Abstract
A 12-year-old domestic Shorthaired cat developed a multinodular exophytic mass on the dorsal surface of the nose. The skin surrounding the mass was nonpigmented, and actinic keratosis had been diagnosed in this area 3 years previously. Histologic examination revealed hyperkeratosis, epidermal hyperplasia, papillomatosis, koilocytosis, and possible intranuclear viral inclusions. Polymerase chain reaction amplified papillomaviral deoxyribonucleic acid from formalin-fixed samples of the lesion. Sequencing of the amplicon revealed 98% similarity to human papillomavirus (HPV) type 9. To the authors' knowledge, this is only the second reported feline cutaneous viral papilloma. In addition, this is the first report of a feline papilloma being associated with an HPV.
Papillomaviruses are small, double-stranded deoxyribonucleic acid (DNA) viruses that infect humans and numerous animal species. Papillomaviruses cause disease by stimulating epithelial proliferation. Papillomaviruses tend to be highly species-specific with most species infected by multiple different papillomavirus types. 10, 13 There are over 100 different types of human papillomaviruses (HPVs), including some types that cause cutaneous papillomas. 5 Although papillomaviruses are an established cause of skin disease in cats, 13 only 1 exophytic cutaneous viral papilloma has been reported in this species. 3 Papillomaviral involvement in the previous case was confirmed using immunohistochemistry, and the type of papillomavirus could not be determined. 3 The present case report describes a feline cutaneous viral papilloma from which HPV type 9 DNA was amplified using polymerase chain reaction (PCR).
A 12-year-old domestic Shorthaired cat developed an exophytic 1.5 × 1 cm mass that extended caudodorsally from the right external nares. The mass was black and multinodular and was raised approximately 1 cm above the surrounding skin. The mass had been first observed 3 months prior to presentation and had been reported to have grown steadily during this time. However, the mass developed on a nonpigmented area of skin in which actinic keratosis had been diagnosed 3 years previously. The diagnosis of actinic keratosis in this cat was based on clinical examination without confirmatory biopsy. Sun screen had been applied to the actinic keratosis, and lesion progression had not been observed. The cat was reported to be clinically healthy, and no other skin lesions were observed. Due to concerns of a squamous cell carcinoma, the mass was surgically excised and fixed in formalin.
The mass was routinely processed for histologic evaluation. Examination revealed a focal area of markedly thickened epidermis. The hyperplastic epidermis was arranged within prominent folds forming exophytic projections (papillomatosis) (Fig. 1). The epidermis was covered by increased keratin, which was predominantly orthokeratotic, although some areas of parakeratosis were also visible. Follicular ostia were dilated with keratin. The keratin also extended from follicles forming prominent keratin spikes. Approximately half of the keratinocytes within superficial layers of the epidermis demonstrated koilocytosis (Fig. 2). Affected keratinocytes were mildly enlarged and had prominent perinuclear vacuolation. Additionally, affected cells often had small dark nuclei or, less commonly, nuclear enlargement. Also visible were small numbers of massively enlarged keratinocytes with large quantities of foamy pale basophilic cytoplasm (Fig. 3). Nuclei within this cell population were large and basophilic, with a homogeneous or finely granular appearance. Small numbers of keratinocytes contained deeply eosinophilic spheric bodies suggestive of papillomaviral inclusions (Fig. 4).
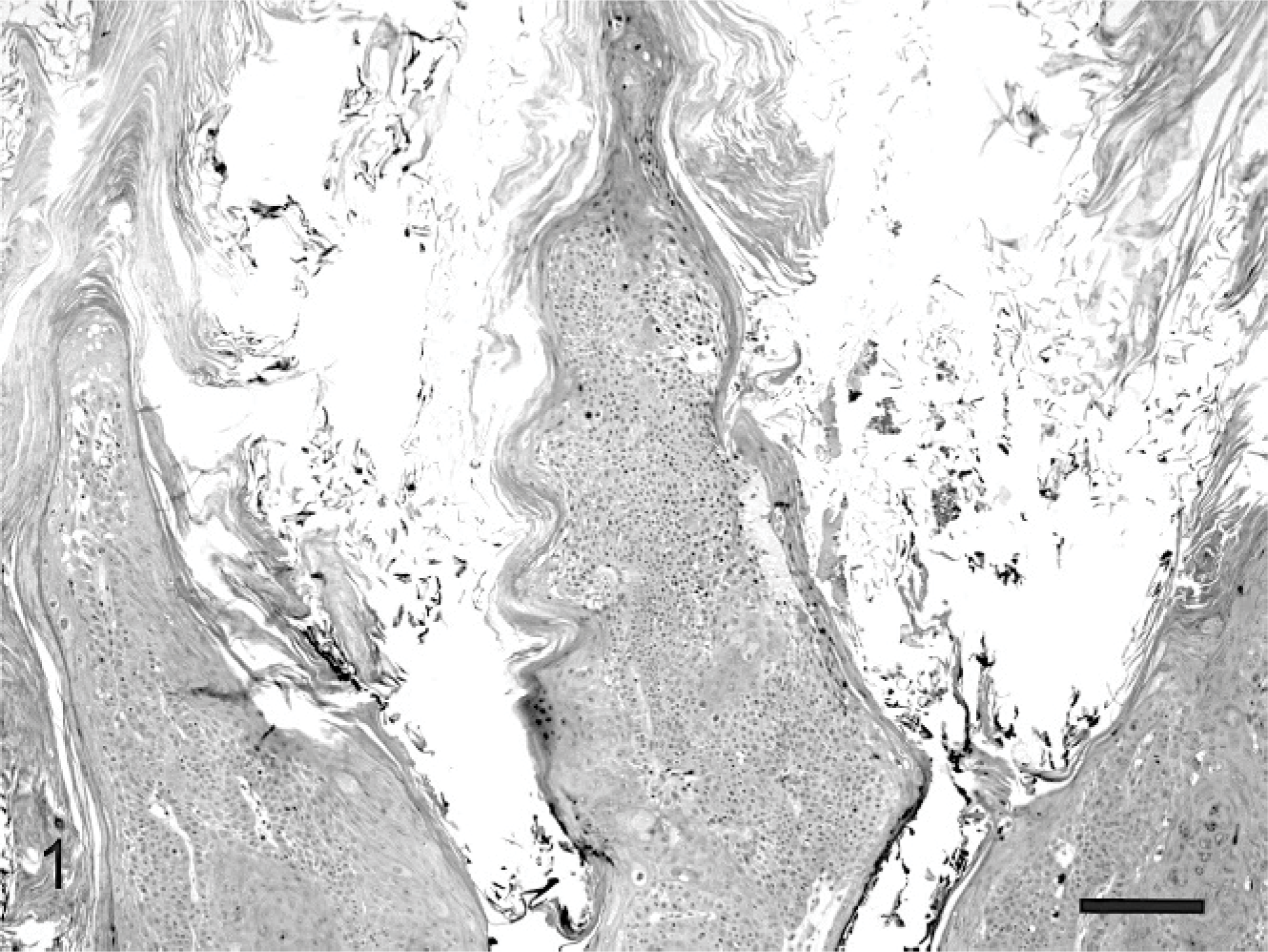
Feline cutaneous viral papilloma. Hyperplastic epidermis is arranged within prominent folds that are covered by large quantities of predominantly orthokeratotic hyperkeratosis. HE. Bar = 90 μm.
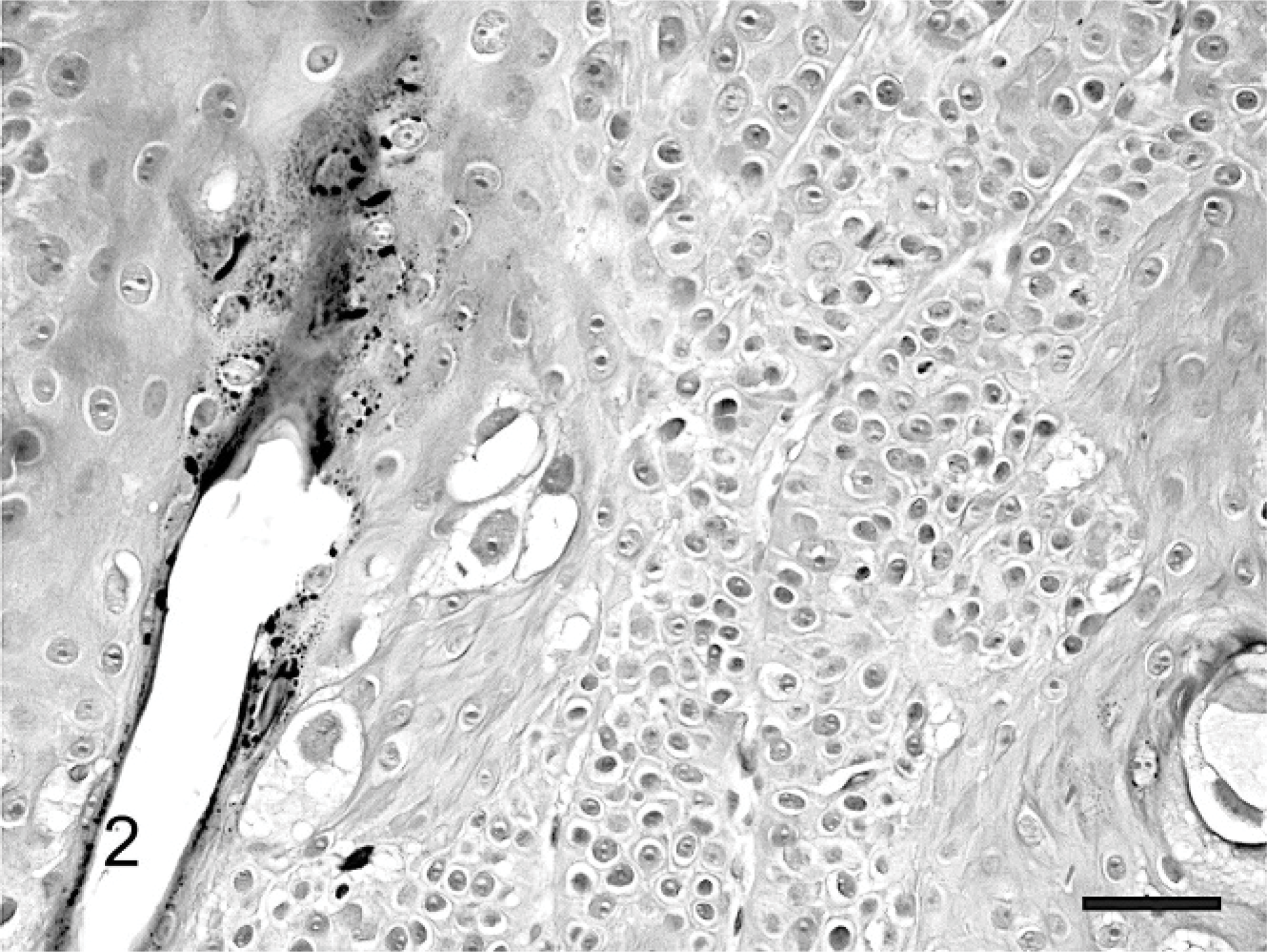
Feline cutaneous viral papilloma. Numerous koilocytes containing a small dark nucleus surrounded by prominent perinuclear vacuolation are visible within the superficial layers of the epidermis. HE. Bar = 25 μm.
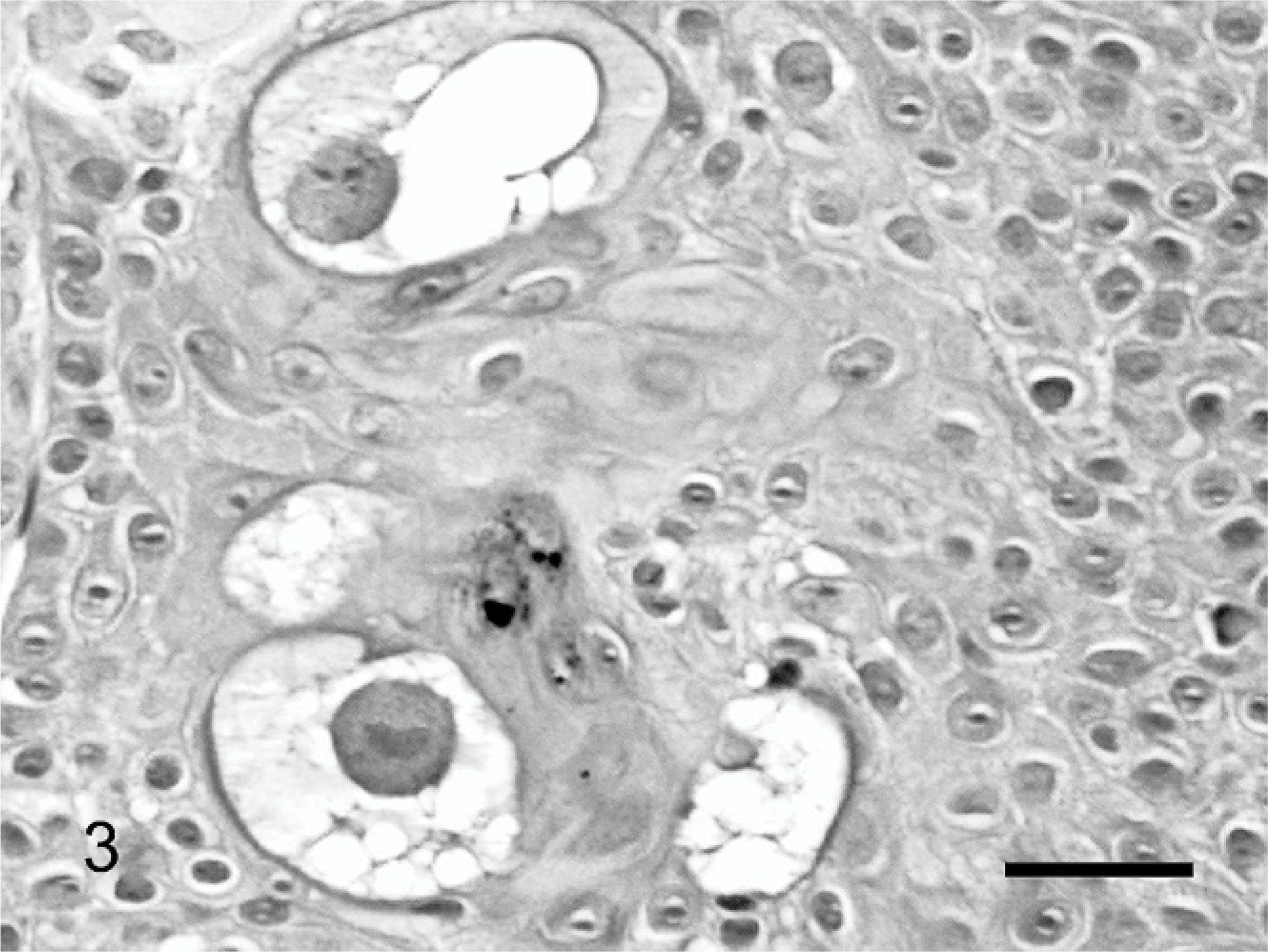
Feline cutaneous viral papilloma. Small numbers of massively enlarged cells with large nuclei containing granular chromatin and prominent cytoplasmic vacuolation are scattered within the papilloma. HE. Bar = 17 μm.
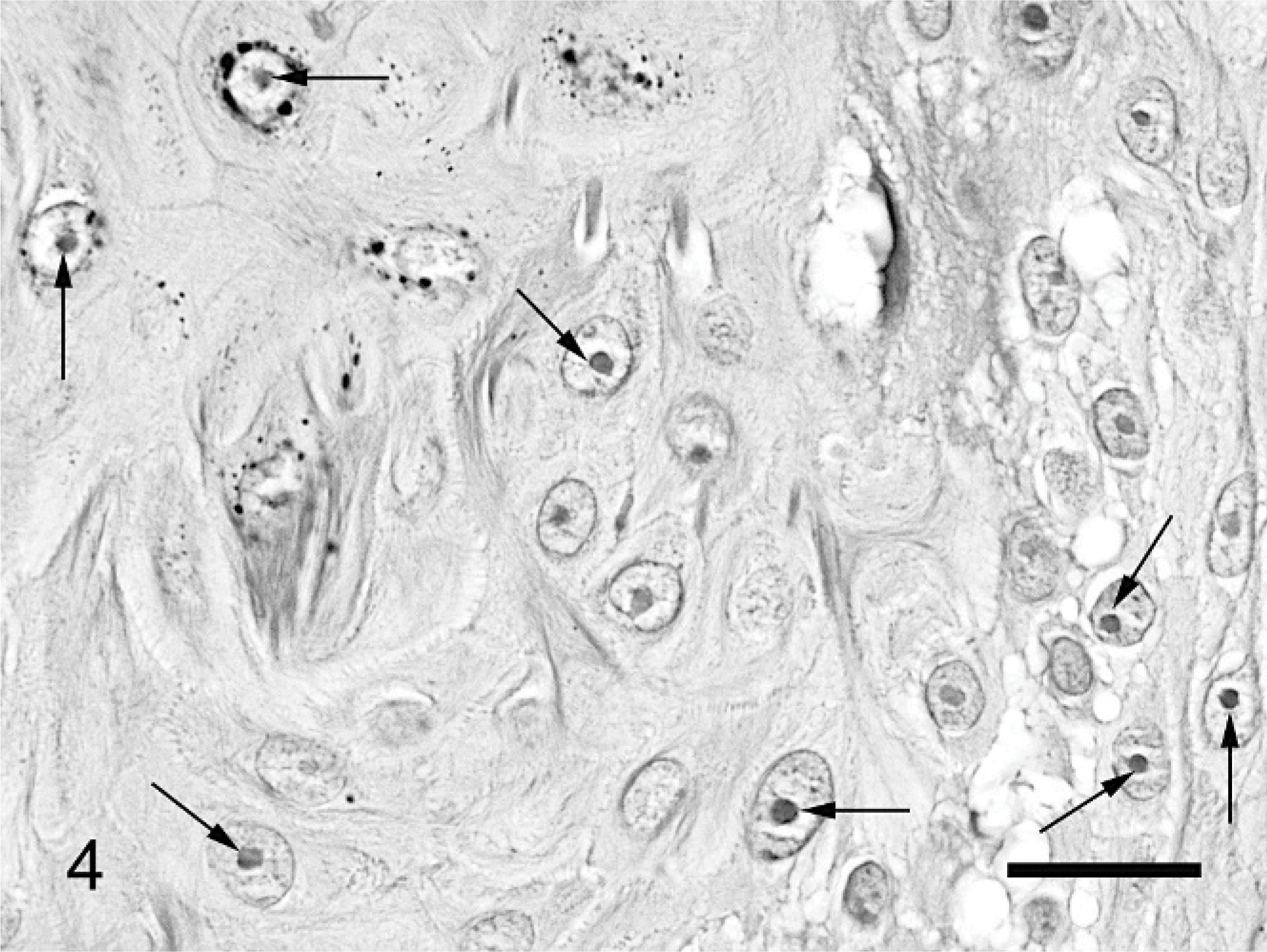
Feline cutaneous viral papilloma. Small numbers of keratinocytes contain small, round, deeply eosinophilic intranuclear bodies suggestive of papillomaviral inclusions (arrows). HE. Bar = 10 μm.
The papillomatosis, koilocytosis, and possible viral inclusions were consistent with a viral papilloma. PCR was used to detect papillomaviral DNA within the lesion. A 10-μm section of formalin-fixed tissue was deparaffinized with xylene and then washed with ethanol. DNA was extracted using a DNeasy Blood and Tissue Kit (Qiagen GmbH, Hilden, Germany) according to manufacturer's instructions. The primer set FAP59/FAP64 was used to amplify conserved regions of the papillomavirus L1 gene. 4 These primers have been previously reported to detect a broad range of human and animal cutaneous papillomaviruses. 4 Final concentrations of the reaction products were 1X PCR buffer, 1.5-mM magnesium chloride, 200-μM deoxynucleotide triphosphate, 0.25 μM of each primer, 1.25-U Platinum Taq DNA polymerase (Invitrogen Corporation, Carlsbad, CA), and 2.5-μl template DNA in a final reaction volume of 50 μl. Amplification was carried out in a Px2 Thermal Cycler (Thermo Electron Corporation, Milford, MA) by 94°C for 10 minutes followed by 45 cycles of 94°C for 1.5 minutes, 50°C for 1.5 minutes, and 72°C for 1.5 minutes, with final extension at 72°C for 5 minutes. Electrophoresis in a 1% agarose gel containing ethidium bromide was used to detect the amplified fragment. Samples of canine oral papilloma and equine sarcoid were used as positive controls, while 18 feline skin samples (3 fibrosarcomas, 3 biopsies of allergic dermatitis, 3 biopsies of normal skin, 2 cases of feline leprosy, 2 feline mast cell tumors, 2 apocrine gland cystadenomas, 2 melanomas, and a follicular cyst) were used as negative controls. To sequence the DNA amplified from the papilloma, we purified the PCR product (Qiaex II 150 Gel extraction kit, Qiagen GmbH) and subjected it to automatic dye-terminator cycle sequencing with a BigDye Terminator, Version 3.1. Ready Reaction Cycle Sequencing kit and using a ABI3730 Genetic Analyzer (Applied Biosystems Inc., Foster City, CA). Results were compared with known sequences from GenBank (see http://www.ncbi.nlm.nih.gov/genbank/; using the basic local alignment search tool (http://www.ncbi.nlm.nih.gov/blast).
PCR amplified a 480-base pair fragment from samples of the feline cutaneous papilloma, canine oral papilloma, and equine sarcoid. Papillomaviral DNA was not amplified from the 18 control samples of feline skin. Sequencing revealed that the papillomaviral DNA amplified from the feline cutaneous papilloma had 98% similarity to HPV type 9 genomic DNA (Genbank accession X74464.1). The sequence amplified from the presently described feline papilloma was Genbank accession EF60823.
Examination of the surgical site after papilloma removal revealed multiple small dark plaques covered by mild crusting. These were clinically diagnosed as actinic keratosis. Treatment with topical sun screen has continued, and the lesions have not progressed during the 13 months since surgery. One person within the cat's household has had a plantar wart for the last 5 years. None of the other family members have visible skin disease. The affected cat has contact with another cat and a dog, neither of which have evidence of skin disease.
To the authors' knowledge, this is the second report of an exophytic cutaneous viral papilloma in a cat. Cutaneous papillomas due to papillomavirus infection were also reported to develop in association with feline papillomavirus 1 (FdPV-1). 13 However, these lesions were described as multiple scaly plaques 13 and may be more consistent with the feline hyperkeratotic plaques that were initially described in Persian cats. 2 The present case has some similarities to the previously described exophytic feline viral papilloma. 3 Papillomas developed on the face of the cat in both cases, in the present cat on the nose, in the previously described cat on the eyelid. 3 Both papillomas consisted of exophytic folds of epidermis covered by a thick layer of keratin. 3 Intranuclear viral inclusions were described in the previous cat. 3 While intranuclear bodies that were suggestive of viral inclusions were visible in the present cat, their identity could not be confirmed histologically. Koilocytosis was a prominent lesion only in the present cat. 3 In both cats, the papilloma developed in an area of damaged skin. In the previous cat, the papilloma developed at the site of a prior surgical removal of a xanthoma. 3 In the present cat, the papilloma developed within an area of actinic keratosis. Surgical excision of both papillomas was curative. 3 In the previous cat, papillomaviral involvement was confirmed using immunohistochemistry. 3 Immunohistochemistry detects a wide range of papillomaviral antigens, so the type of papillomavirus could not be determined. 3 In the present cat, papillomaviral association with the lesion was confirmed using PCR. Sequencing of the amplicon revealed 98% similarity to HPV type 9. Although only a segment of the total viral DNA was sequenced, the close similarity over this segment suggests the sequence amplified was from HPV type 9. In comparison, the second-most similar sequence was HPV type 14, with just 87% similarity.
The histologic evidence of papillomatosis, koilocytosis, and possible intranuclear viral inclusions strongly suggests that this lesion was induced by a papillomavirus. Additionally, this lesion was clinically and histologically similar to a previously described feline viral cutaneous papilloma. 3 Furthermore, no other papillomaviruses were amplified from this lesion. Therefore, while causation cannot be definitively proven, these results are considered strong evidence that infection with HPV type 9 resulted in papilloma formation in this cat.
There are over 100 human papillomavirus types with numerous new types still being identified. 1, 5, 7 Eighty percent of humans asymptomatically carry one, or multiple, papillomavirus types. 1 Human papillomaviruses are divided into 3 broad groups. First, around one third of papillomaviruses is adapted to mucosal surfaces. 7 The second group of HPVs causes human cutaneous viral papillomas. 11 The third group of HPVs is the epidermodysplasia verruciformis (EV)-associated HPVs. EV is a rare genetic disease that results in increased susceptibility to HPVs. 9 As well as causing disease in EV patients, EV-associated HPVs commonly asymptomatically infect non-EV humans. 1 Human papillomavirus type 9 is considered an EV-associated HPV. 9 No people in contact with this cat had skin lesions consistent with EV. Therefore, it appears possible that this cat was infected from a person asymptomatically carrying this papillomavirus. Unfortunately, testing of the cat's owners for the presence of papillomaviruses was not possible.
Considering the type of virus amplified from this lesion, it appears likely that this cat was infected by human contact. However, when 80% of people asymptomatically carry papillomaviruses, 1 it is surprising that feline cutaneous viral papillomas are so rare. There are 3 possible explanations for this. First, both the presently described and the previously reported feline cutaneous viral papillomas developed in areas of pre-existing skin disease. 3 In the present cat, it is possible that actinic keratosis impaired the skin defenses and allowed infection and subsequent papillomatosis due to HPV type 9. In humans, EV-associated HPVs asymptomatically infect non-EV people but require immunosuppression to produce disease. 9 Second, of the HPVs, HPV type 9 may be uniquely able to infect cats. Human papillomavirus type 9 appears to be rare within people. 1, 4, 5, 11 Isolation of HPV type 9 from the owners of the cat, but not other people, would support the hypothesis that HPV type 9 is rare within humans but has an unusually high zoonotic potential. Third, it is possible that cats are the natural host for HPV type 9. The virus could be a common asymptomatic infection in cats that only rarely causes disease in this species. It would, therefore, be possible that human EV patients are infected with HPV type 9 due to contact with cats. However, because no papillomaviral DNA was detected in any of the 18 nonpapilloma feline skin samples, this hypothesis appears less likely. Additionally, a previous study of 50 feline vaccine-site sarcomas also did not detect papillomaviral DNA. 6
Papillomaviruses are generally considered to be highly host-specific. 10, 13 However, cats appear unusually susceptible to zoonotic papillomaviral infection. In addition to cutaneous viral papillomas, papillomaviruses have been reported to cause 3 other skin diseases in cats. First, feline cutaneous fibropapillomas (also referred to as feline sarcoids) are associated with a papillomavirus most closely related to bovine papillomavirus. 12, 15 Second, sessile cutaneous hyperkeratotic lesions have been associated with FdPV-1 and usually develop in immunosuppressed cats. 2, 13, 14 Feline papillomavirus type 1 has been shown to be most closely related to canine oral papillomatosis virus 14, 16 Third, papillomaviruses have been associated with feline bowenoid in situ carcinomas (BISCs). 17 Six papillomaviruses have been amplified and sequenced from feline BISCs; one had 97% similarity to HPV isolate FAIMVS14, while the others were all a novel unique virus that was most closely related to HPV type 17. 8 Considering the close contact of cats with humans and the large number of HPV types, it is perhaps not surprising that HPVs can occasionally infect cats. A recent study of papillomaviruses identified from dogs also reported papillomaviruses that were closely related to HPVs. 18
In conclusion, this is the second reported exophytic feline cutaneous viral papilloma. PCR and subsequent sequencing revealed the presence of HPV type 9 within the lesion. While it cannot be definitively confirmed, HPV type 9 was likely to have caused this lesion. It is unknown whether underlying alterations in the skin or the specific type of HPV was the main factor resulting in disease development in this cat. Additionally, it is unknown whether HPV type 9 was passed from human to cat or whether HPV type 9 normally lives asymptomatically in cats and occasionally causes disease in people.
