Abstract
Hepatoblastoma (HB) is an uncommon pediatric liver tumor in humans and horses. In humans, HB is most frequently diagnosed in fetuses, neonates, and young children, whereas hepatocellular carcinoma (HCC) affects juvenile and adult humans. Hepatoblastoma in the horse is rare, with only 9 reported cases. Affected horses ranged in age from late-term aborted fetuses to 3 years. The current study describes 3 new cases of primary liver tumors in horses and reviews findings in relation to other reports on this condition. Tumors classified as HB were identified in a male Standardbred aborted fetus and in a 4-year-old Thoroughbred filly. Hepatocellular carcinoma was diagnosed in a 15-month-old Paint filly. In the Standardbred fetus, the tumor was only present in the liver. In the Thoroughbred and Paint fillies, primary tumors were in the right liver lobe and at the hilus, respectively, and there were metastases to other lobes (HB) and mesenteric lymph nodes (HCC). Tumors were sharply demarcated from adjacent tissue, nonencapsulated, compressive, and invasive. Consisting of cords and nests, or disorganized sheets of epithelial cells, tumors had variable stromal and vascular components. The fetal tumor contained areas of smaller, less differentiated cells with a pronounced mesenchymal component interpreted to be embryonal hepatic tissue. Diagnoses were based on tumor histomorphologic features, resemblance to hepatocyte developmental stages, age of the animal, and patterns of metastasis. Tumors classified as HB were α-fetoprotein immunoreactive. Primary hepatic tumors in the horse are diverse in morphology and include subtypes compatible with classification criteria applied to human tumors.
Introduction
Primary malignant liver tumors are uncommon in young animals and humans. Most are classified as hepatoblastoma (HB) or hepatocellular carcinoma (HCC). 38 Hepatoblastoma is more common among neonates or young children, while HCC occurs at a higher frequency in juveniles. 40 Fetal and embryonal subtype HB are thought to arise from bipotential hepatocyte and cholangiocyte precursors and are, in part, reflective of successive hepatic developmental stages. However, the origin of small cell undifferen-tiated HB is contested. 28,40 Hepatocellular carcinoma is thought to arise from more differentiated hepato-cytes or, in some cases, from hepatic stem cells. 6 Hepatic oval cells are considered to be adult stem cells that are activated during regeneration, but evidence regarding their malignant transformation is scarce. 28 Different histomorphologic schemes described for classification of HB and HCC of humans are associated with specific prognoses. 6,7,38,40 The causes of HB are largely unknown, but relation to genetic conditions such as abnormal gene imprinting in Beckwith–Wiedemann syndrome or mutated adeno-matous polyposis coli tumor suppressor genes has been reported. 34 Hepatocellular carcinoma typically occurs in individuals with preexisting chronic hepatic disease from viral infections or toxin exposure or from metabolic storage disease. 16 Neoplastic hepato-cytes may revert to production of proteins not normally produced postnatally, such as α-fetoprotein (AFP). Detection of AFP has utility as an immuno-histochemical (IHC) tumor marker for diagnosis, and serum concentration is useful in both diagnosis and assessment of response to therapy. 5,10,12,16,38
Primary equine liver tumors are rare, with only 17 cases reported, of which 9 were classified as HB (Table 1). 3,4,8,13,18,19,21,22,24,27,29,32,33,35 Other reports of primary hepatic tumors in fetuses and juvenile horses included mixed hamartoma, 33 mesenchymal hamartoma, 4 and HCC. 18,32 In mature horses, cholangiocel-lular carcinoma 24 and combined HCC and cholan-giocarcinoma have also been diagnosed (Table 2). 19 Horses with primary hepatic tumors ranged in age from late-term aborted fetuses to 3 years. Because such tumors are rare in horses, no classification scheme has been devised, but adaptations from their human correlates are applied. Genetic influences on tumor formation in horses are unknown, although 1 case was associated with a facial malformation. 36
Hepatoblastoma in humans most commonly involves the right liver lobe. 16 The number of reports of equine HB is too low to make generalizations, but the primary tumor was in the left liver lobe in 6 out of 9 equine HB cases. 3,8,13,21,22,27,29 Ultrasonographically, human HB typically appeared as a lobulated, vascular, solid mass with calcification and hypoechoic areas that may have indicated necrosis or hemorrhage. 34 Metastases to abdominal lymph nodes are considered rare in HB but not in HCC. 12 Hepato-blastoma features commonly reported in horses included hepatomegaly, single to multifocal masses with heterogeneous echogenicity, encapsulation, and cavitation. 3,8,13 Hepatoblastomas in humans occasionally elaborate cytokines or hormones resulting in polycythemia, hypoglycemia, thrombocytosis, 2,15,25 precocious puberty and virilization, 26 osteoporosis, 1 or opsoclonus–myoclonus syndrome. 39 Some similar paraneoplastic effects such as polycythemia 3,13,21 and persistent hypoglycemia 13 have also been described in horses.
The classification scheme used by the International Childhood Liver Tumours Strategy Group indicates that the histologic diagnosis of HB is based on the presence of neoplastic cells with the appearance of fetal or embryonal hepatocytes in narrow to broad trabeculae, small undifferentiated cells, or any of these admixed with mesenchymal cells, with or without areas of teratoid differentiation. 40 In general, HB cells are smaller than benign hepatocytes, while HCC cells are larger. 40 In children, cure for HB depends on completeness of tumor excision. Chemotherapy may be used to reduce the size of the tumor prior to surgery but, by itself, is not curative. 14 If complete resection is not possible, then liver transplantation may be considered. 9 Neither surgery nor chemotherapy has been attempted in equine hepatic tumors, and the prognosis of such tumors in horses appears, therefore, uniformly extremely poor. The current study describes 3 cases of primary hepatic neoplasia in horses and compares the cases with those previously reported. The cases in the present study comprised all primary equine hepatic tumors identified by computerized search of medical records from 1984 to 2009 at the University of Guelph (Guelph, Ontario, Canada), the University of Prince Edward Island (Charlottetown, Prince Edward Island, Canada), and the University of Saskatchewan (Saskatoon, Saskatchewan, Canada). Protocols in place throughout this period consisted of 24-hr fixation in formalin followed by paraffin embedding. Fresh sections were cut prior to each IHC staining procedure.
Because the diagnosis of human HB can be difficult, IHC panels may be used, but there is no one pattern specific to HB. 12,37 Commonly used markers include low-molecular-weight cytokeratin, AFP, and α-1-antitrypsin, among others. Hepatocyte paraffin-1 (HepPar-1), though not commonly used in childhood tumor IHC panels, 31 is a sensitive and specific marker of differentiated hepatocytes that reacts with liver cell mitochondria and has been used to identify primary hepatic neoplasms. 20 HepPar-1 has also been shown to stain some HB. 31
Cases
Tissues were received from an 8.5-month-old aborted male Standardbred fetus (horse no. 1). No clinical information on the dam was received. Tissue samples included lung, kidney, liver, testis, and spleen. Liver sections contained a biphasic, nonen-capsulated, but clearly demarcated tumor. The superficial components of the tumor were composed of small polygonal epithelial cells organized into packets by loosely arranged streams of mesenchymal cells, interspersed with larger epithelial cells (Fig. 1A). The small epithelial cells had indistinct cell borders with scant, pale, basophilic, vesicular cytoplasm, and small nuclei with multiple prominent nucleoli. Anisokaryosis was 5-fold, but there was less than 1 mitotic figure per 10× field. The deeper portion of the tumor, forming the majority of the mass, contained sheets of larger polygonal epithelial cells, small sinusoids, and multifocal collections of arteries and veins, consistent with primitive portal areas lacking bile ducts (Fig. 1B). The latter were surrounded by wide bands of loosely arranged mesenchymal cells that connected multifocally with the subcapsular population. Cells comprising the majority of the mass resembled mature hepatocytes with pale eosinophilic cytoplasm, a central round nucleus, and single to multiple prominent nucleoli. Multifocal clusters of the small neoplastic cells were admixed with the larger population and were noted within blood vessels. There was occasional extra-medullary hematopoiesis in sinusoids and scattered single cell necrosis. The interpretation from the case was HB, most similar to human subtype “wholly epithelial type, mixed fetal and embryonal subtype.” 37,40
Horse no. 2, a 4-year-old Thoroughbred (TB) filly, had a history of melena and was treated with omeprazole. Presence of frank blood in feces initiated referral to the Teaching Hospital, University of Guelph. On admission, the filly was depressed with moderate tachycardia, tachypnea, and mildly injected mucous membranes. A complete blood cell count (CBC) was unremarkable except for lymphopenia (0.71 × 109/l; reference interval: 1.3–4.7 × 109/l) and thrombocytosis (542 × 109/l; reference interval: 83–270 × 109/l). Serum biochemical evaluation indicated elevated glutamate dehydrogenase (GLDH), alkaline phosphatase, and gamma-glutamyltransferase (GGT) activities as well as increased bilirubin (65 μmol/l; reference interval: 21–57 μmol/l) and bile acids (68 μmol/l; reference interval: 0–28 μmol/l). Cholesterol and triglycerides were also slightly increased. These findings were consistent with hepatocellular necrosis, cholestasis, and reduced enterohepatic clearance. The horse was treated with a plasma transfusion, antibiotics, and vitamin K. Abdominal ultrasound examination revealed a markedly enlarged liver with mixed echogenicity. Two percutaneous liver biopsies were obtained for histopathologic assessment. Examination of the sections revealed mild to moderate portal fibrosis and biliary proliferation. A small area containing an unencapsulated population of disorganized, large polygonal cells was noted. The cells of this population had poorly defined cellular margins, abundant eosinophilic vacuolated cytoplasm, and round to oval nuclei. Anisokaryosis was 2-fold, and the mitotic rate was high (5 per 40x field). A diagnosis of undifferentiated carcinoma was made, and the filly was euthanized based on an extremely poor prognosis.
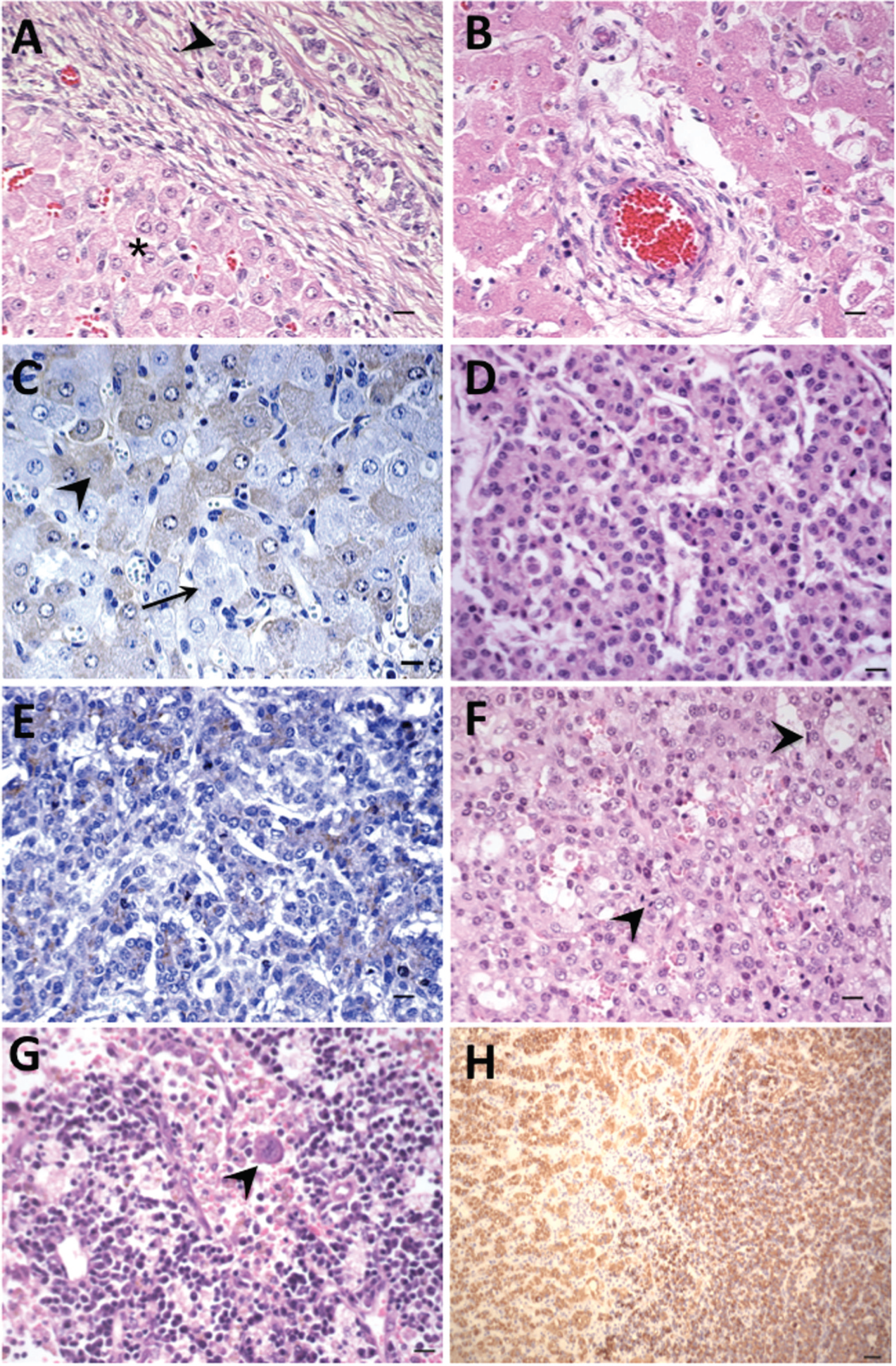
Primary liver tumors; horse no. 1 (A–C), horse no. 2 (D, E), horse no. 3 (F–H).
At necropsy there was subcutaneous edema in the ventral abdomen and perineum. The liver was markedly enlarged (36.8 kg, 6.7% of body weight) with rounded edges. Seventy percent of the right liver lobe, and less of all other hepatic lobes, contained numerous 5 mm to 3 cm diameter, multifocal to coalescing, irregular, and slightly raised yellow foci. On cut surface, the foci were yellow and friable and extended deeply to involve the parenchyma of affected areas. The gastric wall was edematous, and 60% of the glandular mucosa had superficial, widespread erosions and hemorrhages. There were no gross abnormalities in other organs.
On histopathology, the liver contained multiple lobules of an unencapsulated, well-demarcated, expansile, and infiltrative epithelial neoplasm. Lobules were composed of nests and anastomosing cords of cells delineated by a fine fibrovascular stroma. Neoplastic cells were polygonal and slightly smaller than normal hepatocytes, and there were 3 mitotic figures per 40x field. Nuclei were central and round with a single prominent nucleolus. There was 6-fold anisokaryosis, and the nuclear to cytoplasmic ratio ranged from 1:1 to 1:2. Tumor cells were evident in vessels of adjacent, unaffected tissue. Small foci of inflammatory cells, and low numbers of smaller cells with scant cytoplasm and hyperchromatic nuclei (consistent with a matrix population) were scattered throughout the neoplasm. There were multifocal areas of necrosis as well as large cystic areas filled with homogenous eosinophilic material. For horse no. 2, the morphologic diagnosis was HB, most similar to human “wholly epithelial type, embryonal subtype” (Fig. 1D). 37,40
Horse no. 3, a 15-month-old Paint filly with a history of quietness progressing to lethargy 2 weeks prior to presentation, was admitted to the Veterinary Teaching Hospital, University of Guelph. Prior to referral, an increase in the packed cell volume (PCV), strongyle infestation, fever, elevated serum protein, and icterus were noted. The referring veterinarian had treated the filly with intravenous electrolytes and calcium, and with oral fenbendazole and ivermectin. A course of ceftiofur sodium was begun 4 days prior to admission, at which time the PCV had been within normal limits. On admission, blood gas analysis revealed metabolic acidosis and hyponatremia, and from the CBC, a normocytic anemia (hematocrit: 0.18 l/l; reference interval: 0.28–0.44 l/l) with discolored plasma was identified. There was a mild leukocytosis with a neutrophilia, left shift, and monocytosis. Morphologic changes on the blood smear included agglutination of erythrocytes and increased Howell–Jolly bodies. Serum methemoglobin was 17.1% of total hemoglobin. Serum biochemical evaluation indicated markedly elevated GLDH and GGT activity, hypercholesterolemia, and hyper-bilirubinemia. Serum urea nitrogen and creatinine were also markedly increased. There was hypopro-teinemia (47 g/l; reference interval: 50–75 g/l) with a mild decrease in albumin (21 g/l; reference interval: (22–35 g/l). On day 3, urine methemoglobin was 89.8% of total urine hemoglobin. On day 5, urine specific gravity was 1.008, pH was 5.0, there was a trace positive protein reaction, and hematuria. A direct Coombs's test was negative. The filly developed marked ventral edema and icteric sclerae. Clinical findings were deemed consistent with antibiotic-induced, immune-mediated hemolytic anemia; hepatocellular damage; cholestasis; and renal failure. Percutaneous liver biopsies were obtained. One section had loss of normal liver architecture and contained sheets of polygonal cells with indistinct cytoplasmic borders, and basophilic, occasionally vacuolated, cytoplasm. Nuclei were round, central, with indistinct nucleoli. Cells were generally smaller than adjacent hepatocytes, and had minimal anisokaryosis and a high mitotic rate (3 per 40x field). Differential diagnoses were regeneration following severe injury, or hepatoma. Another liver section showed normal architecture with mild lymphocytic infiltrates and neutrophils clustered around portal triads, and hepatocyte changes suggestive of toxic or metabolic hepatopa-thy. The horse developed bloody diarrhea over the next 2 days and was euthanized.
At necropsy, the horse had icteric sclerae, ventral edema, and the abdomen contained 5 liters of serosanguineous fluid. Affecting approximately 60% of the liver were extensive, multifocal, coalescing, 2–20 mm foci centered on the hilus and the left lobe of the liver, with scattered foci in the right lobe. Foci were yellow, soft, and raised. Similar appearing tissue was found in the vena cava. The lung had scattered small white foci, and in the renal cortex, there were small, depressed areas. Microscopically, the liver masses were unencapsulated, multinodular, infiltrative, and composed of neoplastic epithelial cells arranged in disorganized sheets and trabecula. Neoplastic cells were polygonal, had indistinct cytoplasmic borders, and had variable amounts of pale eosinophilic cytoplasm (Fig. 1F). Nuclei were round, central, and had single nucleoli. An occasional binucleate form was noted. The nuclear to cytoplas-mic ratio ranged from 1:1 to 1:2, anisokaryosis was 8fold, and there was an average of 5 mitotic figures per 40x field. Neoplastic cells were generally smaller than normal hepatocytes. There were scattered, single, small cells with oval nuclei throughout the masses. Intersecting the mass at regular intervals were broad bands of spindle cells with variable amounts of eosinophilic fibrillar cytoplasm intermixed with ba-sophilic amorphous matrix. There were multifocal areas of hemorrhage, and scattered macrophages containing hemosiderin. The mesenteric lymph node was congested and contained neoplastic cells, which distended the subcapsular sinuses and formed focal aggregates in the medulla. Neoplastic cells were of similar appearance to those in the liver, but occasional giant cells with bizarre and multiple nuclei were observed (Fig. 1G). The lung contained small ab-scesses centered on pigmented fungal hyphae, and a diagnosis of phaeohyphomycosis was made. The kidney had subacute tubular necrosis with multifocal infarcts. The morphological diagnosis in this case was HCC.
Additional studies
Neoplastic tissues from each case were immuno-histochemically assessed for expression of vimentin, a cytokeratin 7 (CK7), a neuron-specific enolase a (NSE), S100, a synaptophysin, a pan-cytokeratin a (pan-CK, AE1/AE3), HepPar-1 (clone OCH1E5.2.10), a AFP, a and beta-catenin. b Heat-induced epitope retrieval was used to unmask antigens as necessary, and slides were stained with an automated immunostainer. c Protocols involved detection of diluted primary antibody with a polymer-linked labeled secondary antibody and Nova Red chromogen. In negative control slides, non-immune rabbit serum or buffer replaced the primary antibody. Positive controls consisted of normal equine and human liver sections, and human HB sections. Serum AFP concentration was measured in horse No. 2 with an enzyme-linked immunosorbent assay. d
In all 3 cases, tumor cells were negative for vimentin, NSE, S100, synaptophysin, and CK7. Bile duct epithelium labeled positively for CK7 in horses no. 2 and 3. Bile ducts were not evident in tumor sections from horse no. 1, and no labeling for CK7 was apparent. Application of antibodies to pan-CKs did not identify epithelial immunoreactivity. Tumors of horses no. 1 and 2 were negative for HepPar-1, but nontumor areas of horse no. 2 and all areas of sections from horse no. 3 had strong immunoreac-tivity for HepPar-1 (Fig. 1H). Tissues from horses no. 1 and 2 demonstrated immunoreactivity for AFP with diffuse faint and strong cytoplasmic labels, respectively (Fig. 1C, 1E). Serum AFP concentration in horse no. 2 was within the normal reference interval for humans. Tissues from horse no. 3 did not label with antibodies to AFP. Beta-catenin only labeled bile duct epithelium in unaffected liver tissues but was not detected in neoplastic cells. Findings of equine HB and HCC cases previously reported, and in the current study, are summarized in Tables 1 and 2, respectively.
Discussion
Hepatoblastoma has been diagnosed in stillborn fetuses, neonates, and juvenile horses less than 4 years of age, and there is no clear age-dependent occurrence of the subtypes of this tumor. 3,8,13,21,22,27,29 Traumatic rupture of these tumors as a result of dystocia was reported in equine neonates 22 and have also been reported in humans. 34 Juvenile horses frequently presented with a nonspecific history of lethargy, fever, inappetence, weight loss, diarrhea, and mucous membrane congestion. 3,8,13,21,29 Icterus, dyspnea, and pleural effusion were among the more specific signs noted. 3 Laboratory findings were indicative of inflammatory conditions, liver dysfunction or hepato-cellular necrosis, and erythrocytosis. 3,8,13,21,29 Serum erythropoietin was elevated in horses with secondary polycythemia and was a useful adjunct to diagno-sis. 3,13 Serum AFP concentration was elevated in a case of HCC 3 but not in horse no. 2 with HB despite tumor immunoreactivity for AFP. Possible reasons are limited AFP release into the blood stream from neoplastic hepatocytes or pulsatile release. Ultraso-nographically, markedly enlarged livers containing a mass of mixed echogenicity have been reported. In 6 out of 9 cases, at postmortem examination, the left liver lobe contained the primary tumor with metas-tases to other lobes. 3,8,13,21,22,27,29 In the cases presented herein, the affected lobe was not recorded in horse fetus no. 1, and in horse no. 2 the primary site involved the right liver lobe. Numerous metastatic sites outside the liver have been reported, 3,8,13,21,22,27,29 including 1 case that reported omental and mesenteric tumors. 29 The latter finding is inconsistent with those reported for humans, where HB was rarely, if ever, metastatic to abdominal lymph nodes. 12 In the present report, mesenteric lymph node metastasis was present only in the case classified as HCC (horse no. 3).
The histologic diagnosis of human HB is based on the similar appearance of the neoplastic cells to hepatoblasts 17,40 and falls into 1 of 2 broad categories: wholly epithelial type or mixed epithelial and mesenchymal type HB. 37,40 Wholly epithelial tumors have 4 subtypes, including fetal, embryonal (including mixed fetal and embryonal), macrotrabecular, and small cell undifferentiated. Mixed epithelial and mesenchymal pattern tumors contain osteoid and are further classified as those with or without teratoid features. 40 Fetal subtype tumors frequently have extramedullary hematopoiesis and accumulations of fat and glycogen, whereas embryonal type tumors have none of these features. 17 This latter feature of human HB is consistent with the findings of a report of 3 equine mixed epithelial and mesenchymal HB, where periodic acid–Schiff's reaction and oil red-O staining revealed areas containing glycogen or lipid, respectively. 22 In the cases presented herein, extensive lipid vacuoles were evident in tumor cells, and the presence of extramedullary hematopoiesis and the histomorphologic features further supported classification as mixed fetal and embryonal subtype.
In animals, HCC has a histologically variable appearance, and in the simplest classification scheme, 3 variants are described: trabecular, adenoid, or solid. 23 Trabecular HCC is the most frequent form and may be difficult to discern from normal liver. In adenoid HCC, rudimentary acini are noted that have lumina of variable widths, which may contain proteinaceous material. 23 Finally, solid HCC is composed of sheets of poorly differentiated and pleomorphic cells that do not form sinusoids and have a variable amount of stroma. 23 In the latter case, tumor cells may be difficult to recognize as being of hepatocyte origin, and giant cells may be present. This contrasts with HB in humans, where giant cells are an exceedingly rare finding. 21 Neither of the first 2 cases described in the current study contained giant cells; however, such cells were noted in the third case, which was consistent with classification as HCC. There are more cases of equine HB than HCC reported in the veterinary literature; however, given the rarity of HB in any species, whether that is reflective of true incidence of these tumors is questionable.
Immunohistochemistry is another diagnostic technique used in both human and equine hepatic tumor classification. 3,8,13,21,22,31,38,40 However, no IHC panel is distinctive for HB, as other tumors, including HCC, may also express markers such as AFP. 6,31 Human HB of the fetal and embryonal subtypes tend to have strong immunoreactivity for AFP, areas of mixed tumors may have some positive staining, and small cell undifferentiated forms do not stain. 37 Immuno-reactivity for AFP in equine tumors is faint to strongly positive in wholly epithelial type HB, and variably positive in mixed epithelial and mesenchymal tumors. 3,13,22 In the cases presented herein, there was faint cytoplasmic reactivity in the mixed fetal and embryonal subtype HB, and strong immunostaining in the cytoplasm of the embryonal subtype HB, which is similar to the patterns observed in human tumors.
Tumor tissue from horse no. 1 was uniformly negative for CK7 and pan-CK. Because biliary epithelium is expected to express these antigens, this confirmed an absence of bile ducts, as is common in human fetal subtype HB. 40 Interestingly, a section of normal liver from horse no. 1 contained bile ducts and demonstrated positive immunoreactivity for pan-CK, confirming normal development of liver in this area. HepPar-1 is a marker of hepatocellular differentiation that has been used to diagnose canine and human HCC, 11,30 but human HB may also be strongly positive for HepPar-1. 11 Tissues from horse no. 1 and tumor areas in horse no. 2 were negative for HepPar-1. The lack of HepPar-1 staining may indicate a phenotype less differentiated toward mature hepato-cytes. Both neoplastic and nonneoplastic hepatocytes in horse no. 3 had strong cytoplasmic reactivity for HepPar-1, indicative of hepatocellular differentiation. These findings further support the interpretation of tumors in horses no. 1 and 2 as HB, a less differentiated tumor compared with the more differentiated HCC in horse no. 3. Signal decay was not considered to be a factor in HepPar-1 reactivity, as horse no. 3 was the oldest case in the series. Human HB is associated with excessive Wnt/beta-catenin signaling. Expression and cellular location of beta-catenin are of diagnostic and prognostic value. 7 Lack of detection in equine neoplastic tissues may indicate that either beta-catenin signaling is not of importance in hepatocytic carcinogenesis of horses or that the antibody utilized had insufficient sensitivity for equine tissues.
Summary of findings reported for primary equine hepatoblastoma (HB).

IHC = immunohistochemistry; TB = Thoroughbred; EPO = serum erythropoietin concentration; AFP = α-fetoprotein; CK = cytokeratin; HepPar1 = hepatocyte paraffin-1; SB, Standardbred.
Summary of findings reported for equine primary hepatocellular carcinoma (HCC). *
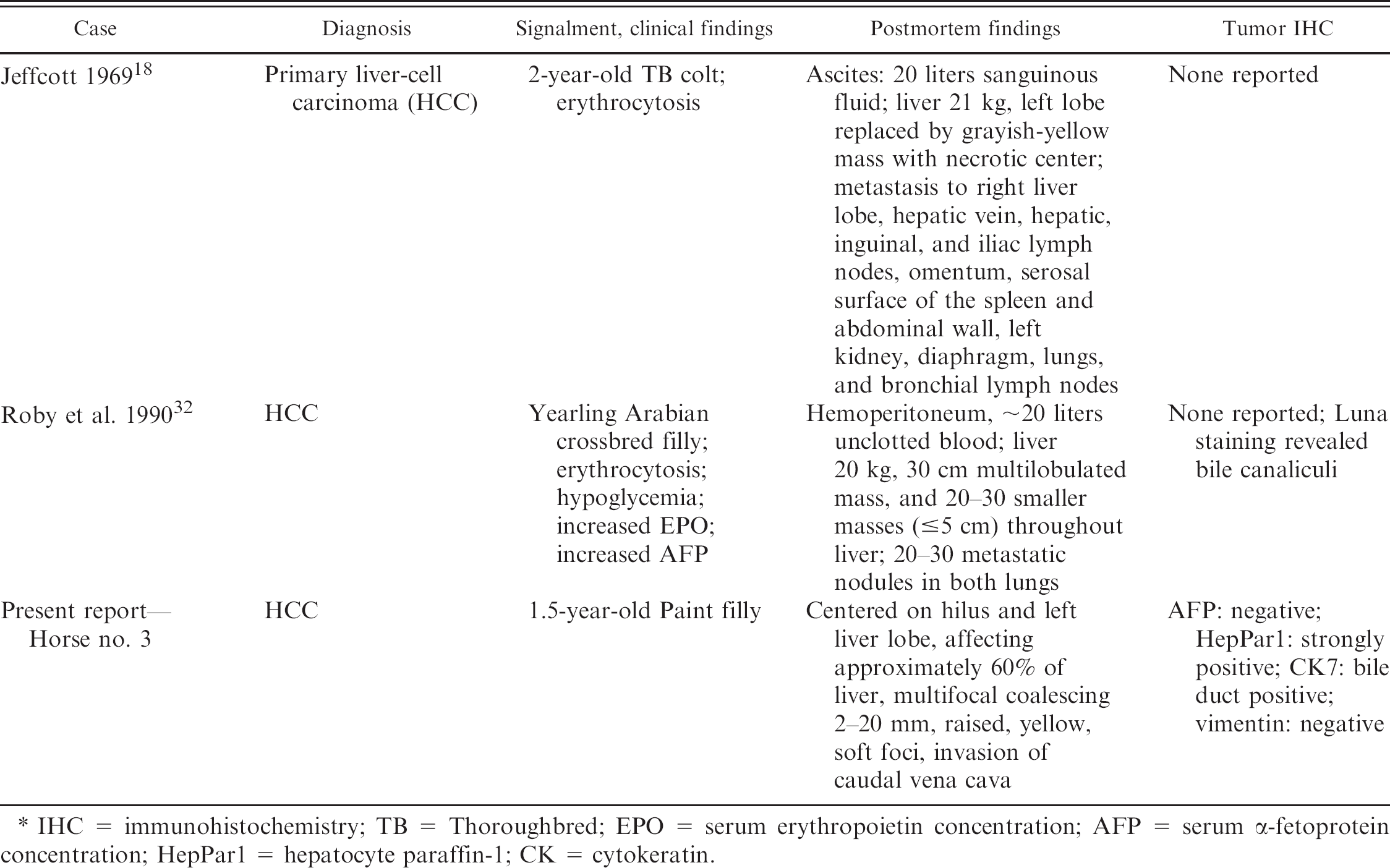
IHC = immunohistochemistry; TB = Thoroughbred; EPO = serum erythropoietin concentration; AFP = serum α-fetoprotein concentration; HepPar1 = hepatocyte paraffin-1; CK = cytokeratin.
Despite its rarity and the apparent grave prognosis, it is of value to identify histologic and morphologic features of these complex hepatic tumors to allow accurate classification. The cases reported herein, along with others previously reported, suggest that equine and human HB share histomorphologic and IHC features. Thus, the scheme currently used to classify human tumors might also be applicable to equine liver tumors. However, the clinical significance of such classification schemes in equine patients awaits assessment of more cases.
Footnotes
a.
Dako Canada Inc., Mississauga, Ontario, Canada.
b.
Abcam Inc., Cambridge, MA.
c.
Dako Canada Inc., Mississauga, Ontario, Canada.
d.
Laboratory Reference Centre, Hamilton, Ontario, Canada.
