Abstract
This report describes the use of polymerase chain reaction and DNA in situ hybridization to diagnose Aleutian mink disease parvovirus DNA in various tissue specimens from 2 companion striped skunks. Clinical, laboratory, and microscopic findings also support a clinical diagnosis of Aleutian disease in these mink.
Keywords
Aleutian mink disease parvovirus (ADV) is known to infect several members of the Mustelidae family, with mink and ferrets being the most commonly reported hosts. Aleutian disease (AD) in adult animals is characterized by hypergammaglobulinemia and immune-complex disease. 2, 5 The most common form of AD involves a persistent infection due to the inability of the host immune response to neutralize and eliminate the virus. 2 Lymphoplasmacytic infiltration of various organs, immune complex deposits in blood vessels, and severe mesangioproliferative glomerulonephritis ultimately lead to death. 2
Natural ADV infection and disease have been described mink and ferrets, although raccoons, otters, and other mustelids also have been reported to have ADV-induced lesions on histopathology and anti-ADV antibody seropositivity. 1, 4, 6, 10 Ferrets are the only known companion animals capable of developing natural disease as a result of ADV infection. 7– 9 Along with several other free-ranging members of the Mustelidae family, striped skunks (Mephitis mephitis) may be experimentally infected with ADV. 4, 6 Anti-ADV seropositivity has been reported in free-ranging skunks, but natural ADV infection and disease have not been documented in either wild or companion striped skunks. Pet striped skunks remain uncommon as companion animals and information regarding diseases in these animals is limited. This report presents two naturally acquired cases of AD in pet skunks. The disease was diagnosed using DNA in situ hybridization and polymerase chain reaction (PCR).
Case 1
A 6.5-year-old male striped skunk died unexpectedly and was necropised by the referring veterinarian. The original source of this animal was not known. Gingival hyperplasia and chronic dental disease were noted 1 year prior to death. The most recent serum biochemical profile, performed within ∼6 months of death, revealed normoproteinemia (7.7 g/dl; reference interval: 4.4–8.2 g/dl) with normoalbuminemia (2.5 g/dl; reference interval: 2.3–4.5 g/dl), hyperglobulinemia (5.2 g/dl; reference interval: 1.2–4.8 g/dl), and decreased albumin : globulin (A : G) ratio (0.48 : 1; reference interval: 0.6–1.1 g/dl). Increased aspartate aminotransferase (AST) activity (134 IU/l; reference interval: 47–124 IU/l) also was present. The most recent complete blood cell count had been performed ∼14 months prior to death and was within reference intervals for the species. The clinical pathology data suggested probable antigenic stimulation resulting in hypergammaglobulinemia and increased hepatocellular permeability.
Abnormal necropsy findings included ventricular wall thickening and a small quantity of peritoneal fluid. Samples of lung, liver, spleen, stomach, kidney, and heart were fixed in 10% neutral-buffered formalin and submitted to the University of Georgia Veterinary Diagnostic Laboratory. Fixed tissues were routinely processed, sectioned, and stained with hematoxylin and eosin (HE).
Microscopically, large foci of myocardial fibrosis were observed in the right ventricular free wall, interventricular septum, and apex of the left ventricular free wall. Associated inflammation consisted of small aggregates of lymphocytes and plasma cells and scattered hemosiderin-laden macrophages, indicating previous hemorrhage. The renal interstitium was diffusely infiltrated with large numbers of plasma cells (Fig. 1). Scattered plasma cells contained prominent eosinophilic cytoplasmic droplets (Russell bodies), indicative of active antibody production. Some renal tubules contained eosinophilic hyaline casts, whereas a few other tubules contained neutrophils. The liver contained scattered aggregates of hemosiderin-laden macrophages. Remarkable microscopic abnormalities were not observed in sections of spleen, stomach, or lung.
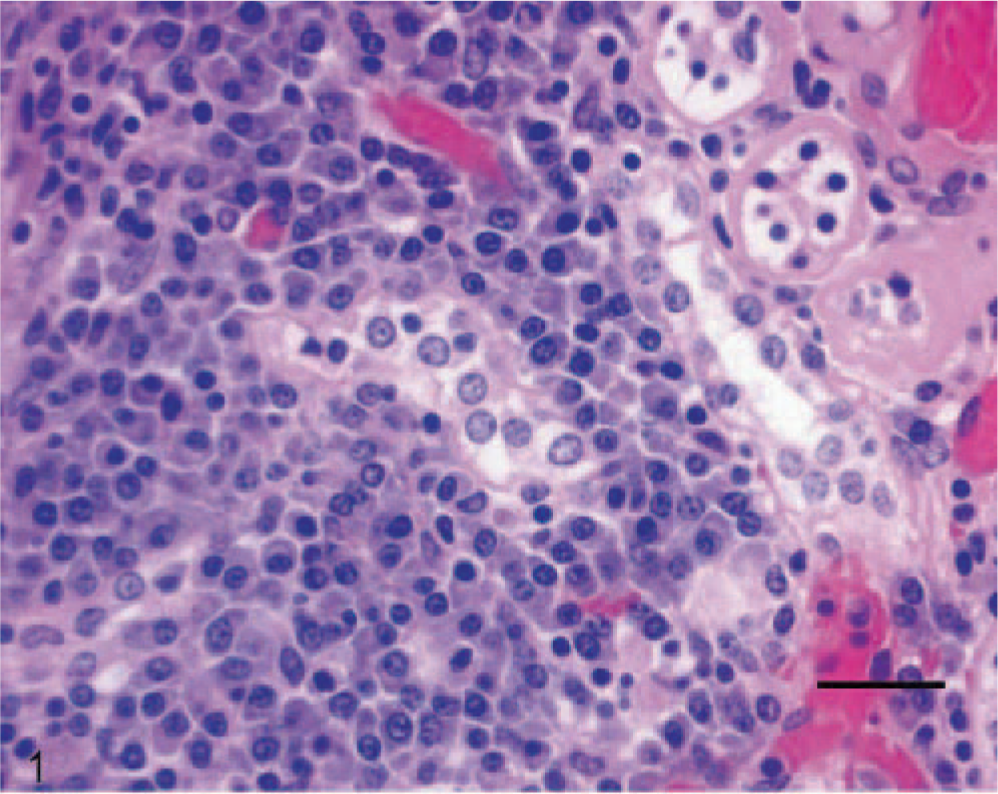
Kidney; skunk; case 1. Lymphoplasmacytic inflammation in the renal interstitium of a skunk with Aleutian disease. HE. Bar = 20 μm.
The histologic diagnosis was focally extensive myocardial fibrosis and severe, lymphoplasmacytic to purulent, tubulointerstitial nephritis. Immunohistochemical staining of a replicate section of kidney was negative for Leptospira spp.
Sections of formalin-fixed tissues were analyzed for the presence of ADV by DNA in situ hybridization. The tissues were incubated with a DNA probe consisting of a PCR-amplified, double-stranded clone of ADV-G DNA labeled with digoxigenin (PCR DIG Labeling Mix; Roche Applied Science, Indianapolis, IN). 3, 6 Sites of probe hybridization were visualized by high-affinity immunocytochemistry using antidigoxigenin antibody (Digoxigenin Detection Kit; Roche Applied Science) conjugated to alkaline phosphatase (AKP) and a chromagen solution (Digoxigenin Detection Kit) of nitroblue tetrazolium dye. The presence of DNA–DNA hybrids was identified by the deposition of blue-black insoluble pigment (formazan). Positive and negative tissue controls were archived sections of liver from ferrets with and without ADV infection, respectively. DNA in situ hybridization was strongly positive for ADV in the periarteriolar lymphoid sheaths and lymphoid follicles of the spleen, gastric submucosa, hepatic parenchyma, renal interstitium and tubules, and myocardium. Some pulmonary alveolar macrophages were also positive for ADV following DNA hybridization. ADV staining in the liver was present in a nuclear pattern within scattered hepatocytes, but it also was present in foci of lymphoplasmacytic inflammation within the parenchyma. In the kidney, staining for ADV was present in a nuclear to diffuse pattern within renal tubular epithelial cells (Fig. 2).
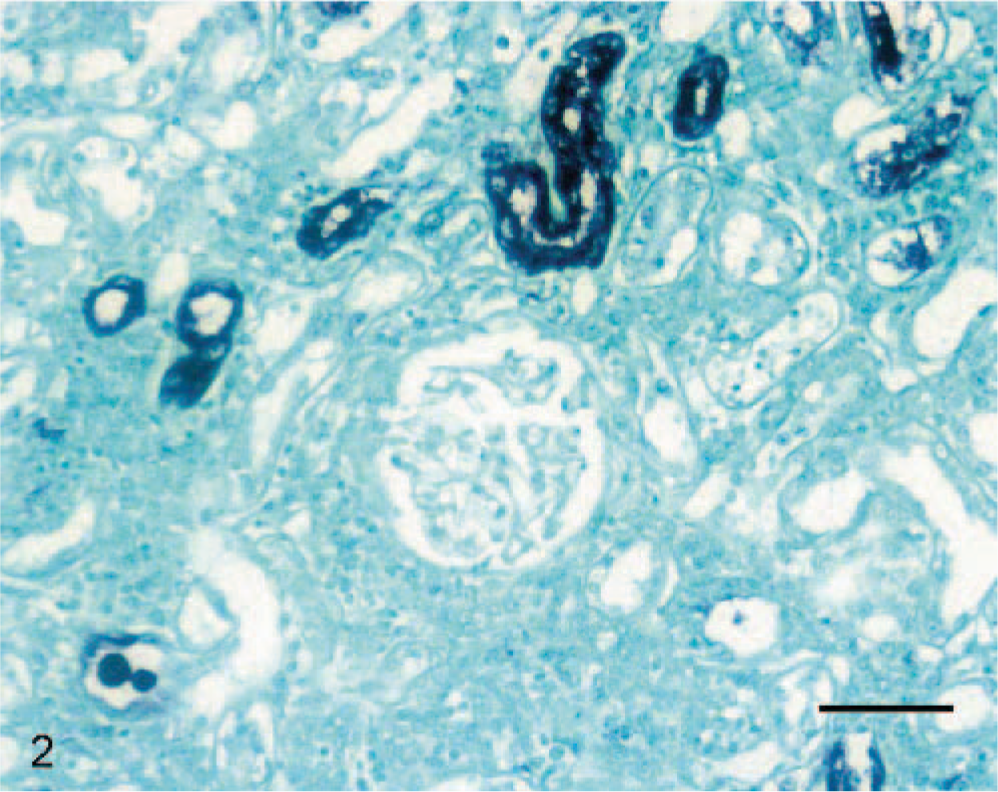
Kidney; skunk; case 1. Aleutian disease viral nucleic acid (blue-black formazan pigment deposition) is present within nuclei of scattered renal tubular epithelial cells. Some degenerating renal tubular epithelial cells have more diffuse staining. DNA in situ hybridization for ADV with formazan chromagen and fast green counterstain. Bar = 20 μm.
DNA subsequently was isolated from paraffin-embedded tissues and amplified via PCR. The ∼615-bp DNA fragment was equivalent to that generated by a known ADV positive control. A BLAST search revealed that the nucleic acid sequence of this fragment was 92% homologous to reported GenBank sequences of the ADV capsid protein VP 2 gene.
Discussion
Case 2
A 3-year-old spayed female, striped skunk was presented to the referring veterinarian with an oral infection. This animal was obtained from a commercial breeder at 5.5 weeks of age and was housed with 20 other skunks at a private residence. The skunk had pyrexia (99°F; reference interval: 95–97°F), dehydration, and lethargy. Facial tremors were observed during physical examination. Intravenous fluids were administered, but the skunk died 48 hours later.
A complete blood count and serum chemistry profile were performed 2 days prior to death. Hematologic abnormalities included increased hemoglobin (16.8 g/dl; reference interval: 9–15 g/dl) and mean corpuscular hemoglobin (21.01 pg; reference interval: 12–20 pg) concentrations. Serum chemistry abnormalities included increased AKP activity (159 IU/l; reference interval: 14–111 IU/l), hyperphosphatemia (8.7 mg/dl; reference interval: 3.1–7.5 mg/dl), hyperproteinemia (11.2 g/dl; reference interval: 5.7–8.9 g/dl), normoalbuminemia (3.8 g/dl; reference interval: 2.3–4.5 g/dl), hyperglobulinemia (7.4 g/dl; reference interval: 2.8–5.1 g/dl), and a decreased A : G ratio (0.51 : 1; reference interval: 0.6–1.1 g/dl). Hypoglycemia also was present (37 mg/dl; reference interval: 71–159 mg/dl). The clinical pathology data suggested hemoconcentration, probable antigenic stimulation, and cholestasis. Hyperphosphatemia may have been secondary to decreased glomerular filtration. Neither hemolysis nor nutritional imbalances could be identified. Hypoglycemia suggested possible endocrinopathy or neoplasia.
The animal was submitted to the Ohio Department of Agriculture for necropsy. Gross alterations were minimal and included tan mottling of the liver; icterus involving the subcutis, omentum, and mesenteric fat; and pulmonary and pancreatic congestion. Tentative diagnoses of hepatopathy and jaundice were made. Portions of cerebrum, cerebellum, brainstem, heart, lung, lymph nodes, liver, pancreas, intestine, abdominal fat, spleen, thyroid glands, adrenal glands, kidney, and urinary bladder were collected for subsequent histologic examination. The formalin-fixed tissues were routinely processed, sectioned, and stained with HE.
Microscopic examination of HE-stained tissue sections revealed extensive hepatocellular necrosis. A moderate infiltrate of neutrophils was present within the parenchyma, whereas the portal triads contained many lymphocytes and plasma cells that extended into the periportal parenchyma. Bile ductular hyperplasia was present, and some of these structures contained neutrophils within the lumina. Scattered hemosiderin-laden hepatocytes and Kupffer cells also were observed. In the kidney, Bowman's spaces within glomeruli were distended with eosinophilic, flocculent to finely fibrillar material. Extensive coagulative necrosis of proximal renal tubular epithelial cells was present, and many epithelial cells contained large amounts of orange-brown granular pigment, suggestive of hemosiderin or bile. Minimal leukocyte infiltrates were noted. The urinary bladder contained a subserosal, circumferential, neutrophilic infiltrate that occasionally extended into the fibromuscular wall. Most of the urothelium had sloughed, but remaining epithelial cells in the lumen were devoid of cytoplasmic and nuclear inclusions. Mild lymphocytic perivascular cuffing was present within the brainstem. Sections of cerebrum had a few aggregates of microglial cells and astrocytes within the gray matter and multifocal infiltrates of small lymphocytes more superficially. The myocardium had multifocal, lymphoplasmacytic, perivascular infiltrates within the left ventricle and epicardial fat. Microsocpically, sections of lung were characterized by accumulation of proteinaceous fluid within alveoli and hyperplasia of bronchial-associated lymphoid tissue (BALT). Sections of spleen were expanded by increased numbers of lymphocytes and plasma cells. Scattered hemosiderin-laden macrophages were present. The thyroid gland had moderate multifocal lymphoplasmacytic infiltrates. The adrenal glands had multifocal cortical necrosis and hemorrhage. The pancreas contained scattered small aggregates of lymphocytes and plasma cells within the interstitium.
The histologic diagnoses included severe necrotizing suppurative and lymphoplasmacytic cholangiohepatitis with bile ductular hyperplasia and mild hepatic hemosiderosis, marked multifocal renal tubular necrosis and presumed hemosiderosis or bile accumulation within tubular epithelial cells, mild subserosal purulent cystitis, mild multifocal cerebral gliosis and encephalitis with lymphoplasmacytic perivascular cuffing, multifocal lymphoplasmacytic myocarditis and steatitis, pulmonary edema with BALT hyperplasia, splenic lymphoplasmacytic hyperplasia and hemosiderosis, moderate multifocal lymphoplasmacytic thyroiditis, acute bilateral adrenal hemorrhage and necrosis, and mild multifocal lymphoplasmacytic interstitial pancreatitis.
Warthin-Starry staining of the liver and kidney was negative for spirochetes, and a pooled tissue culture was negative for Leptospira spp. Fluorescent antibody staining of brain was negative for rabies virus. In addition, immunohistochemical staining of small intestine and lung was negative for canine parvovirus and canine distemper, respectively.
Specimens of frozen liver and spleen were submitted for DNA in situ hybridization and PCR analysis. DNA in situ hybridization was strongly positive for ADV in both organs, despite the presence of mild autolysis. The presence of autolysis caused the ADV staining reaction to be less localized than in well-fixed tissue, but staining was distinct and unquestionable. Following PCR, extracted DNA from the liver and spleen generated a DNA fragment that was ∼615 bp in length. This fragment was similar in size to that amplified from the positive control specimen for ADV (Fig. 3). The nucleotide sequence of the amplified fragments was 92% homologous to reported sequences of the ADV capsid protein VP 2 gene, as determined by a BLAST search of the GenBank database.
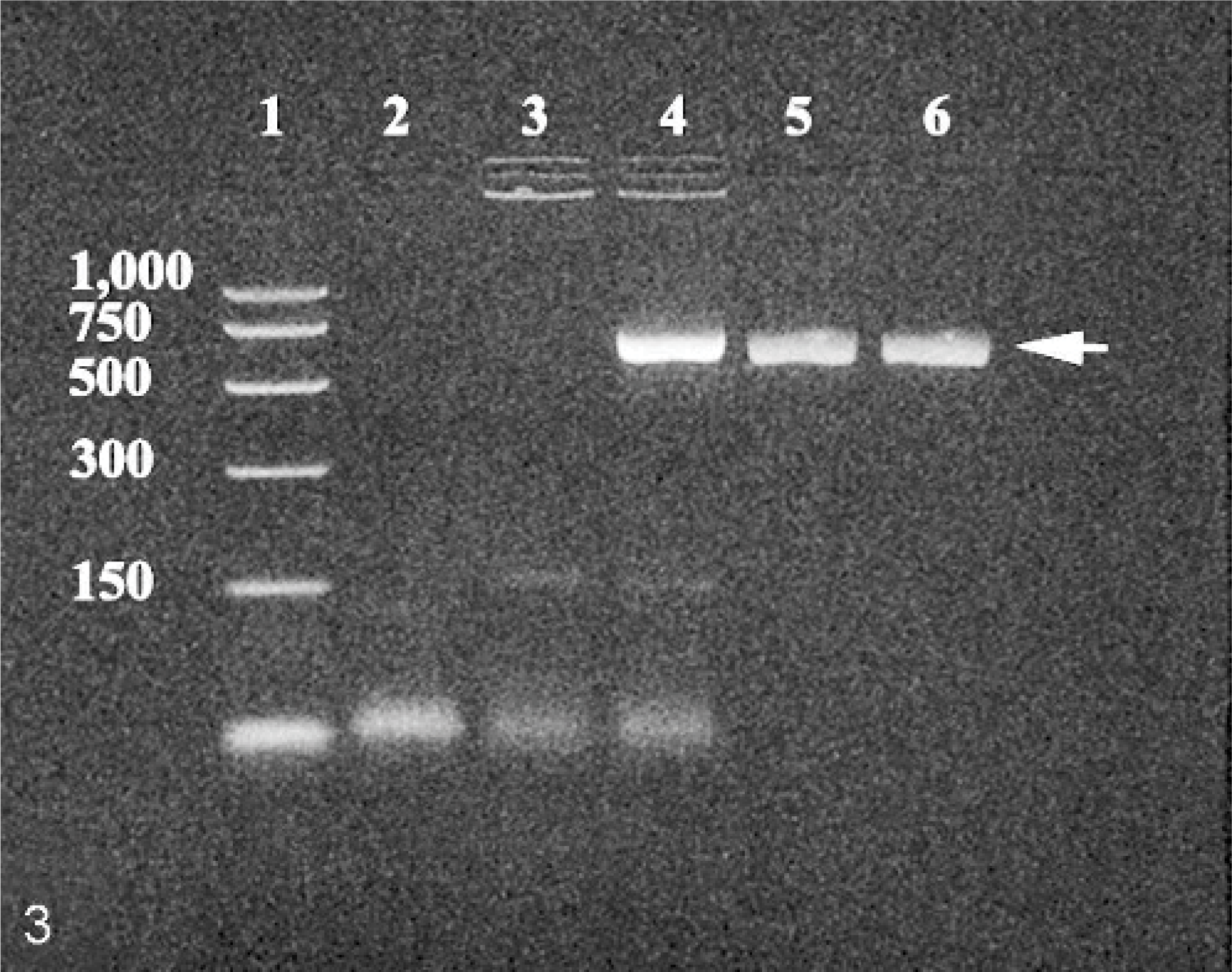
Ethidium bromide–stained agarose gel of reagents and PCR-amplified DNA products; skunk; case 2. Lane 1 = PCR size markers; lane 2 = distilled water used in the reaction (negative control to exclude contamination); lane 3 = ADV negative control; lane 4 = ADV positive control; lane 5 = DNA extract from frozen, autolytic liver; and lane 6 = DNA extract from frozen, autolytic spleen. Arrow (right) indicates the position of the ∼ 615 bp DNA fragment amplified from the ADV target DNA.
Similar clinical chemistry findings in both skunks included hyperglobulinemia with a decreased A : G ratio and increased activity of hepatobiliary enzymes, such as AST and AKP, indicative of hepatocellular leakage or cholestasis, respectively. Hyperglobulinemia is considered to be one of the hallmarks of AD and is secondary to viral stimulation of humoral immunity. Increased activities of AST and AKP indicate the presence of hepatobiliary disease, which may have resulted from lymphoplasmacytic inflammation, viral infection of hepatocytes, and/or the accumulation of immune complexes associated with persistent ADV infection. The etiology of the hypoglycemia in the second skunk was not determined; however, improper sample handling was not involved. Other potential explanations for the hypoglycemia could include excess insulin release following pancreatic inflammation or reduction of hormonal regulation of glucose homeostasis due to hypothyroidism (lymphoplasmacytic thyroiditis) and/or hypoadrenocorticism (adrenal cortical hemorrhage and necrosis). Histologic changes for both skunks were typical of previous lesion descriptions in ADV-infected animals, especially mink, where the lymphoplasmacytic infiltrates are typical of AD.
Routine histology of these skunks would have been unable to document ADV infections in these animals. This problem is further compounded in that formalin fixation precludes virus isolation and may interfere with fluorescent antibody staining techniques. In addition, severe autolysis may obscure subtle histologic changes. Definitive diagnosis of ADV infection was made by DNA in situ hybridization and PCR testing using selected primers for the nonpathogenic, laboratory-adapted G strain of ADV. 6 These techniques were able to diagnose ADV infection despite formalin fixation, routine processing, paraffin embedding, autolysis, and freezing of the tissue specimens. Lymphoplasmacytic inflammation may have many causes and is not specific for a particular virus. Although immune complexes were not demonstrated, the biochemical and histologic changes are considered typical of AD in mink and ferrets. The presence of these changes in these skunks also suggests the presence of AD in the ADV-infected animals. The source of the naturally acquired ADV infection in these skunks is speculative, but it would probably be associated with infected breeding facilities, commercial pet supply facilities, or their ultimate housing, where they could be exposed to other ADV-infected mustelids.
Footnotes
Acknowledgements
This work was supported in part by donations of striped skunk enthusiasts to the Emerging Diseases Research Group and the UGARF Animal Health Fund at the University of Georgia. We thank Dr. Donald DeLong, Dr. Jennifer Sandy, Dr. Frank Krupka, Ms. Robin Newsom, Ms. Deborah Cipriani, and Ms. Deborah Rogers for the medical histories and collection of clinical samples.
