Abstract
A 1.5–year-old male Feist dog was presented to a veterinarian for reluctance to stand on the hind legs. Treatment included dexamethasone and resulted in a favorable initial response, but posterior paresis returned and progressed to recumbency, hyperesthesia, and attempts to bite the owner. The dog was euthanized. The brain was negative for rabies by fluorescent antibody analysis. Multiple foci of encephalitis were found in the cerebrum and particularly in the cerebellum. Protozoa morphologically consistent with Sarcocystis sp. were identified at sites of intense inflammation and malacia. Additionally, multiple schizonts were identified in areas without inflammation. Immunohistochemistry using both polyclonal and monoclonal antibodies specific for Sarcocystis neurona was strongly positive. No reaction to polyclonal antisera for Toxoplasma gondii or Neospora caninum was found. Polymerase chain reaction confirmed that the protozoa were S. neurona. Additional aberrant hosts for S. neurona other than horses have been identified, but S. neurona encephalitis has not been documented previously in the dog.
Keywords
Knowledge of Sarcocystis neurona has expanded dramatically since the initial recognition of a clinical neurologic syndrome in horses by Rooney in 1970 and the association of spinal cord lesions with protozoa in 1974. Identification of the definitive host, the opossum, and multiple intermediate hosts involved in the predator-prey life cycle has mostly occurred since 1998. The number of potential intermediate hosts has progressively expanded, and neurologic disease resembling equine protozoal myeloencephalitis (EPM) in horses has been reported in a variety of other mammals. These mammals include those that have been shown to be natural intermediate hosts (horse, raccoon, domestic cat, skunk, southern sea otter), other Equidae (pony, zebra), and miscellaneous species including mink, Pacific harbor seal, and Canadian lynx. 3, 5, 6 A single case report documented the dog as a potential intermediate host for S. neurona. 14 However, no previous reports of EPM-like infection in the dog have been published. This report presents a naturally occurring case of protozoal encephalitis attributed to S. neurona in the dog.
A 1.5-year-old, 20-pound male Feist crossbred dog was presented to a veterinarian with a complaint of reluctance to stand on the rear legs. This inability was a distinct deviation from the dog's conditioned response to stand and beg when offered a treat. The attending veterinarian detected no injury and initially suspected a herniated lumbar intervertebral disk, based on a survey radiograph. The dog was treated with dexamethasone and responded favorably for approximately 3 days. Subsequently, the hind limb weakness gradually returned over the next week. By day 10, the dog was recumbent and unable to rise, although he remained alert and responsive and continued to eat and drink normally. Examination on day 11 revealed hyperesthesia and attempts at biting when the dog was placed on the right side; no such response was elicited on the left side. Dexamethasone injections were repeated on day 11 with no response. Due to progressive deterioration, the dog was euthanized on day 15.
Prior history indicated that the dog had received both rabies and canine distemper virus booster vaccinations 4 months prior to the onset of neurologic signs. The dog had been owned since 7 weeks of age and had been in good health. One other dog was in the household and did not experience any abnormality. Both dogs mostly lived indoors but were turned out for about 1 hour at least twice daily in a large fenced enclosure in a small rural North Carolina town.
Necropsy examination was conducted by the referring veterinarian. Half of the brain was submitted for rabies fluorescent antibody examination. Sections of heart, lung, liver, kidney, pancreas, small intestine, colon, and brain were submitted in neutral buffered 10% formalin. Skeletal muscle and spinal cord were not submitted. Tissues were processed routinely, embedded in paraffin, sectioned at 5 μm, and stained with hematoxylin and eosin. Formalin-fixed, paraffin-embedded brain sections were examined by immunohistochemistry with antibodies for Toxoplasma gondii, Neospora caninum, Sarcocystis falcatula (polyclonal), and S. neurona (polyclonal) as previously described 12 using the anti-rabbit EnVision+ system, and S. neurona (2G5 monoclonal supernatant; provided as a courtesy by Dr. Antoinette Marsh and the University of Missouri, College of Veterinary Medicine, Columbia, MO) 10 at a 1:700 dilution using the anti-mouse EnVision+ system (DAKO Corp, Carpinteria, CA). Positive controls included tissues containing the respective parasites to which the antibodies were directed in either experimentally infected or naturally infected host tissues. These same infected tissues were used as negative controls for closely related protozoans (i.e., T. gondii and N. caninum served as negative controls for Neospora and Toxoplasma polyclonal antisera, respectively), and S. falcatula–infected tissues served as a negative control for S. neurona antibodies.
The immunohistochemistry control for the S. neurona polyclonal rabbit antisera was mouse brain from nude/nude mice inoculated with 107 culture-derived merozoites of the University of California, Davis SN-1 isolate and euthanized on postinfection day (PID) 23. 7 The control for the monoclonal S. neurona antibody was equine spinal cord from a naturally occurring case of EPM confirmed previously by immunohistochemistry. The control for the S. falcatula polyclonal rabbit antisera was lung from a budgerigar inoculated with 105 culture-derived merozoites of S. falcatula. 9 The T. gondii control was brain and lung from Swiss Webster mice inoculated with 103 to 105 culture-derived tachyzoites of strain ME49. The control for N. caninum was brain from Swiss Webster mice preinoculated with 6 mg of methylprednisolone intraperitoneally, inoculated with 106 tachyzoites of the BPA-1 isolate from the University of California, Davis, and euthanized on PID 20 to 43. 1
Gross necropsy lesions were not seen. The dog was reported to be in good body condition at the time of death. Rabies fluorescent antibody examination was negative. Histologic lesions were confined to the brain; the spinal cord was not submitted for examination. Histopathologic examination revealed widespread asymmetric encephalitis predominantly in gray matter and often associated with necrosis in the cerebrum, cerebellum, and brainstem. Inflammation was most pronounced in the cerebellum (Fig. 1). Within the neuropil of the cerebrum, foci of necrosis were associated with focally intense collections of macrophages, neutrophils, a few lymphocytes, and occasional eosinophils (Fig. 2). Small blood vessels with hypertrophied endothelial cells marginated and extended into these areas of necrosis and were frequently cuffed by numerous lymphocytes, a few plasma cells, and macrophages. Small discrete collections of macrophages, probable microglia, and some intermingled neutrophils occurred predominantly in gray matter (Fig. 3). Similar dense inflammatory cell accumulations, often with neutrophils predominating, were found in necrotic foci in the cerebellar cortex. These inflammatory infiltrates often expanded the leptomeninges of the cerebellum, particularly at sites overlying malacic folia. Scattered small foci of necrosis with mixed inflammatory cell infiltrates including neutrophils and macrophages, nodules of microglia, and a few lymphocytes predominated in gray matter nuclei in the brainstem.
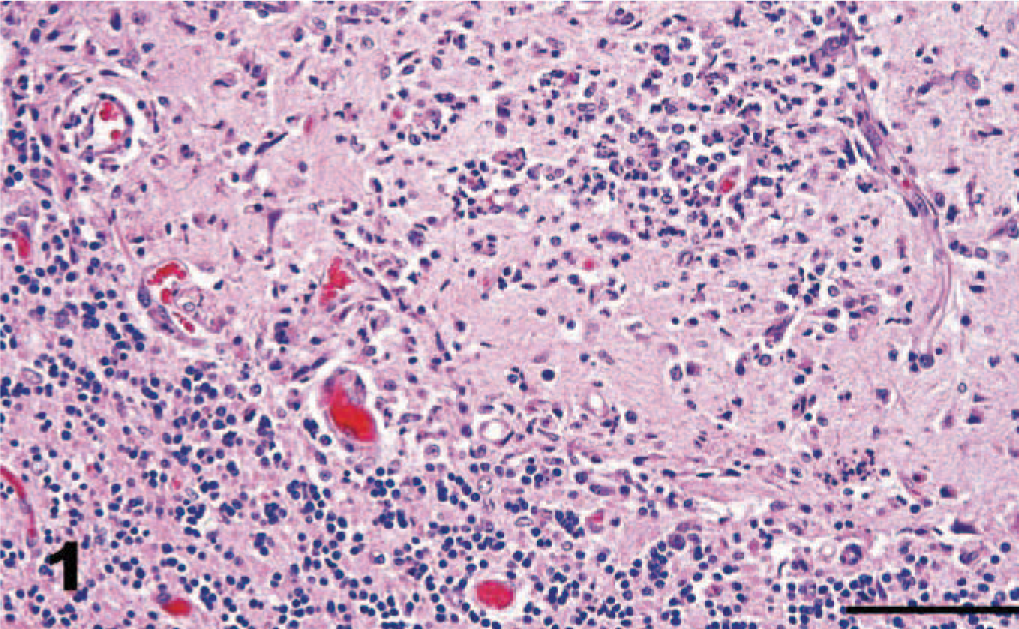
Cerebellum, dog. Dense inflammatory cell accumulations, associated with necrosis, in the cerebellar cortex include predominantly neutrophils intermingled with fewer macrophages and a few lymphocytes. HE. Bar = 100 μm.
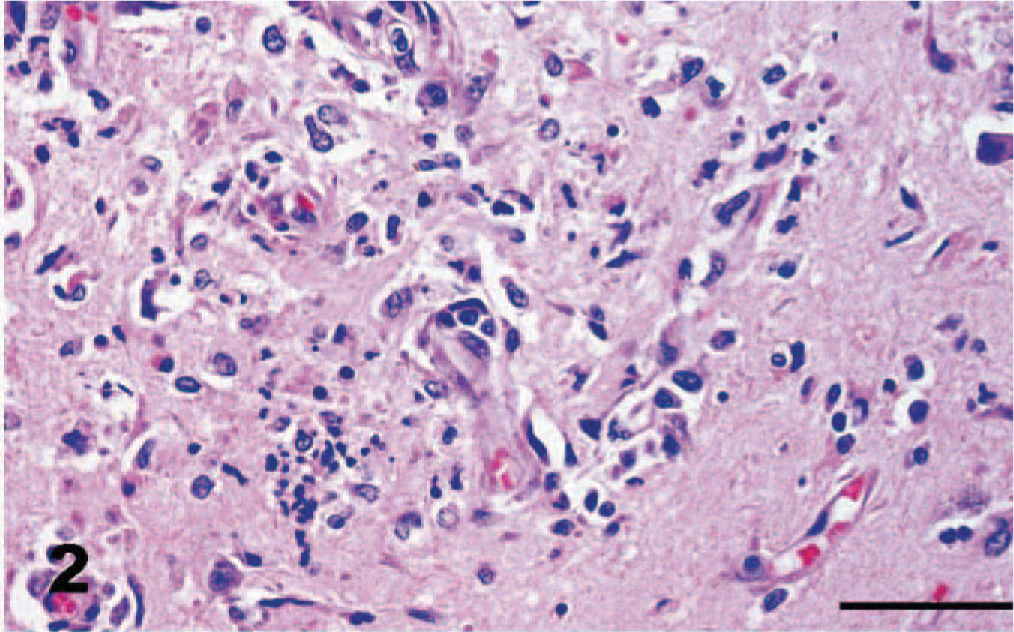
Cerebrum, dog. Small blood vessels with hypertrophied endothelial cells marginate and extend into zones of necrosis and inflammation. HE. Bar = 50 μm.
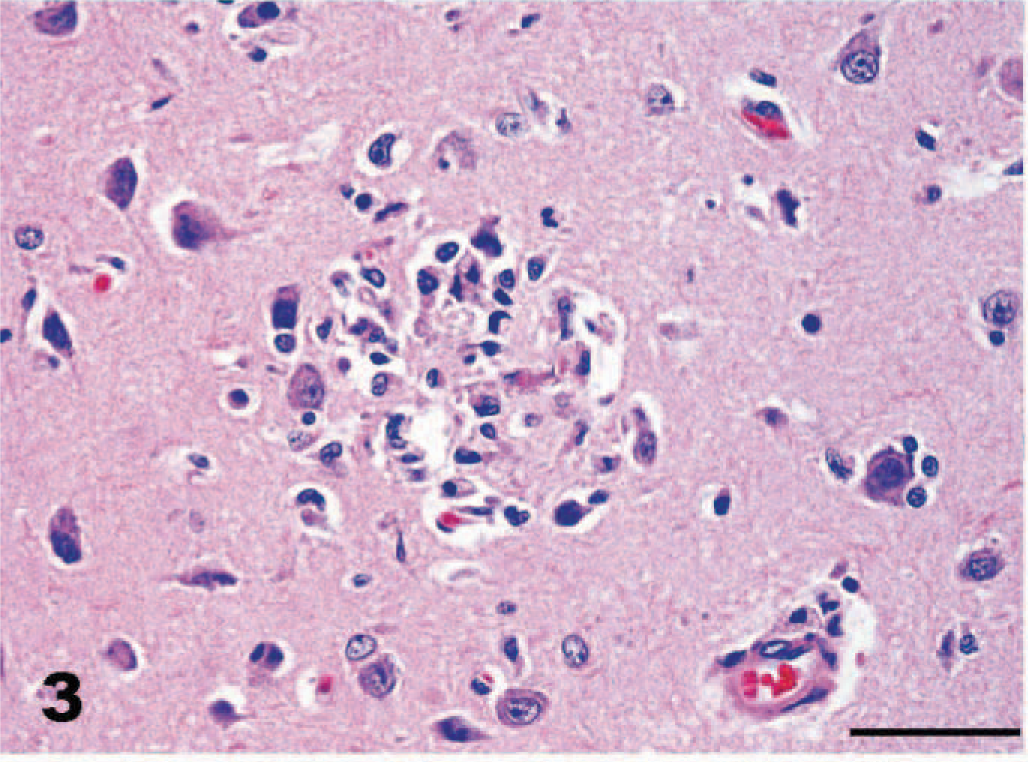
Cerebrum, dog. Small discrete focus of necrosis with accumulation of macrophages and microglia in cortical gray matter. HE. Bar = 50 μm.
Protozoal organisms were numerous and generally occurred within areas of intense inflammation; occasionally, schizonts were identified at the periphery of foci of inflammation. Most of the schizonts were made up of clusters of haphazardly arranged, oval to slightly elongated or crescentic protozoal zoites, approximately 3 × 2 μm, based on examination with light microscopy of paraffin-embedded tissue (Fig. 5). Schizonts measured up to 20 μm in diameter. Some schizonts formed distinct rosettes with radially arranged protozoal merozoites surrounding central granular acellular material consistent with residual bodies (Fig. 4). Additionally, larger uninucleate ovoid organisms with prominent nuclei, consistent with immature schizonts, as well as immature schizonts with large multilobated nuclei were identified, particularly on immunohistochemistry (Figs. 6, 7). Schizonts and merozoites appeared to lie within neural cells and glial cells, although often this could not be determined with certainty. Individual free merozoites were also commonly seen with HE-stained sections or by immunohistochemistry. Neither protozoa nor inflammation were found in any visceral organ tissue in HE sections; skeletal muscle was not available for examination.
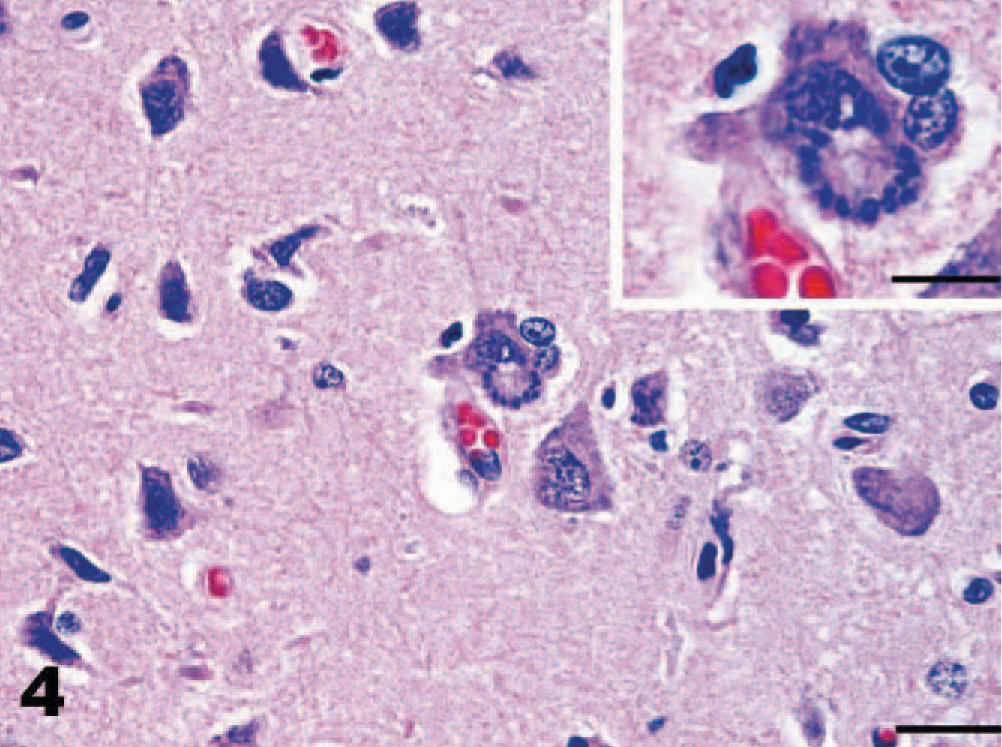
Cerebrum, dog. Occasional schizonts dividing by endopolygeny in cerebral neurons. Notice budding rosette-like arrays of merozoites surrounding granular acellular material consistent with a residual body. Bar = 20 μm. Inset of higher magnification of affected neuron. HE. Bar = 10 μm.
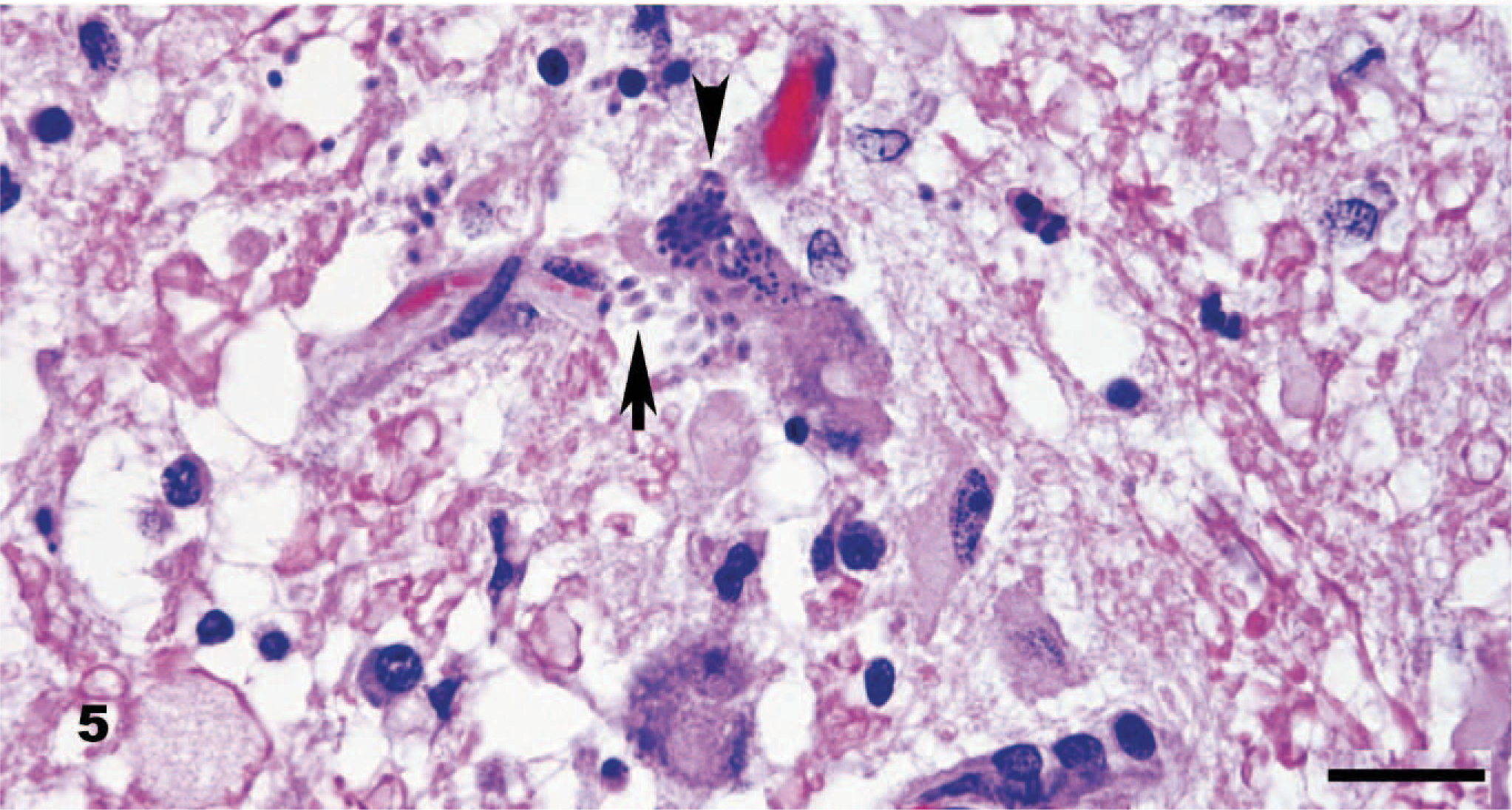
Brainstem, dog. Intracellular schizonts in a neuron (arrowhead) and adjacent free merozoites (arrow) are typical of the numerous protozoal organisms identified. Elongated merozoites range from 3 to 5 × 2 μm and have identifiable nuclei. HE. Bar = 20 μm.
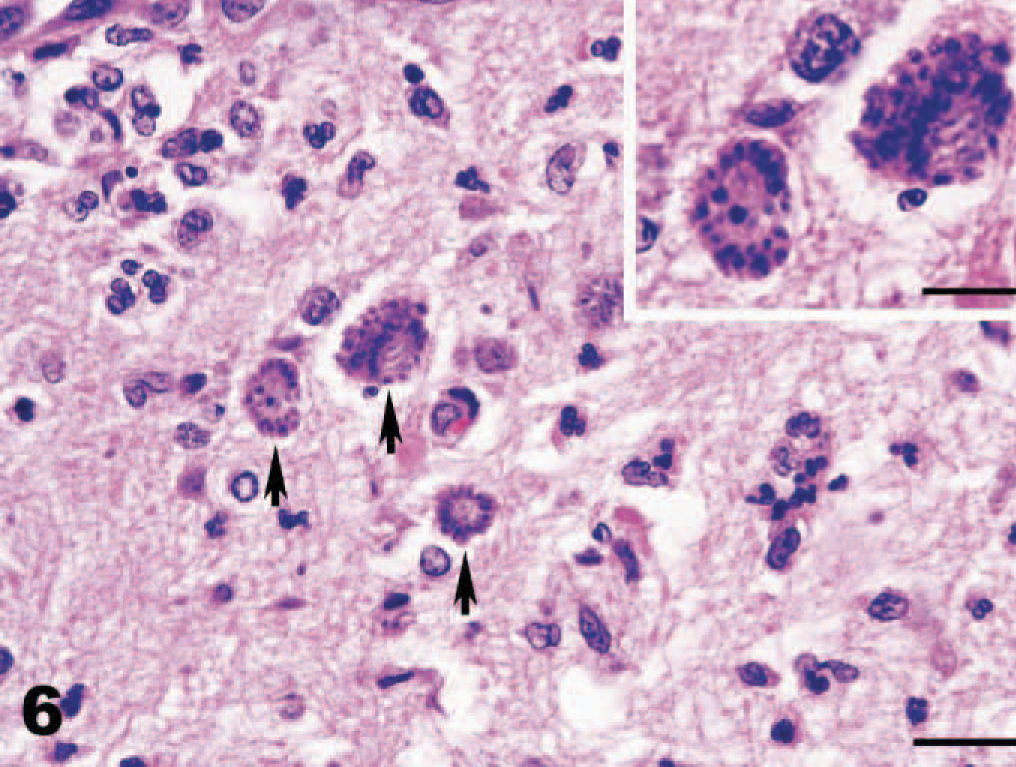
Cerebellum, dog. Multiple schizonts dividing by endopolygeny forming distinct rosettes of merozoites (arrows). Bar = 20 μm. Inset of higher magnification. HE. Bar = 10 μm.
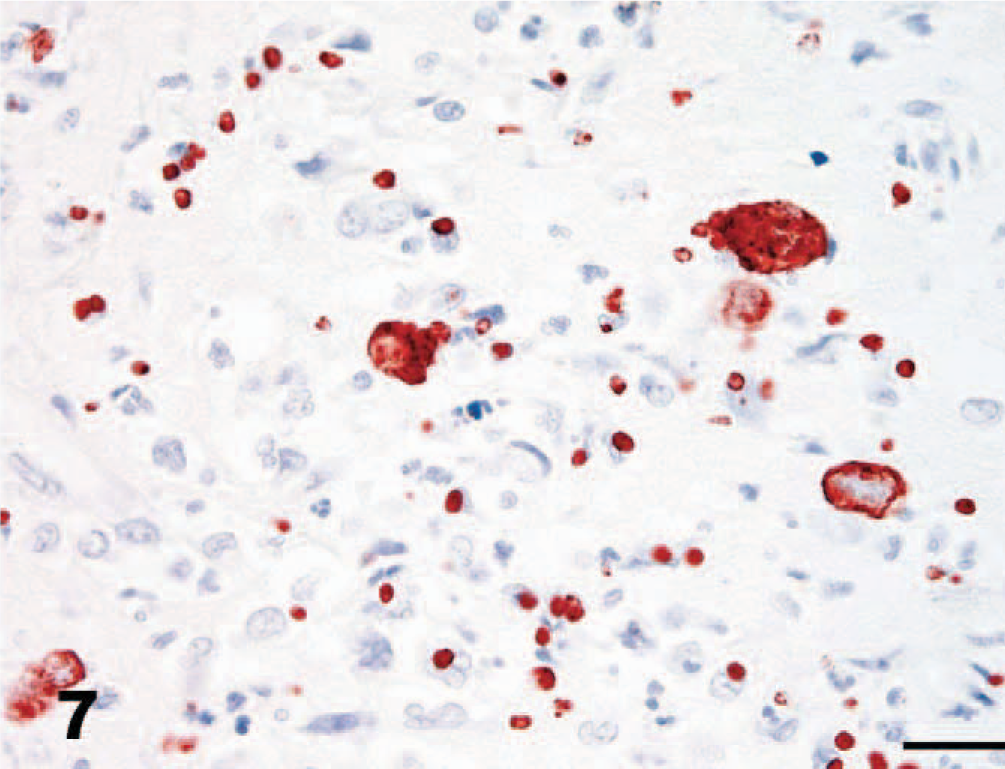
Cerebellum, dog. Immunohistochemistry for S. neurona. Protozoal organisms stain intensely with monoclonal S. neurona antibody. Notice intense specific staining of large schizonts and especially of the immature schizonts and merozoites, which are not apparent or recognized only with difficulty in Fig. 6. S. neurona (2G5 monoclonal) and EnVision+ system. Bar = 20 μm.
Protozoal organisms stained intensely with both polyclonal and monoclonal S. neurona antibodies (Fig. 7). No staining occurred with antibodies to T. gondii or N. caninum. Occasional minimal faint positive staining was detected with the S. falcatula antibody.
For further characterization of the organism, DNA was extracted and amplified by polymerase chain reaction (PCR) from formalin-fixed and paraffin-embedded tissues. Briefly, paraffin tissue sections were placed in 2.0-ml microfuge tubes and heated at 70°C for 15 minutes followed by the addition of 180 μl of ATL buffer (QIAGEN, Valencia, CA). The tubes were heated at 70°C for several hours with frequent vortexing and then centrifuged at 14,000 rpm (20,800 × g) for 3 minutes. Forty microliters of proteinase K was added, and a tissue extraction protocol (QIAGEN) was followed except that the DNA was eluted with 60 μl of 95°C Millipore H2O in the final step.
Two microliters of eluted DNA was used in the first round of a nested 50-μl PCR reaction. The PCR amplifications were carried out in a GeneAMP 2400 thermocycler (Perkin Elmer, Waltham, MA) under the following reaction conditions: 5.0 μl of 10X PCR buffer with MgCl2 (15 mM), 200 μM dNTPs, 1 μM each primer: ITS-5 (5′-GGAAGTAAAAGTCTAACAAGG-3′), ITS-2 (5′-GCTGCGTTCTTCATCGATGC-3′), 7 and 1.5 U of Taq polymerase. After initial denaturation of templates and primers (94°C for 3 minutes), 35 cycles of the following conditions were used: 95°C for 40 seconds, 58°C for 40 seconds, 72°C for 90 seconds, and followed by a 5-minute extension at 72°C. In the second round of the nested reaction, 1 μl of product DNA from the first round was used as the DNA template. Reaction times and conditions were identical to the first round, except the following primers were used: ITS-snFi (5′-GTACTAGTGCTGCTCACC-3′) and ITSbFor (5′-TCGCTGAAAATAGTAAGTTC-3′). 8
Ten microliters of PCR product were electrophoretically separated on a 2% agarose gel stained with ethidium bromide and visualized under ultraviolet light. PCR products from positive bands were cleaned using the ExoSAP-IT PCR clean-up system (USB Corp., Cleveland, OH) and sequenced at the Division of Biological Sciences sequencing facility at the University of California, Davis. The following 321-bp partial ITS-1 DNA sequence was obtained: TGAGTTACCCGGGATGATGTCGTCAAGGGTATCATGATGATGATGTTGGTGTGGTTTATAATATGAAGGCGTGCTTGGGTCTTTTCCTTCTGAGAAATTAGGGTGAAGCCAGGGATCTGATGATGATAAAAGATATACAGGCTGTGTCACATGTAGAGAGTAGAACGTTCTCTTGTGTGTGCCCCTACACATGGTACATGTTCAAAATGAACGTGTCTATGTGTGAGGTACGTGCATGTGCGCGTTGTTACCCCCTTCTTTGGCCGCCTTTTTATCGTCAGCAATCCTTCGGCATCCTCCTTTTTCTTTTTTTTTTCCCAC. A search of GenBank showed the sequence to be 100% identical to several published S. neurona ITS-1 sequences (GenBank accession numbers DQ084486.1, DQ084484.1, and AY082644.1).
The diagnosis of S. neurona encephalitis was supported by the limitation of the infection to the central nervous system, the presence of an EPM-like encephalitis with necrosis, morphologically consistent schizonts and merozoites, strong positive immunoreactivity to S. neurona–specific antibodies and negative immunoreactivity to Neospora and Toxoplasma, and PCR analysis. The faint reaction to the polyclonal S. falcatula antibody is not unusual, reflecting some cross-reactivity between these two protozoans. This cross-reactivity has been speculated to be attributed to factors such as shared antigens. S. neurona and S. falcatula are known to be closely related but biologically different organisms. 2 The rosette-like array of merozoites is consistent with the division of the parasite by the process of endopolygeny, a pattern consistent with a Sarcocystis species. According to Dubey et al., 3 this form of division involves lobulation of the nucleus with interconnections of the lobes by chromatin strands, resulting in formation of more than 4 merozoites and arrangement of the merozoites in groups.
The possibility of induction of clinical neurologic disease in this dog subsequent to systemic corticosteroids is plausible. Several stressors such as injury, surgery, and parturition have been suggested as possible causes of transient immunosuppression resulting in progression of clinical EPM in horses. The higher incidence of EPM in horses, particularly Thoroughbreds and show horses, in association with the stressors of racing, strenuous exercise, transport, and other use-associated factors, is well established. Stress-associated effects on the immune system have been implicated. Use of exogenous corticosteroids in S. neurona–infected horses is controversial at best. Dubey et al. 3 states that corticosteroids may worsen clinical signs and should be avoided in horses suspected to have EPM. Alternatively, other authors have reported that clinical disease is less severe in horses given corticosteroids, due to abatement of associated inflammation. 11 In the present case, initial clinical neurologic signs began before corticosteroids were administered. It is impossible to know with certainty whether the initial clinical signs of the dog were truly related to protozoal infection. Although the corticosteroid therapy was rather short in duration, it may have enhanced protozoal proliferation and thus the clinical progression of the disease in the dog. Initial transient improvement was likely associated with some reduction of inflammation or edema.
A recent report confirmed the presence of a S. neurona–like infection in a 6-year-old Labrador Retriever with myositis. 14 The polymyositis was associated with marked and severe muscle atrophy. Sarcocysts were identified in multiple skeletal muscles, chiefly in myocytes peripheral to areas of inflammation. DNA studies, including PCR analysis with differentiating primers, confirmed the identity of S. neurona. The sarcocysts identified were categorized as immature microcysts, and therefore the possibility was raised that the Sarcocystis infection could have been a sequela of immunosuppressive therapy for polymyositis rather than the cause of the myositis. Nonetheless, the muscle stages of S. neurona were verified. This Labrador Retriever did not have a history of neurologic disease, and central nervous system infection was not found.
Among other Sarcocystis species, Sarcocystis canis is also reported to cause infections in dogs. This protozoan was first described and named in 1991. 4 Only the schizont stage has been identified. Acute canine sarcocystosis due to S. canis has predominantly been reported in very young dogs, often as fatal hepatitis and often in association with concurrent diseases such as canine distemper and blastomycosis. Intralesional protozoa have predominated in visceral tissues, including lungs, liver, kidneys, spleen, skin, and lymph nodes; brain involvement with meningoencephalitis has been reported in 3 S. canis cases. The protozoan seen in this dog did react with both polyclonal antisera and a monoclonal antibody raised against S. neurona protozoa. PCR excluded the possibility of S. canis. Furthermore, the protozoan seen in this dog was not found outside the brain, unlike reports with S. canis–like protozoans. 13, 15
To our knowledge, S. neurona encephalitis has not been documented previously in the dog. The morphology, central nervous system tissue tropism, immunohistochemical reactivity, and DNA analysis support the definitive diagnosis of S. neurona in this dog. The findings suggest that the dog likely represents one further susceptible intermediate host species of S. neurona.
Footnotes
Acknowledgements
We thank the California Animal Health and Food Safety Laboratory Davis histology technicians for their technical assistance in performing the immunohistochemistry and the North Carolina Rollins Diagnostic Laboratory histology technicians for histopathologic sections. We acknowledge the University of Missouri, College of Veterinary Medicine in collaboration with Antoinette Marsh, The Ohio State University, for use of the 2G5 monoclonal Sarcocystis neurona antibody.
