al. and Oikawa, i.e., hemosiderophages and mild fibrosis, are similar to the findings in the lower HS score lung fields in our study; 20, 22 this suggests that the variation between our results and these previous studies reflects the vagaries of sampling the large equine lung.
Clearly at this stage of our investigations, we do not know if the veno-occlusive remodeling is the cause of hemorrhage or a consequence of the accumulation of hemosiderin. One of the possible causes of veno-occlusive remodeling in EIPH lungs is regional pulmonary venous hypertension, primarily within the dorsocaudal lung field. It is known that, in addition to marked increases in pulmonary arterial blood pressure, there is a significant rise in mean pulmonary artery wedge pressure (measure of pulmonary venous pressure when horses experience rapid-onset high intensity exercise as during racing. 12, 13 The normal pulmonary artery wedge pressure in resting horses has been measured at 18 mm Hg; 12 during exercise, the wedge pressure increases to between 56 and 80 mm Hg. 12, 13 In humans, diseases that increase pulmonary venous pressure can result in vascular pathology similar to EIPH and PVOD. 5 Chazova et al. documented veno-occlusive remodeling in a cohort of patients with mitral stenosis and fibrosing mediastinitis. 5 The extent of vascular pathology was more pronounced in PVOD than in mitral stenosis or fibrosing mediastinitis, but shared the essential features of thickening of the intima and adventitia and arterialization of veins. 5 The authors of the study conclude that increased pulmonary venous pressure plays a role in the veno-occlusive remodeling and pathology of PVOD. 5 Pressure increases in veins are capable of eliciting remodeling of the vessel wall. In experimental systems, supra-physiologic (300 mm Hg for 2 minutes) increases in pressure in distended jugular vein grafts results in upregulation of matrix degrading metalloproteinases (MMP-2, MMP-9) and formation of new intima in the affected veins, which occasionally can occlude the vessel. 6
If pulmonary hypertension is responsible for the veno-occlusive remodeling, the distribution of the lesions in EIPH implies that there are particular lung regions experiencing higher pressures, and that the flow is not uniform within the lung. Indeed equine pulmonary blood flow distribution is heterogeneous and becomes more so during exercise with redistribution directed primarily to the caudodorsal lung fields. 9 Not only are there broad regional blood flow distribution patterns within the equine lung, but also micro-environmental perfusion heterogeneity within lung fields that may account for more focal veno-occlusive remodeling. As was shown by Bernard et al., blood flow in the equine lung can vary by as much as 2-fold within very small regions of lung. 1
Once the initial veno-occlusive lesion develops, either as a consequence of an initial bleed of as yet unknown etiology or as a consequence of focal venous distension, a vicious cycle may then develop as a consequence of vascular congestion upstream from the occluded veins. The resulting capillary distension likely causes stress failure and focal hemorrhage that gives rise to hemosiderin accumulation and its associated features of fibrosis and bronchial neovascularization, and perhaps further venous remodeling. The numerous small arterioles detected within the EIPH lungs were only present within the high HS lung sites, along with the veno-occlusive remodeling, hemosiderin, and fibrosis. These vessels likely represent foci of bronchial arterial neovascularization, as was reported by O'Callaghan et al. 18– 20 The bronchial circulation is the primary source for significant vascular proliferation in the lung. 4 Most of the neovascularization was adjacent to the pleura and septa; these vessels likely arose from branches of the bronchial circulation that run within the pleura and interlobular septa of the horse. 14 In the horse, the bronchial circulation drains into the pulmonary circulation; 14 thus this increase in the bronchial vascular bed could increase the volume of blood within the pulmonary capillaries and exacerbate the bleeding from the capillaries, or directly bleed into the lung.
In summary, we have identified heretofore unreported pulmonary venous remodeling within the lungs of EIPH horses. As yet, we do not know if the remodeling is primary and the cause of EIPH or is simply a consequence of an initial bleed into the lung. However, once it develops, regional veno-occlusive remodeling will lead to regional vascular congestion and hemorrhage, with subsequent accumulation of hemosiderin, fibrosis, and bronchial angiogenesis.
Footnotes
Acknowledgements
We wish to thank Dr. Deryck Tan Yang, Donnelle Glen, and Nordiana bte Kasmuri at the Singapore Turf Club for collecting and shipping the samples from Singapore to Michigan State University. They would also like to acknowledge Lisa Bartner for providing the diagram of the equine lung in Figures 1 and ![]() .
.
