Abstract
Mycobacterioses can produce nonspecific clinical signs in dogs and cats that make diagnosis difficult. Furthermore, the full characterization of mycobacterial agents is not always possible or practical. We characterized mycobacteria detected through cytology in 12 dogs and 7 cats with generalized clinical signs from the province of Buenos Aires in Argentina. In dogs, molecular testing confirmed the presence of Mycobacterium avium subsp. hominissuis (MAH) in 8 cases and M. fortuitum in 1 case. All dogs were Miniature Schnauzers, suggesting that this breed may be more susceptible to M. avium than other dog breeds. The cat isolates were 2 M. bovis, 1 M. fortuitum, and 1 MAH. Mycobacterial interspersed repetitive unit–variable-number tandem repeat patterns suggested possible links with cattle, swine, and humans studied previously in Argentina. The results show that pets may act as susceptible hosts with the potential risk of transmitting the infection to humans and other animals.
Companion animals, particularly cats and dogs, are susceptible to infections caused by various mycobacteria. 9 Typically, mycobacterial infections produce nonspecific clinical signs, thus impairing diagnosis. 9 In cats, tuberculosis (TB) is most commonly caused by Mycobacterium bovis because cats are naturally resistant to infections with Mycobacterium tuberculosis. 11 As well, in places where bovine TB is endemic, such as Argentina, cats may be fed infected food.2,20 Systemic signs of TB in cats usually include weight loss, anorexia, and fever. 11 In diseases caused by mycobacteria other than M. bovis, cats usually have previous injuries such as bites by other cats, scratches and abrasions, and the most common source of infection is the environment. In M. avium infections in cats, there are generally localized lesions of the skin and subcutaneous tissue. 11 In Argentina, although mycobacterioses have been reported in cats,23,24 the prevalence of these diseases is still unknown. In dogs, TB is uncommon and generally caused by infection with M. tuberculosis or M. bovis. 9 Although dogs may become infected by other mycobacteria, they are classified as relatively resistant to these organisms.6,13 During autopsies, however, granulomatous lesions in organs, including generalized lymphadenopathy, may be evident.6,9,13 We report confirmed cases of mycobacterioses in cats and dogs from Buenos Aires, Argentina, and evaluated possible links between those cases and mycobacterial isolates from other hosts, including humans, from the same region.
Between 2013–2015, the Veterinary Diagnostic Laboratory (VDL) of the School of Veterinary Medicine, University of Buenos Aires, the largest veterinary hospital in Buenos Aires, received 12 Miniature Schnauzer dogs and 7 European cats (all deceased) that were suspected as having suffered from mycobacterioses. All deceased animals came from Buenos Aires and the surrounding suburbs. Two dogs had signs only of digestive problems, with occasional bouts of vomiting and diarrhea. The other cases also had signs of chronic disease and starvation that included anemia, anorexia, progressive weight loss, fever, and lethargy. Most cases had enlarged mesenteric and superficial lymph nodes. Although most cats had systemic signs and generalized lymphadenopathy, 4 of 6 also had skin lesions. The digestive tract was the system most affected in cats, with vomiting and diarrhea, whereas 1 case also had dyspnea and coughing, which suggested a respiratory form of the disease (Table 1).
Breed, age, sex, clinical signs, case history, and techniques used to characterize mycobacterioses in dogs and cats in Argentina.*
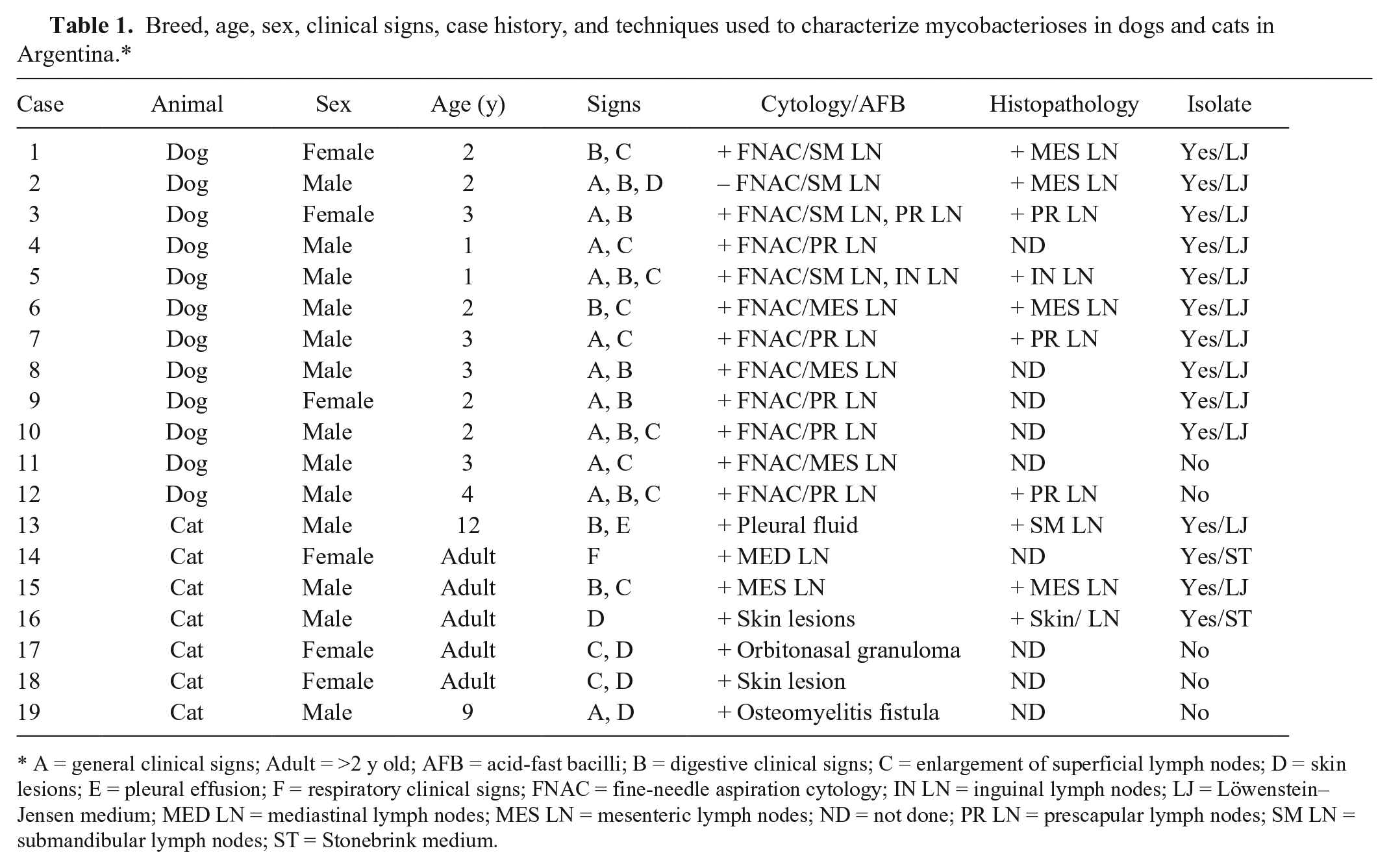
A = general clinical signs; Adult = >2 y old; AFB = acid-fast bacilli; B = digestive clinical signs; C = enlargement of superficial lymph nodes; D = skin lesions; E = pleural effusion; F = respiratory clinical signs; FNAC = fine-needle aspiration cytology; IN LN = inguinal lymph nodes; LJ = Löwenstein–Jensen medium; MED LN = mediastinal lymph nodes; MES LN = mesenteric lymph nodes; ND = not done; PR LN = prescapular lymph nodes; SM LN = submandibular lymph nodes; ST = Stonebrink medium.
Fine-needle aspiration cytology from lymph nodes revealed increased numbers of neutrophils and macrophages in all animals. Additionally, direct smear examinations with Ziehl–Neelsen staining were performed. All animals but one had acid-fast bacilli morphologically consistent with Mycobacterium species within macrophages (Table 1). All animals had deteriorated rapidly and then died or were euthanized.
Postmortem examinations 1 revealed granulomatous inflammation of lymph nodes and numerous organs, including intestine, spleen, and liver. Histologic examination revealed granulomatous inflammation in lymph nodes, tonsils, liver, and spleen, as well as areas of caseous necrosis (Table 1).
Samples from granulomatous lesions were cultured on Löwenstein–Jensen and Stonebrink media as described previously. 15 The bacteria were identified by PCR, with the M. avium complex was identified using the target sequences IS1245 10 and IS901, 18 whereas IS6110 12 was used for the M. tuberculosis complex. Cultures that could not be identified by PCR were subjected to 16S ribosomal (r)RNA gene fragment sequencing. 16 Spoligotyping was carried out as described previously 14 using a commercial spoligotyping kit (Mapmygenome, Madhapur, Hyderabad, Andhra Pradesh, India). To complete the typing of Mycobacterium avium subsp. hominissuis (MAH) and M. bovis strains, mycobacterial interspersed repetitive unit–variable-number tandem repeat (MIRU-VNTR) typing was performed as described previously. 21 The positive controls for the spoligotyping and the MIRU-VNTR typing were M. tuberculosis H37Rv (ATCC 27294), M. bovis BCG (Bacillus Calmette–Guérin; ATCC 27289), and M. avium subsp. avium D4ER.
Only 10 of 12 samples from dogs and 4 of 7 samples from cats yielded positive results in culture and PCR, indicating the presence of different mycobacterial species (Table 2). The 16S rRNA gene fragment sequence alignment used for samples 7 and 15 had an identity of 99.7% and 99% with M. fortuitum, respectively. The MIRU-VNTR typing results for the confirmed M. avium and M. bovis isolates are presented in Table 2, with the exception of strain 2 for which amplification failed because of poor DNA quality.
PCR, spoligotyping, and mycobacterial interspersed repetitive unit–variable-number tandem repeat (MIRU-VNTR) typing of mycobacterial isolates from dogs (cases 1–9) and cats (cases 13–16).*
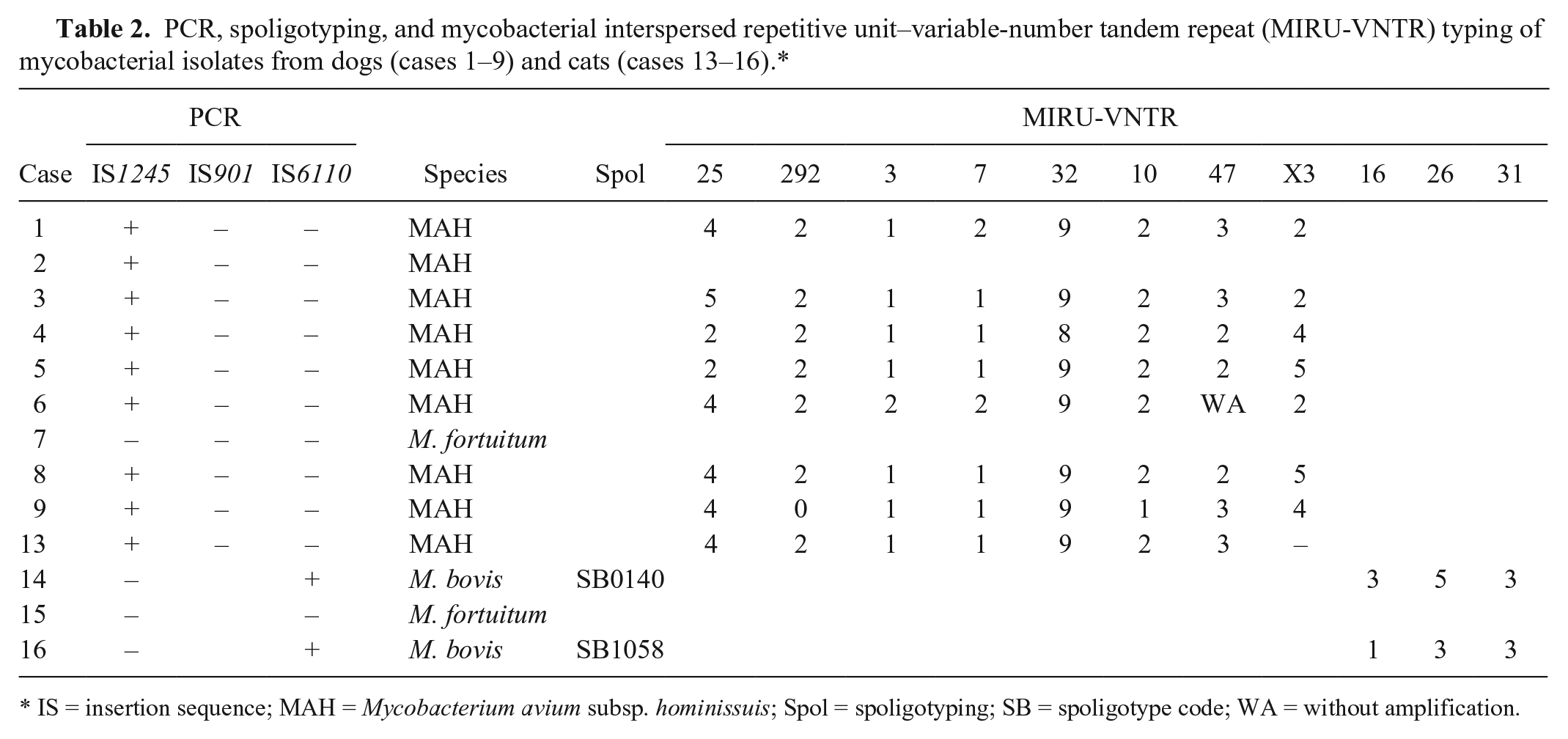
IS = insertion sequence; MAH = Mycobacterium avium subsp. hominissuis; Spol = spoligotyping; SB = spoligotype code; WA = without amplification.
The canine isolates were confirmed as M. avium (9 isolates, with 8 of the MAH subspecies) and 1 M. fortuitum. In 7 dogs, the signs of vomiting and diarrhea, and gastrointestinal lesions, suggested an oral route of Mycobacterium infection, which would increase the zoonotic potential, given that this microorganism can be shed in feces. 9
In our study, all dogs suspected of mycobacterioses received at the VDL were Miniature Schnauzers and came from different breeders, all of them confirmed as suffering from M. avium infection. The literature suggests an increased susceptibility of some dog breeds to M. avium, mainly Miniature Schnauzer and Basset Hounds.6,9,13
Among the isolates from 7 cats, 2 were identified as M. bovis, 1 as M. fortuitum, and 1 as MAH. The source of the M. bovis infections might have been food, given that feeding cats raw bovine viscera is still a popular practice in Argentina. 23 Moreover, bovine TB is still endemic in Argentina. 24 Spoligotype SB0140 from case 14 is the most frequent pattern in Argentina, and has been detected in different hosts, including humans.2,23 In contrast, spoligotype SB1058 from case 16 has been identified at a very low frequency. 23 Although the risk of M. bovis being transmitted to humans is believed to be negligible, it still remains a risk for public health. 11 In Buenos Aires, a case of human septic arthritis of the glenohumeral joint caused by M. bovis has been reported. 7 In that case, the only epidemiologic association was that the person owned several cats that were fed raw bovine lungs, and M. bovis was isolated through bronchoalveolar lavage of one of the cats. 7
M. fortuitum was present in case 15. This mycobacteria is usually identified in infections of the subcutis and skin and is uncommon in feline systemic diseases.9,11 Interestingly, MAH was detected in case 13, and is rarely reported in the literature.4,17
The VNTR patterns (MIRUs 16, 26, and 31) seen in our study were compared to those detected in other hosts in Argentina. The M. bovis strains isolated from cats had a pattern similar to that previously observed in cattle, swine, and humans, and differed from the main pattern in the country (2-5-3) in MIRU 16.2,20 Patterns identified in MAH strains matched those found in strains from swine and humans previously reported in Argentina. 3 In addition, we observed the presence of alleles in locus 25 and locus 292 related to the differentiation of the M. avium complex subspecies.19,22
The identification of Mycobacterium species involved in a disease scenario in domestic pets is important because the implications for public health vary depending on the species involved.5,11 Genotyping contributes to understanding aspects of the epidemiology of the disease, including potential links and routes of transmission and spread. Similarly, an appropriate treatment regime is crucial to avoid selection of drug-resistant mutants, which would represent a risk for public and animal health. 8 Our study highlights the importance of submitting samples from companion animals suspected of suffering from mycobacteriosis to an appropriate diagnostic laboratory for confirmation of the Mycobacterium species involved.
Footnotes
Acknowledgements
We thank Dr. Julia Sabio y García and Pablo Crescentini for the critical reading of this manuscript. Martín José Zumárraga and Soledad Barandiaran are career members of Consejo Nacional de Investigaciones Científicas y Técnicas, Argentina.
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
This work was supported by University of Buenos Aires (UBACyT 20020100100203) grant.
