Abstract
A 4-year-old male Labrador Retriever dog was presented with a 10-day history of tetraplegia, depression, and absent postural reflexes. The cerebrospinal fluid was positive for Leishmania spp. DNA. At necropsy, a 2-cm long mass was observed adhered to C7 and C8 left spinal nerves. Microscopically, nerve fiber destruction together with mixed inflammatory infiltration was observed in the spinal nerves. Cervical spinal cord sections showed multifocal, diffuse granulomatous inflammation in the white matter. In the brain, perivascular infiltrates were observed in some areas together with subtle pallor of the parenchyma. Immunohistochemistry for Leishmania infantum confirmed the presence of amastigotes in the spinal nerves, spinal cord, brain parenchyma, and choroid plexuses. The current study describes the presence of Leishmania amastigotes in nervous tissue inciting radiculoneuritis, myelitis, and mild meningoencephalitis, suggesting a likely route by which L. infantum amastigotes reach and affect the central nervous system parenchyma.
Canine leishmaniasis (CL) is a cutaneous, mucocutaneous, or visceral disease caused by intracellular Leishmania protozoan parasites that are transmitted by the bite of female sand flies. Domestic dogs are the main reservoir hosts. The clinical signs are very variable, ranging from subclinical infections to generalized disorders characterized by hyperthermia, anemia, weight loss, polydipsia, hepatomegaly, splenomegaly, hypergammaglobulinemia, generalized lymphadenopathy, cutaneous lesions, and other lesions.2,4
In visceral CL, parasites and their lesions are found throughout the host organism, producing proliferative inflammatory reactions causing an immune imbalance. Among the inflammatory infiltrates, macrophages predominate, and occasionally amastigotes are found within them. Many organs have been observed to be affected, especially the lymph nodes, liver, spleen, and skin. 2 It is thought that a humoral response and deposition of immune complexes are likely responsible for the pathogenesis2,4 including the systemic necrotizing vasculitis that has been described in affected dogs. 17
Occasionally, neurological signs and/or histopathological lesions have been observed affecting the central nervous system (CNS) in CL,2,9,11 giving rise to the term cerebral leishmaniasis. 8 However, except for the choroid plexuses 13 and meninges, 19 Leishmania amastigotes, to the authors’ knowledge, have not been observed in either the CNS parenchyma or peripheral nerves in CL. The current study described a dog with radiculoneuritis, myelitis, and mild meningoencephalitis associated with Leishmania spp. amastigotes in the peripheral and central nervous systems.
A 4-year-old male Labrador Retriever suffered an episode of left hemiparesis 9 months before presentation. Blood analysis at that time showed thrombocytopenia and mild hyperglobulinemia. Enzyme-linked immunosorbent assay (ELISA) a for anti-Leishmania antibodies and indirect fluorescent antibody test (IFAT) b for Leishmania were negative. The animal responded favorably to chronic treatment with prednisolone. c Seven months later the animal relapsed, and blood analysis revealed anemia, hyperproteinemia, hypoalbuminemia, and hyperglobulinemia. A second ELISA a test was performed and the result was again negative. Serology for Toxoplasma and Neospora was negative. At that time, treatment was begun with clindamycin, d and an improvement in the clinical signs was noted.
Forty-five days later, the dog was referred to the Al Sur Veterinary Hospital with a 10-day evolution of tetraplegia. Clinical examination showed hypothermia, dehydration, tachypnea, and cachexia. The animal was depressed, and postural reactions were absent. Carporadialis and flexor reflexes in the forelimbs were absent; bilateral Horner syndrome was also observed. Atrophy of the supra and infraspinous temporal, deltoid, bicep, and triceps brachii muscles was observed. Based on the clinical presentation, a lesion was suspected in the cervicothoracic spinal segment C6-T2. Differential diagnoses included disc disease, fibrocartilaginous embolism, infectious meningoencephalomyelitis, and neoplasia.
Blood analysis revealed anemia, leukocytosis, and hyperproteinemia. Cerebrospinal fluid (CSF) analysis showed mixed pleocytosis (23 cell/µl, proteins: 128.91 mg/dl), and Pandy test was positive. The CSF was positive by IFAT b performed against Leishmania infantum (1/80) and also for antigen detection by quantitative polymerase chain reaction e (1.67 × 106 copies of kDNA/ml, with an estimation of 1–2 parasites per µl). Finally, the animal died due to deterioration of health. A complete necropsy was performed.
A 2 cm × 1 cm × 0.4 cm white mass with a 5 mm in diameter dark-brownish spot was observed adhering to the C7 and C8 left spinal nerves (Fig. 1). Because no gross lesions were observed in other organs, the CNS was the only material sent to the laboratory of histopathology. Along with the lesions, the entire spinal cord and brain were fixed in neutral buffered 10% formalin and routinely processed for histological evaluation. Four micrometer–thick paraffin sections were stained with hematoxylin and eosin. For immunohistochemistry (IHC), anti–L. infantum antibodies f were used at 1/1,600 dilution to detect the organism, as previously described. 7
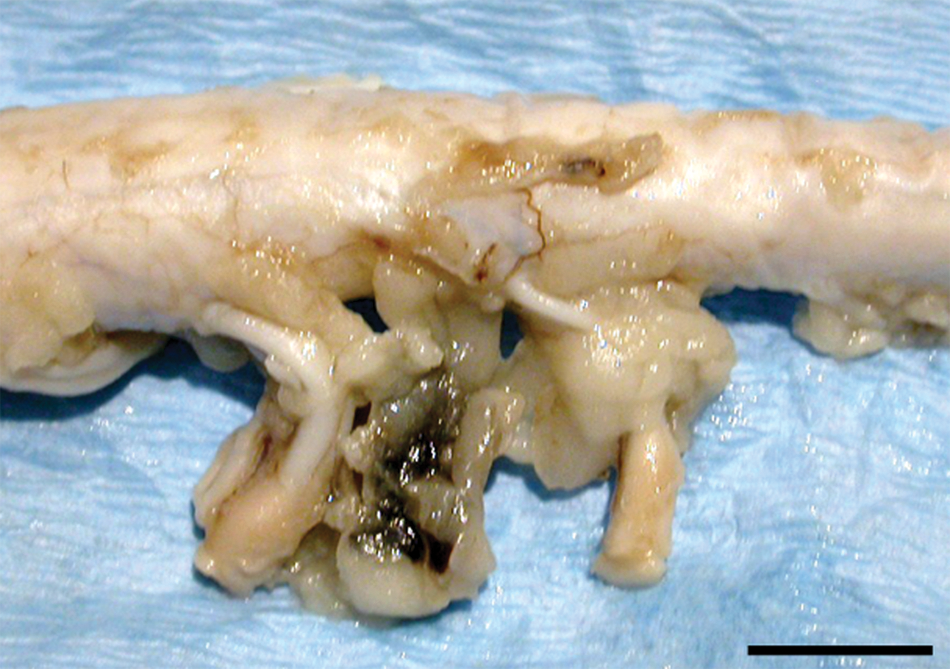
Cervical spinal cord section of a dog, showing the dark brownish mass between C7 and C8 left spinal nerves. Bar = 1 cm.
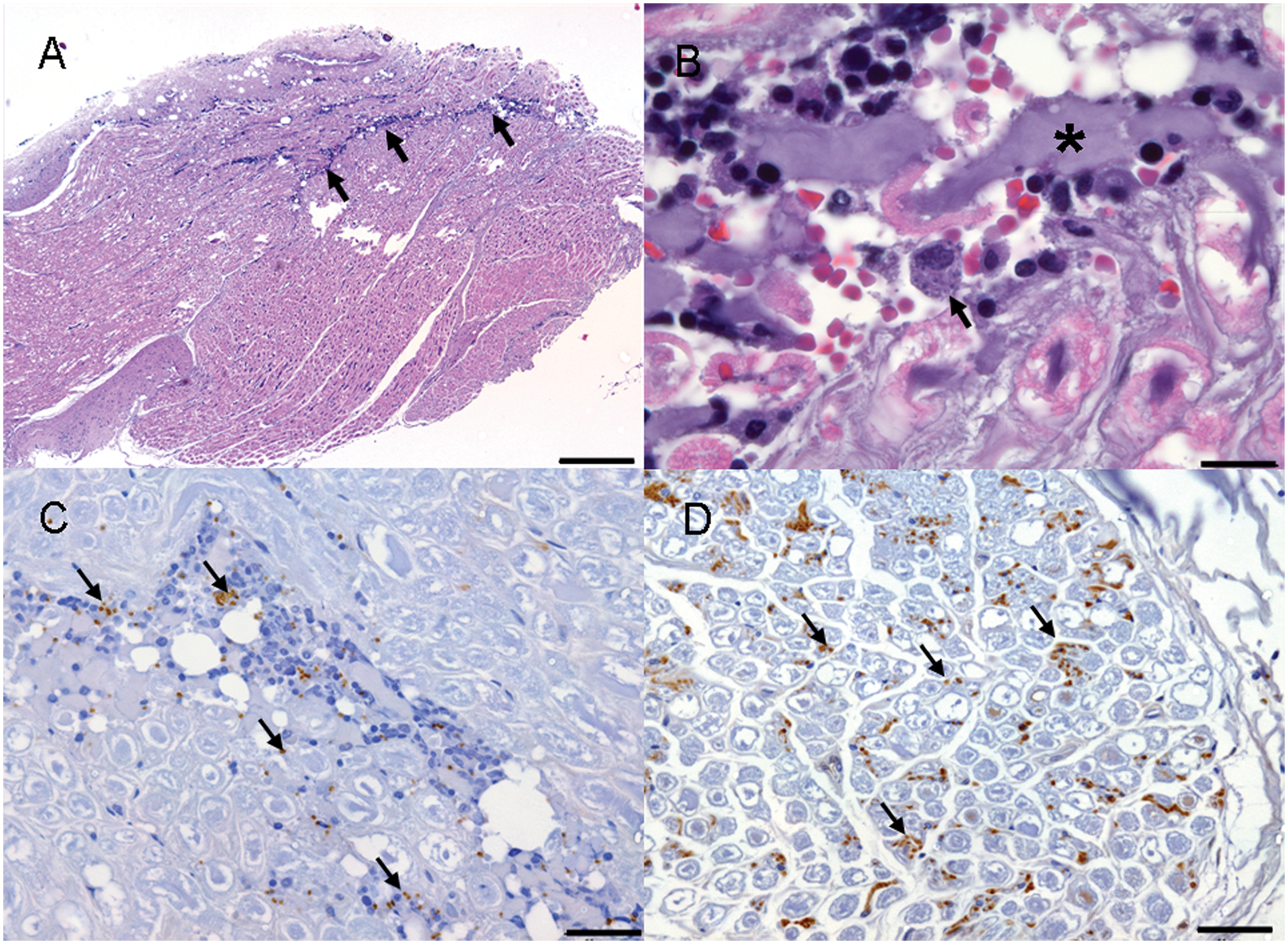
Microscopically, the mass adhering to the cervical spinal nerves corresponded to adipose tissue and the dark-brownish spot to a focus of hemorrhage. In the adjacent spinal nerves, a mixed inflammatory infiltration mainly composed of macrophages, lymphocytes, and plasma cells, and fewer multinucleated cells, neutrophils, and eosinophils (Fig. 2A), was observed. Some macrophages showed a large cytoplasm filled with 1.5 µm in diameter, round, pinpoint structures composed of a round nucleus, light cytoplasm, and an often visible kinetoplast that were compatible with Leishmania amastigotes (Fig. 2B). The nerves presented asymmetrical bilateral nerve fiber destruction in the form of fragmented fibers, axonal swellings (spheroids; Fig. 2B), and the presence of Renaut bodies. Cervical spinal cord sections presented severe necrosis of the dorsal funiculi with multifocal, diffuse presence of activated macrophages with foamy cytoplasm (gitter cells). Small perivascular mononuclear cuffs were observed in this area near the surface of the dorsal horn. A few gitter cells and perivascular cuffs were also observed in the lateral and ventral funiculi. No lesions were observed in the rest of the spinal cord.
In the brain, perivascular cuffs, composed mainly of gitter cells and lymphocytes, were observed in some areas, together with a slight pallor of the parenchyma (Fig. 3A). The white matter was mainly affected in the internal capsule, corona radiata, optical tracts, cerebral peduncles, and right dorsal parahippocampal gyrus. In some areas, such as the neocortical periventricular white matter and corpus callosum, small mononuclear perivascular cuffs were observed. A discrete mononuclear infiltrate was observed surrounding a few leptomeningeal blood vessels.
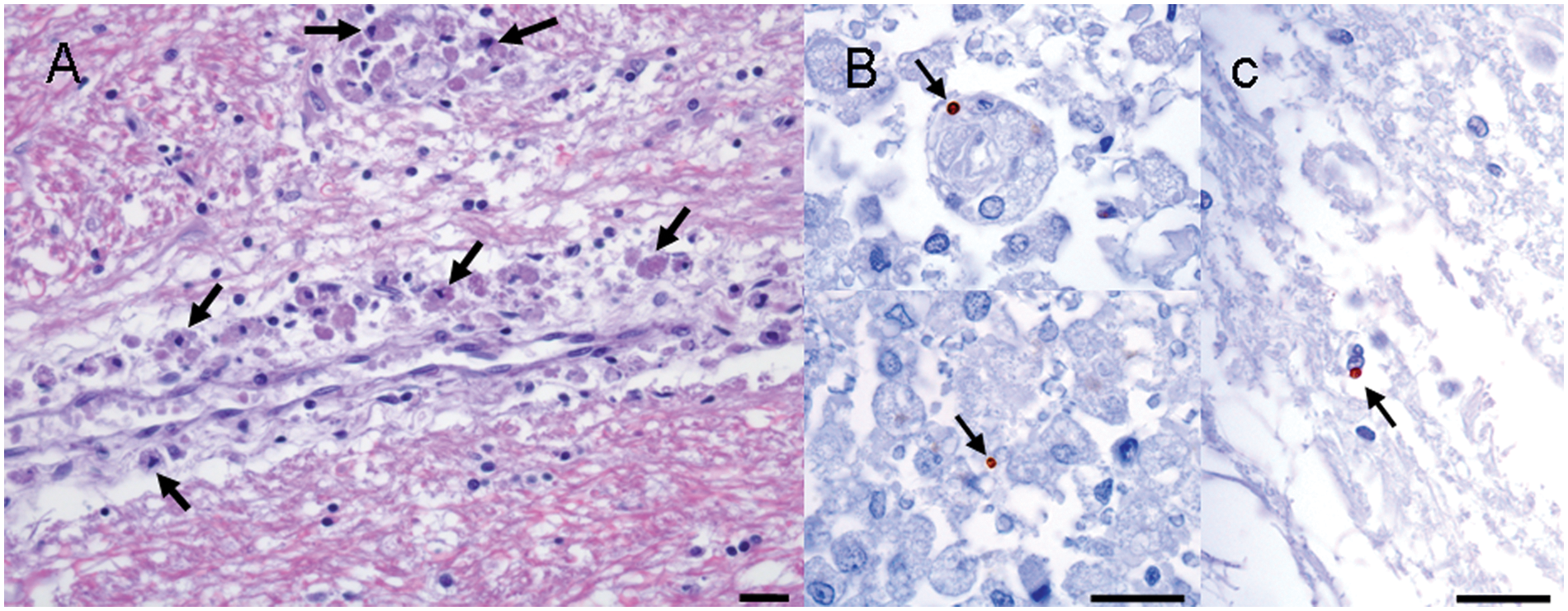
Leishmania IHC confirmed the presence of abundant extra- and intracellular (within macrophages) amastigotes in the peri- and endoneurium of the spinal nerves (Fig. 2C). It is worth noting that free amastigotes infiltrating the endoneurium were also observed in spinal nerve areas without the presence of inflammatory infiltrate (Fig. 2D). Sporadic amastigotes were also observed in the parenchyma of the spinal cord (Fig. 3B) and thalamus (Fig. 3C) and in choroid plexuses. Parasites were observed in blood vessels in the leptomeninges and the brain parenchyma.
Herein the authors present a CL case of peripheral and central nervous systems infection caused by L. infantum with presence of amastigotes in spinal nerves, spinal cord, brain parenchyma, and choroid plexuses. In previously reported spontaneous CL cases, neurological symptoms such as generalized seizures, motor deficiencies, vestibular and cerebellar involvement, visual alterations, signs of paralysis of cranial nerves, circling, vocalization, paraparesis, and, in the last stages, paralysis of the hind limbs, have been observed.2,9,11 However, only in a few of those cases have associated histopathological findings, such as neuronophagia, gliosis, leptomeningitis, spongiosis, vascular congestion, perivascular lymphoplasmacytic infiltrate, and focal microhemorrhages been observed.3,8,9,12 In spontaneous CL cases, amastigotes have been detected in brain blood vessel walls, suggesting the presence of circulating Leishmania antigens, 9 and anti-Leishmania immunoglobulin Gs have also been found in the CSF.8,11,12,19 However, except for the meninges 19 and choroid plexuses,8,13 in none of the previously reported immunohistochemical studies has Leishmania amastigotes been detected in the CNS.8,9,11,13 Because Leishmania antibody titers have been observed to be very similar in CSF and blood, some authors have suggested that antibodies found in CSF might be produced in lymphoid tissue from which the antibodies access the CNS.8,11 A study published in 2011 6 reported a dog with paraparesis caused by a Leishmania extradural inflammatory granuloma in the vertebral canal, with extension to the epidural space and compression of the spinal cord. However, amastigotes were only observed inside macrophages in the extradural granuloma and free in the interstitium. 6 Detection of amastigotes in nervous tissue is very difficult; over the course of many years in the authors’ laboratory, the authors have diagnosed thousands of CL cases, and except for a case of systemic vasculitis, 17 the presence of other nervous lesions or Leishmania amastigotes in nervous tissue has never before been observed. In human leishmaniasis, CNS involvement is rare; it has been observed in immunosuppressed people as a consequence of extension of a contiguous infection, most often in the paranasal sinuses, and usually in conjunction with disseminated infection. 20
Microbes seem to use several routes to enter the CNS including pathogen-directed penetration of choroid plexus epithelial cells, leading to the CSF; cerebral capillary endothelial cells, leading to the brain parenchyma; disrupting the blood-brain or blood-CSF barriers; and, finally, being transported across these barriers within infected leukocytes. 18 Researchers in a previous study 1 infected mice with subcutaneous injection of L. amazonensis amastigotes. The researchers showed nervous parenchyma infection with presence of amastigotes, thus demonstrating the ability of these organisms to reach the brain, and it was suggested that parasites might have arrived to the CNS via loaded leukocytes. 1 In CL, no amastigotes have been observed in CSF. However, in human beings, the migration of parasites into the CSF was reported in a 10-year-old boy suffering from visceral leishmaniasis for more than 2 years. 16 In the present study, Leishmania amastigotes observed inside blood vessels from the brain parenchyma and leptomeninges would indicate hematogenous entry of the organism into the CNS. Furthermore, the disruption of the blood-CSF barrier would lead amastigotes to spread also though the CNS, which would explain the presence of Leishmania antigens in the CSF in this case.
The current study presents a case of CL showing radiculoneuritis with nerve destruction associated with abundant Leishmania amastigotes. In human beings, the most frequent neurologic manifestation of leishmaniasis is peripheral neuropathy. 20 Peripheral nerve involvement has been observed in some cases of human cutaneous leishmaniasis with inflammation and presence of amastigotes inside nerves, and even destruction of the nerve in 1 case. 10 The fact that neuritis with nerve destruction was observed unilaterally in the current case could suggest a previous lesion affecting this area that might have attracted amastigote-carrying inflammatory cells to the area, thereby enhancing the spread of the parasite in the nerves. However, a focal reaction cannot be ruled out because other cases with focal infection have been reported, such as that of extradural granuloma. 6
In summary, the authors report herein the presence of Leishmania spp. amastigotes in nervous tissue producing radiculoneuritis, myelitis, and meningoencephalitis. The presence of amastigotes inside brain blood vessels and CSF suggests disruption of the blood-brain and blood-CSF barriers, allowing the organism to enter the nervous parenchyma. However, since cutaneous lesions were not observed near the spinal nerves or in the rest of the body, and amastigotes were observed inside blood vessels, the authors consider the present case to be one of visceral CL with spread to spinal nerves and CNS. It has been argued that treatment with anti-Leishmania drugs could led the parasite to escape from the drug by taking refuge in the CNS.9,16 In the present case, no anti-Leishmania drugs were used; however, chronic treatment with corticoid drugs would have produced an exacerbation of the disease. 14 Glucocorticoids, through the blockade of cytokine expression, would affect several cytotoxic T-cell functions 5 thus facilitating the progression of the infection. In fact, it has been observed that dogs experimentally infected with L. infantum, and treated with corticosteroids, presented enhanced parasite burden, clinical symptoms, and serological responses than untreated infected dogs. 15 The present case demonstrates the ability of Leishmania parasites to reach and affect the CNS parenchyma.
Footnotes
Acknowledgements
The authors wish to thank L. Pérez and E. Blasco of the veterinary neuropathology group of the Universitat Autònoma de Barcelona for technical assistance.
a.
(ELISA) Snap-Leishmania, IDEXX Laboratories, Westbrook, ME.
b.
Leishmania infantum Zimodeme MON-1, Universidad de Granada, Granada, Spain.
c.
Dacortin, Merck España, Madrid, Spain.
d.
Robeclin, Pfizer PGM, Pocé-sur-Cisse, France.
e.
Quantitative PCR for the detection of kinetoplast DNA using primers for the detection of Leishmania donovani complex (Leishmania infantum + donovani + chagasi), Scanelis, Colomiers Cedex, France.
f.
Rabbit polyclonal anti–Leishmania infantum antibody strain PB75, Instituto de Salud Carlos III, Madrid, Spain.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
