Abstract
Cytokeratin 5 and p63 have been described as basal and myoepithelial cell markers in human breast. Mixed tumors of the canine mammary gland have been associated with a myoepithelial origin. Cytokeratin 5 expression has not been evaluated in these tumors. We investigated the relation between cytokeratin 5 and p63 double-immunohistochemical expression in 23 mixed tumors of the canine mammary gland (10 benign mixed tumors and 13 carcinomas arising from benign mixed tumors) and their origin. Cytokeratin 5 and p63 co-expression was observed in myoepithelial cells of benign mixed tumors, as well as in squamous differentiation of carcinoma arising from benign mixed tumors. Though a few interstitial spindle cells of the mesenchymal components expressed both p63 and cytokeratin 5, the basal epithelial cells were labeled only by cytokeratin 5. The co-expression of p63 and cytokeratin 5 in myoepithelial cells and squamous differentiation suggest that, like in human breast, cytokeratin 5 can also be considered a myoepithelial- and squamous-cell differentiating marker in canine tumors. The presence of some interstitial spindle cells stained for p63 and cytokeratin 5 might be associated with a myoepithelial origin of the mesenchymal component of mixed tumors of the canine mammary gland. Moreover, contrary to p63, basal epithelial cells were labeled by cytokeratin 5, indicating that cytokeratin 5 may not represent an exclusive myoepithelial cell marker but also a basal epithelial cell marker in canine mixed tumors. According to these data, basal epithelial cells may be related to the origin of the epithelial component of mixed tumors of the canine mammary gland.
The mixed tumors of the canine mammary gland are a heterogeneous group of neoplasms with incompletely known histogenesis, which frequently leads to different classifications and terminologies. It seems likely that mixed tumors are of myoepithelial origin. 9, 10 Benign mixed tumors (BMT) are neoplasms composed of benign cells morphologically resembling epithelial components (luminal and/or myoepithelial) and mesenchymal cells that produce cartilage and/or bone and/or fat, eventually in combination with fibrous tissue. 17 The carcinomas arising from BMTs (CBMT) of the canine mammary gland are neoplasms composed of carcinomatous cells occurring in a benign mixed tumor. 17
In the normal mammary gland, 3 cell populations can be recognized, each with a distinct immunoprofile. The luminal layer is composed of glandular epithelial cells, which are labeled by cytokeratins 7, 8, 18, and 19; and basal epithelial cells, which are labeled by cytokeratins 5, 14, 17, but not smooth muscle alpha-actin (αSMA). The basal layer is composed of myoepithelial cells expressing cytokeratins 5, 14, 17, in addition to p63 and αSMA. 13 In the mammary neoplasms, these 3 cell types can become limited to 2 or 1, characterizing particular kinds of neoplasms. 1, 13 Recently, cytokeratin 5 (CK5) has been reported to be not only a myoepithelial cell marker but also a progenitor cell marker in breast neoplasms. 5, 6 However, CK5 expression has not yet been evaluated in mixed tumors of the canine mammary gland.
p63, a homologue of the p53 gene, has been described as a nuclear transcriptional factor, with diverse functions in the cell cycle ranging from apoptosis to cell differentiation. 7, 15, 28, 32 p63 has also been associated with the development of various epithelial neoplasms. 2, 7, 21, 31 p63 expression was observed in myoepithelial cells of benign neoplasms and in low differentiated carcinomatous cells of human breast. 3, 4, 19, 20, 23, 24, 27, 30 Moreover, p63 has been proposed to be an important myoepithelial cell marker that serves to define benign and malignant lesions and may indicate a myoepithelial histogenesis in some tumors of the canine mammary gland. 9 Nevertheless, the cellular origin of mixed tumors of the canine mammary gland and the reason why carcinomas develop into benign mixed tumors are still unclear. Therefore, the aim of the present study was to determine the relation between p63 and CK5 expressions in mixed tumors of the canine mammary gland in an attempt to clarify the histogenesis of these neoplasms.
Materials and Methods
Casuistic
Twenty-three cases of canine mammary gland tumors were retrieved from the files of the Laboratory of Comparative Pathology, Department of General Pathology, Institute of Biological Science, Federal University of Minas Gerais, Brazil. The selection was based on the histopathologic diagnosis according to World Health Organization criteria. 17 The histologic types selected were as follows: benign mixed tumors (n = 10) and carcinoma arising from benign mixed tumors (n = 13). All dogs were females, ranging in age from 5 to 15 years (mean, 11 years); the diameter of the mammary tumors ranged from 4 to 25 cm (mean, 11 cm). However, the age of the dogs was not related to the size of the neoplastic masses. All dogs presented with single neoformations. The dogs were not sterilized. None of the animals had received any treatment before the biopsy procedure.
Immunohistochemistry
All tissue samples were routinely fixed in 4% neutral formalin and embedded in paraffin. Briefly, 3-µm-thick sections were cut from paraffin blocks containing representative tumor samples. Paraffin sections were dewaxed in xylene, rehydrated through a graded alcohol series, placed in 10 mM citrate buffer, and submitted to heat retrieval by using a vapor lock for 40 minutes. After heating, the slides were allowed to cool to room temperature and were briefly washed with Tris-buffered saline solution. Endogenous peroxidase activity was blocked with 3% hydrogen peroxide in methanol for 5 minutes. Normal serum (Novostain Super ABC kit, Novocastra Laboratories, Newcastle Upon Tyne, UK) was used for 30 minutes to block nonspecific immunoassaying. Immunohistochemical staining was performed by using an avidin-biotin peroxidase system (Novocastra). The following primary antibodies were incubated overnight at room temperature: p63 (1 ° 200, clone 4A4, Santa Cruz, USA), CK5 (1 ° 200, clone XM26; Novocastra), multicytokeratin (AE1/AE3) (1 ° 100, clones AE1 and AE3; Novocastra), alpha smooth muscle actin (αSMA) (1 ° 100, clone αsm-1, Novocastra, UK), and vimentin (VIM) (1 ° 100, clone V9, Novocastra, UK). After washes in phosphate-buffered saline solution (PBS), biotinylated universal secondary antibody (Novocastra) was applied for 30 minutes. The sections were incubated with the avidin-biotin complex reagent (Novocastra) for 30 minutes and were developed with 3.3-diaminobenzidine tetrahydrochloride (DAB) in PBS, pH 7.5, containing 0.036% hydrogen peroxide, for 5 minutes. Light Mayer's hematoxylin was applied as a counterstain. The slides were then dehydrated in a series of ethanols and mounted with Permount (Fischer, Fairlawn, NJ, USA).
Cytokeratin 5-positive tumors were submitted to a double-immunolabeling study according to the manufacturer's protocol (EnVision Doublestain System, Dako, Denmark). For this purpose, we used CK5 (1 ° 200, clone XM26; Novocastra) with p63 (1 ° 200, clone 4A4; Santa Cruz Biotechnology, Santa Cruz, USA). p63 (nuclear marker) was developed with DAB (brown), and the CK5 (cytoplasmic marker) was developed with fast red (red).
Normal skin was used as the positive control for p63, and normal mammary tissue was used as the positive control for CK5 and AE1/AE3. A uterine leiomyoma was used as the positive control for αSMA and VIM. The external controls were obtained from canine tissue. Regardless of the external controls, all samples exhibited internal controls: normal skin and mammary tissue. Negative controls for immunostaining were prepared by omission of the primary antibody.
Tumors were considered to be positive when distinct brown nuclear (p63) or cytoplasmic (CK5, AE1/AE3, αSMA, and VIM) staining was present homogenously. The number of immunoreactive cells was assessed semiquantitatively, in 30 fields chosen randomly, at high power (×400): 0 = no stained cells; ± = less than 10% positive cells; + = 10–50% positive cells; and ++ = more than 50% positive cells. 9 AE1/AE3, αSMA, and VIM were used only to confirm the histologic patterns of the tumors.
Statistical analysis
Statistical analysis was performed by using the Graph Pad Prism v.3 software (San Diego, CA). The relation between p63 and CK5 expression and the histologic diagnosis was determined by the Fisher test. All tests were 2 tailed, and a P value of <0.05 was considered to be significant.
Results
Benign mixed tumors were characterized as tumors composed of benign cells morphologically resembling epithelial components (luminal and/or basal) and mesenchymal cells that have produced cartilage and/or bone in association with fibrous tissue. Carcinomas arising from BMTs were distinguished as individual nodules of malignant epithelial cells, the majority of them presenting a squamous appearance, with individual keratinization and pearls formations, occurring in BMTs. The data regarding immunolabeling for AE1/AE3, αSMA, and VIM in BMT and CBMT are presented in Table 1. The immunolabeling for p63 and CK5 is presented in Table 2.
immunoreactivity for cytokeratins (AE1/AE3), alpha smooth muscle actin (αSMA), and vimentin (VIM) of benign mixed tumors (BMT) and carcinomas arising from benign mixed tumors (CBMT) of the canine mammary gland.∗
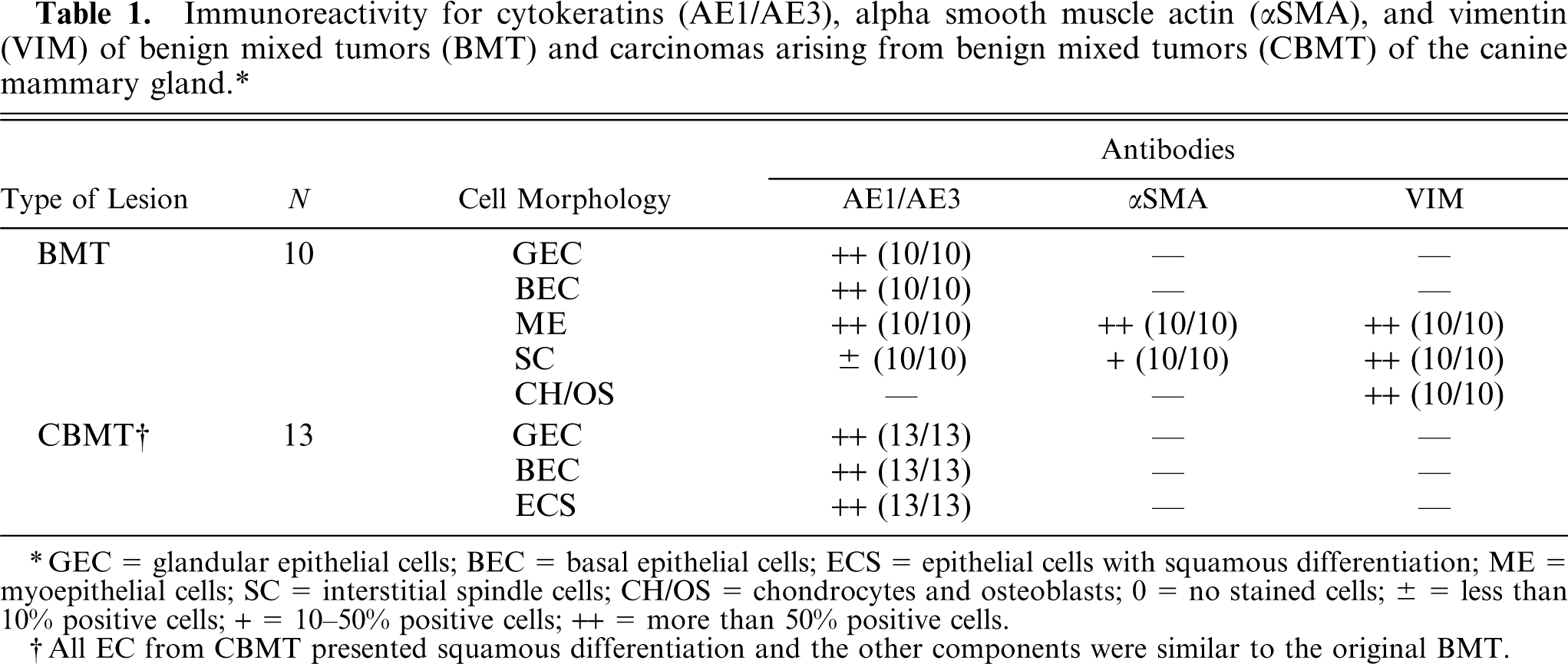
GEC = glandular epithelial cells; BEC = basal epithelial cells; ECS = epithelial cells with squamous differentiation; ME = myoepithelial cells; SC = interstitial spindle cells; CH/OS = chondrocytes and osteoblasts; 0 = no stained cells; ± = less than 10% positive cells; + = 10-50% positive cells; ++ = more than 50% positive cells.
All EC from CBMT presented squamous differentiation and the other components were similar to the original BMT.
immunoreactivity for p63 and CK5 antibodies of benign mixed tumors (BMT) and carcinomas arising from benign mixed tumors (CBMT) of the canine mammary gland.∗
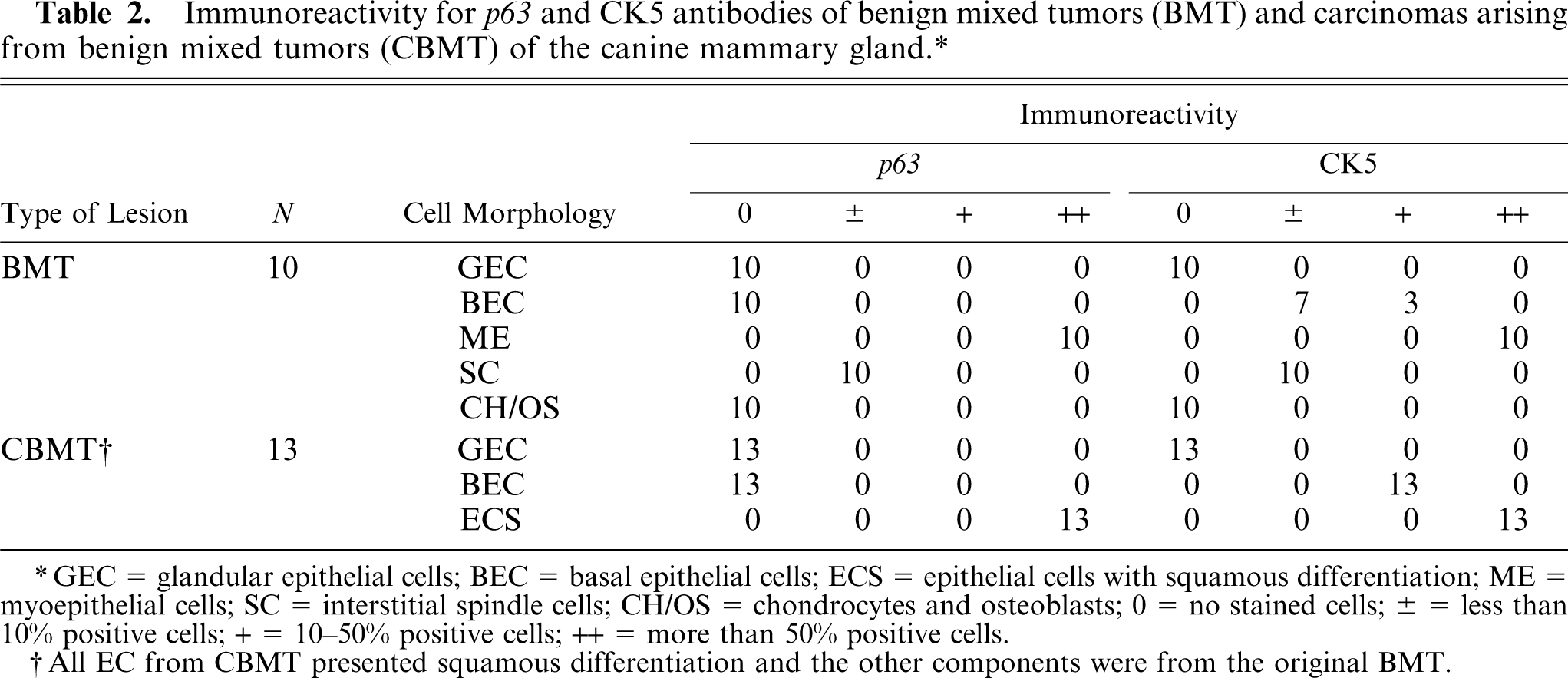
GEC = glandular epithelial cells; BEC = basal epithelial cells; ECS = epithelial cells with squamous differentiation; ME = myoepithelial cells; SC = interstitial spindle cells; CH/OS = chondrocytes and osteoblasts; 0 = no stained cells; ± = less than 10% positive cells; + = 10-50% positive cells; ++ = more than 50% positive cells.
All EC from CBMT presented squamous differentiation and the other components were from the original BMT.
In BMT, most epithelial cells (glandular and basal) stained for the pool of cytokeratins AE1/AE3. Only the basal cells, however, showed a weak immunolabeling for CK5. All the myoepithelial cells from BMT expressed CK5, AE1/AE3, αSMA, and VIM. The majority of interstitial spindle cells in the mesenchymal component expressed αSMA and VIM, but only some of them expressed CK5 and AE1/AE3. Chondrocytes and osteoblasts were labeled only by VIM.
In CBMT, many epithelial cells (glandular and basal phenotype) were labeled by AE1/AE3. Only the basal cells stained for CK5. The areas of squamous differentiation in CBMT presented immunolabeling for both AE1/AE3 and CK5. The interstitial spindle cells, chondrocytes, and osteoblasts from the mesenchymal component of CBMT, which is benign, had an immunolabeling profile similar to BMT.
There was a strong relation between CK5 and p63 expression in the myoepithelial cells of BMT (P = 0.041), as well as in areas of squamous differentiation of CBMT (P = 0.026). In BMT and CBMT, a few spindle cells of the mesenchymal component expressed both p63 and CK5, but these markers were not expressed by chondrocytes or osteoblasts. Moreover, some epithelial cells with a basal phenotype expressed CK5 in BMT and CBMT, but none of them presented immunolabeling for p63 (Figs. 1–4).
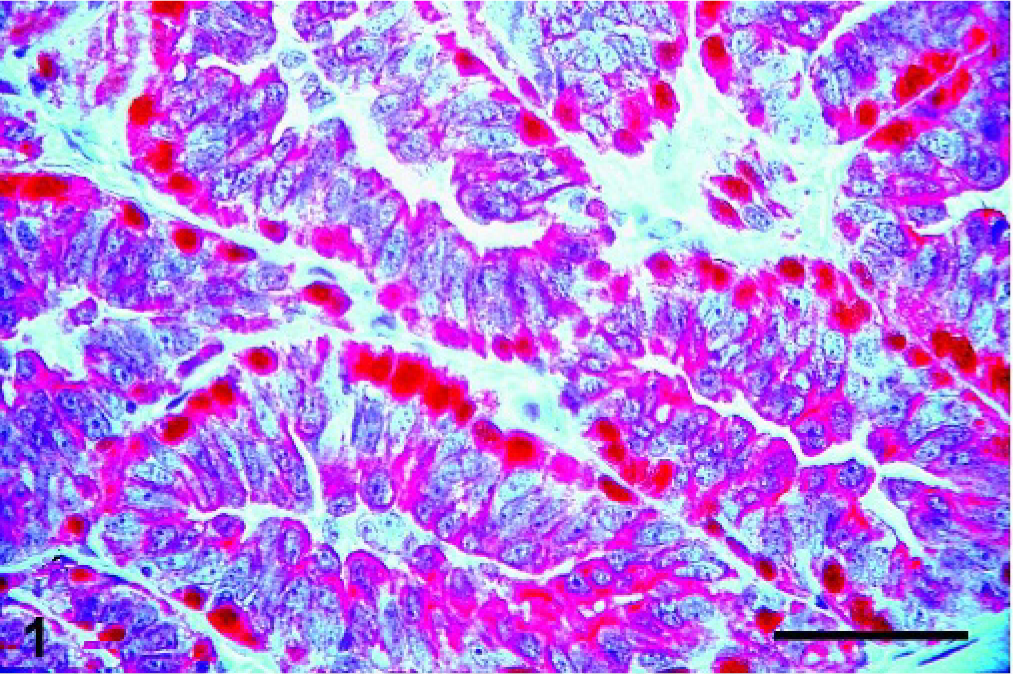
Mammary gland; Canis familiaris, case No 4. Benign mixed tumor with both basal epithelial and myoepithelial CK5-positive cells stained in red (cytoplasm), and p63-positive myoepithelial cells stained in brown (nucleus). Double immunolabeling method, CK5 and p63. Bar = 100 µ.
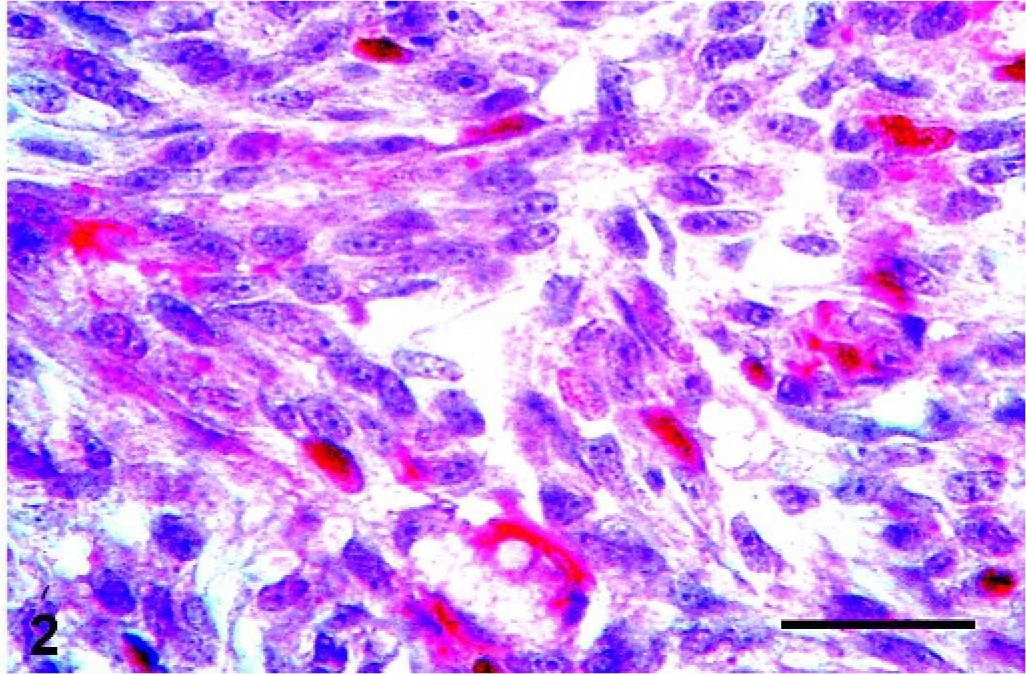
Mammary gland; Canis familiaris, case No 4. Benign mixed tumor showing some interstitial spindle cells in the mesenchymal component stained for CK5 (cytoplasmic staining, red) and p63 (nuclear staining, brown). Double immunolabeling method, CK5 and p63. Bar = 100 µ.
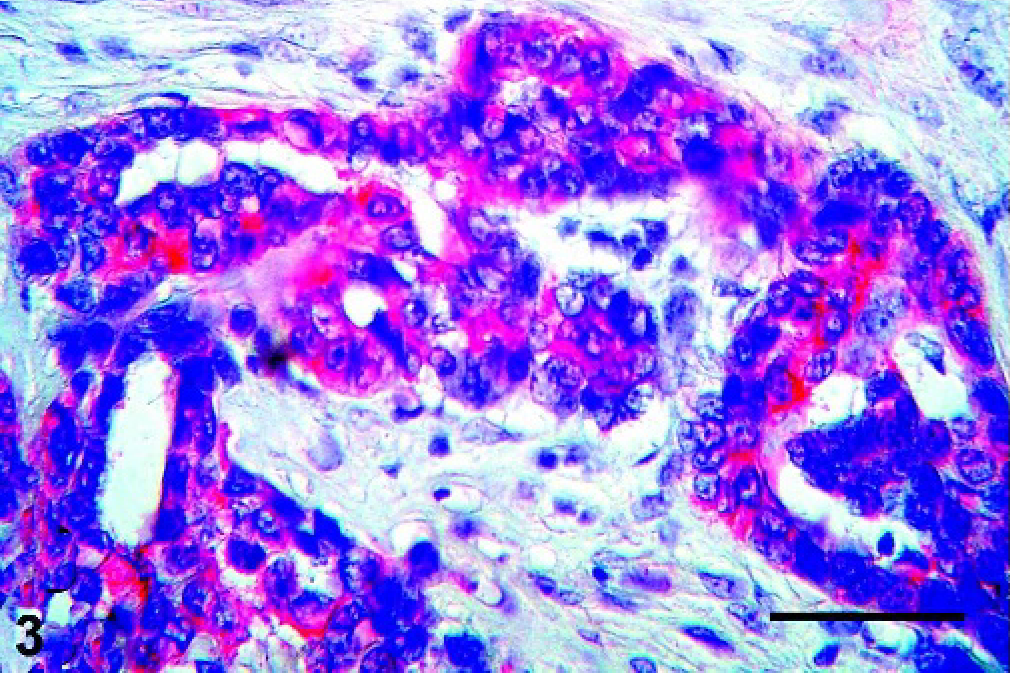
Mammary gland; Canis familiaris, case No 11. Carcinoma arising from benign mixed tumor showing positivity for CK5 only in the basal epithelial cells (cytoplasmic staining, red). Double immunolabeling method, CK5 and p63. Bar = 100 µ.
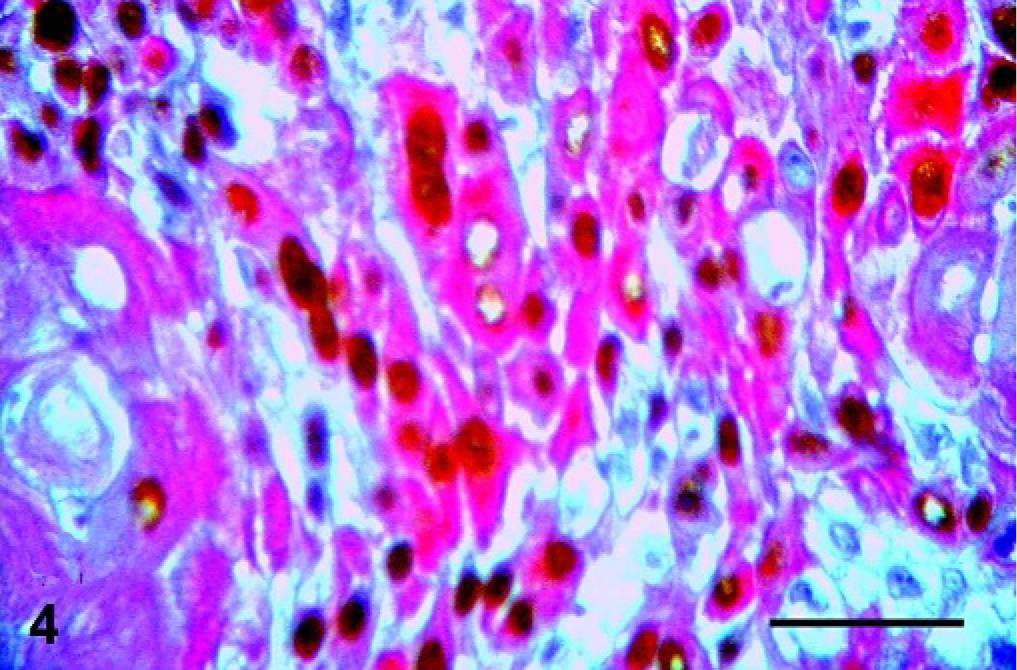
Mammary gland; Canis familiaris, case No 11. Carcinoma arising from a benign mixed tumor showing cells with squamous differentiation positive for both CK5 (cytoplasmic staining, red) and p63 (nuclear staining, brown). Double immunolabeling method, CK5 and p63. Bar = 100 µ.
Discussion
p63, a homologue of the p53 gene, is located on chromosome 3q27. This gene product is crucial for the maintenance of a stem-cell population in several epithelial tissues and is necessary for the normal development of epithelial organs, 16 including mammary glands. 31 Besides being expressed in the basal epithelial cells of other organs (e.g., skin, cervix, and prostate), 7, 25, 31 p63 is also expressed in the nuclei of myoepithelial cells of normal breast ducts and lobules. 3, 4, 19, 20, 23, 24, 27, 30 Cytokeratin 5, also known as CK5, is a 58 kD protein found in basal epithelial cells of different tissues, including epidermis, hair follicles, sebaceous and sweat glands, tongue, vagina, urothelium, prostate, and breast. 1 Cytokeratin 5 can also be detected in myoepithelial cells of normal breast. 8, 13
p63 is overexpressed in several primary human epithelial tumors, a fact indicating a possible role for p63 in the growth and the development of these tumors. 2, 7, 21, 31 This overexpression occurs mainly in squamous carcinomas, suggesting that p63 may act as an oncogene in this kind of neoplasm. 11 Like p63, CK5 stained the poorly differentiated areas, as well as the foci of squamous differentiation, suggesting that these markers may be used together in immunohistochemical panels to characterize squamous differentiation in poorly differentiated carcinomas. 11 In the present study, the strong relation between the expression of p63 and CK5 in myoepithelial cells of BMT, as is the case for human breast, 14 suggests that CK5 can also be considered a myoepithelial cell marker in the canine mammary gland and may be useful to discriminate benign neoplasms from malignancies in canine tumors.
p63 and other myoepithelial cell markers have been described in matrix-producing and metaplastic carcinomas of the human breast, suggesting that these tumors exhibit myoepithelial cell differentiation. 12, 14 p63 has been reported to be a valuable marker of myoepithelial cells in tumors of the canine mammary gland, suggesting a myoepithelial histogenesis of these tumors. 9 Our data show that the presence of some interstitial spindle cells of the mesenchymal component in BMT and CBMT expressing p63 and CK5, both known as myoepithelial cell markers in human breast, may reinforce the hypothesis of a myoepithelial origin of the mesenchymal component in mixed tumors of the canine mammary gland.
Though CK5 is considered to be a myoepithelial and a basal epithelial cell marker in the normal human breast, there is a subset of human-breast carcinomas that express CK5. The expression of CK5 indicates a basal epithelial phenotype 1, 5, 6, 18 and is associated with a poor prognosis in human breast carcinomas. 22, 26, 29 These CK5-positive cells were shown to represent progenitor or adult stem cells that give rise to the glandular and myoepithelial cell lineage. 5, 6 A strong relation between CK5 and p63 expression was recently observed in human-breast carcinomas, indicating that p63, like CK5, may be considered a marker for the basal phenotype of breast cancer. 22 However, our findings did not demonstrate co-expression of p63 and CK5 in basal epithelial cells in either BMT or CBMT. Moreover, basal epithelial cells were labeled by CK5 in BMT and mainly in CBMT, with no p63 labeling. CK5 may not represent an exclusive myoepithelial-cell marker in canine mixed tumors but rather a basal epithelial cell marker, such as occurs in the human breast. Probably basal epithelial cells may be related to the origin of the epithelial component of mixed tumors of the canine mammary gland.
In conclusion, because the three cellular phenotypes observed in normal mammary glands can be lost and converted to new phenotypes in mammary cancer, they may or may not present their original immunoprofile. It is possible that in breast cancer the basal phenotype preserves a myoepithelial immunoprofile, in contrast to mixed tumors of the canine mammary gland, in which p63 may represent a myoepithelial origin of the mesenchymal component and the epithelial component may originate from basal epithelial cells that exhibit CK5 expression. It is possible that mixed tumors of the canine mammary gland have components originating from common stem cells with a higher capacity for divergent differentiation than human breast cancer.
Footnotes
Acknowledgements
This research was supported by a grant from Fundação de Amparo à Pesquisa do Estado de São Paulo (FAPESP 03/02532-8). The authors wish to thank Auristela de Mello Martins for technical assistance.
