Abstract
We describe a 10-month-old, intact female American Cocker Spaniel with pulmonary lymphomatoid granulomatosis (PLG). On clinical examination, this dog presented with nonproductive dry cough, serous nasal discharge, dyspnea, and lack of appetite. Radiography showed a consolidated lesion in the left cranial lung lobe. Histopathologic examination showed a mixed population of atypical lymphoid cells that had infiltrated into the pulmonary blood vessels angiocentrically. The lymphocytes were CD3 positive, consistent with a pan-T-cell phenotype. The lymphoid cells in the lesion were also positive for CD20cy and CD79a, indicative of the presence of B cells. We also observed large Reed-Sternberg–like cells that were positive for CD15 and CD30, similar to observations in human pulmonary Hodgkin's disease (PHD). In conclusion, canine PLG in this Cocker Spaniel was associated with B and T cells, which is first identified in a case of canine PLG. It was histopathologically similar to human lymphomatoid granulomatosis and immunophenotypically similar to human PHD.
Canine pulmonary lymphomatoid granulomatosis (PLG) is a rare pulmonary lymphoproliferative disease of dogs. 1, 4, 7, 8, 9, 10 It is characterized by multiple pulmonary nodules that consist of sheets of atypical lymphoreticular and plasmacytoid cells admixed with fewer mature eosinophils and lymphocytes. 8 PLG has a predilection for the caudal lung lobes and frequently metastasizes to hilar lymph nodes. 4 Canine PLG shows many similarities to human pulmonary Hodgkin's disease (PHD) and lymphomatoid granulomatosis (LYG). 8
We report here the clinical, histopathologic, and immunophenotypic features of a dog diagnosed with PLG. Our results provide new insights into canine PLG, as well as confirm previous findings in this disease. A 10-month-old, intact female American Cocker Spaniel, kept exclusively indoors, was referred to the Veterinary Medical Teaching Hospital of Konkuk University, Seoul, Korea, because of a 1-week history of nonproductive, dry cough, serous nasal discharge, dyspnea, and lack of appetite. Physical examinations revealed that the dog was dyspneic and lethargic. On auscultation, harsh sounds and wheezes were detected in the left lung. A complete blood count showed abnormal leukocytosis (28.14 × 103/μl; reference range, 6–17 × 103/μl) and eosinophilia (5.628 × 103/μl; reference range, 0–1.4 × 103/μl). Thoracic radiography showed total consolidation of the left lung lobes with a diffuse interstitial pattern. The dog was treated with prednisolone and antibiotics. Seven months after the therapy, the dog died because of severe respiratory distress.
A necropsy revealed a complete consolidation of the left cranial lung lobe. The left lung contained a well-circumscribed mass (6 × 12 cm), which was firm on palpation. On cut section, the mass was white and contained multiple foci of necrosis and hemorrhage. Other organs, including the regional lymph nodes, were unremarkable in appearance. Tissues collected at necropsy were fixed in 10% neutral buffered formalin, embedded in paraffin, sectioned at 4 μm and stained with hematoxylin and eosin (HE).
For immunohistochemical (IHC) analysis, slides were incubated with antibodies to CD3, CD79a, CD20cy, CD15, and CD30, all purchased from DakoCytomation, Fine Life Science Co. (Seoul). The antibodies were diluted 1 : 100 (CD3 and CD20cy) or 1 : 200 (CD79a, CD15, and CD30). CD3 was assayed by using the 2-step Envision system-AP (alkaline phosphatase; DakoCytomation) and the others were assayed by using the Envision system-HRP (horseradish peroxidase; DakoCytomation), according to the manufacturer's instructions. To assay for colocalization of T and B cells, we used a double-labeling IHC technique for single-tissue sections.
Histopathologic examination showed a mixed population of atypical lymphoid cells and eosinophils in the pulmonary blood vessels. The atypical lymphoreticular cells, which consisted of monotypic and pleomorphic populations of anaplastic lymphoid cells, infiltrated angiocentrically with adjacent fibrinoid necrosis. Mitosis varied from 1 to 4 per high-power microscopic field (Fig. 1). The infiltrate also contained some large cells with large pleomorphic and binucleated nuclei resembling Reed-Sternberg cells in PHD. These clinical and histopathologic findings led to a diagnosis of PLG.
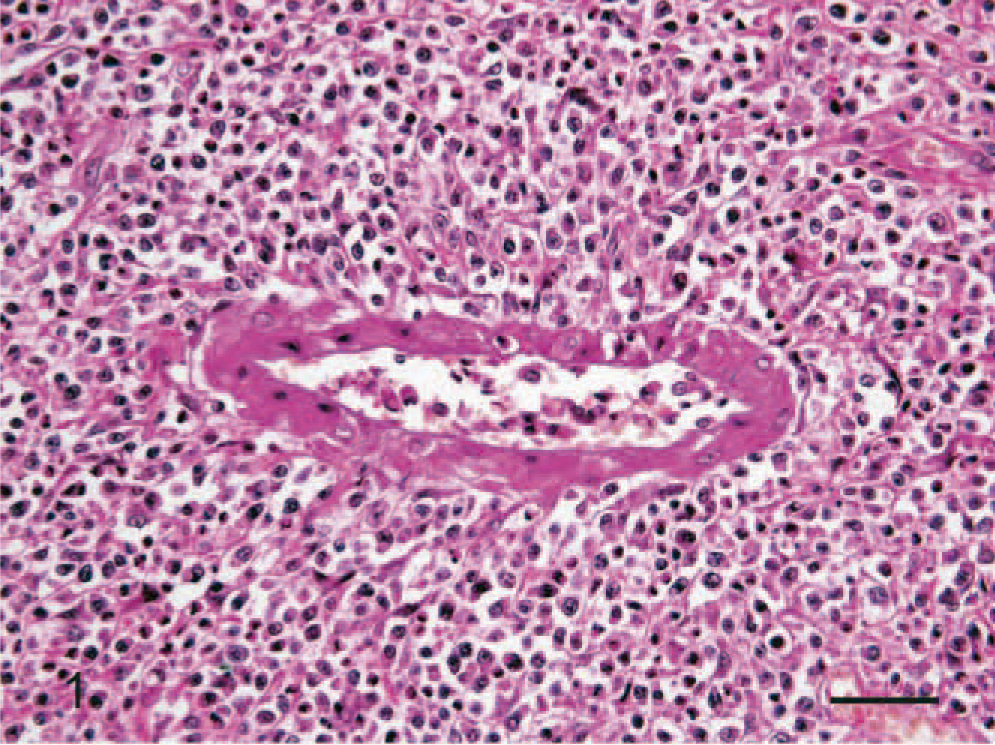
Lung; dog. Angiocentric infiltrate of an atypical and pleomorphic population of lymphoid cells with fibrinoid necrosis. HE. Bar = 30 μm.
IHC examination of the lymphoid infiltrates showed that they consisted primarily of T cells. Clusters of atypical lymphocytes, however, were consistently stained with antibodies to the B-cell markers, CD79a and CD20cy. Double-labeling IHC showed that these pulmonary lymphatic nodules contained both T and B cells (Fig. 2). The large, Reed-Sternberg–like cells were positive for CD15 (Fig. 3) and CD30. These results are summarized in Table 1, comparing with that of human LYG and PHD.
CD classification of human LYG, Hodgkin's disease, and canine PLG.∗

∗ + = positive, − = negative, ND = not determined; superscript numbers refer to sources in the References section.
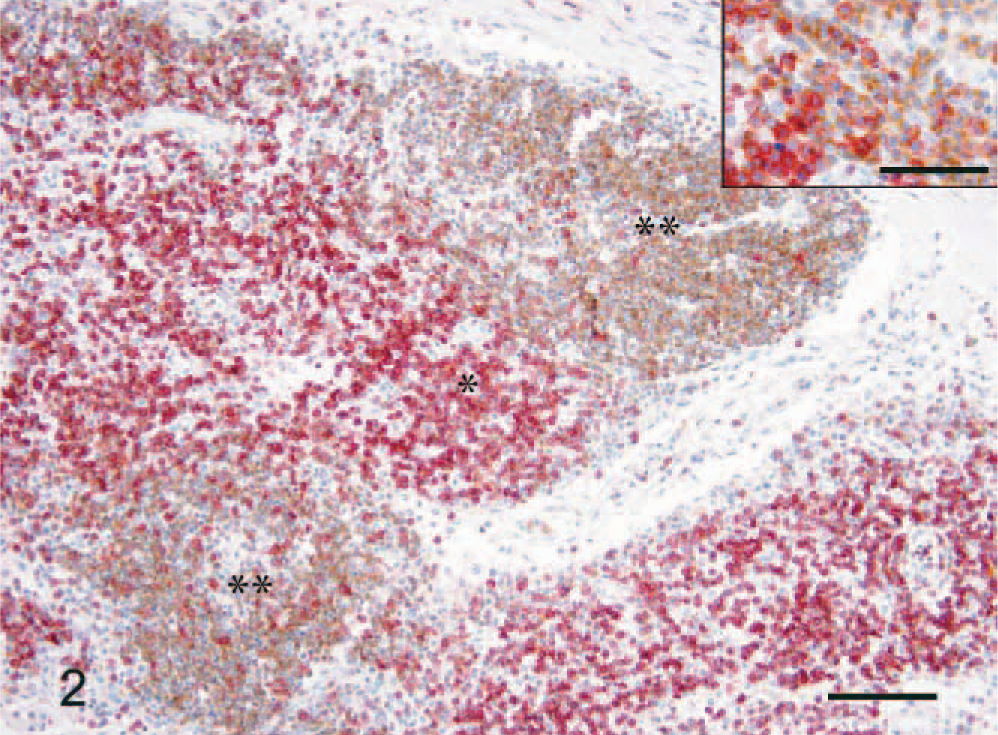
Lung; dog. Double labeling of CD3 and 79a expression. Strong immunohistochemical reaction to CD3(∗) and CD79a(∗∗). Envision system-AP and HRP, Harris hematoxylin counterstain. Bar = 85 μm.
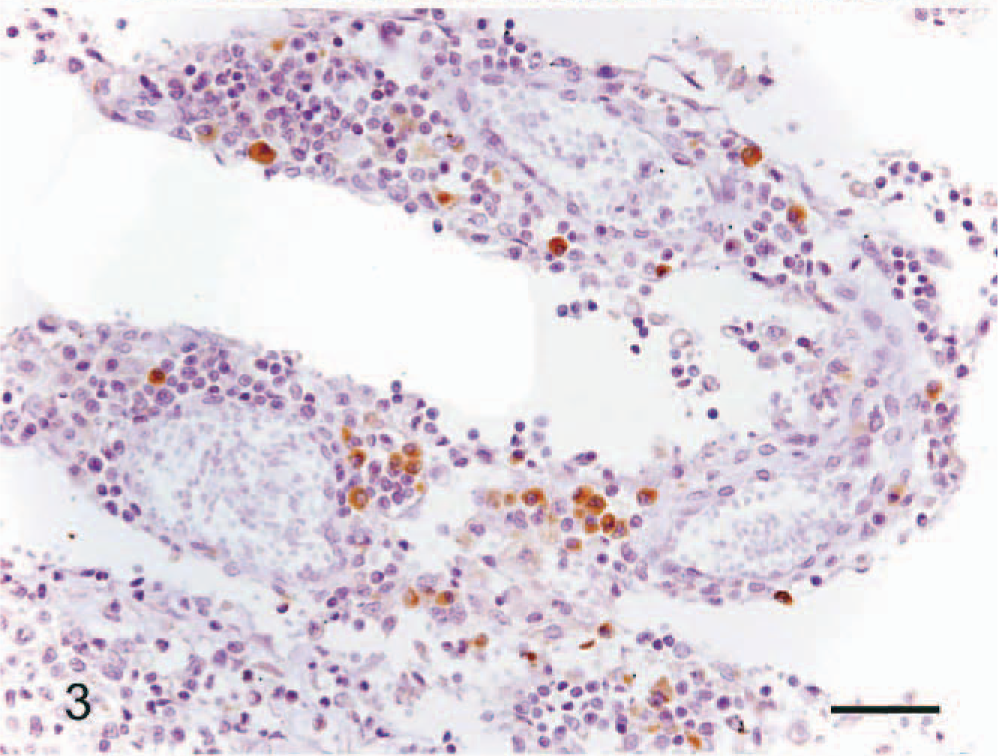
Lung; dog. Immunohistochemical reaction to CD15. Positive labeling of the Reed-Sternberg cells. Envision system-HRP, Harris hematoxylin counterstain. Bar = 30 μm.
The lesion in this dog was histologically similar to that in previous reports, in that atypical lymphohistiocytes had proliferated and infiltrated into the pulmonary blood vessels angiocentrically. 4, 8, 10 Immunophenotypic analysis, however, showed that, although CD3 positive lymphocytes predominated, numerous lymphocytes strongly positive for CD20cy and CD79a, were also present. This indicates that PLG in this dog is of both T- and B-cell lineage. This finding differs from that of a previous report, which found that T-cell markers, e.g., CD3, were expressed in 2 of 3 dogs, whereas B-cell markers were not expressed. 10 In our dog, some Reed-Sternberg–like cells, positive for CD15 and CD30, were also present.
Canine PLG has features similar to those of human LYG and PHD. 8 Human pulmonary LYG is characterized as a form of systemic vasculitis, as shown by prominent alteration of the vessels, coagulative necrosis, and the presence of angiocentric granulomatous infiltrates with neoplastic lymphocytes. 2 Human pulmonary LYG is positive for CD3, CD20cy, and CD79a, but negative for CD15 and CD30. 5 PHD includes polymorphous pulmonary infiltrates, composed of sheets of atypical and variable cells, including small mature lymphocytes, eosinophils, and plasma cells, as well as Reed-Sternberg cells. 3 Reed-Sternberg cells are positive for CD15 and CD30, and neoplastic lymphocytes consist of mixed populations of CD3 and CD20cy positive cells. 3
Histopathologically, the canine PLG described here has characteristics of human LYG, in that pleomorphic and anaplastic lymphoid cells had infiltrated into the vessels angiocentrically. Atypical lymphocytes positive for CD3, CD20cy, and CD79a, and Reed-Sternberg–like cells positive for CD15 and CD30 were also present. PLG in this dog was quite similar to human Hodgkin's disease immunophenotypically.
In summary, our IHC analysis showed that this dog with PLG had neoplastic B-cell coexpression, indicating that canine PLG is a pan-T-cell neoplasm strongly associated with neoplastic B-cell phenotype. We also found that canine PLG in this dog had clinical, histopathologic features similar to those of human LYG and immunophenotypic features similar to those of human PHD.
Footnotes
Acknowledgements
The study was supported by the Konkuk University Research Fund 2005. We thank Ms. R.-W. Jang for technical assistance. D.-N. Hwang and B.-T. Kang contributed to this paper as the co-first authors.
