Abstract
A case of xanthogranulomatous inflammation of the small bowel in a 12-year-old male American Staffordshire Terrier is described. Disseminated yellow-white nodules 2 to 3 mm in diameter bulging on the serosal surface of the small bowel, as well as on mesenteric tissue, were detected. Histopathologic examination revealed a nodular collection of foamy cells, mainly involving serosal and muscular layers, associated with necrotic areas, hemorrhages, neovascularization, variable numbers of reactive spindle cells, neutrophils, lymphocytes, plasma cells, and rare multinucleated giant cells. Transmural lymphangectasia and mucosal lymphoplasmacytic inflammation were also observed. Both Oil Red O stain and ultrastructural study revealed lipid droplets in the cytoplasm of foamy cells. Lysozyme immunoreactivity was detected in single as well as in clustered foamy cells, while smooth muscle actin was positive in spindle cells and scattered foamy elements. Lymphangectasia associated with lymphoplasmacytic enteritis suggests a component of lymphatic fluid stasis in the pathogenesis of such lesions.
Xanthogranulomatous inflammation is an uncommon but well-recognized disease process in the human kidney 6 and gallbladder, 17 characterized by aggregations of lipid-laden foamy macrophages and parenchymal destruction. It has also been reported less often at many other sites in human body, including ovary, fallopian tube, endometrium, lymph node, bone, lung, appendix, 5 colon, 13, 16 stomach, 11 and parotid gland. 3 To the best of our knowledge, such process has not yet been described in veterinary literature. This represents the first report on a xanthogranulomatous reaction occurring in the small bowel of a dog.
A 12-year-old, neutered male American Staffordshire Terrier was presented with chronic diarrhea unresponsive to pharmacologic therapy, of 15 days in duration, associated with anorexia, weight loss, and asthenia. Exploratory laparotomy evidenced disseminated to coalescing yellowish-white nodules 2 to 3 mm in diameter bulging on the serosal surface of the small bowel, as well as on mesenteric tissue. The intestinal wall was diffusely hyperemic and thickened, with scattered areas of hemorrhages on serosa. Lymphatic vessels along the serosal surface of small bowel and throughout the mesentery were white and prominent. The dog was euthanatized because of the poor body conditions, but the owner did not consent to necroscopic examination.
A tissue sample from the small bowel was fixed in 10% neutral buffered formalin, embedded in paraffin wax, and sectioned at a thickness of 5 μm. Deparaffined sections were stained with hematoxylin and eosin (HE), periodic acid–Schiff (PAS), Grocott's methenamine silver, Ziehl-Neelsen, Giemsa and Gram's stains, while Oil Red O staining was performed on fixed frozen sections. Additional sections were also subjected to immunohistochemistry using primary antibodies directed against vimentin (1 : 50, V9, mouse monoclonal; DAKO, Glostrup, Denmark), myeloid/histiocyte antigen (1 : 50, MAC387, mouse monoclonal; DAKO), lysozyme (1 : 400, rabbit polyclonal; DAKO), smooth muscle actin (1 : 400, 1A4, mouse monoclonal; DAKO) or von Willebrand factor (1 : 400, rabbit polyclonal; DAKO). Immune complexes were treated with secondary biotinylated Goat anti-Mouse&Rabbit antibody (ready-to-use, Biospa, Milan, Italy) and subsequently detected using streptavidin-peroxidase (Biospa) or avidin–biotin–peroxidase complex method (ABC kit; Vector Laboratories, Burlingame, CA). Peroxidase activity was detected using 0.1% hydrogen peroxide in 3-3′-diaminobenzidine solution (Sigma). Sections were finally counterstained with Mayer's hematoxylin counterstain. For electron microscopic analysis, small pieces of formalin-fixed tissue sample of small bowel were washed in cacodilate buffer, re-fixed in 1% osmium tetroxide, and embedded in epoxy resin. Ultrathin sections were counterstained with uranyl acetate and lead citrate and were examined using an EM 900-Zeiss transmission electron microscope (Zeiss, Oberkochen, Germany).
Histopathologic examination revealed nodular collection of foamy cells, with a large, finely vacuolated cytoplasm, mainly involving serosal and muscular layers with focal extension to mesenteric fat tissue and submucosal layer. Such nodules were usually surrounded by fibrous tissue and characterized by necrotic areas, lakes of amorphous material with occasional presence of small cholesterol clefts, hemorrhages, neovascularization, variable numbers of neutrophils, lymphocytes, plasma cells, and rare multinucleated giant cells (Figs. 1–3). Scattered suppurative foci constituted by neutrophils clustered in small abscesses were also observed. Nodular lesions were frequently located around lymphatics, which appeared to be dilated in all layers of the intestinal wall (Fig. 4). The mucosa, including the lamina propria and the muscularis, was uninvolved by the above-described process; however, the lamina propria showed a diffuse lymphoplasmacytic infiltration (Fig. 4, inset). Grocott, Ziehl-Neelsen, Giemsa and Gram's stains failed to detect microorganisms. The content of foamy cells was negative or slightly positive on periodic acid–Schiff (PAS), while Oil Red O staining demonstrated the presence of lipids in the cytoplasm of such cells (Fig. 5). Immunohistochemically, nodules appeared to be intensely reactive with vimentin, while myeloid/histiocyte antigen strongly stained nonfoamy macrophages, intermingled with faintly and focally positive foamy cells (Fig. 6). Lysozyme immunoreactivity was detected in single as well as in clustered foamy cells (Fig. 7), whereas smooth muscle actin was positive in spindle cells and scattered foamy elements (Fig. 8). Von Willebrand factor clearly demonstrated neovascularization associated with nodular lesions. Ultrastructural study also evidenced numerous nonmembrane-bound medium-density lipid droplets of variable sizes replacing the cytoplasm of foamy elements (Fig. 9). In addition, varying amounts of cytoplasmic lipid droplets were observed in neutrophils, lymphatic endothelial cells and spindle cells, the latter frequently showing abundant rough endoplasmic reticulum (Fig. 10). On the basis of the above results, a diagnosis of xanthogranulomatous inflammation of the small bowel was made.
Xanthogranulomatous inflammation is a pathologic entity characterized by aggregation of numerous lipid-laden macrophages (xanthoma cells), admixed with a minor component of acute and chronic inflammatory cells. Intermingling of foamy macrophages and plasma cells is the most typical finding of the process. The presence of suppurative foci, necrosis, and hemorrhages are other characteristic findings, which play a role in eliciting this inflammatory response. However, in some cases a fully xanthomatous (mosaic-like) pattern becomes extremely prominent, while the other inflammatory cells decrease in number. 5 In this respect, xanthomatosis of the small bowel 4 has also been considered a lesion with xanthogranulomatous genesis. 5 The precise pathogenesis of this type of inflammation is still uncertain. It has been regarded as a secondary phenomenon of chronic suppuration, but other proposed mechanisms include impaired cellular immunity, infection by low-virulence organisms, reaction to a specific infectious agent, ineffective antibiotic therapy, vascular occlusions, defective lipid transport, and lymphatic obstruction. 1, 16
In the present case, transmural lymphangectasia associated with lymphoplasmacytic enteritis suggests an involvement of lymphatic blockage in the pathogenesis of the xanthogranulomatous reaction. In fact, even though microbiologic exams were not performed, histochemical stains failed to detect microorganisms and, as well, suppurative foci represented a minor component of the process, while the fully xanthomatous pattern predominated. In this respect, in human-isolated xanthomatosis of the small bowel, the dilated lacteals observed microscopically also suggested a component of lymphatic obstruction with distension. 4 Likewise, primary lymphoedema has also been associated with the onset of xanthomatosis. 9 In dogs, large focal accumulation of lypophages surrounding a remnant of a dilated lymphatic containing amorphous material, sometimes accompanied by lymphocyte aggregates, has occasionally been found in association with intestinal lymphangectasia. 2, 12, 14, 18 Such lesions, described as lipogranulomatous lymphangitis, probably represent a consequence of a lymphatic fluid stasis with chronic leakage of lipid-laden chyle. 2 Similar pathogenetic mechanisms can also be hypothesized for the case reported here. However, in our case, the histological pattern appears to be rather different from the previously described cases of lipogranulomas, and, in this respect, the presence of nodular aggregates of foamy cells associated with infiltration of acute and chronic inflammatory cells, necrosis, microabscesses, hemorrhages, and neovascularization allows us to define this process as a xanthogranulomatous inflammation, with similar characteristics to those described in humans. 5 Our immunohistochemical, as well as ultrastructural findings, also reflect those obtained in various human reports, 5, 8, 10, 15 thus further supporting such histologic diagnosis. Although medium-density lipid droplets found in the present case differ from the electron-lucent lipid droplets, which have been observed in human xanthogranulomatous appendicitis, this is not surprising because the density of lipid droplets as seen in electron micrographs can vary from electron-lucent to markedly electron-dense, depending on the saturated and unsaturated fatty acid content, as well as their degree of unsaturation. 7
Finally, as far as the genesis of foamy cells is concerned, the immunoreactivity for histiocytic markers confirms the origin of most of such cells from macrophages. Nevertheless, the ultrastructural evidence of lipid droplets in the cytoplasm of spindle cells characterized by abundant rough endoplasmic reticulum indicates that cells other than histiocytes, such as fibroblasts, can also accumulate lipids. In this respect, lipid droplets were also observed in some fibroblasts in human xanthogranulomatous pyelonephritis. 10 At the same time, positivity for smooth muscle actin detected in scattered foamy elements suggests that they could also derive from smooth muscle cells. Even though a similar finding has not yet been described in human cases, the ultrastructural evidence of accumulation of lipid droplets in degenerated muscle cells within muscularis mucosae has been reported in human xanthogranulomatous appendicitis. 15 In this connection, the ability of smooth muscle cells to accumulate lipid globules in their cytoplasm has been established in canine atherosclerotic lesions. 19
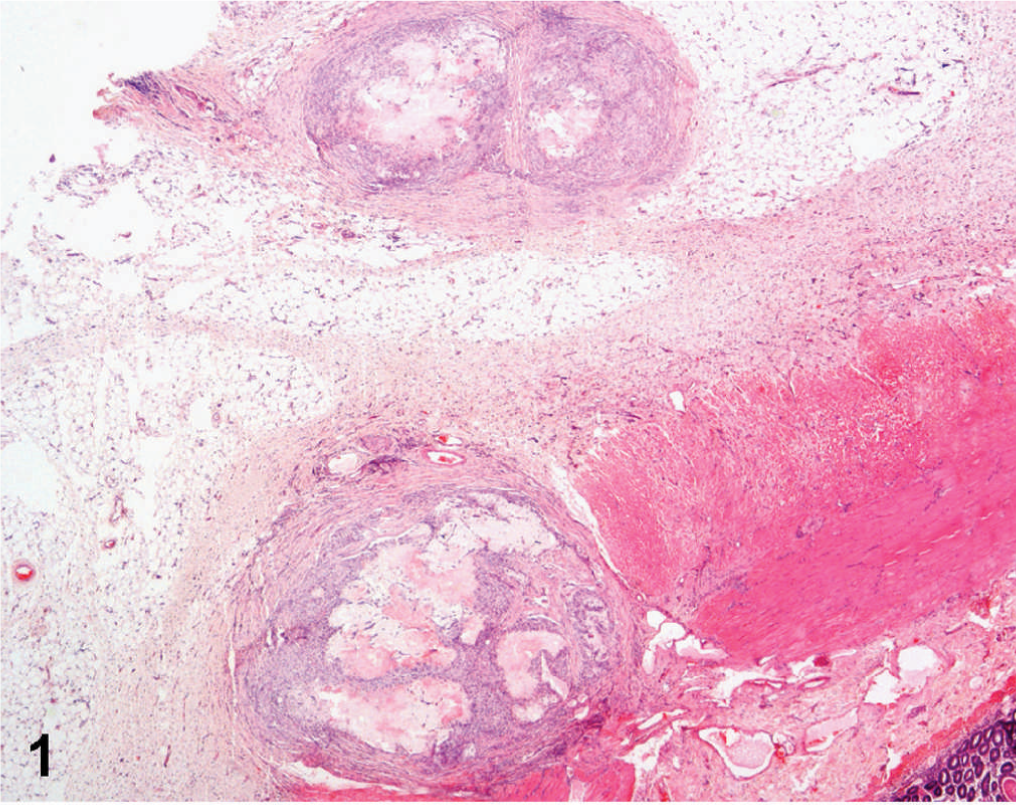
Small bowel; dog. Nodular lesions surrounded by fibrous tissue, involving serosal and muscular layers, as well as mesenteric fat tissue. HE stain.
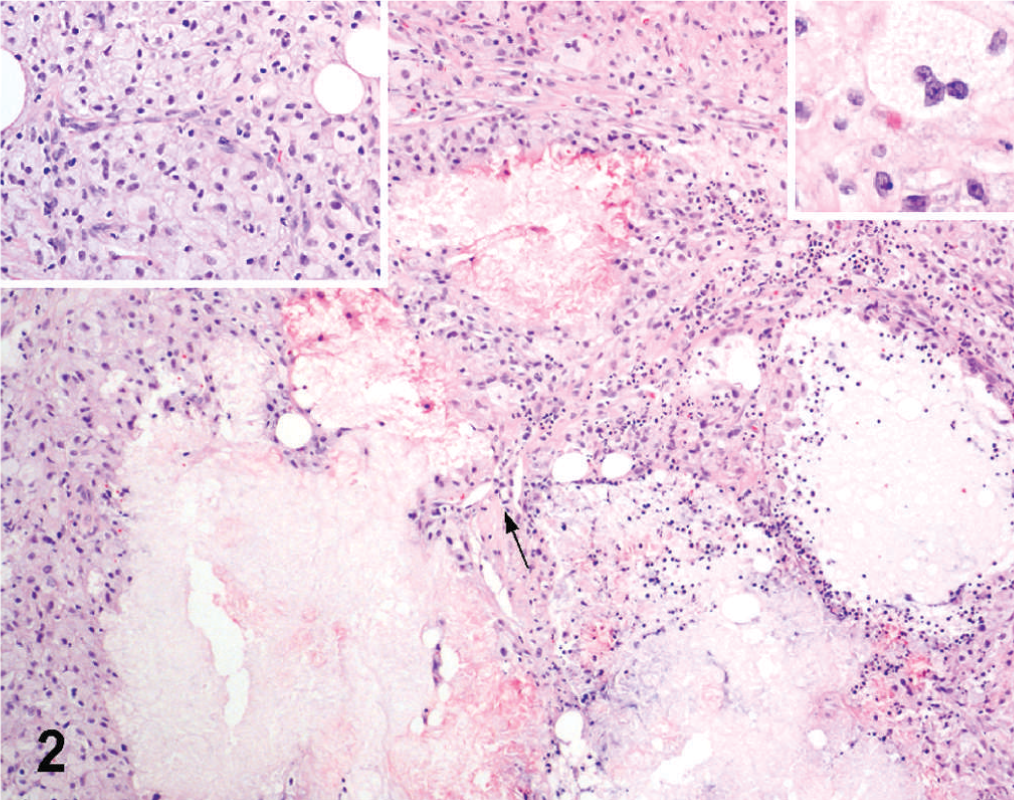
Small bowel; dog. Nodular aggregates of foamy cells around necrotic debris and lakes of amorphous material with small cholesterol clefts (arrow). Inset, upper left: Higher magnification of foamy cells. Inset, upper right: Higher magnification of a multinucleated giant cell. HE stain.
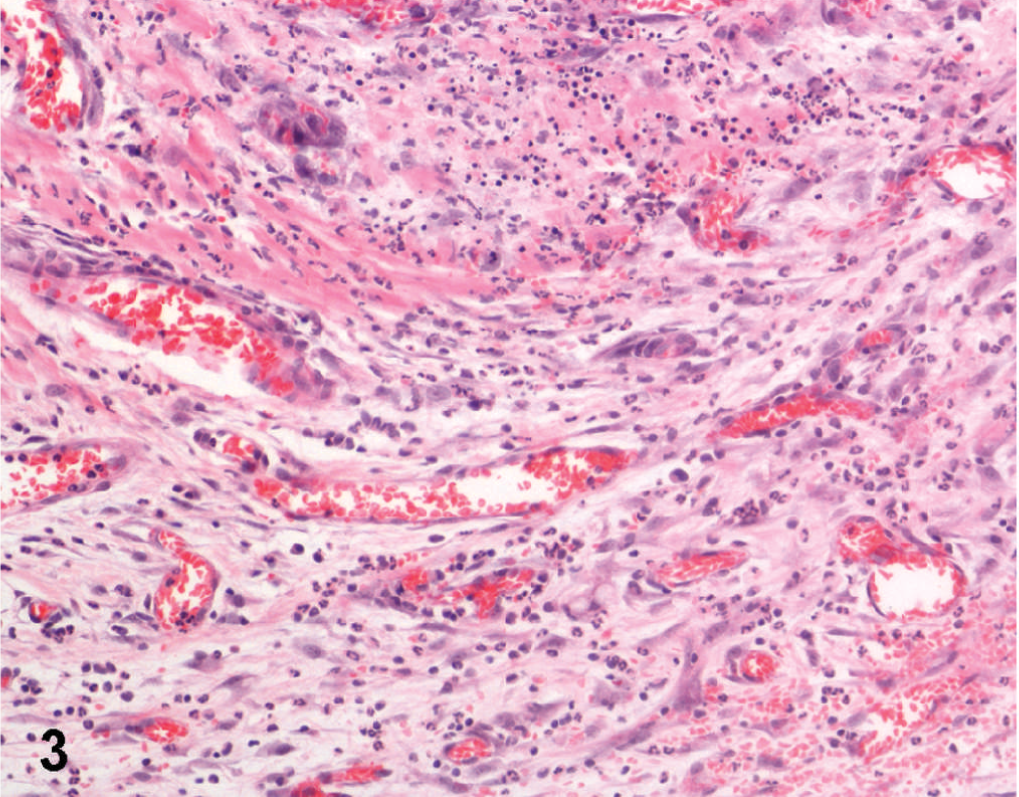
Small bowel; dog. Suppurative foci associated with diffuse mixed inflammatory infiltrate, hemorrhages, and neovascularization. HE stain.
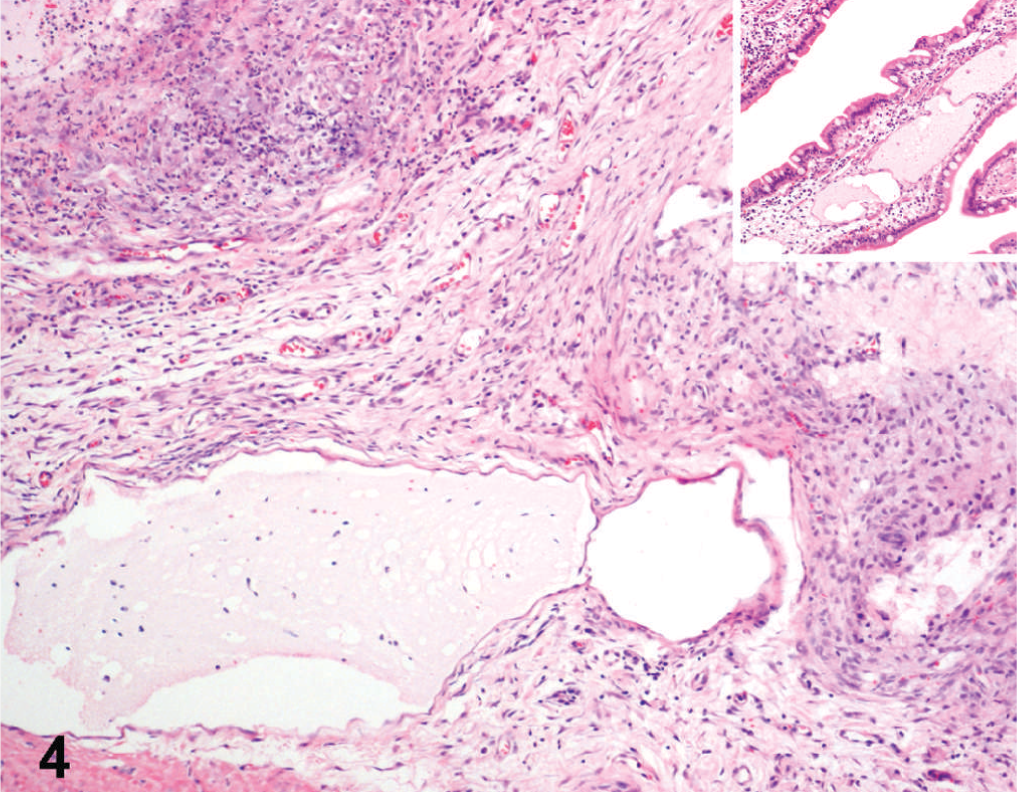
Small bowel; dog. Nodular lesions adjacent to dilated lymphatics. Inset: Lymphoplasmacytic infiltration and lymphatic dilation in the lamina propria. HE stain.
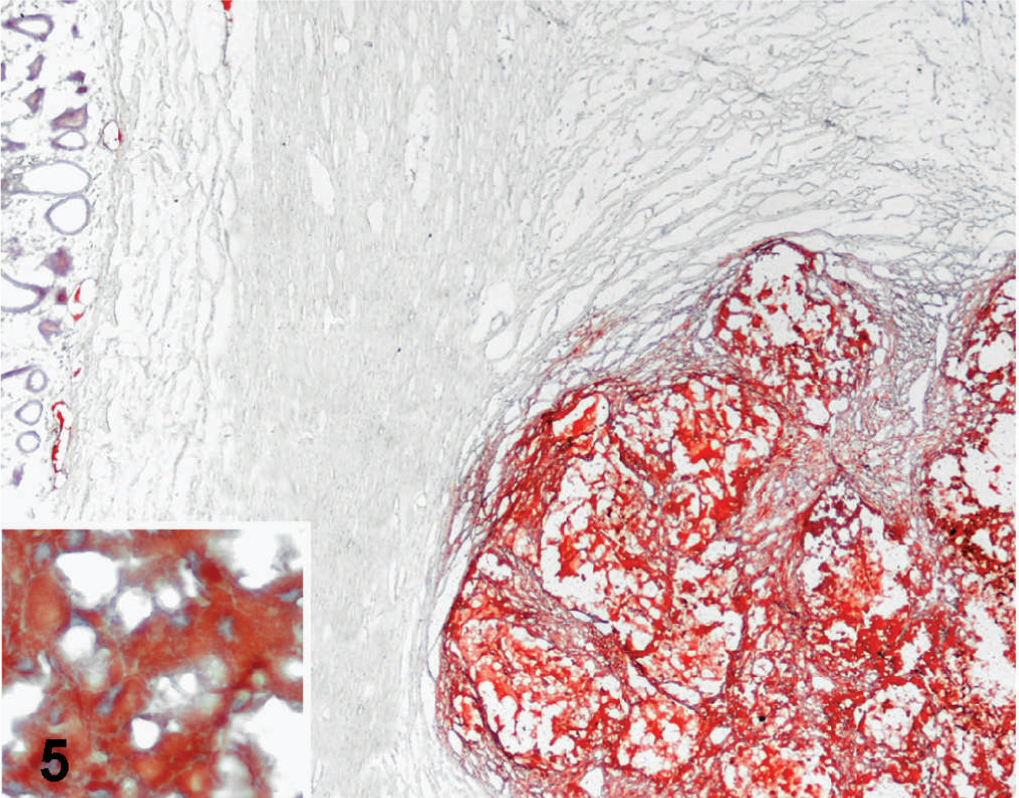
Small bowel; dog. Nodular lesions intensely stained with Oil Red O. Inset: Higher magnification of strongly stained cytoplasm of foamy cells. Oil Red O staining, Mayer's hematoxylin counterstain.
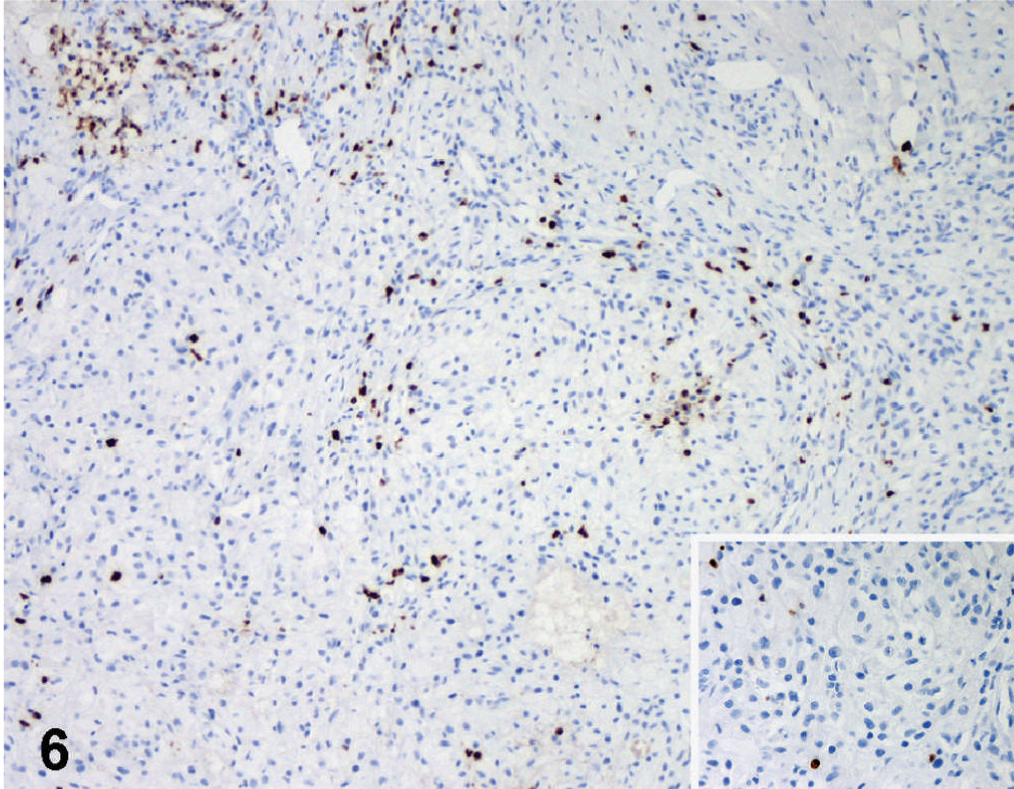
Small bowel; dog. Intense immunoreactivity to myeloid/histiocyte antigen in macrophages intermingled with slightly and focally positive foamy cells. Inset: Positive cells at higher magnification. ABC method, Mayer's hematoxylin counterstain.
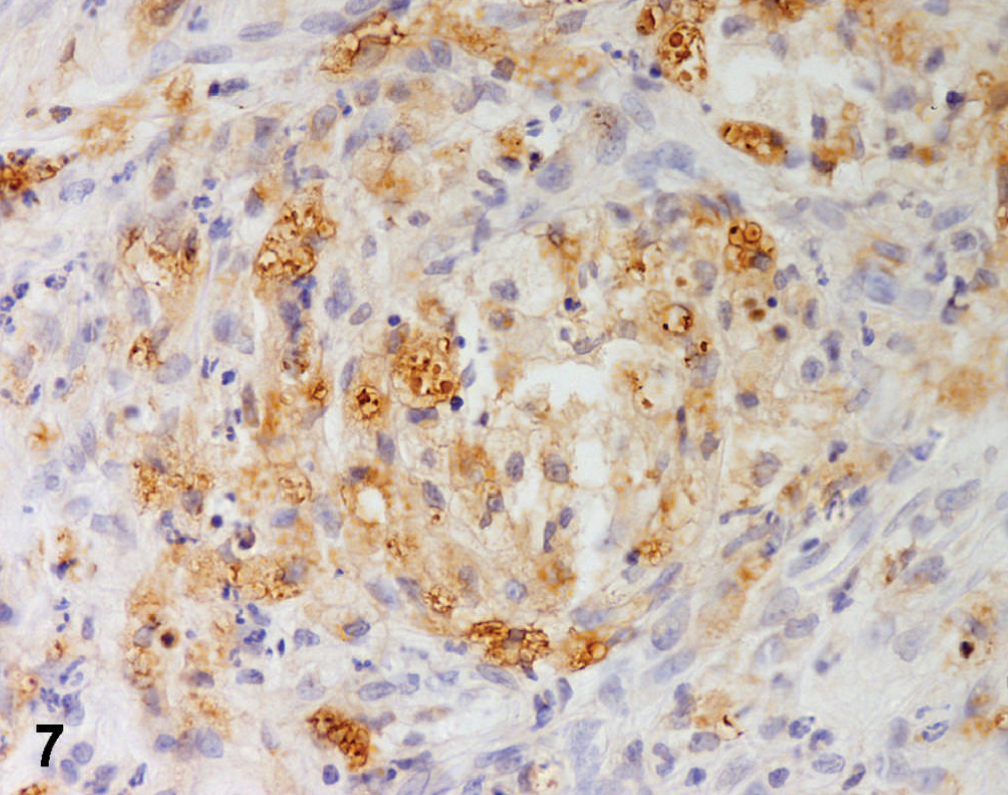
Small bowel; dog. Diffuse lysozyme immunostaining in clustered foamy cells. ABC method, Mayer's hematoxylin counterstain.
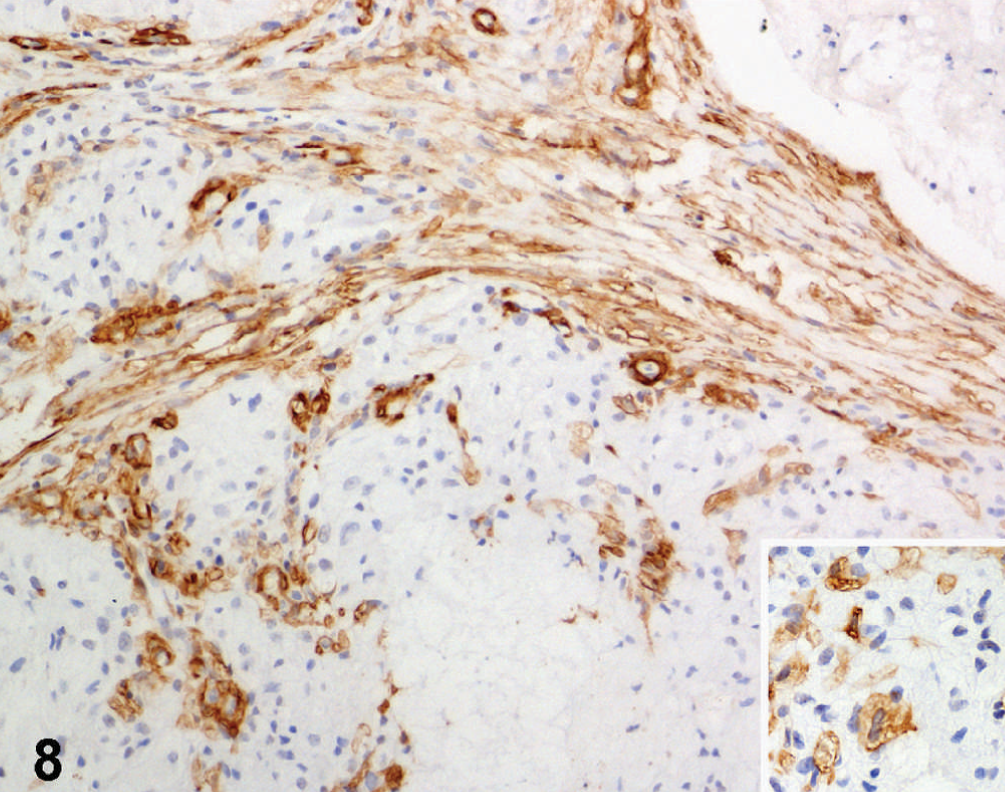
Small bowel; dog. Positivity for smooth muscle actin in spindle cells and in scattered foamy elements (Inset). Streptavidin-peroxidase method, Mayer's hematoxylin counterstain.
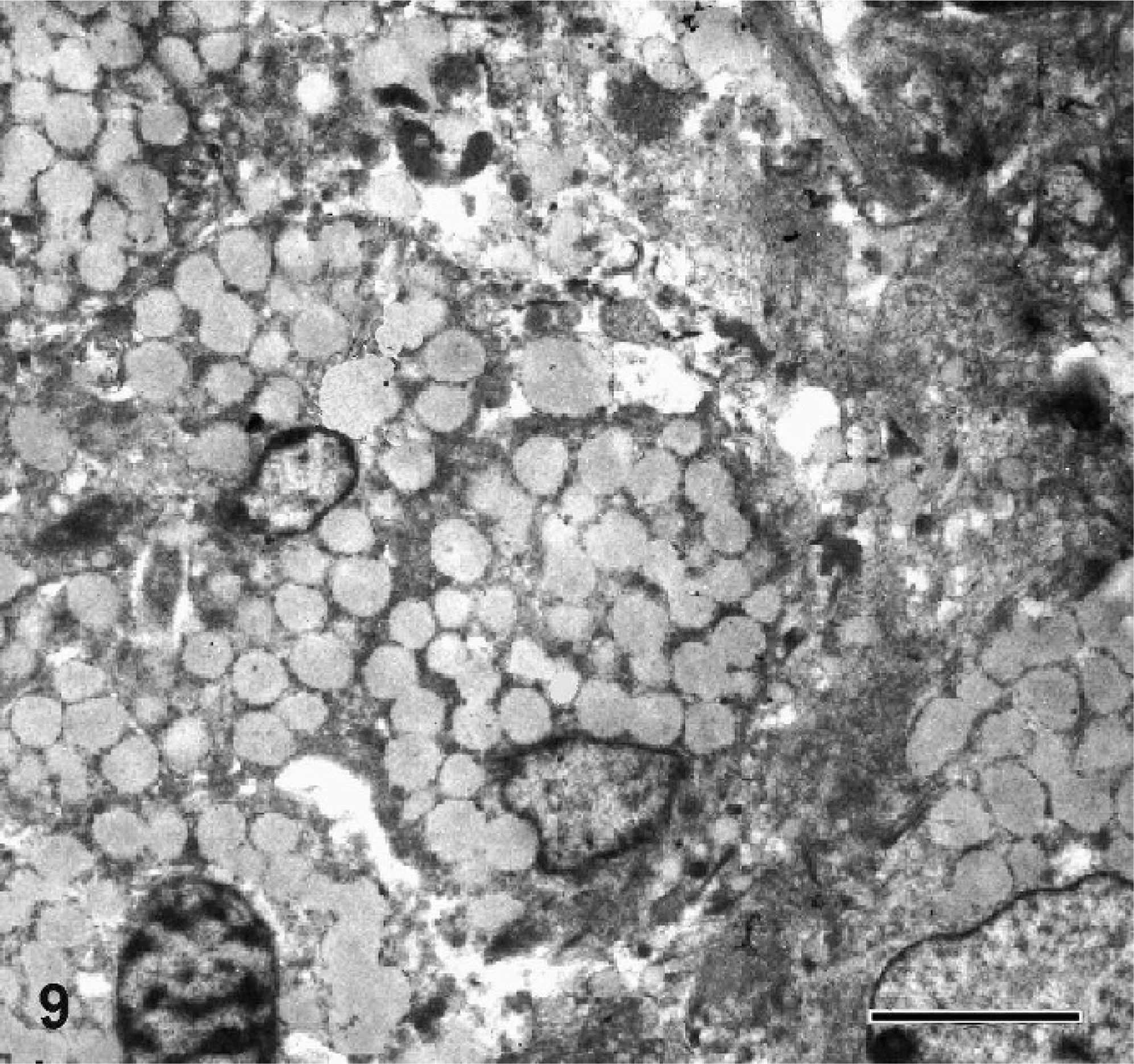
Small bowel; dog. Numerous lipid droplets filling the cytoplasm of foamy cells. Transmission electron microscopy; uranyl acetate and lead citrate counterstain. Bar = 5 μm.
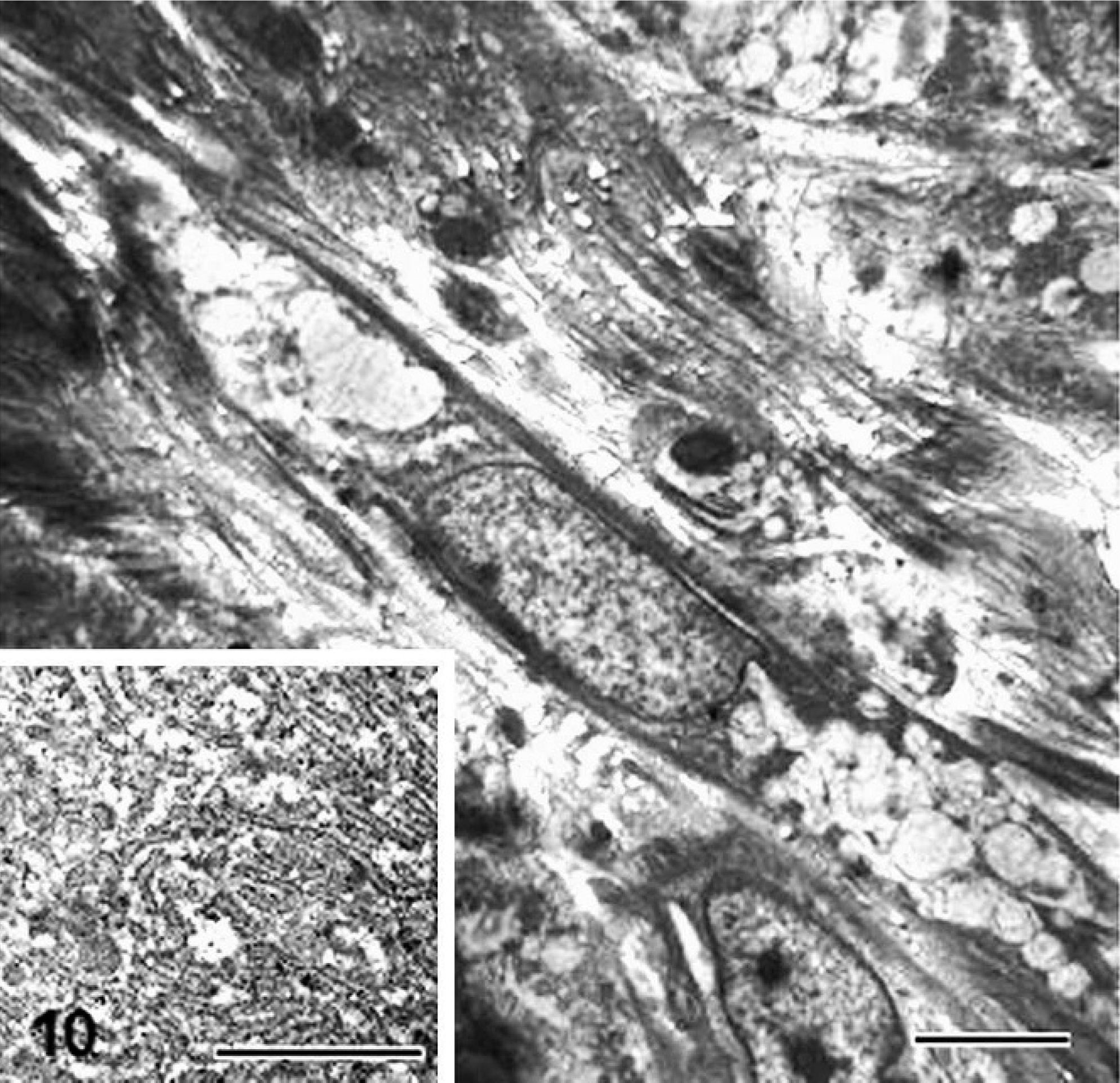
Small bowel; dog. Lipid globules in the cytoplasm of spindle cells (bar = 5 μm) showing abundant rough endoplasmic reticulum (Inset, bar = 2 μm). Transmission electron microscopy; uranyl acetate and lead citrate counterstain.
