Abstract
From 2002 to 2007, 23 ferrets from Europe and the United States were diagnosed with systemic pyogranulomatous inflammation resembling feline infectious peritonitis (FIP). The average age at the time of diagnosis was 11 months. The disease was progressive in all cases, and average duration of clinical illness was 67 days. Common clinical findings were anorexia, weight loss, diarrhea, and large, palpable intra-abdominal masses; less frequent findings included hind limb paresis, central nervous system signs, vomiting, and dyspnea. Frequent hematologic findings were mild anemia, thrombocytopenia, and hypergammaglobulinemia. Grossly, whitish nodules were found in numerous tissues, most frequently the mesenteric adipose tissue and lymph nodes, visceral peritoneum, liver, kidneys, spleen, and lungs. One ferret had a serous abdominal effusion. Microscopically, pyogranulomatous inflammation involved especially the visceral peritoneum, mesenteric adipose tissue, liver, lungs, kidneys, lymph nodes, spleen, pancreas, adrenal glands, and/or blood vessels. Immunohistochemically, all cases were positive for coronavirus antigen using monoclonal antibody FIPV3-70. Electron microscopic examination of inflammatory lesions identified particles with coronavirus morphology in the cytoplasm of macrophages. Partial sequencing of the coronavirus spike gene obtained from frozen tissue indicates that the virus is related to ferret enteric coronavirus.
The genus Coronavirus comprises three serogroups of viruses that can infect many mammalian and avian species. Group 1 viruses include feline coronavirus (FCoV), canine coronavirus (CCV), transmissible gastroenteritis virus (TGEV) of swine, and human coronavirus 229E. Group 2 coronaviruses include bovine coronavirus and mouse hepatitis virus. Group 3 virus includes avian infectious bronchitis virus and its variants. 25 In domestic carnivores, two major clinical presentations can be observed with coronaviral infections. One is self-limiting infection, such as CCV 21, 27 and feline coronaviral enteritis, caused by FCoV. 1 The other one is a severe systemic disease, feline infectious peritonitis (FIP), also caused by FCoV. 25 Recently, in dogs, severe visceral disease associated with a variant of CCV 4 and fatal enteric disease due to CCV 7 have also been recognized. An enteric coronavirus serologically related to TGE and associated with preweanling diarrhea has been described in mink. 14
Two diseases caused by coronaviruses have been described in ferrets: severe acute respiratory syndrome (SARS) 19 and epizootic catarrhal enteritis (ECE). 30, 31 SARS coronavirus has been experimentally transmitted to ferrets, although, to the authors' knowledge, no natural infections have been reported in this species. Virus was isolated from the respiratory, urinary, and gastrointestinal tracts, and was associated with pulmonary lesions, but no damage to other internal organs was reported. 30, 31
Epizootic catarrhal enteritis was first described in 1993 associated with diarrhea in young and adult ferrets. The causative agent was found to be a coronavirus, 30, 31 now referred to as ferret enteric coronavirus (FECV). 31 No lesions other than those affecting the gastrointestinal tract have been described, and FECV could be detected in saliva, feces, and enterocytes, but not in serum, spleen, or lymph nodes. 31 Recent studies have established that FECV is most closely related to FCoV. 31 Recently, a disease has been recognized in ferrets with gross, histologic, and immunohistochemical features that are very similar, if not identical, to FIP. 15, 20 For the purposes of this manuscript, this disease is herein referred to as ferret systemic coronavirus infection (FSCV), and this report describes this condition in 23 ferrets.
Materials and Methods
Animals
Criteria for inclusion in the study included the presence of histologic changes typical of FSCV, absence of other infectious agents based on special stains, and immunohistochemical demonstration of coronavirus antigen in lesions. Lesions typical of FSCV included pyogranulomatous inflammation and necrosis with or without perivasculitis and vasculitis, occurring in a variety of abdominal and visceral organs. All data regarding signalment, history, blood and serum chemistry values, serum protein electrophoresis, serology for Aleutian disease virus (ADV) and FCoV, and gross and histologic lesions were included when available.
Histopathology
All tissues obtained by biopsy or at necropsy were preserved in 10% neutral-buffered formalin for up to 5 days prior to being processed routinely. Tissues were sectioned at 5 μm, mounted on frosted glass slides, and stained with hematoxylin and eosin (HE). For all cases, at least one tissue section containing the lesion was stained by Fite acid-fast (AF) technique. Sections from select cases also were stained with Brown and Brenn (B&B), Gomori methylamine silver (GMS), and Warthin Starry (WS) techniques.
Immunohistochemistry
Immunohistochemical analyses were performed using anti-FCoV monoclonal antibody FIPV3-70 as the primary antibody (Custom Monoclonals International, Sacramento, CA) and a streptavidin-biotin procedure using an automated immunostainer (Dako Autostainer Universal Staining System; Dako, Carpinteria, CA) as previously described, 3 with some modifications. Briefly, sections were deparaffinized in xylene and graded ethanols and rinsed in Tris-buffered saline (TBS [0.05 M Tris-HCl, 0.15 M NaCl, pH 7.6]; Dako TBS; Dako). Prestaining heat-induced antigen retrieval consisted of steam heating sections for 30 minutes in high-pH buffer (Target Retrieval Solution, pH 10; Dako) using a standard vegetable steamer.
Following antigen retrieval, all incubations were performed at room temperature. Blocking steps included incubation for 15 minutes with 3% H2O2 in methanol to block endogenous peroxidase, and 5 minutes with 5% normal goat serum (Dako). Critical immunostaining steps occurring with the autostainer included incubation of sections with anti-FIP primary antibody (FIPV3-70), biotinylated linker antibody, detection reagent, and chromagen interceded by washes with TBS buffer containing 0.2% Tween-20. Monoclonal antibody FIPV3-70 was used at a concentration of 6 μg/ml for 30 minutes. Biotinylated goat anti-mouse/rabbit IgG linker reagent (Covance Research Products, Berkeley, CA) and horseradish peroxidase–conjugated streptavidin-biotin complex detection reagent (Ultra Streptavidin, Covance Research Products, Berkeley, CA) were incubated on sections for 30 minutes each. The 3-amino-9-ethyl-carbazole (AEC) chromogen (Dako) was incubated on sections twice for 4 minutes each. Following AEC incubation, sections were rinsed with deionized water, counterstained manually with Mayer hematoxylin, coverslipped with aqueous mounting medium, and examined with a light microscope.
Positive control tissue consisting of liver and lymph node from a cat with confirmed FIP based upon histopathology and immunohistochemistry was included in each run to confirm immunoreactivity of the appropriate pattern (cytoplasm of macrophages within pyogranulomatous infiltrates). Negative antibody control, consisting of an irrelevant isotype-matched primary antibody (anti–Babesia bovis Mab 23/28.57), was reacted with each test slide to ensure the lack of nonspecific binding by linker or signal amplification reagents to tissue sections. Slides classified as FCoV positive showed immunoreactivity in a pattern consistent with positive control slides and previous publications. 17 Slides classified as FCoV negative showed no specific immunoreactivity. Additionally, multiple cases of granulomatous disease attributed to mycobacteriosis or nocardiosis based on special stains or cultures were stained by this technique.
Electron microscopy
Formalin-fixed mesenteric tissue containing granulomatous lesions from case 4 was subsequently retrimmed and placed into modified (half-strength) Karnovsky fixative. 17 Following Karnovsky immersion, tissues were further postfixed in 2% osmium tetroxide reduced with 2.5% potassium ferrocyanide. 26 Following osmification, ferret tissue was rinsed in 0.2 M sodium cacodylate, dehydrated through a graded ethanol series, transitioned through propylene oxide, and infiltrated and embedded in Eponate-12 epoxy formulation (Eponate-12; Ted Pella Inc., Redding, CA). Thick sections were cut, mounted on glass slides, stained by Toluidine blue O, and examined by light microscopy. Thin sections were mounted on bare 150-mesh copper grids, stained in 4% uranyl acetate in 75% ethanol followed by poststaining in Reynold lead citrate, 24 and examined in a Zeiss 906E transmission electron microscope at 60 kV accelerating voltage (Carl Zeiss SMT, Peabody, MA).
Virus isolation
Selected fresh-frozen tissue samples from 2 ferrets with confirmed FSCV were homogenized in both sterile minimal essential medium (MEM) with antibiotics and a 10% solution inoculated onto 4 cell lines: Crandell feline kidney (CrFK), Madin-Darby canine kidney (MDCK), Vero cells, and rabbit kidney (RK-13b). The cells were passaged at weekly intervals for 4 passages.
Detection of coronavirus in ferret tissues by reverse transcription–polymerase chain reaction (RT-PCR)
Total RNA was extracted using the RNeasy Mini Kit (Qiagen, Valencia, CA) following the manufacturer's protocol. The samples were initially tested with an SYBR Green real-time consensus RT-PCR assay that broadly detects the group 1 animal coronaviruses FCoV, CCV, and TGEV of swine. The assay was developed at the Diagnostic Center for Population and Animal Health at Michigan State University (East Lansing, MI) for routine diagnostic testing. The primer sequences (forward primer: 5′-GGTCATCGCGCTGTCTACTCT-3′, and reverse primer: 5′-GCTCGTCATAGCGGATCTTTA-3′, with nucleotide positions 29022–29042 and 29170–29150 [minus strand], respectively, in the 3′ untranslated region of FCoV strain C1Je; GenBank accession no. DQ848678) are conserved among FCoV, CCV, and TGEV strains. The real-time RT-PCR was performed with the QuantiTect SYBR Green RT-PCR kit from Qiagen with a final primer concentration of 0.5 μM in a 50-μl reaction volume. The assay was run in the iCycler iQ System (Bio-Rad Laboratories, Hercules, CA). Cycling conditions were: reverse transcription of 50°C for 30 minutes followed by 95°C for 15 minutes, then 40 cycles of 94°C for 30 seconds, 53°C for 30 seconds, and 72°C for 30 seconds. A post-PCR melt curve analysis was incorporated in the run to determine product specificity. A melting temperature peak at approximately 78°C was expected for a positive sample. The assay was also performed in real-time SYBR Green format under the same conditions as described above. A melting temperature peak at approximately 79°C was expected for a positive sample. Third, degenerate consensus primers that will amplify a portion of the spike gene of any coronavirus 28 were used as previously described. 30
Sequencing and sequence analyses
PCR products were purified from agarose gels using the QIAquick Gel Extraction Kit (Qiagen). Purified amplicons were sequenced bidirectionally at the Research Technology Support Facility at Michigan State University (East Lansing, MI). Sequence assembly and analyses were done with the Lasergene biocomputing software (DNASTAR Inc., Madison, WI). Sequence data were subjected to BLAST analysis. 2
Results
Animals
A total of 23 ferrets were included in the study based on histologic findings and results of immunohistochemistry. Table 1 summarizes the signalments, history, clinical signs, and pertinent blood values.
Signalment, history, and blood values of ferrets with FIP-like disease.∗
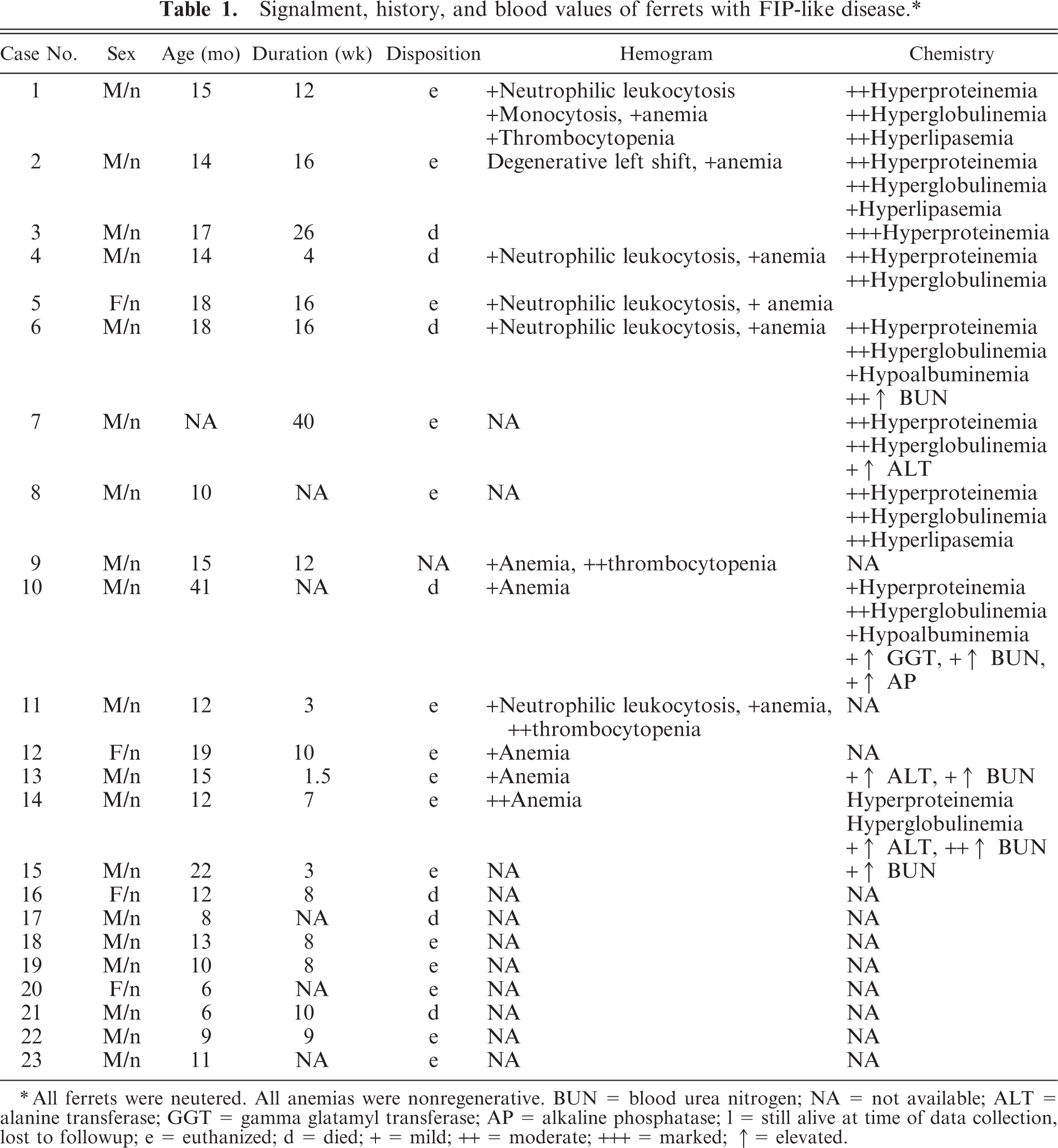
All ferrets were neutered. All anemias were nonregenerative. BUN = blood urea nitrogen; NA = not available; ALT = alanine transferase; GGT = gamma glatamyl transferase; AP = alkaline phosphatase; 1 = still alive at time of data collection, lost to followup; e = euthanized; d = died; + = mild; ++ = moderate; +++ = marked; ↑ = elevated.
All cases were submitted between the years 2002 and 2007 from private veterinary practices throughout the United States (11 cases) and Spain (12 cases). A total of 18 were male, and 5 were female. All ferrets were neutered. Ages at onset of disease ranged from 2 to 36 months, and average age was 11 months. All ferrets were in private pet homes at the time they became ill, except one ferret, which lived in a ferret shelter. A total of 11 ferrets had been housed with other ferrets that were clinically healthy, and 12 ferrets did not live with other ferrets. Three ferrets lived in the same household with at least 1 cat. Two ferrets lived in households with at least 1 dog. Five ferrets lived in households with no other pets. Other pet information was not known for 13 ferrets. Eleven ferrets had been vaccinated at least once against canine distemper virus, and vaccine history was not known for the other ferrets. The disease was progressive in all cases; the duration of clinical disease ranged from 1 to 195 days, and the average duration was 69 days. The duration of illness was not known for 3 ferrets. Clinical signs included weight loss (21), palpable intra-abdominal mass or masses (16), lethargy (12), anorexia (11), thin body condition or emaciation (8), vomiting (8), splenomegaly (8), decreased consumption of water (7), dehydration (5), sneezing (5), bruxism (4), renomegaly (4), nasal discharge (3), systolic murmur (3), greenish urine (2), labored breathing (2), peripheral lymphadenomegaly (2), reddened rectal mucosa (2), and rectal prolapse (1). Signs of central nervous system disease were seen in 12 ferrets and included acute or progressive hind limb paresis or paraparesis (6), ataxia (2), seizures (2), pallor (2), and one each of wide hind-end stance, opistothonus, abnormal gait, and proprioceptive deficits. Seven ferrets were pyrexic, and fevers ranged from 39.4°C to 40.8°C. A total of 7 ferrets died, 15 were euthanized, and 1 was still alive at the time of the study.
Table 1 summarizes abnormalities in hemograms and serum chemistry anylates, compared with published values. 9 Mature neutrophilic leukocytosis was noted on at least one occasion (4), and on two occasions (3). The leukocyte count was within normal limits for ferrets on at least one occasion (7), and on two occasions (2). Mild nonregenerative anemia was noted in 11 ferrets on one occasion and in 6 ferrets on two occasions. Thrombocytopenia was noted in 3 ferrets and varied from mild (1) to severe (2). Platelet counts were not reported for 10 ferrets, and hemograms were not known for 6 ferrets. Serum chemistry profiles were available at least once for 13 ferrets. Important abnormalities included hyperproteinemia (n = 12; 6.9–13.4 g/dl) and hyperglobulinemia (n = 9; 6.0–8.4 g/dl). Globulin levels were not available for 4 ferrets. Additional abnormalities included elevations in serum lipase (3), blood urea nitrogen (2), serum alanine transferase (2), alkaline phosphatase (1), and serum gamma glutamyl transferase (1). Serum protein counterelectrophoresis was negative for exposure to Aleutian disease virus in all 8 ferrets tested. Testing for FIP was performed on two ferrets and was negative by serum enzyme-linked immunosorbent assay (ELISA; n = 1) and PCR (1).
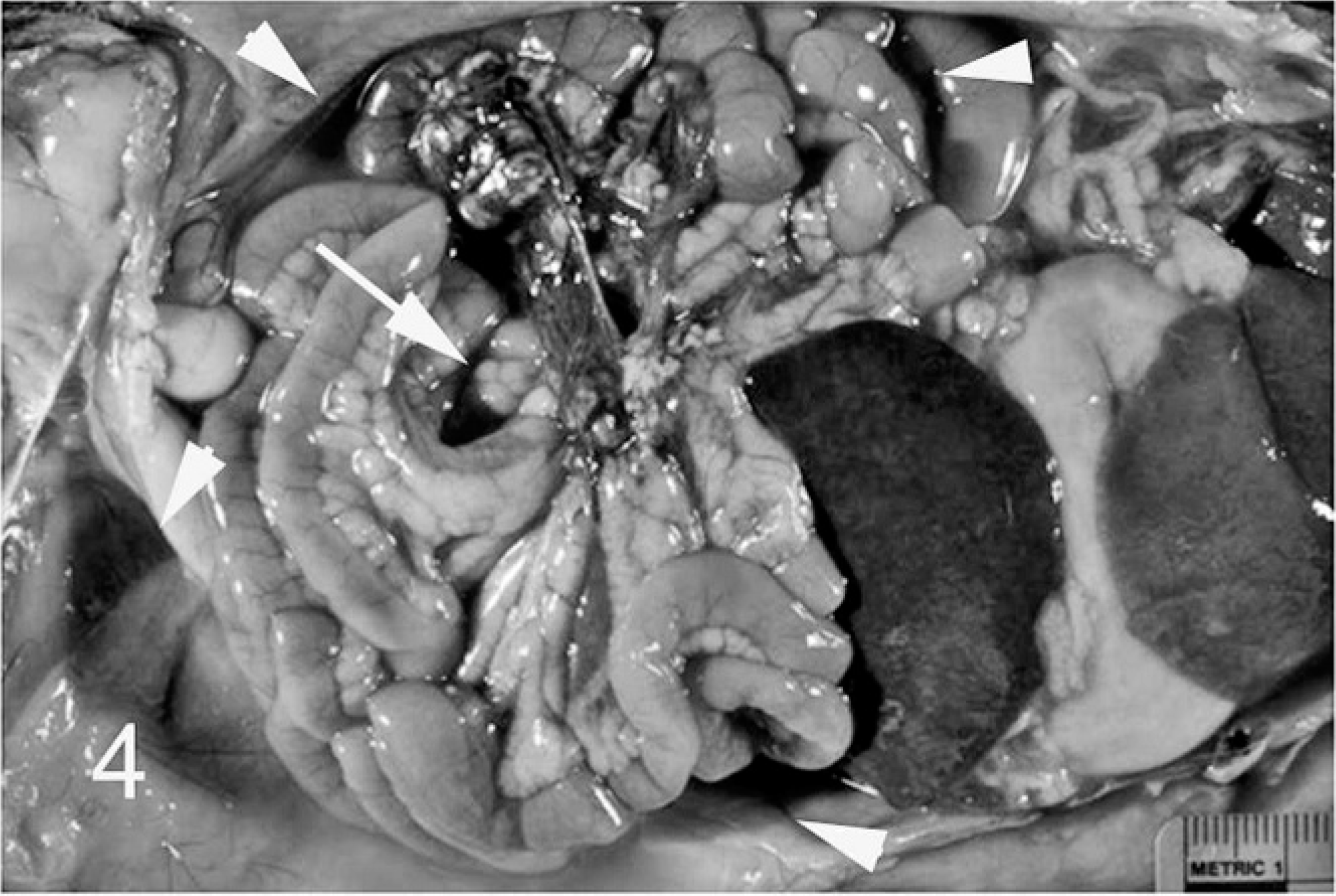
Serum protein electrophoretograms were available for 8 ferrets, and they revealed polyclonal gammopathy (7). Urinalysis was performed for 4 ferrets. Noted abnormalities included greenish urine (2), proteinuria (2), and blood and rare bilirubin crystals (1). Gross lesions detected during biopsy or necropsy were reported for 7 ferrets. The primary gross lesion was circumscribed to coalescing white, tan, or slightly pink irregular nodules or foci of white discoloration ranging from 5 to 30 mm in greatest dimension on the surface and within the parenchyma of spleen (4), liver (2), kidneys (1), lung (2), mesentery (2), and lymph nodes (n = 4; Figs. 1 2 3 4) Also noted were splenomegaly (4), renomegaly (3), hepatomegaly (1), and ascites (1). Serum counterimmune electrophoresis (CEP) for Aleutian disease virus (ADV) was performed for 8 ferrets and was negative in all cases.
Histopathology
The typical histologic lesion for this condition was severe pyogranulomatous inflammation within affected tissues. Inflammation varied from regionally diffuse pyogranulomatous inflammation to organized microgranulomas, and it frequently involved the adventitial and medial tunics of small veins and venules. Inflammation consisted predominantly of neutrophils and macrophages, with fewer lymphocytes and plasma cells and rare multinucleated giant cells. Inflammation was sometimes associated varying degrees of necrosis and with scant fibrin deposition (Figs. 5 6 7 8). Histologic lesions were detected in the mesentery/peritoneum (13), lymph nodes (12), spleen (10), kidneys (10), liver (7), lung (5), intestine (4), pancreas (3), stomach (2), brain (1), and adrenal gland (1). Other notable lesions included nonsuppurative meningoencephalitis (5) and suppurative or nonsuppurative tubulointerstitial nephritis (4). No organisms were detected in the lesions using AF, B&B, GMS, and WS staining techniques.
Immunohistochemistry
Positive staining for coronavirus antigen was detected in all cases and in all foci of pyogranulomatous inflammation in which the stain was applied, although staining reaction was most prominent in the lymph node and splenic lymphoid follicles and inflammatory lesions. The positive reaction was apparent within foci of necrosis and in the cytoplasm of macrophages and unidentified cells (Fig. 9). Ferrets with granulomatous inflammation due to other causes did not have a positive staining reaction for coronavirus antigen.
Electron microscopy
Electron microscopic examination of inflammatory foci revealed macrophages that frequently contained multiple intracytoplasmic particles resembling virions, both within cytoplasmic membrane-bound vacuoles and free in the cytoplasm (Fig. 10). Enveloped particles were somewhat pleomorphic and spherical, 70–140 nm in diameter, and frequently had a central lucent zone 20–30 nm in diameter. Rarely, these structures had roughly circumferential spikes on the outer wall. Nonenveloped particles were 40–50 nm and did not have a lucent zone.
Virology
No cytopathic effect has been noted to date. Further passage and probing for noncytopathogenic coronaviruses will be conducted using a coronavirus PCR.
PCR and sequencing
The ferret samples were found to be negative for FCoV, CCV, and TGEV by consensus RT-PCRs specific for these three group 1 coronaviral agents. Tissues from the first case examined were also found to be negative for FECV. However, another case examined with the FECV-specific RT-PCR yielded faint bands of the expected size for some of the tissues. The degenerate set of consensus primers, designed to detect a portion of the spike gene of any coronavirus, amplified a product of the expected size in both cases (Fig. 11). A unique 599-bp sequence (excluding primer sequences) was obtained, and BLAST analysis showed significant similarity between the ferret-derived sequence and group 1 coronavirus spike gene sequences. Alignment of the deduced partial spike amino acid sequence (199 residues) to corresponding sequences of known group 1 coronaviruses showed 71% to 73% sequence identities to FCoV, TGEV, and CCV, 77% sequence similarity to FECV, and 51% and 55% similarities to porcine epidemic diarrhea virus and human coronavirus 229E, respectively.
Discussion
Visceral disease caused by mutated coronaviruses that closely resemble host enteric coronaviruses has been recognized for some time in cats as FIP, and a similar syndrome has recently been recognized in dogs. 4 Pathogenic visceral coronavirus infections related to viral mutations also occur in mice, 13 pigs, 18 and humans. 11 Thus, it is not surprising that a visceral disease associated with a coronavirus similar to FECV now has been recognized in the domestic ferret, and clinicopathologic features of this condition very closely resemble those of FIP.
Juvenile and young adult ferrets are most susceptible to developing FSCV infection, and a sex predilection is not apparent. In our study, males were more prevalent, but this may reflect owner preference or coincidence more than a true predisposition, since several females also were affected. Most cats that contract FIP are young, and there is no sex predilection. 12 All affected ferrets were from indoor environments with or without exposure to dogs, cats or other ferrets. Although more common in catteries or multiple cat households, FIP does occur frequently in single-cat households. 12 Indoor cats are more prone to FIP, presumably because this environment facilitates repeated oral exposure to fecal virus in litter boxes. 12 Fecal-oral exposure is the likely route of transmission for FECV and, as with FCoV, persistent infection may facilitate FSCV infection. 12 The clinical course of this disease was progressive and unresponsive to therapy, and duration of the disease course was highly variable and may have been longer than stated for those animals that were euthanized. All but one ferret had died of the disease or was euthanized at the time of this writing, and it is possible that this disease is invariably fatal, as is FIP. 12
As with FIP, no pathognomonic clinical signs were seen in ferrets with FSCV. In general, the clinical signs seen in the ferrets were attributed to morbidity or organ damage associated with the inflammatory cell infiltrates. The most common signs were similar to those of cats with FIP and included weight loss, lethargy, anorexia, diarrhea, and the presence of palpable intra-abdominal masses. 12, 17 Some cats with FIP have a fever, 12 and fever also was observed in several of the ferrets in our study for which body temperature was recorded.
Diagnostic abnormalities were not identified in hemograms from affected ferrets. Trends observed in the hemogram values, although not diagnostic, mirror those that may be encountered with FIP. These include normal leukogram, mature neutrophilic leukocytosis, mild regenerative or nonregenerative anemia, and thrombocytopenia. 12 In FIP, the leukogram values likely reflect peripheral demand for leukocytes associated with inflammation. Erythrograms reflect anemia of chronic disease when nonregenerative, and possibly reflect malabsorption of vitamin B12 due to inflammation in the gut in cases of regenerative anemia. Thrombocytopenia is attributed to platelet consumption associated with disseminated intravascular coagulation due to vasculitis. 12 It is possible that similar mechanisms exist for the hemogram abnormalities in FSCV. High fever and marked neutrophilic leukocytosis are features of myofasciitis (MF), a recently described condition of domestic ferrets believed to be an immune-mediated disorder; however, although some overlap exists, generally the fevers and leukograms have much higher values in MF patients. Additionally, morphologic features and distribution of lesions in MF differ considerably from those of FSCV. 10
The most consistent serum chemistry abnormality in FSCV was hyperproteinemia, attributed to hyperglobulinemia, and this also is the most consistent chemistry abnormality with FIP. Serum electropherograms were performed on 8 of the ferrets, and in 7 cases the hyperglobulinemia was due to polyclonal gammopathy, as seen with most cases of FIP. 12 Polyclonal hypergammaglobulinemia also has been observed in ferrets with ADV infection, 22, 23 but CEP for ADV was negative in all tested cases, and the microscopic lesions did not resemble those of ADV infection in ferrets, the latter being primarily a plasmacytic inflammatory cell infiltrate. 22, 23 Serologic tests for FIP using ELISA and PCR techniques were performed on 5 ferrets and were negative in all cases. Evaluation of serum antibody titers for the diagnosis of FIP is a highly controversial topic, and the significance of antibody titers is not well understood. 12 Titers can be positive in cats without clinical disease and can be negative in cats that die of FIP. 12 Currently, there is no serologic test for detecting antibodies to FSCV. Therefore, the significance and applicability of serologic tests for FIP in ferrets with FSCV are questionable.
Urinalyses were performed on only 4 ferrets, and few values were observed that may be relevant to FSCV infection. Greenish urine, proteinuria, and detection of blood and bilirubin crystals may reflect renal parenchymal damage due to inflammation, or protein leakage through glomeruli with membranoproliferative changes. Interestingly, the hyperbilirubinemia and icterus sometimes associated with FIP was not a feature of FSCV, so spillover of bilirubin into the urine due to hyperbilirubinemia seems unlikely. The hyperbilirubinemia seen with FIP is not well understood, and it has been attributed to hepatic necrosis, hemolysis, and compromised metabolism and excretion of bilirubin. 1 Based on the morphologic similarities of these two diseases, it seems likely that at least some cases of FSCV would be associated with hyperbilirubinemia and icterus, and the cause for this difference in presentations is not known.
The most strikingly similar comparisons of FSCV to FIP are in the appearance of the gross and histologic lesions. The widespread nodular foci on serosal surfaces and within the parenchyma of the abdominal and thoracic viscera, as well as the nodular enlargement of the mesenteric lymph nodes very closely resemble the “dry form” of FIP. 12, 29 To date, ferrets with FSCV reportedly do not consistently have serous effusions in the body cavities that are typically seen in the “wet form” of FIP, and only 1 ferret in this report had such an effusion; however, these two forms are somewhat arbitrary and are generally both present to varying degrees at any time in the disease course of FIP. 12 It is possible that ferrets are not being diagnosed when effusions are more prominent in the disease course or that the ferret disease is simply less effusive. With increased awareness of this disease in ferrets, it is possible that effusive forms may be reported more frequently.
Histologic changes in ferrets with FSCV are indistinguishable from those of cats with FIP, which initially inspired investigators to examine the role of coronaviruses in the pathogenesis of the disease. Perivasculitis and vasculitis, predominantly neutrophilic or pyogranulomatous, are hallmark features of the FIP, as are the solid foci of pyogranulomatous inflammation that develop around or adjacent to affected vessels. 17, 29 Granulomatous inflammation also can occur in the uvea and central nervous system of cats with FIP. 12 In none of the submitted cases was eye or spinal cord examined histologically. Although microscopic examination of the brain was limited to 3 cases, the inflammatory lesions were detected in this tissue. It is likely that similar ocular and central nervous system manifestations occur in ferrets with FSCV as occur in cats with FIP, and the central nervous system signs noted in ferrets from this study may have been due to this.
Positive immunohistochemistry for feline coronavirus antigen in lesions was a requirement for inclusion in the study. Antigen was detected in macrophages and in extracellular debris associated with the lesions, as is typical of antigenic distribution with FIP. 17 As with FIP, 12 a positive reaction is considered diagnostic. In the authors' practice, a negative immunohistochemistry result warrants a search using cytochemical stains for other pathogens in the lesions. The foci of granulomatous or pyogranulomatous inflammation particularly resemble mycobacteriosis or nocardiosis, and acid-fast staining was performed in all the study cases to eliminate these organisms as contributing causes of the lesions.
Electron microscopic examination of inflammatory foci identified pleomorphic particles within the cytoplasm of macrophages that morphologically were consistent with coronavirus virions. Enveloped coronavirus particles are spherical, 70–140 μm in diameter, develop within the cytoplasm, and derive their envelope and peplomers from intracytoplasmic organelles, including endoplasmic reticulum, Golgi, and lysozomes. 5, 6 Particles were frequently observed within cytoplasmic vacuoles, most of which resembled lysosomes, but because many cells were degenerative and fixation was not optimal, it was difficult or not possible to determine the origin of many vacuoles containing virions. Naked virions also were seen in the cytoplasm of necrotic cells, in which cellular organelles may have been too degenerative to support full maturation of the virions. The characteristic peplomer spikes on the surface of the virion envelope were seen only rarely, likely due to cellular necrosis or suboptimal tissue fixation.
Viral culture attempts have thus far been unsuccessful for the two cases in which frozen tissues were available for this purpose. The coronaviruses are fastidious by nature and can be difficult to culture in vitro. 8, 27 Other detection methods, such as fluorescent antibody and immunohistochemistry for specific antigen detection, electron microscopy, and polymerase chain reaction (PCR) for specific viral sequence detection, have been used as alternative detection tools. 12 The ferret coronavirus was not readily cultured after multiple passages in established cell lines, such as CrFK, MDCK, RK-13, and Vero cells. Further attempts will be made using ferret-derived cell lines and culture conditions that optimize coronavirus culture, such as use of trypsin or pancreatin in the growth media. 27
Direct sequencing of PCR products obtained from frozen tissue extracts using primers specific for FECV and a generic primer pair for the coronavirus spike gene confirmed the presence of a coronavirus in the affected tissues. Based upon these still-limited sequencing data, the virus associated with the lesions described in this paper does not appear to be a feline coronavirus. The virus present in the samples tested also was not identical to the recently described ferret enteric coronavirus, FECV-MSU1, 31 but appears to be most closely related to it by phylogenetic analysis (data not shown). The observed slight cross-reactivity of some samples with the FECV-specific primers may indicate a higher degree of sequence conservation between the nucleocapsid genes of the systemic ferret coronavirus and FECV. Further genomic sequencing will be required to more definitively characterize this systemic ferret coronavirus. The relatively recent recognition of this disease in pet ferrets suggests the occurence of a recent mutation or shift in the FECV that results in this disease, similar to the mutations that occur in FCoV preceding the development of FIP.
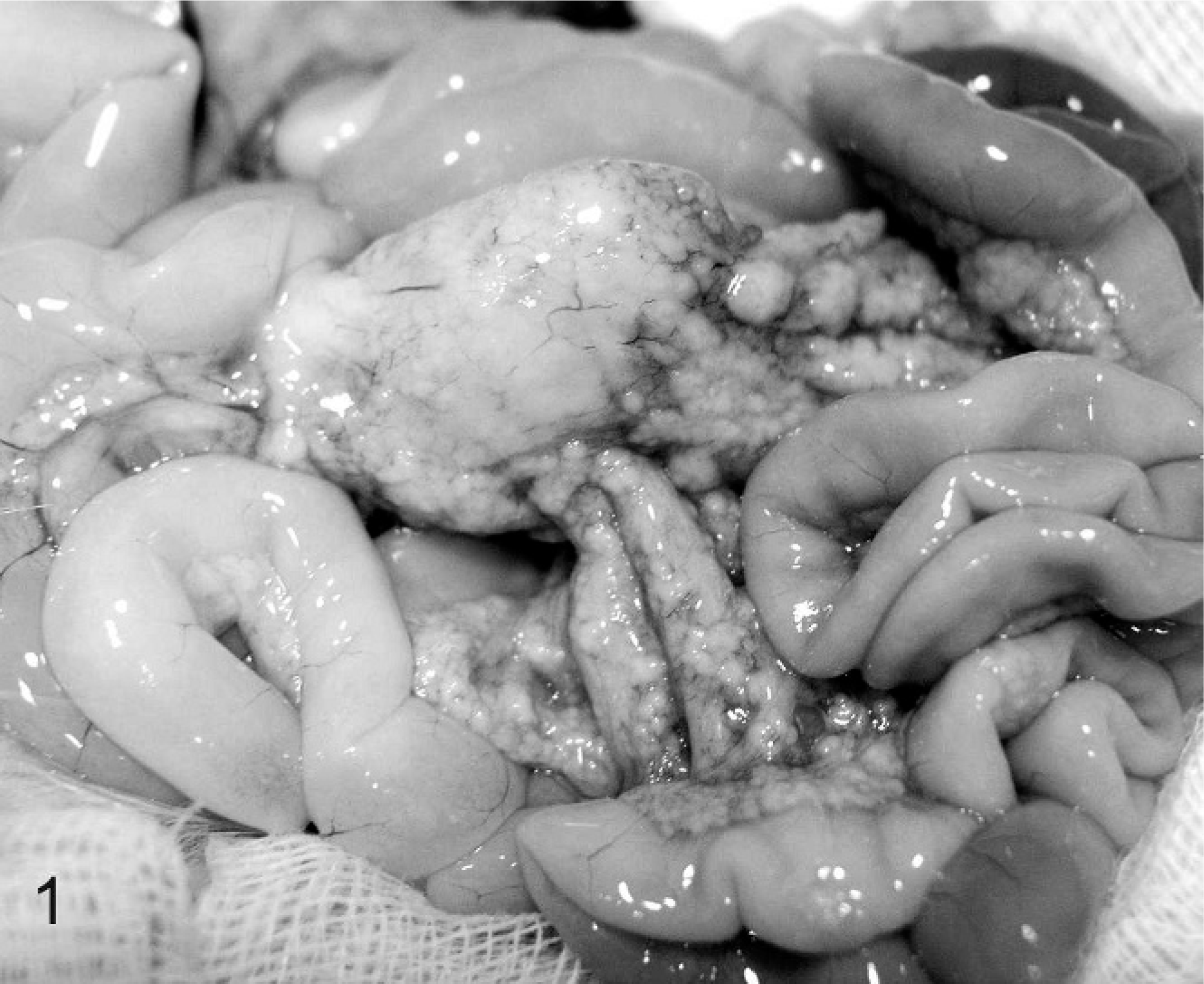
Ferret, coronavirus-associated mesenteritis. Note coalescing pale nodular masses of varying size distributed throughout the mesentery.
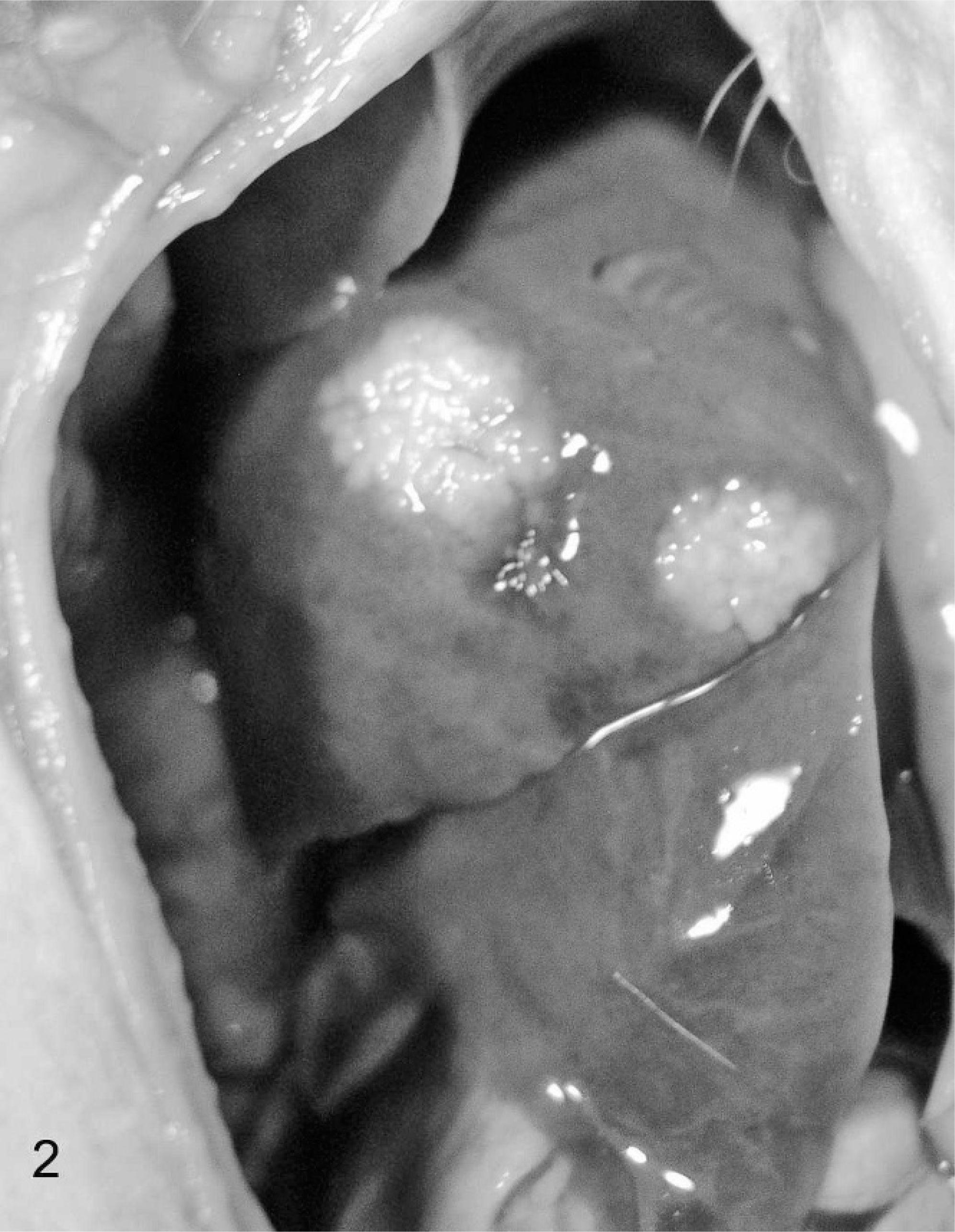
Ferret, coronavirus-associated hepatitis. Note two large, roughly circumscribed, pale, nodular foci with the hepatic parenchyma.
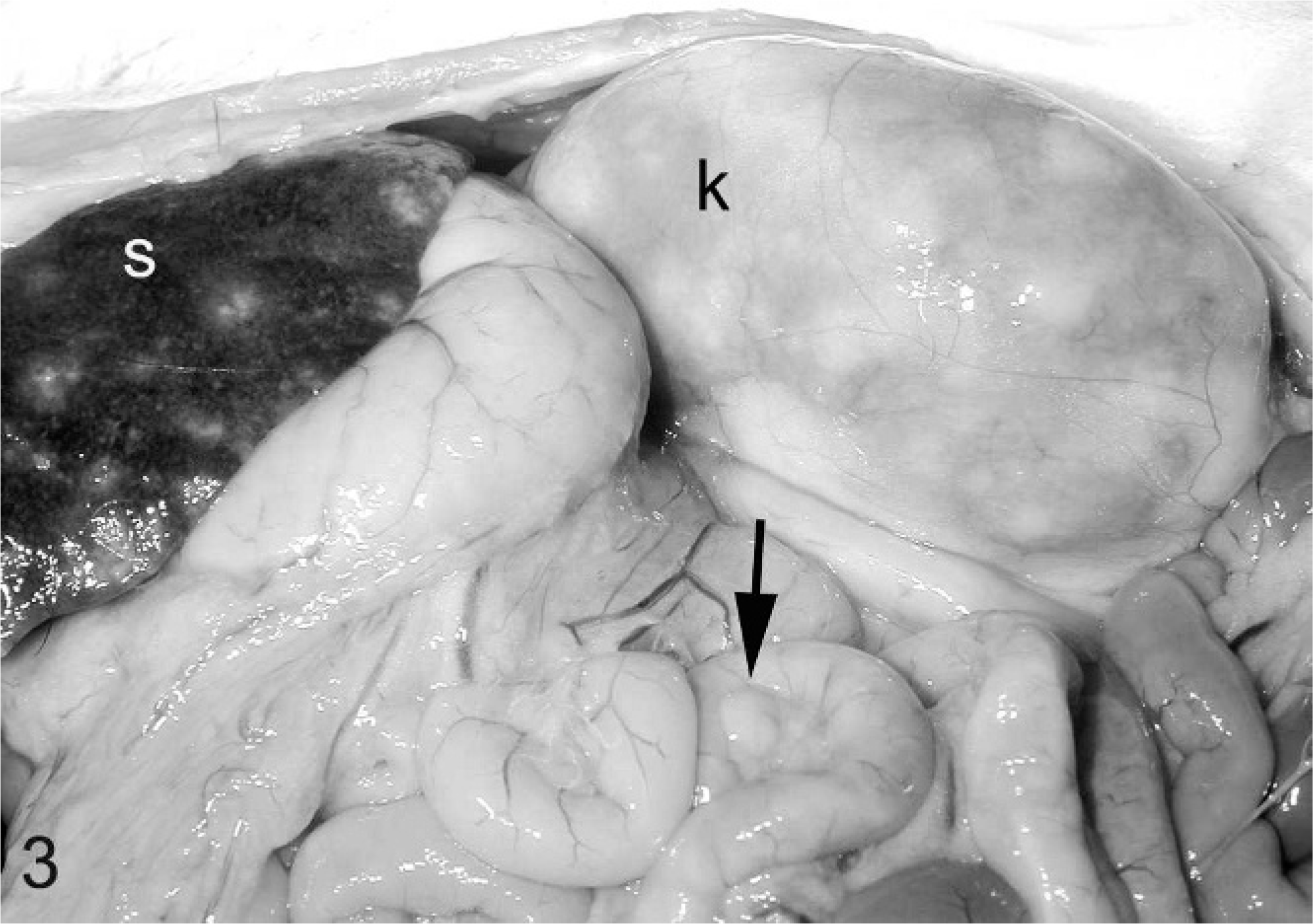
Ferret, abdominal viscera, coronavirus-associated lesions. Note nodular pale foci in spleen (s) and kidney (k). Also note mild mesenteric lymphadenomegaly (arrow) and nephromegaly.
Ferret, coronavirus-associated disease. Note intraabdominal effusion (arrowheads) and pale nodular masses in the mesentery (arrow). Photo courtesy of Dr. Margaret Miller.
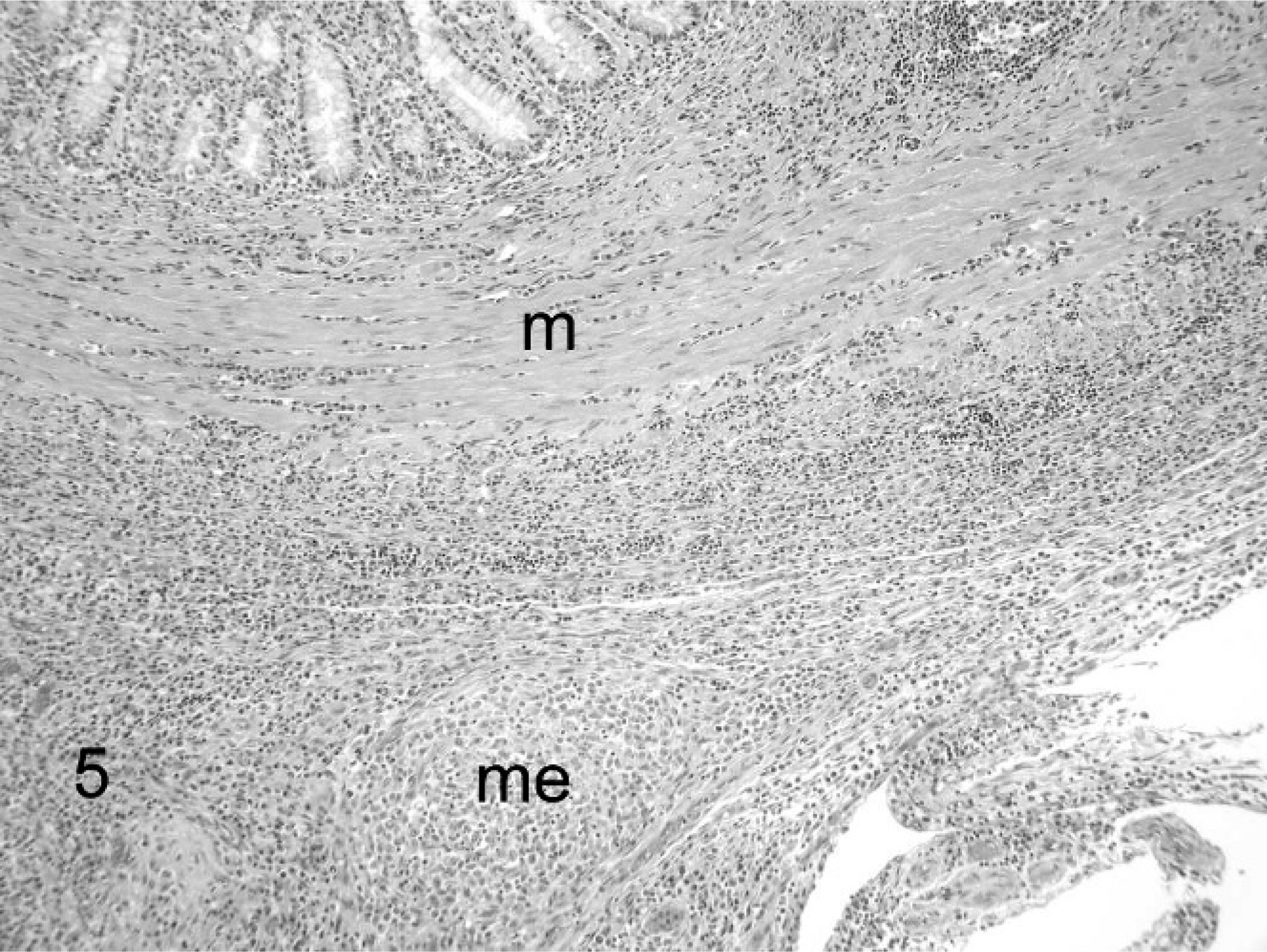
Ferret, coronavirus-associated disease. Low-magnification image illustrates coalescing foci of granulomatous inflammation in the muscular tunics of the duodenum (m) and adjacent mesentery (me). HE.
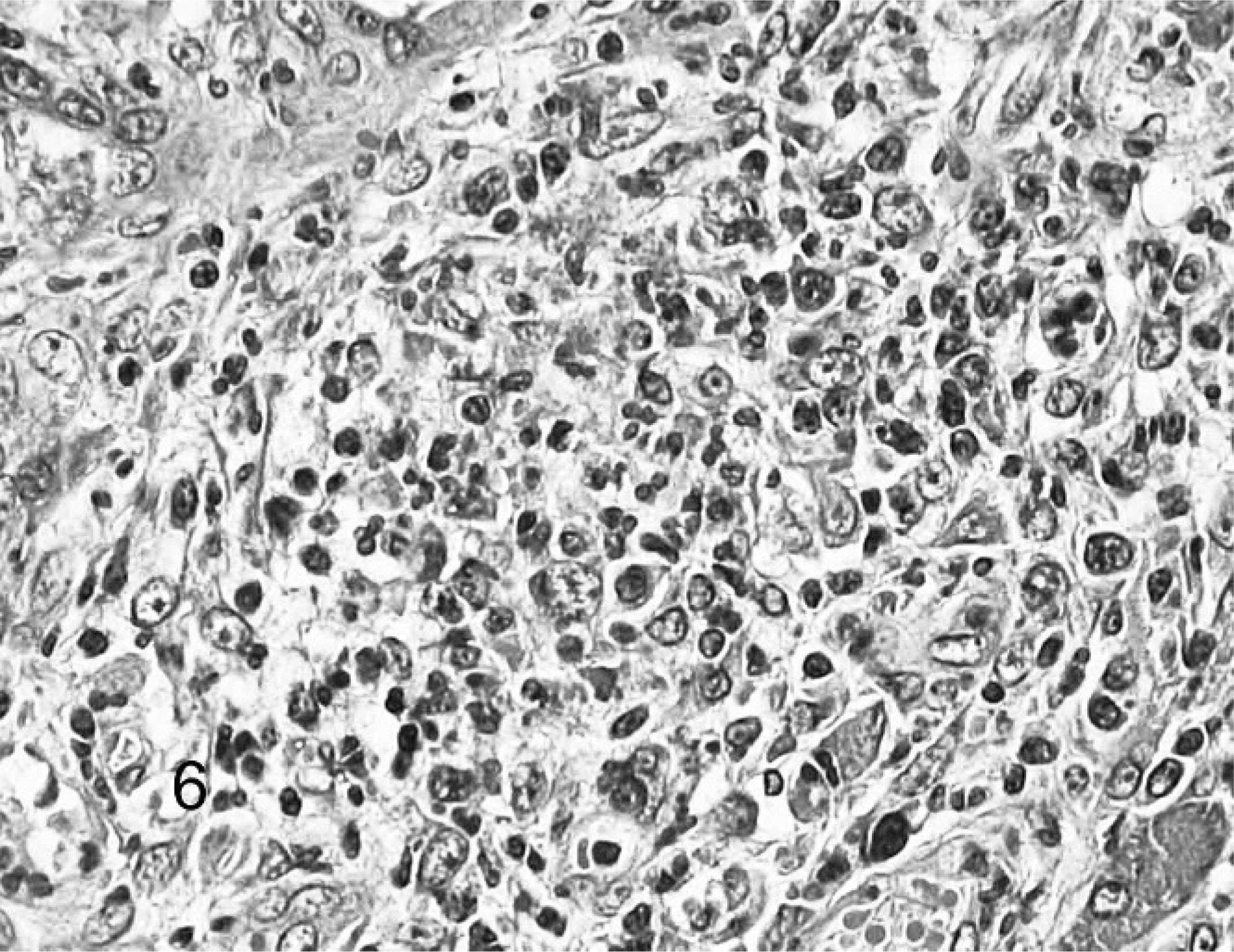
Ferret, coronavirus-associated pancreatitis. Typical inflammatory infiltrate. Note predominance of neutrophils with fewer macrophages and lymphocytes. HE.
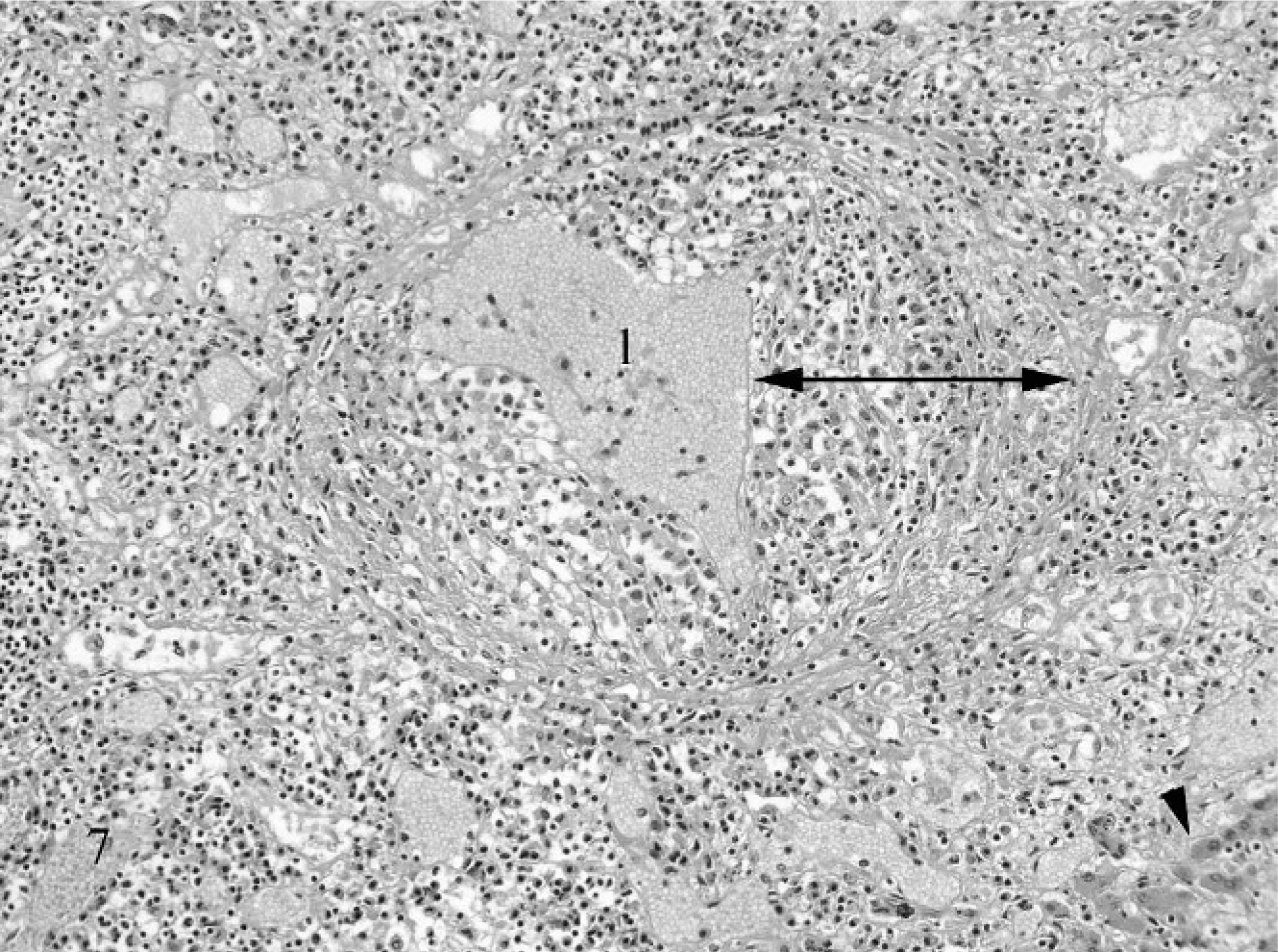
Ferret, coronavirus-associated vasculitis. Note small vein in portal tract with severe circumferential transmural inflammation (doubleheaded arrow). l = vascular lumen. Note that inflammation also is present throughout the tract and extends into the adjacent hepatic parenchyma (arrowhead). HE.
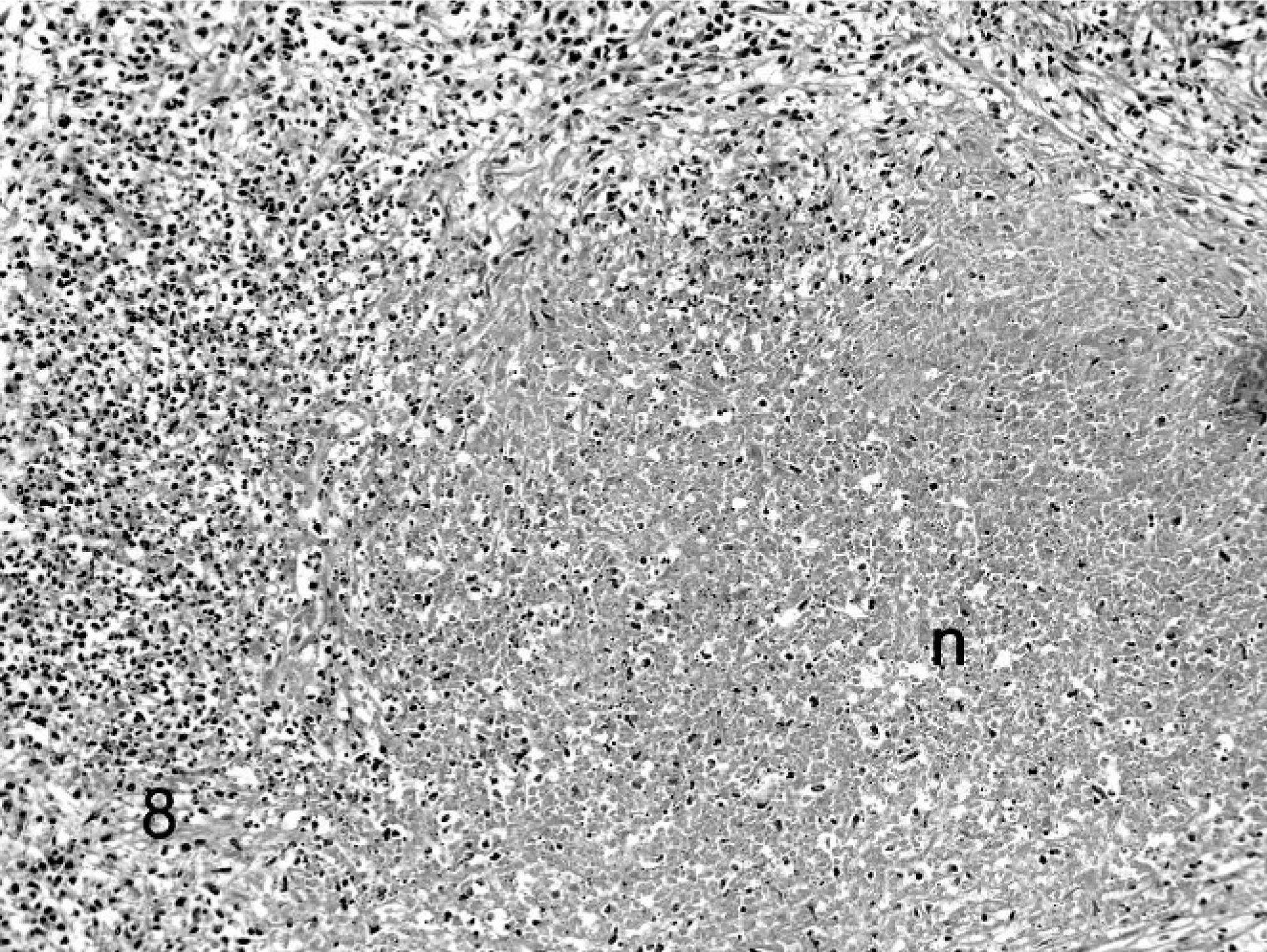
Ferret, coronavirus-associated lymphadenitis. Note large zone of necrosis (n) surrounded by a zone of lymphocytes and histiocytes. HE.
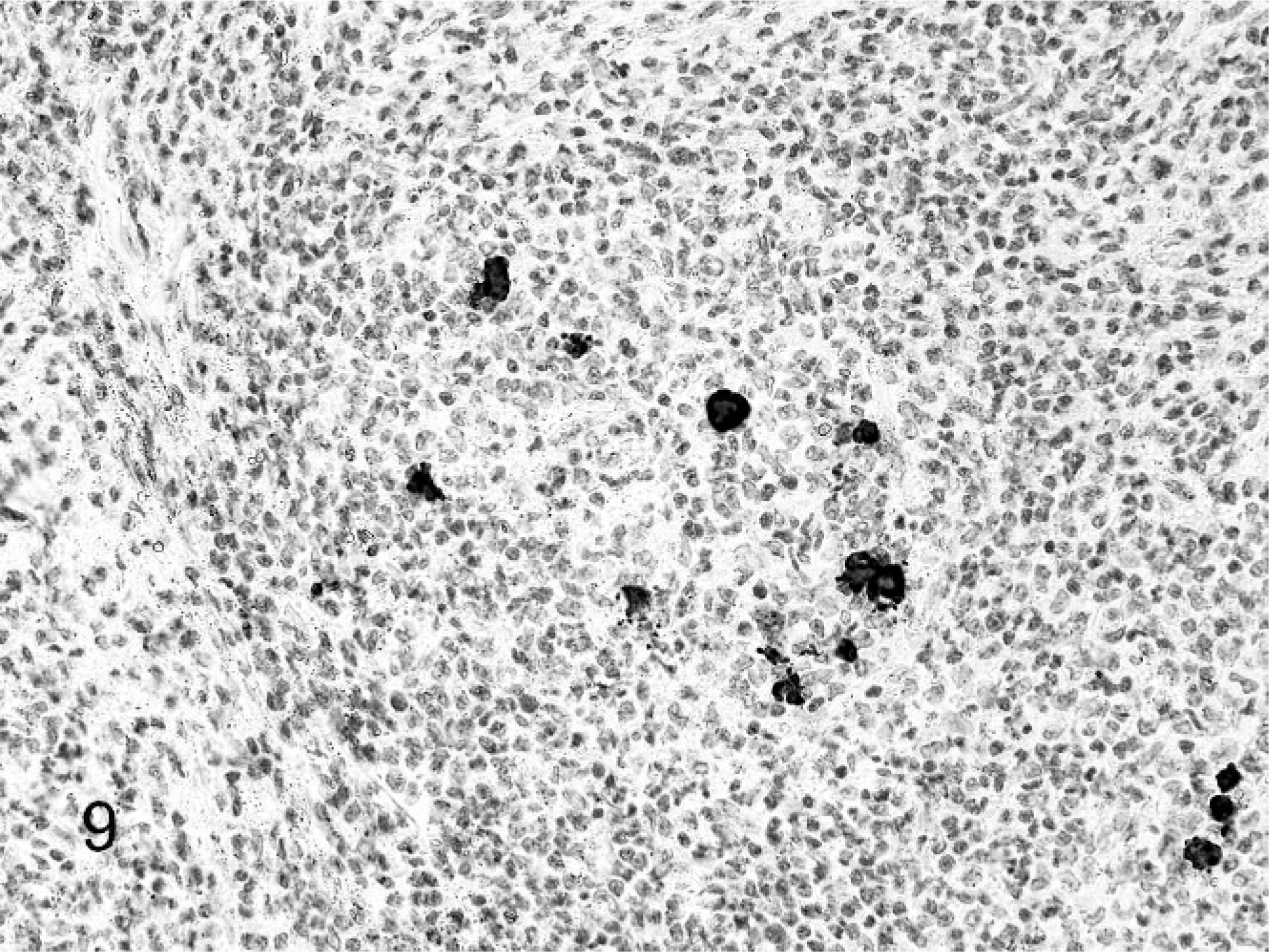
Ferret, coronavirus-associated lymphadenitis. Note intracytoplasmic staining for coronavirus antigen. Streptavidin-biotin, hematoxylin counterstain.
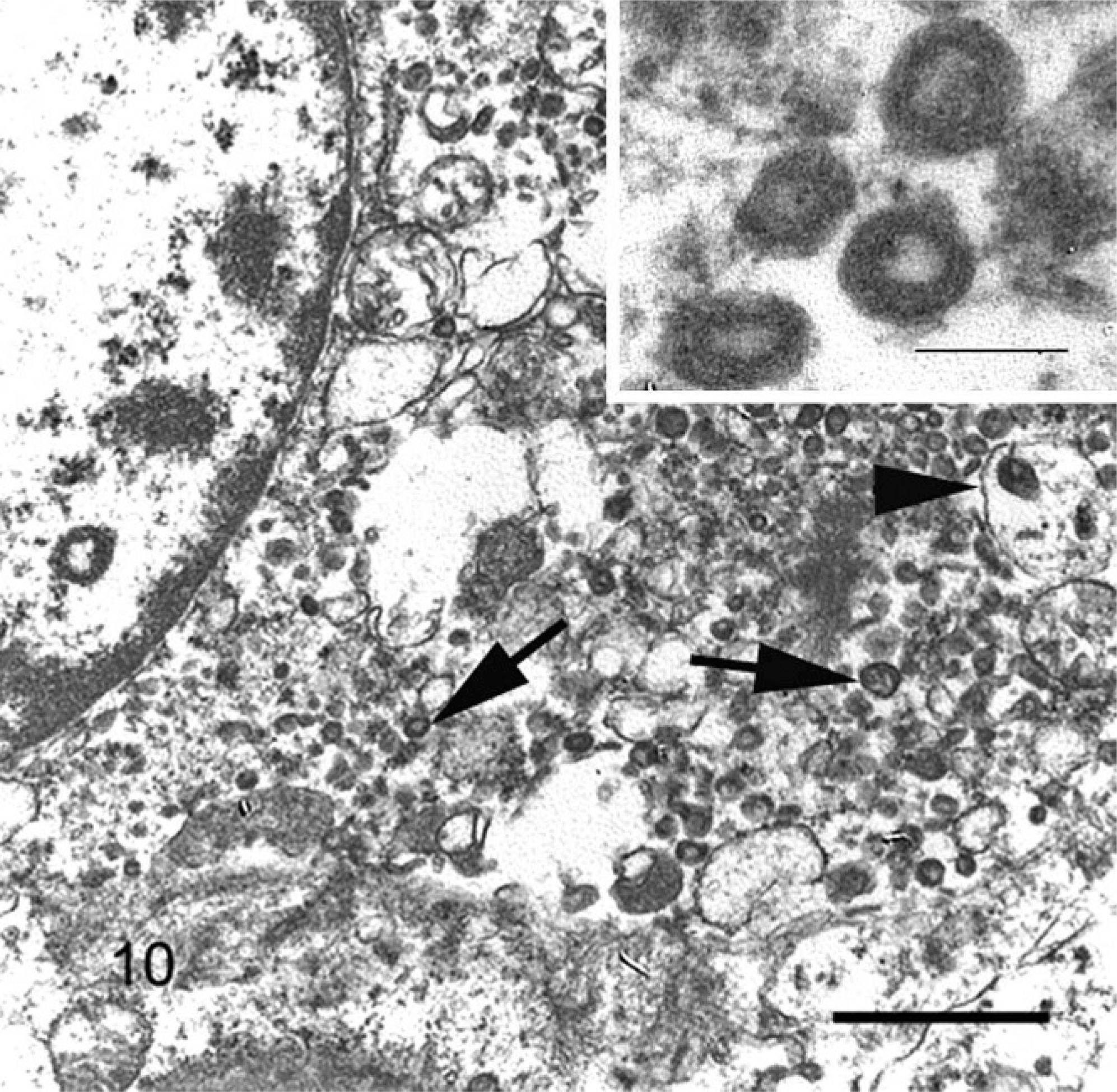
Ferret, coronavirus-associated mesenteritis. Note macrophage containing numerous intracytoplasmic virions free within the cytoplasm (arrows) and within membrane-bound vacuoles (arrowheads). Uranyl acetate and lead citrate, bar = 550 nm. Inset: Enveloped virions containing lucent core. Uranyl acetate and lead citrate, bar = 100 nm.
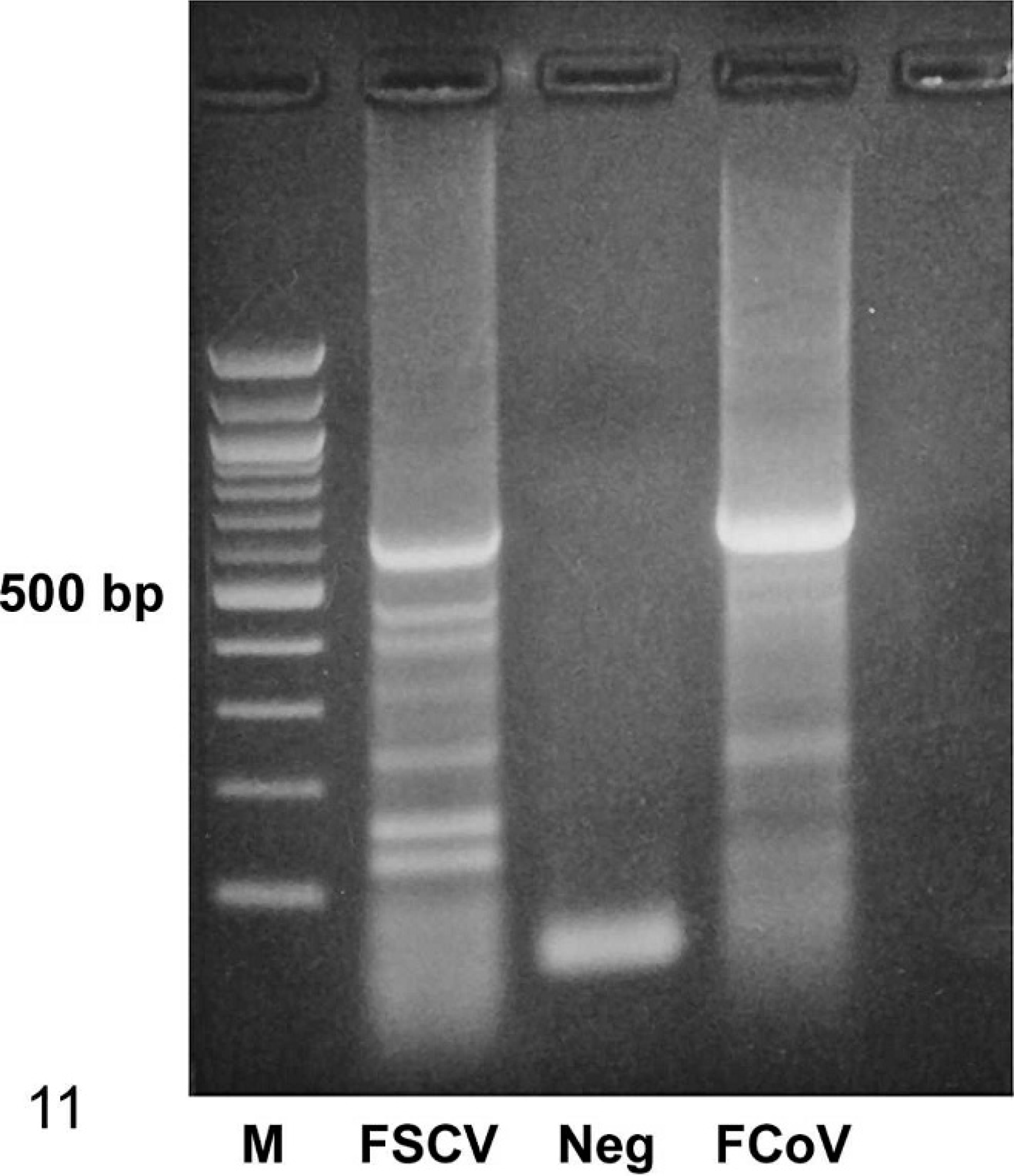
Amplification of a portion of the coronavirus S gene (approximately 600 bp) using degenerate consensus primers. M: 100-bp molecular weight ladder; FSCV: ferret systemic coronavirus template; Neg: negative control, no template control; FCoV: feline coronavirus template, positive control.
Footnotes
Acknowledgements
We thank the following US clinics for submission of cases: A&A Animal Hospital, Franklin Square, NY; All Creatures Animal Hospital, Bremerton, WA; Animal Clinic of Farmers Branch, Dallas, TX; Belle Forest Animal Hospital, Nashville, TN; Foothills Animal Hospital and Franklin Animal Hospital, Beaverton, OR; Old Bridge Veterinary Hospital, Woodbridge, VA; Old Country Animal Clinic, Plainview, NY; and Sno-Wood Veterinary Hospital, Woodinville, WA. We also are indebted to Histology Consulting Service for superb preparation of histology slides; Jamie Kinion, Susan Hinton, and Tera Thompson-Garner for data retrieval; and Christie Buie for photo editing and electronic manuscript submission.
