Abstract
A 2-year-old Hereford cow was presented to the teaching hospital for increased respiratory noise and bilateral bloody nasal discharge. A nodular rhinitis was detected clinically, and the nasal biopsy revealed a granulomatous and eosinophilic rhinitis with intralesional fungi. The cow was euthanized due to financial constraints. Grossly multifocal-to-coalescing, raised, ulcerated firm nodules were present in both nares. Histologically, the lamina propria was expanded by intense infiltrates of eosinophils, epithelioid macrophages, multinucleate giant cells, lymphocytes, and plasma cells. Associated with the inflammatory cells were variably sized, septate hyphae, 5–8 μm in diameter, admixed with numerous, terminal conidia, 6–30 μm in diameter, with a discrete outer wall identified by culture as Pseudallescheria boydii species complex (anamorph Scedosporium apiospermum). The infection was localized to the nasal cavity with no gross or microscopic evidence of a systemic dissemination.
A 2-year-old Hereford cow with a history of chronic (6–8 weeks) upper respiratory dysfunction, increased respiratory noise, and bloody nasal discharge was presented to the teaching hospital. There were no signs of gross trauma, and only 1 animal in the herd was affected. Prior empiric therapy with parenteral oxytetracycline and sodium iodide had not improved the animal's clinical condition. Physical examination revealed normal temperature, heart rate, and body condition. Mild tachypnea (48 breaths/minute), inspiratory stridor, and reduced airflow through the nares were detected. Visual and digital examination of the nasal mucosa revealed multiple polypoid masses, 0.5–3.0 cm in diameter, present in both nares. Nasal biopsy was performed and submitted for histologic examination and mycologic culture. The cow was treated with a parenteral antihistamine pending biopsy results. No improvement was noted following 4 days of antihistamine therapy. Biopsy revealed fungal-induced granulomatous and eosinophilic inflammation. Due to financial constraints, the animal was euthanized.
Grossly, the nasal mucosa was bilaterally expanded by multiple, individual-to-coalescing, occasionally ulcerated, light-grey-to-splotchy-red nodules, 0.5–3.0 cm in diameter. The majority of these nodules was located 3-cm caudal to the nares and extended 15 cm caudally (Figs. 1, 2). On cut section, the submucosal connective tissue was expanded by multiple, discrete, pale-to-dark-red, rough lesions. No significant gross lesions were observed in other organs. Histologically, the nasal mucosa was multifocally ulcerated, and the underlying lamina propria was expanded by intense infiltrates of eosinophils, epithelioid macrophages, numerous multinucleate giant cells admixed with small numbers of plasma cells, lymphocytes, and rare neutrophils (Fig. 3). The inflammatory cells were occasionally arranged in discrete granulomas. Gomori methionine silver (GMS) and periodic acid–Schiff revealed rare, variably sized, septate hyphae, 5–8 μm in diameter, admixed with numerous, intracellular, terminal, round, variably sized conidia, 6–30 μm in diameter, with a discrete outer wall (Fig. 4). Occasional narrow- and broad-based budding was also present. The hyphae and conidia were poorly stained with periodic acid—Schiff but were readily demonstrated by GMS stain.
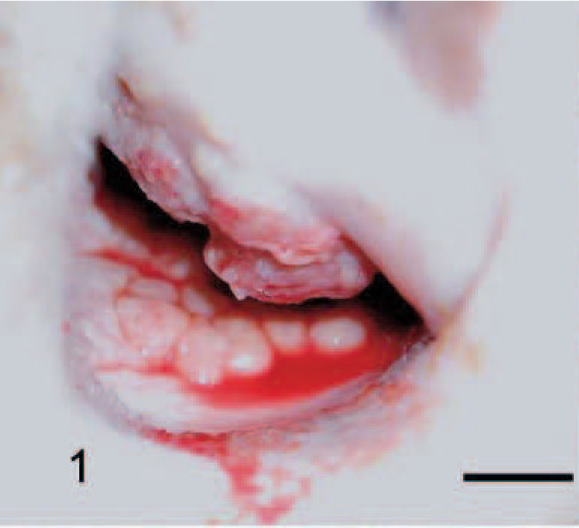
Nasal mucosa; cattle. The nasal mucosa is partially obliterated by cobble-stone–appearing granulomas. Granulomas in the roof are ulcerated. Bar = 2 cm.
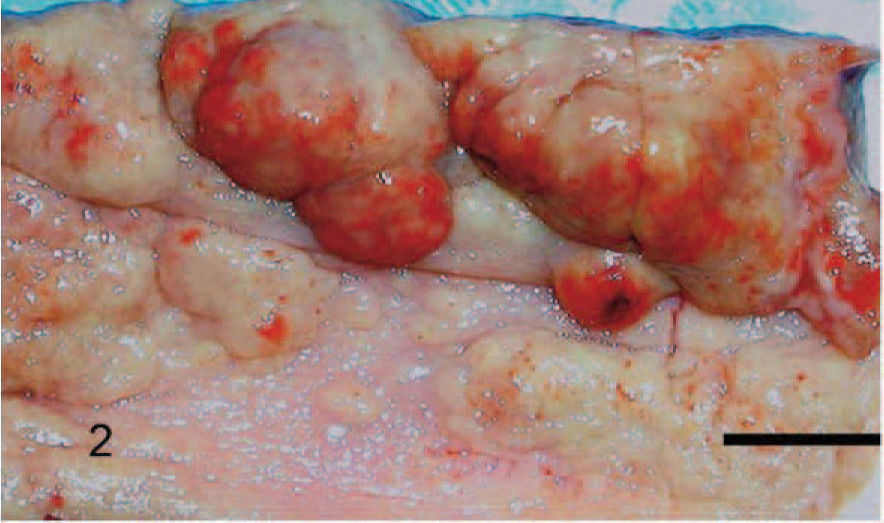
Nasal mucosa; cattle. Multiple, variably sized granulomas expand the nasal mucosa. Bar = 1 cm.
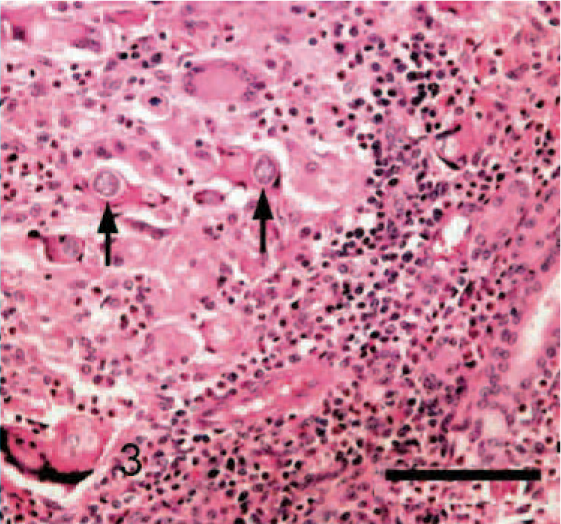
Nasal mucosa; cattle. Epithelioid macrophages, eosinophils, and multinucleate giant cells admixed with small numbers of plasma cells, lymphocytes, and neutrophils are present in the mucosa. Occasionally, phagocytosed round conidia with discrete outer walls are also present (arrows). HE. Bar = 100 μm.
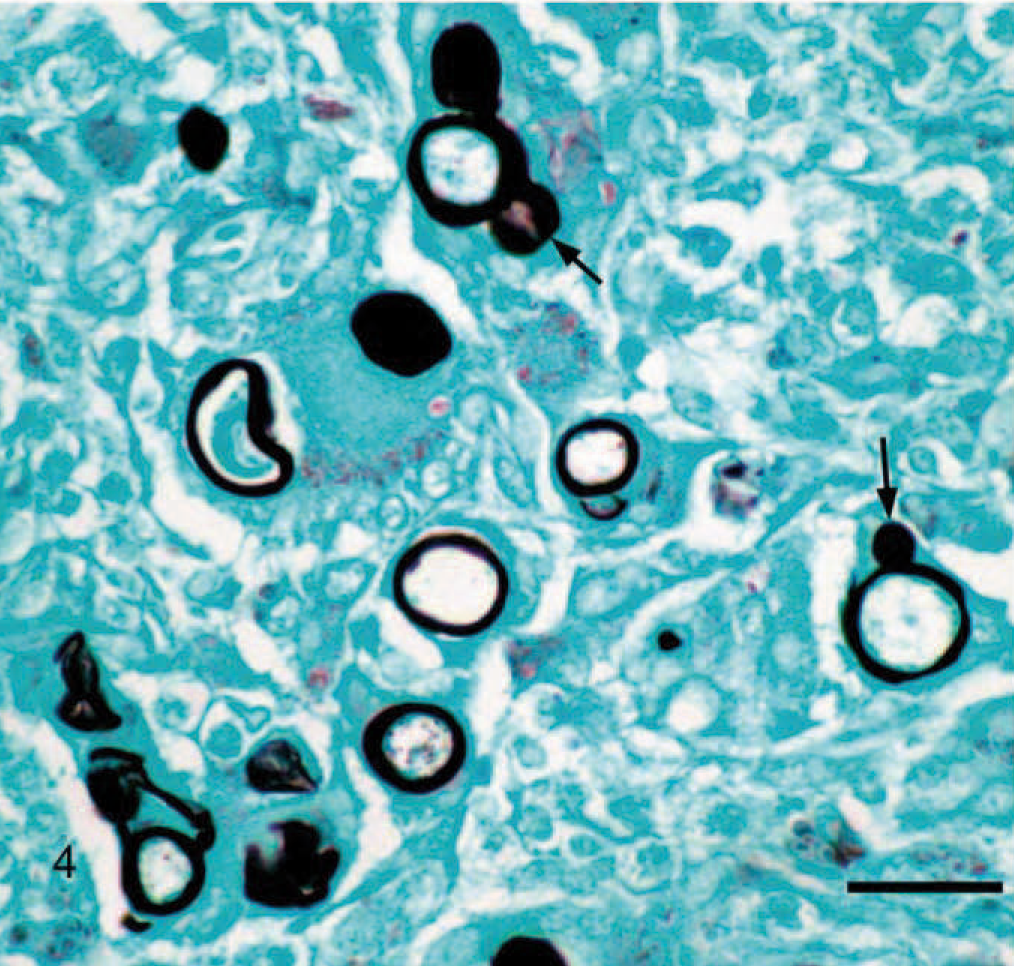
Nasal mucosa; cattle. Note multiple variably sized, phagocytosed, terminal, round conidia, 6–30 μm in diameter, with discrete outer walls. Occasional budding from the mother spore is present (arrows). GMS. Bar = 25 μm.
The overlying intact mucosa was mildly hyperplastic and contained epithelial cells with mitotic figures, intracellular and intercellular edema, and numerous transmigrating eosinophils and neutrophils. The tissue from the nasal mucosa was submitted for mycologic culture. A mycelial fungus was recovered by culture on Sabouraud agar after 5 days incubation at 25°C. The surface colony morphology was spreading white, cottony aerial hyphae with a black reverse pigmentation, which imparted light-grey coloration when conidia were produced after 7 days of incubation. Microscopic morphology revealed septate hyphae with conidiophores bearing single, 5 × 10 μm, unicellular, oval conidia (Fig. 5). The conidia were hyaline, cylindric to clavate. The homothallic nature (the ability to form a sexual stage in culture) of the fungus was demonstrated by the formation of closed, dark-brown, thick-walled, spheric cleistothecia (fruiting bodies) measuring 150–200 μm in diameter, which occurred in the central, mature portion of the culture. 6, 7 When crushed under a coverslip, thin-walled spheric ascocarps were released from these cleistothecia. The ascocarps contained numerous, pale-brown, ellipsoidal-to-oblate, lemon-shaped ascospores measuring 4–5 × 7–9 μm. 6, 7 Growth of the fungus at both 25°C and 37°C was filamentous, and no growth occurred on media containing cycloheximide (Mycosel agar). On the basis of these morphologic characteristics, the fungus was subsequently identified as Pseudallescheria boydii species complex (anamorph Scedosporium apiospermum). For better understanding, P. boydii species complex will be referred to as P. boydii in this report.
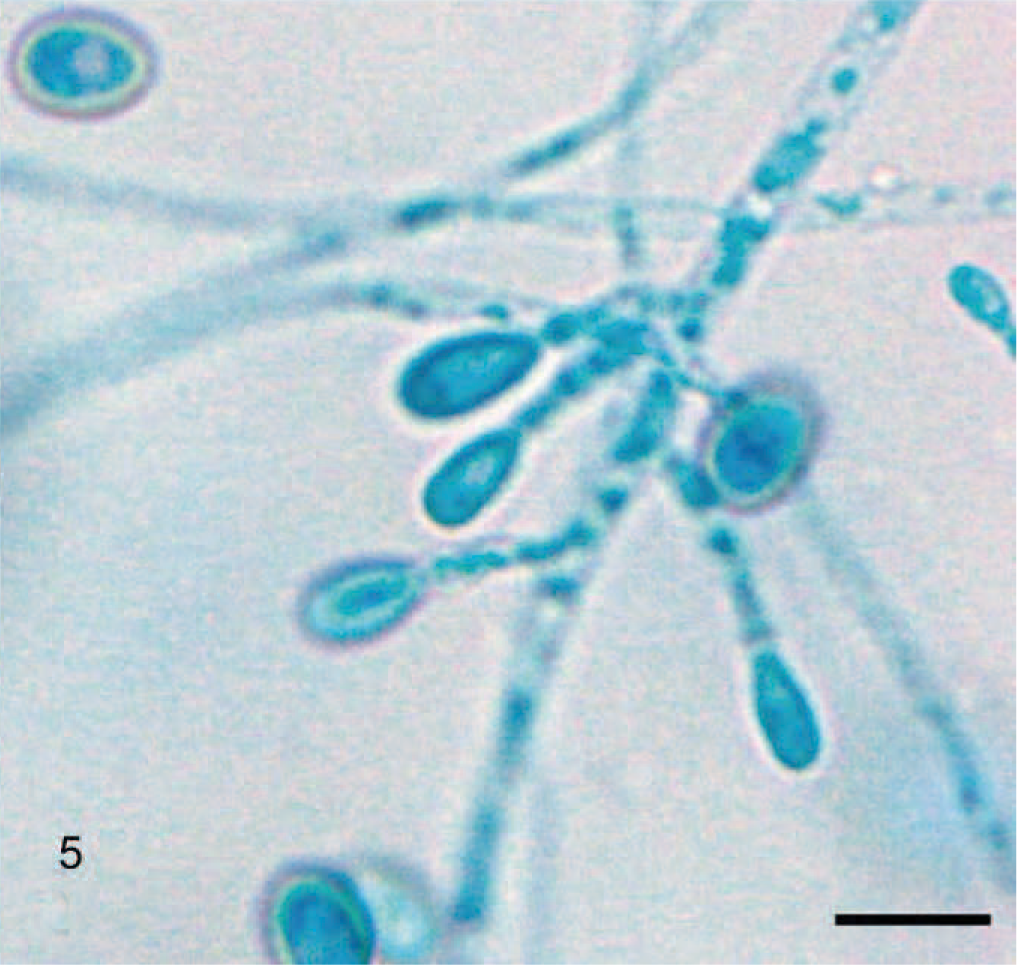
Pseudoallescheria boydii culture reveals septate hyphae with short and hyaline conidiophores bearing single unicellular oval conidia. Unstained colonies on Sabouraud agar. Bar = 10 μm.
P. boydii was previously known as Allescheria boydii and Petriellidium boydii. The asexual form or anamorph is S. apiospermum. Both forms produce conidia in culture, but only P. boydii forms fertile fruiting bodies for sexual reproduction (teleomorph). The 2 forms represent a single species belonging to order Microascales, phylum Ascomycota, and both are associated with disease in animals and humans.
Historically, the nomenclature and taxonomy of P. boydii, which is more correctly characterized as a fungal species complex, is very confusing. 6, 7 This confusion is evidenced by older literature references to the anamorphic form as Monosporium apiospermum and the teleomorphic form as A. boydii or Pteriellidium boydii. Recent phylogenetic analysis has shown that the P. boydii complex is composed of at least 6 species: P. boydii, Pseudallescheria angusta, Pseudallescheria ellipsoidea, Pseudallescheria fusoidea, Pseudallescheria minutispora, and Scedosporium aurantiacum. 6 Distinct morphologic and cultural differences do not exist between all the species. The differences between the species exist at a genetic level that can only be separated through molecular phylogenetics of deoxyribonucleic acid sequences. 6 Therefore, we decided to use the name as P. boydii species complex rather than emphasizing 1 species. Identification of the particular species of the P. boydii complex causing disease is becoming increasingly important as differences in potential invasiveness, antifungal drug susceptibility, and fungal environmental sources may be species-associated. 7
The disease nomenclature for infections by this agent has been and continues to be confusing. Because of the various clinical entities and the ongoing nomenclature revision, it is now recommended to apply a clinical disease name along with the anamorphic strain name in characterizing an infection caused by this group of fungi. 7 Therefore, use of the name Scedosporium is suggested. 7
These fungi are ubiquitous and have successfully adapted to a human-dominated environment. They have been isolated from a variety of substrates including soil, decaying vegetation, polluted streams, sewage sludge, and poultry/cattle manure. Despite their ubiquitous nature, infections by these fungi are extremely rare and are reported most commonly in immunocompromised patients. In humans, these species can cause localized and systemic infections manifested as respiratory, central nervous system, osteoarticular, ocular, endovascular, and lymphocutaneous disease associated with high mortality. 14
In animals, P. boydii is predominantly implicated in the formation of trauma-induced eumycotic mycetomas characterized by granule formation and discharging sinus tracts. 4, 12 P. boydii is also associated with the extremely rare bovine and equine mycotic abortions, fungal pneumonia in a calf, granulomatous rhinitis and onychomycosis in the horse, and eumycotic mycetoma and keratomycosis in the horse and dog. 5, 7– 11, 15, 18 Apart from localized infections, P. boydii is also implicated in the systemic infection in dogs. 1
P. boydii is an emerging pathogen in humans, and most of the infections are reported in immunocompromised individuals undergoing organ transplant or chemotherapy; however, it seems that P. boydii infections in animals can occur in immunocompetent hosts. 17 In all the cases of P. boydii infection in animals that we reviewed, the authors either did not discuss immune status or a predisposing factor was not recognized. No obvious predisposing factors, including bovine viral diarrhea and bovine herpes virus 1 infection and pathogenic bacteria, were demonstrated in this case. Presumably, penetration by a foreign body or, alternatively, damage to the nasal mucosa by other means and subsequent inhalation of pathologic numbers of fungal spores could have resulted in localized development of the nasal granulomas and may be considered as a potential inciting cause. However, the gross and histologic lesions were present in both nares; therefore, this is merely a speculation.
No clinical, gross, or histologic evidence of a systemic dissemination was observed in this case. The infection was limited to the nasal cavity and, similar to horses, granulomas did not invade the underlying bone. Occasionally, nasal infections can invade the vomer bone in dogs. 2 Differential diagnosis for P. boydii rhinitis in cattle includes atopic rhinitis, nasal tumor (e.g., lymphoma, osteosarcoma, squamous cell carcinoma), foreign body, nasal actinobacillosis or actinomycosis, and sinusitis or rhinitis caused by other fungal diseases such as rhinosporidiosis, aspergillosis, and phycomycosis.
The morphology of P. boydii and the accompanying eosinophilic granulomatous host response are distinct and should not be confused with other commonly observed pathogenic fungal species in cattle. The hyphae in tissue sections had some morphologic resemblance with Aspergillus and Fusarium sp., both of which are common pathogenic fungi in cattle. However, no angioinvasion or dichotomous hyphal branching typical of these genera was observed. The broad-based budding suggested Blastomyces dermatitidis, but the size variability and the presence of hyphae were not typical of B. dermatitidis.
A peptidopolysaccharide (rhamnomannan) present in the mycelium of P. boydii is a potent antigen and is capable of generating specific antibodies in rabbits, which can be used for identification of P. boydii, particularly in cases of mixed infections. This antibody recognizes P. boydii species complex–specific peptidopolysaccharide (rhamnomannan) and does not cross-react with other morphologically distinct rhamnomannan containing fungal hyphae such as Sporothrix schenckii. 17 The antibody has been used in ELISA and immunofluorescence for detecting P. boydii fixed mycelia and extract obtained from the mycologic culture. 17 For identification in a mixed infection using immunohistochemical and polymerase chain reaction–based techniques, readers are referred to other articles. 8, 19
A comprehensive review of treatment regimens is beyond the scope of this article; therefore, readers are referred to other articles. 13, 16 Briefly, treatment of P. boydii is frustrating and expensive since the organism exhibits some level of inherent resistance to most antifungal agents. 2 If treatment is attempted, antifungal-susceptibility testing of isolates seems to be imperative. Preferred treatment for a localized lesion is surgical debridement with adjuvant topical clotrimazole or miconazole application. 3, 4 Combination treatment including amphotericin B with itraconazole and fluconazole 13 and posaconazole and voriconazole alone or in combination with caspofungin and terbinafine has been used successfully in humans. 13, 16 Since P. boydii infections occur predominantly in immunosuppressed patients, combining antifungal agents with an immunomodulator (granulocyte colony–stimulating factor and granulocyte-macrophage-colony–stimulating factor) that can restore immune competence may provide a successful outcome. 13
Footnotes
Acknowledgements
We thank Drs. D. A. Sutton and R. Morton for critically reviewing the manuscript. The assistance of Ms. B. Handlin in preparing the manuscript and Ms. J. Ely for performing the special stains is highly appreciated.
