Abstract
A 5-year-old National Show horse mare presented with a soft mass on the left dorsolateral aspect of the tongue. Over the next 2 years, the mare developed numerous, similar, coalescing masses that extended along the left dorsolateral aspect to the tip of the tongue. Microscopically, the bases for these masses were slender, fusiform, mesenchymal cells that formed compact whorls around myelinated and unmyelinated nerves. These cells were labeled by antibodies directed against vimentin but not by S-100. Ultrastructurally, multiple, concentrically arranged, long, slender cell processes, with discontinuous external laminae and many pinocytotic vesicles, helped to accurately phenotype the proliferative element. Whether this unusual perineurial cell proliferative disorder is neoplastic or not remained a matter of conjecture.
Perineurial cell proliferation has rarely been reported in veterinary medicine. Although uncommon in humans, it is being recognized with increasing frequency and diversity. Perineurial cell tumor or perineurioma was first described by Lazarus and Trombetta.11 Bunge et al.1 proved that perineurial cells originate from fibroblasts. Giannini et al.8 later demonstrated that perineuriomas are associated with abnormalities of chromosome 22 in humans. Only intraneural perineurioma is reported in veterinary medicine, and, to date, none have involved the oral cavity.5,9,12,14,15 In this equine case, perineurial cell proliferation was associated with nerves and most closely resembled the intraneural form of perineurioma but was atypical in that it involved the perineurial layer of individual nerves in soft tissue rather than multiple axons within nerves in a nerve trunk.
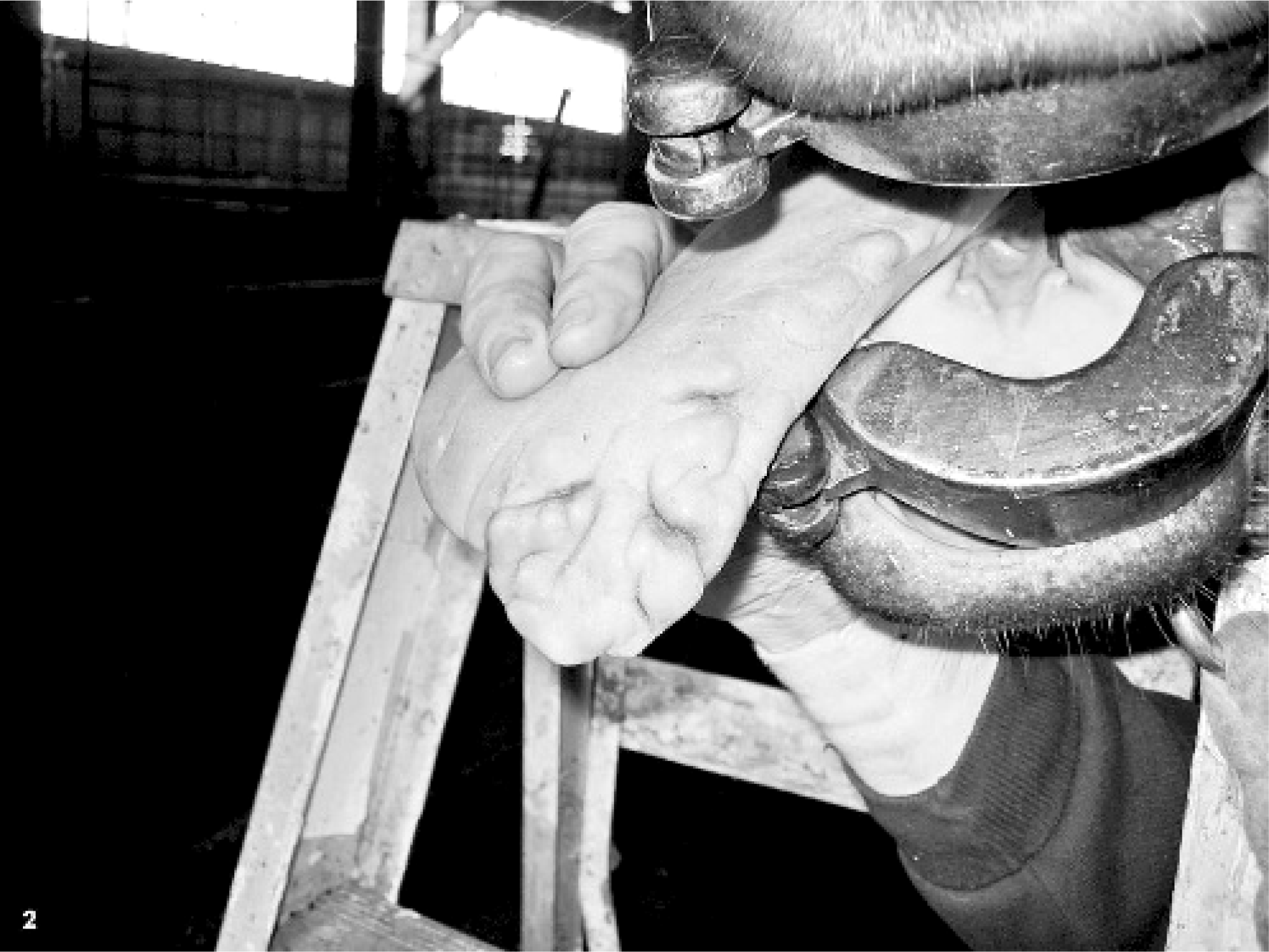
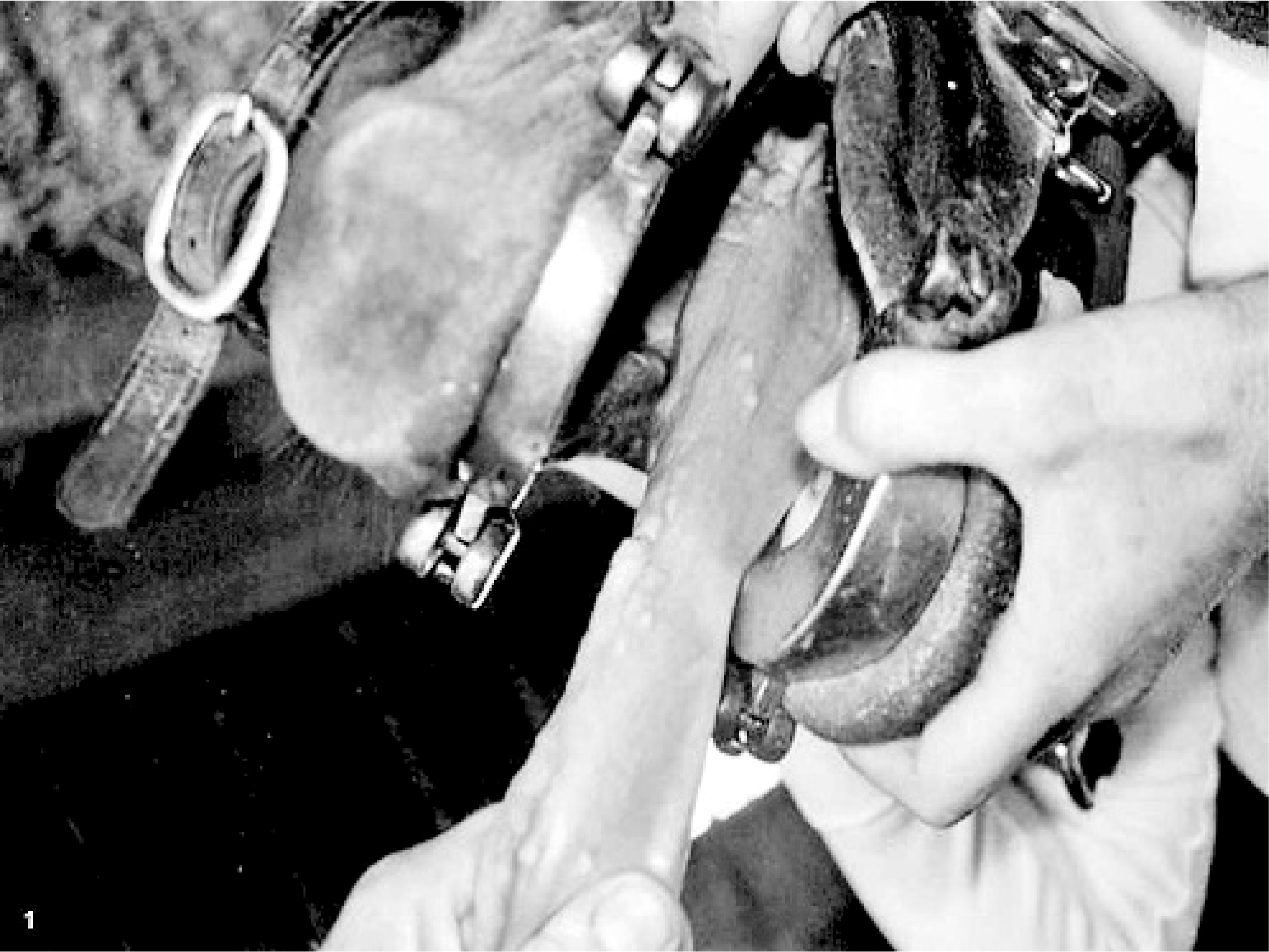
In May 2004, a mass was removed from the left side of the tongue of a 5-year-old National Show horse mare in Illinois. With histopathologic features of marked mucosal hyperplasia and mild inflammatory infiltrate in the underlying submucosa, the mass was diagnosed as a papilloma. In October 2005, the horse was presented for multiple coalescing masses (estimated to be more than 50) on the left side that extended over approximately 20 cm of the tongue. The masses ranged in size from a few mm to 2.5 cm in diameter. All masses were adjacent to the site of the previous mass biopsied 17 months earlier, were located on the left side of the tongue, and had progressed rostrally from the level of the commissure toward the tip of the tongue (Figs. 1 and 2). Clinical evaluation suggested that the masses were physically impeding mastication; the horse had difficulty chewing on the left side, spilled more grain, and drooled saliva from the left side. During clinical examinations and while taking biopsy specimens, it was observed that the horse had reduced sensation in the area spanned by the masses.
Tongue; horse. December 2006: Rostral view depicting progression of lesions on the tongue, with a chain of raised nodules extending to the tip.
Tongue; horse. October 2005: Left lateral view showing multifocal to coalescing masses.
After gross evaluation, formalin-fixed tissue specimens were routinely processed and paraffin embedded for hematoxylin and eosin sections. Immunohistochemical analysis was performed on selected sections, with antibodies directed against S-100, vimentin, neurofilament protein (NFP), smooth-muscle actin (SMA), and von Willebrand factor (vWF) by using the streptavidin-biotin method with a BioGenex i6000 automatic stainer (San Ramon, CA) according to the manufacturer's instructions. The primary antibodies included rabbit polyclonal for S-100 (DakoCytomation, Carpinteria, CA) at a dilution of 1:750, mouse monoclonal for vimentin (BioGenex) at a dilution of 1:1 (of prediluted antibody), mouse monoclonal for NFP at a dilution of 1:4,000, mouse monoclonal for SMA (prediluted from BioGenex), and rabbit polyclonal for vWF (DakoCytomation) at a dilution of 1:800. Secondary antibodies included Super Sensitive (SS) detection system (BioGenex) for S-100, LSAB2 (DakoCytomation) for vimentin, LSAB2 (DakoCytomation) for NFP, SS (BioGenex) for SMA, and LSAB2 (DakoCytomation) for vWF. Epitope retrieval pretreatments consisted of citrate buffer at a pH of 6 for 30 seconds at 125°C followed by 10 seconds at 90°C in a digital decloaking chamber (Biocare Medical, Concord, CA) for S-100, vimentin, and SMA; and 0.1% porcine pancreatic trypsin and calcium chloride treatment at 37°C for 20 minutes for vWF. The chromogen used was 3,3-diaminobenzidine-tetrahydrochloride (BioGenex), and the slides were counterstained with hematoxylin (BioGenex).
For ultrastructural evaluation, glossal masses were re-biopsied by one of the authors (R. Rock) in December 2005. Tissues were fixed in modified Karnovsky's fixative (2.5% paraformaldehyde and 2% glutaraldehyde). Fixation and embedding procedures were enhanced with microwave fixation (http://treefrog.cvm.uiuc.edu/meth_stdMW.html).13 The sample was osmicated with 2% OsO4, followed by treatment with 3% potassium ferrocyanide, uranyl acetate, and dehydration with an ethanol series, ending in acetonitrile. The tissue was infiltrated and embedded by using Lx112 epon substitute (Ladd Research Industries Inc., Burlington, VT) and then polymerized overnight at 90°C. Sections were stained with uranyl acetate and lead citrate, and were viewed with a Hitachi H600 transmission electron microscope (Hitachi instrument Inc., San Jose, CA).
On gross examination of several masses of different sizes, it was noted that the masses elevated the mucosa and were firm and smooth surfaced. Larger masses were ulcerated. When incised, the masses were glistening white and had multiple nodular areas that bulged from the cut surface.
Microscopically, multifocal to coalescing masses were formed by the expansion of glossal submucosa and tunica muscularis by multiple concentric whorls of compact, slender, spindloid cells (Figs. 3 and 4). These whorls ranged in size from 35 microns to 1–2 mm in diameter. Whorling cells were centered on and surrounded nerves, replacing and expanding the perineurium (Fig. 5). Each fusiform cell in a perineural whorl had a central, oval to elongated, plump nucleus and abundant cytoplasm. Peripherally, the whorling cell type blended with the fibrocytes in the connective tissue. Masson stain identified modest amounts of collagen in between the cells that formed the whorls, a finding that was later confirmed ultrastructurally. An additional prominent change that involved the largest mass in the tongue was thickening of the walls of arteries from intimal and medial proliferation. This was viewed as a reactive change. In areas with ulceration, the breach in mucosa was filled by exfoliating fibrovascular tissue, which, toward the surface, was infiltrated by modest numbers of neutrophils and a few macrophages. It was covered by moderate amounts of serocellular debris composed of modest amounts of eosinophilic homogenous to finely granular proteinaceous material, many degenerate neutrophils, a few macrophages, and a little plant material. The superficial submucosal connective tissue adjacent to the site of ulceration was infiltrated by small to moderate numbers of lymphocytes, plasma cells, and macrophages. This tissue also contained a few lymphoid follicles, some of which had active germinal centers (chronic glossitis). The mucosa in most masses was markedly thickened, with multiple layers of stratum spinosum.
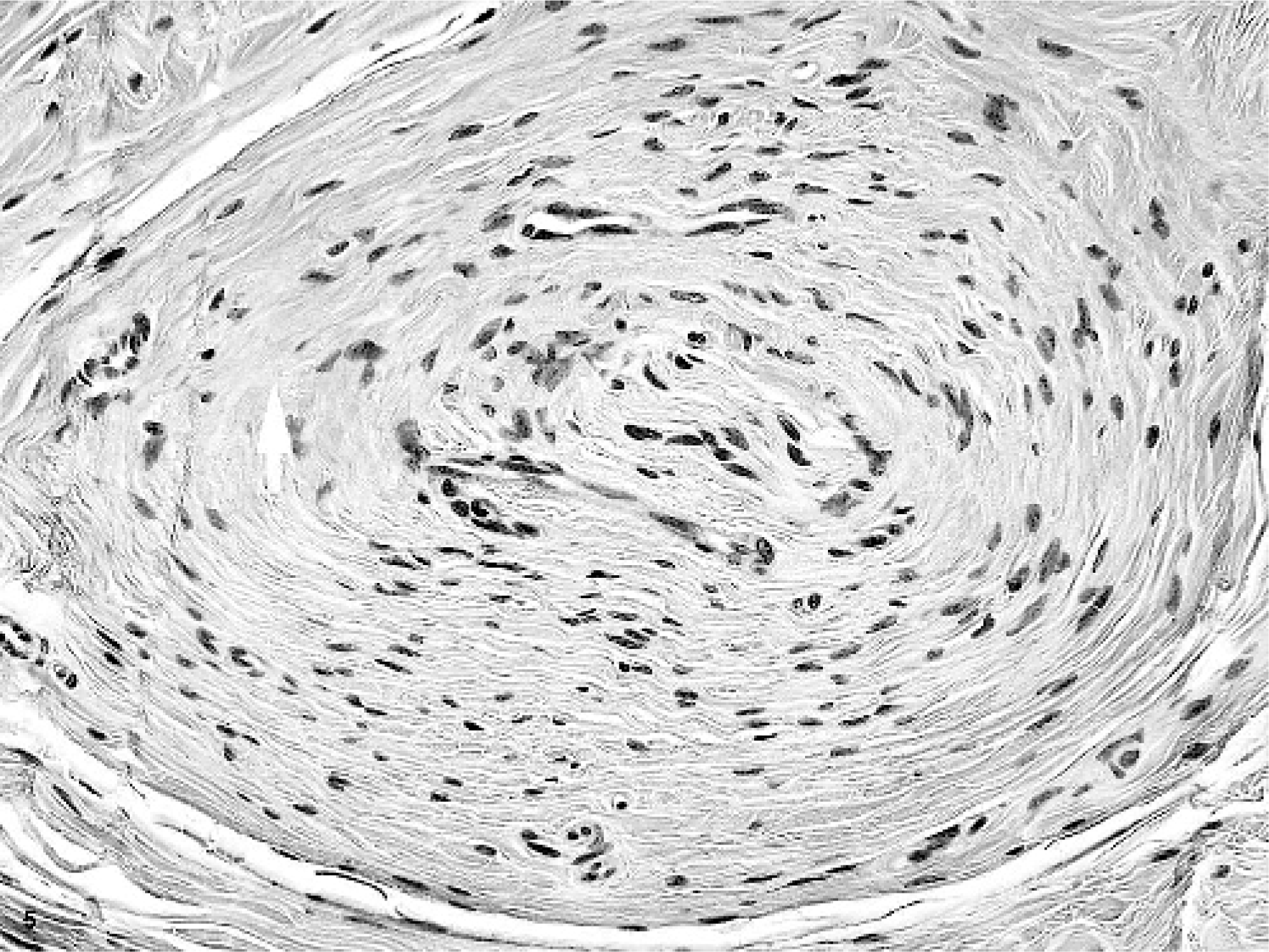
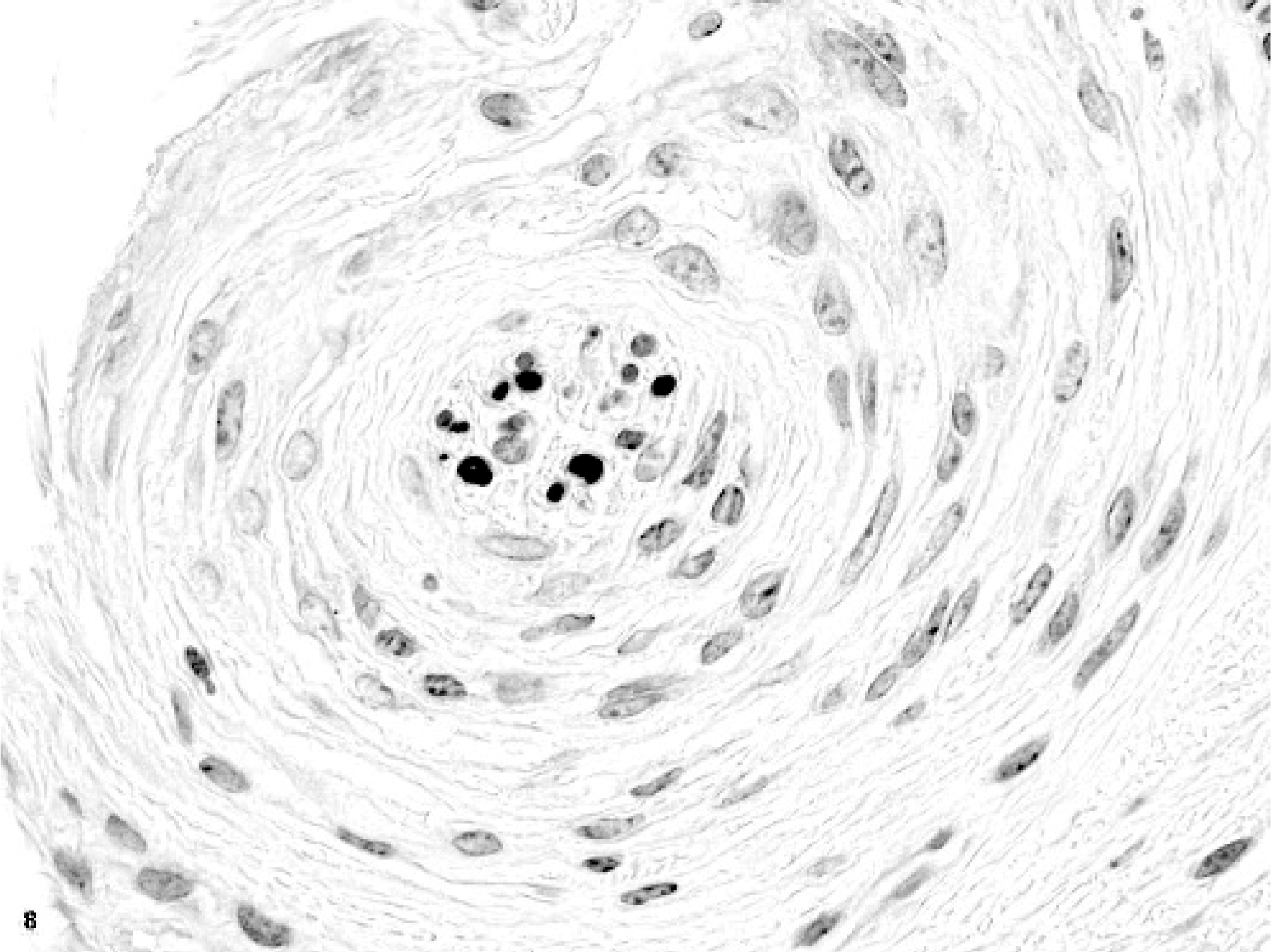
Glossal submucosa; horse. One whorl of spindloid mesenchymal cells surrounding a nerve. HE.
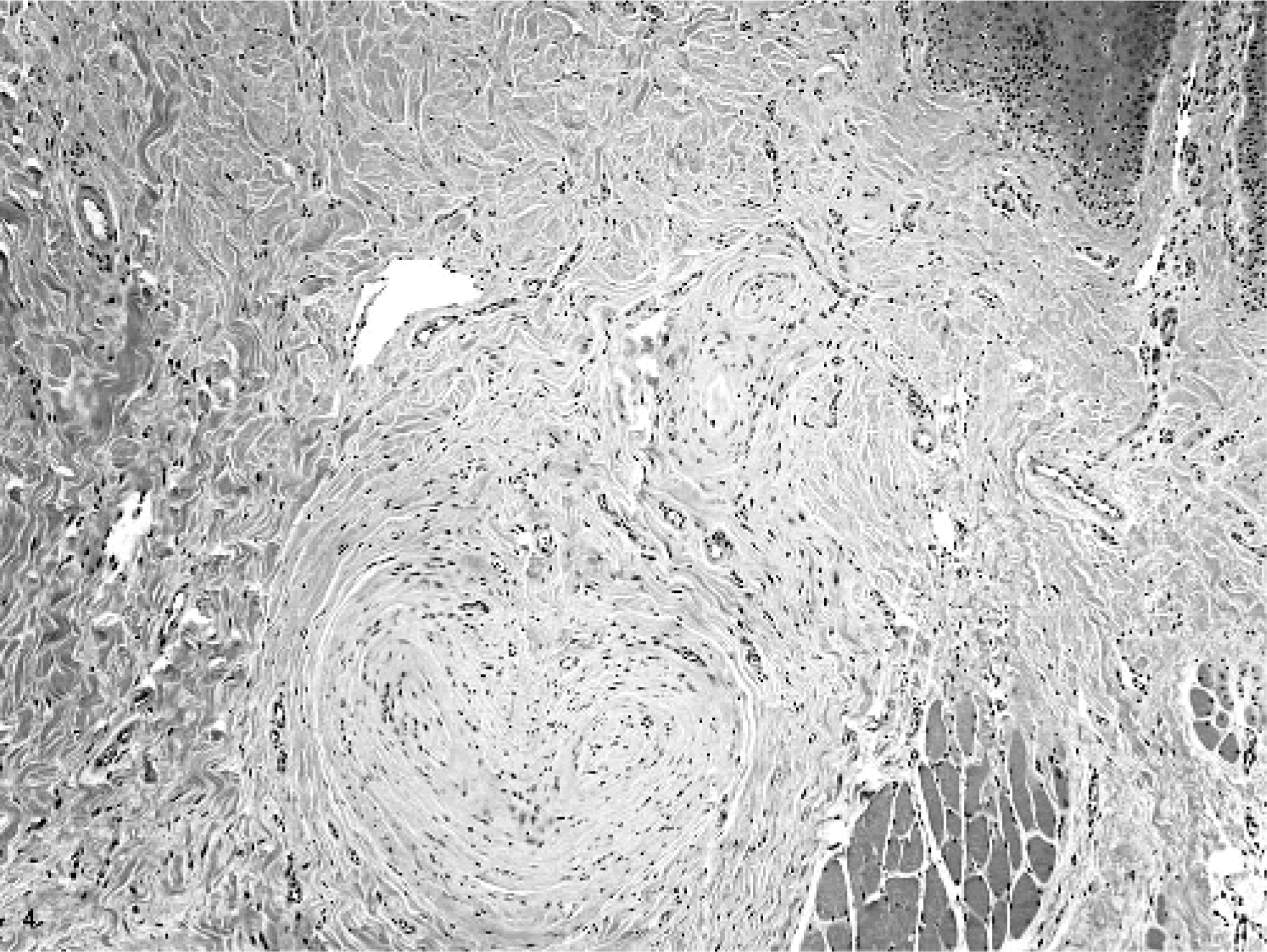
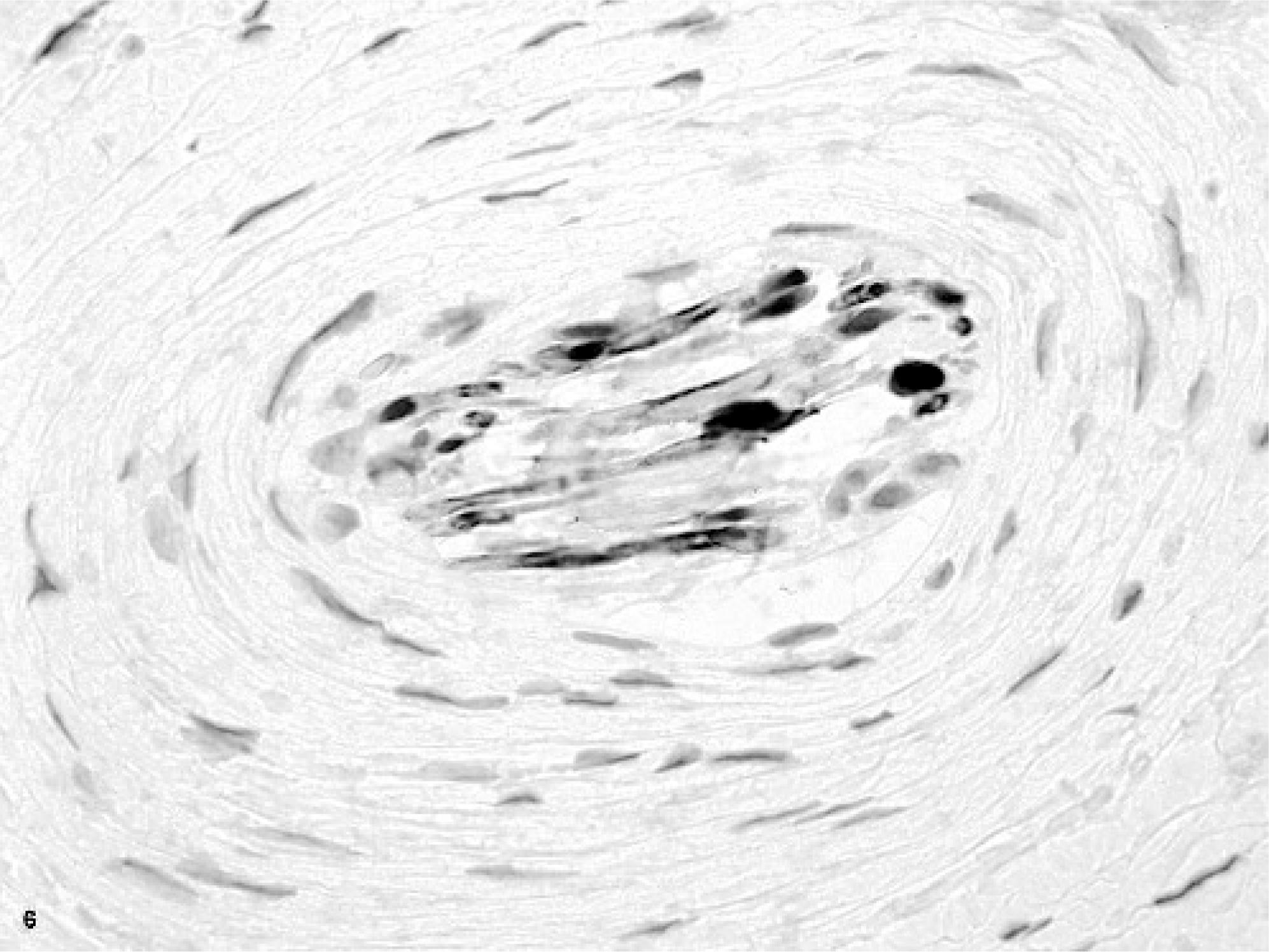
Tongue; horse. Cellular whorls in the glossal submucosa and in between the glossal skeletal myofibers. HE.
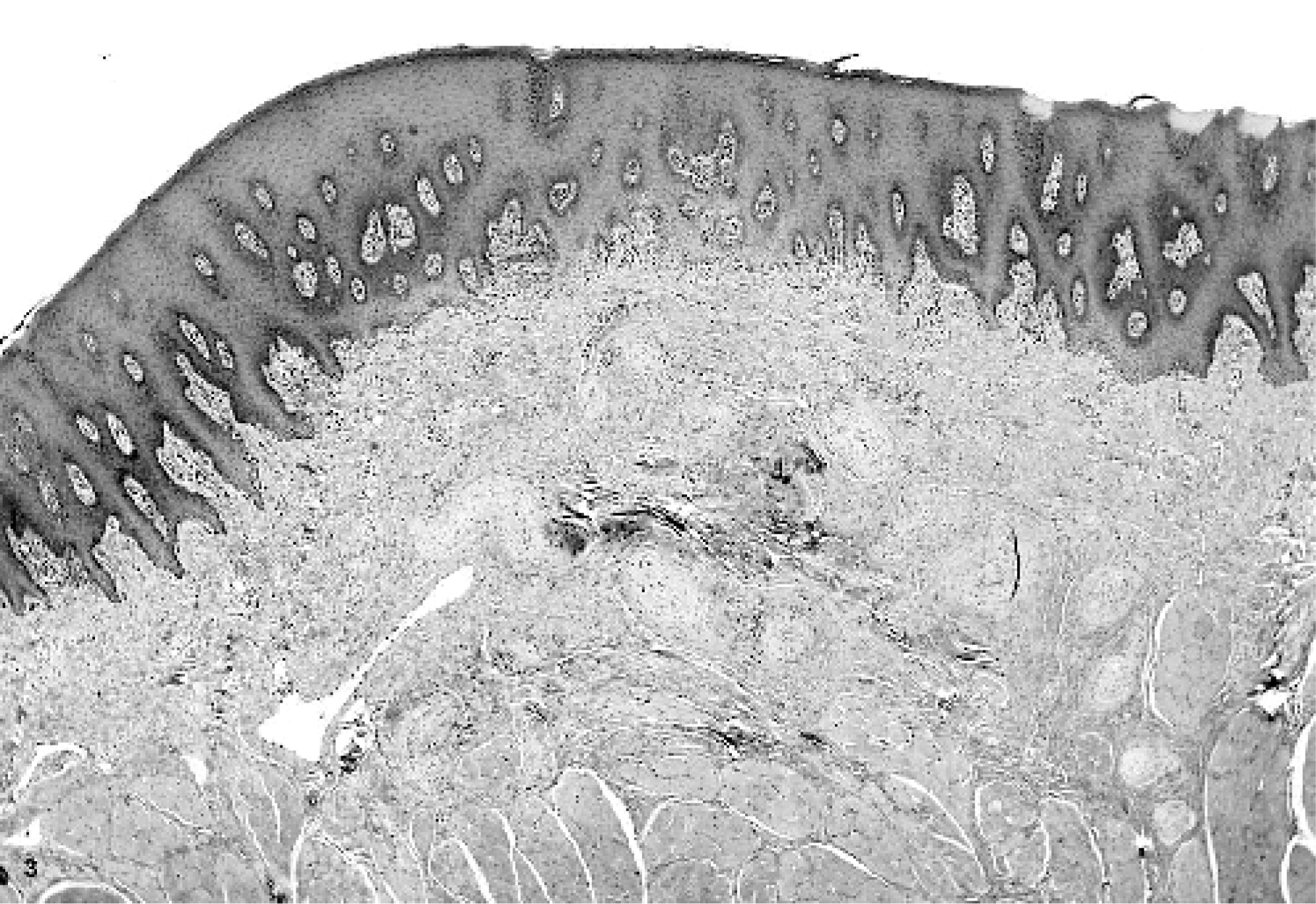
Tongue; horse. Multiple small nodules are seen in the lamina propria. HE.
Antibodies directed against S-100 labeled only Schwann cells and myelin within the centrally located nerves (Fig. 6), whereas antibodies directed against vimentin stained the whorls of proliferative cells (Fig. 7). Antibodies directed against NFP labeled axons and nerve fibers in the center of the whorls (Fig. 8). Cells that formed the whorls were not labeled by antibodies directed against SMA (not shown). Antibodies directed against vWf identified the vasa nervosa, small blood vessels that supply peripheral nerves, and other blood vessels in the tissue (not shown). With normal equine tongue tissue as a control, Dr. C. Fletcher at Brigham and Women's Hospital, Boston, Massachusetts, performed further immunohistochemical stains, in this case, for epithelial membrane antigen and claudin-1, both markers of perineurial cells in humans. Unfortunately, neither reagent identified normal perineurial cells in equine tissue. It was later learned that similar attempts in equine tissue were also unsuccessful at Michigan State University (Matti Kiupel, personal communication).
Glossal submucosa; horse. Antibodies directed against neurofilament protein labeling axons and central nerve fibers within the nerve located in the center of whorls as detected by chromagen 3,3-diaminobenzidene-tetra hydrochloride and counterstaining with hematoxylin.

Glossal submucosa; horse. Antibodies directed against vimentin labeling proliferating cells that formed whorls and Schwann cells within the centrally located nerve, as detected by chromagen 3,3-diaminobenzidene-tetra hydrochloride and counterstaining with hematoxylin.
Glossal submucosa; horse. Antibodies directed against S-100 labeling Schwann cells and myelin within the nerve located in the center of whorls as detected by chromagen 3,3-diaminobenzidine-tetrahydrochloride and counterstaining with hematoxylin.
Histopathologic examination and immunohistochemistry provided some clues as to the cell type in the whorls that formed the masses. However, these analyses were nondiagnostic. Ultrastructurally, the spindloid whorling cells had slender, elongated cytoplasmic processes, occasionally prominent pinocytotic vesicles, patchy external laminae, and cell processes separated by collagen fibrils (Figs. 9– 11). These features were consistent with those described for perineurial cells.3,6 Both, myelinated and nonmyelinated nerves were found to be surrounded by whorling perineurial cells.
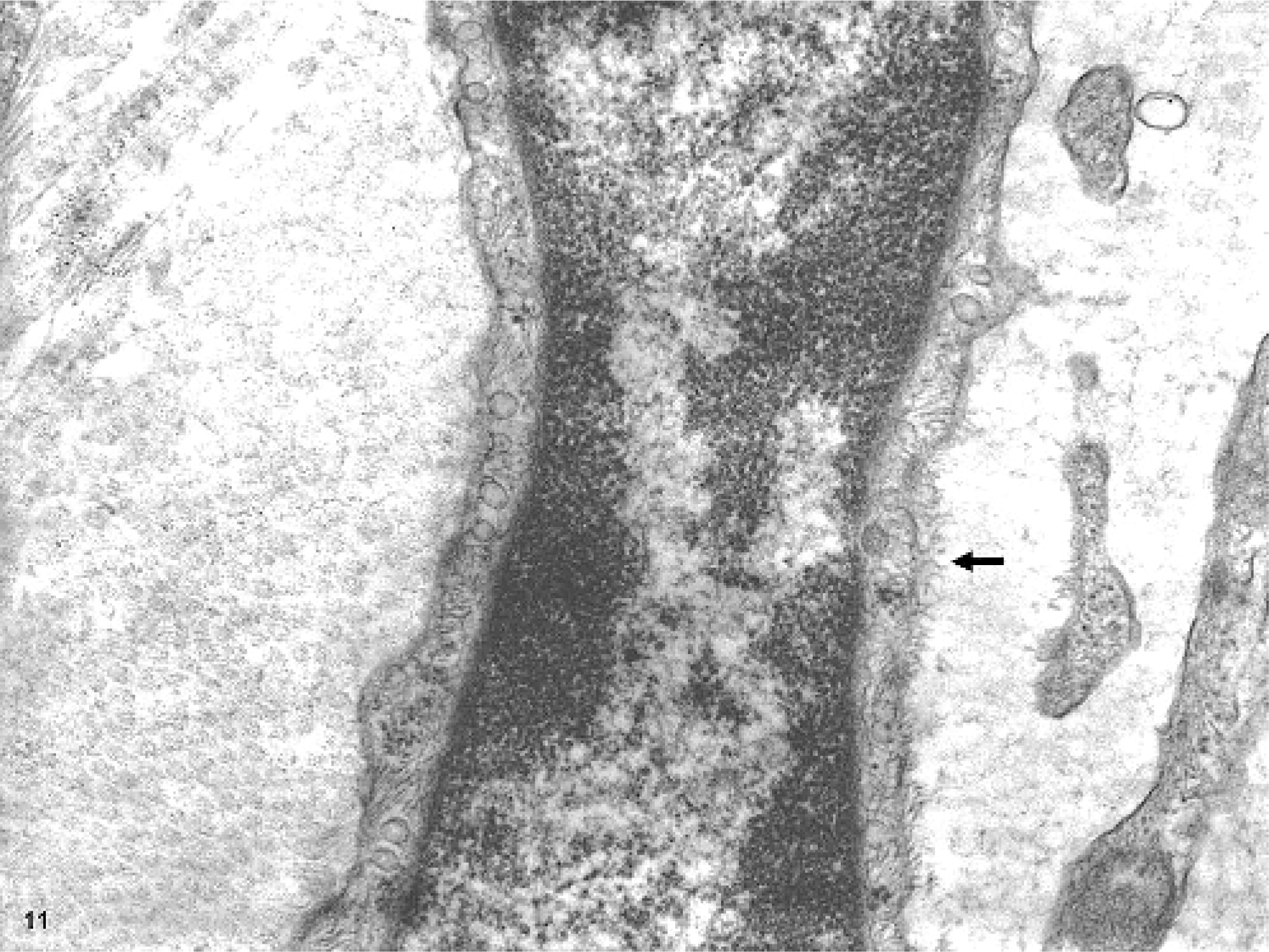
Glossal submucosa; horse. Higher magnification of one-cell process, showing patchy external lamina (indicated with arrow). Transmission electron micrograph.
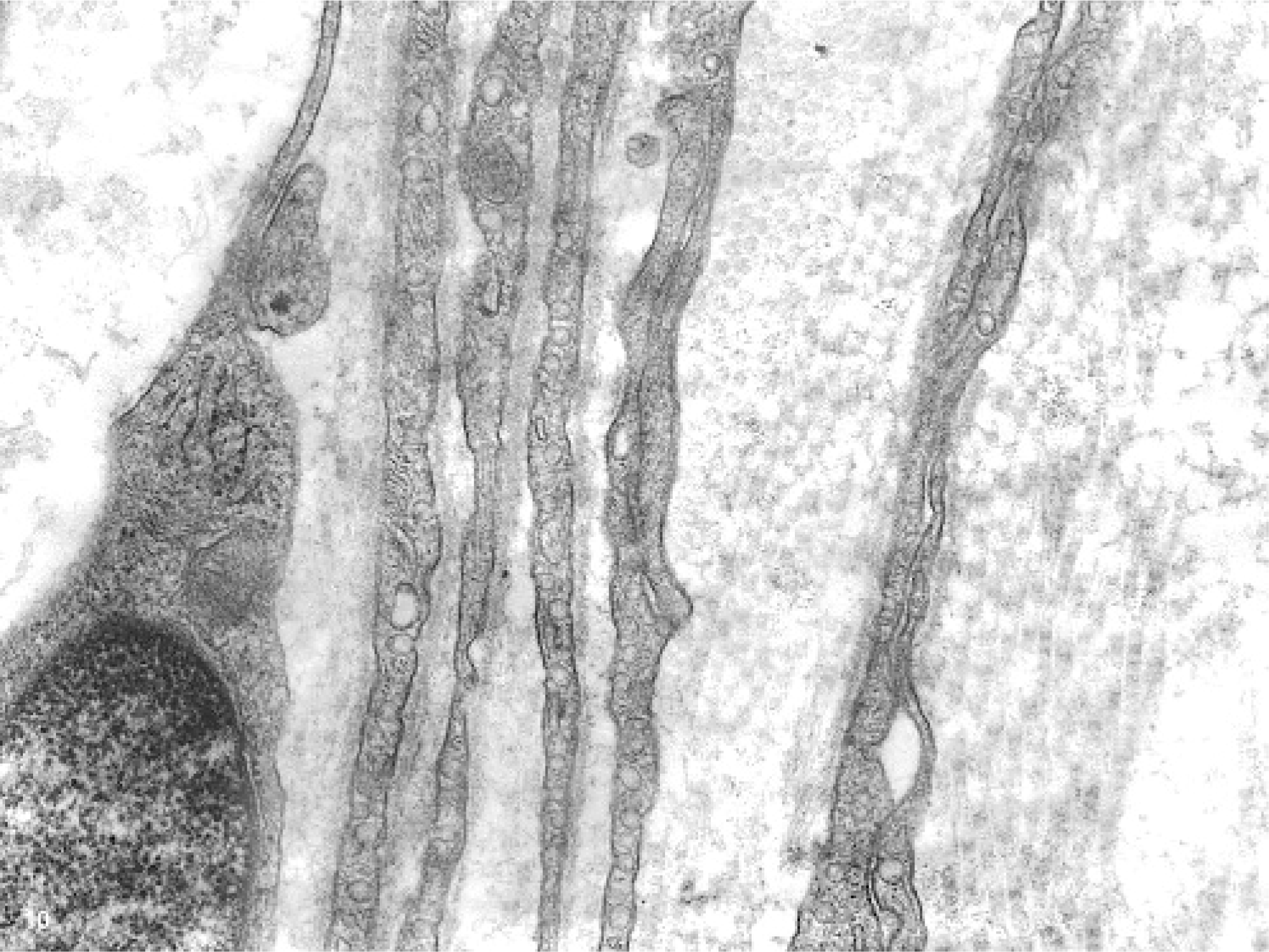
Glossal submucosa; horse. Features of cells that formed whorls consistent with perineurial cells: cell membranes covered by patchy external lamina, occasional presence of prominent pinocytotic vesicles, and cell processes separated by variable amounts of collagen. Transmission electron micrograph.
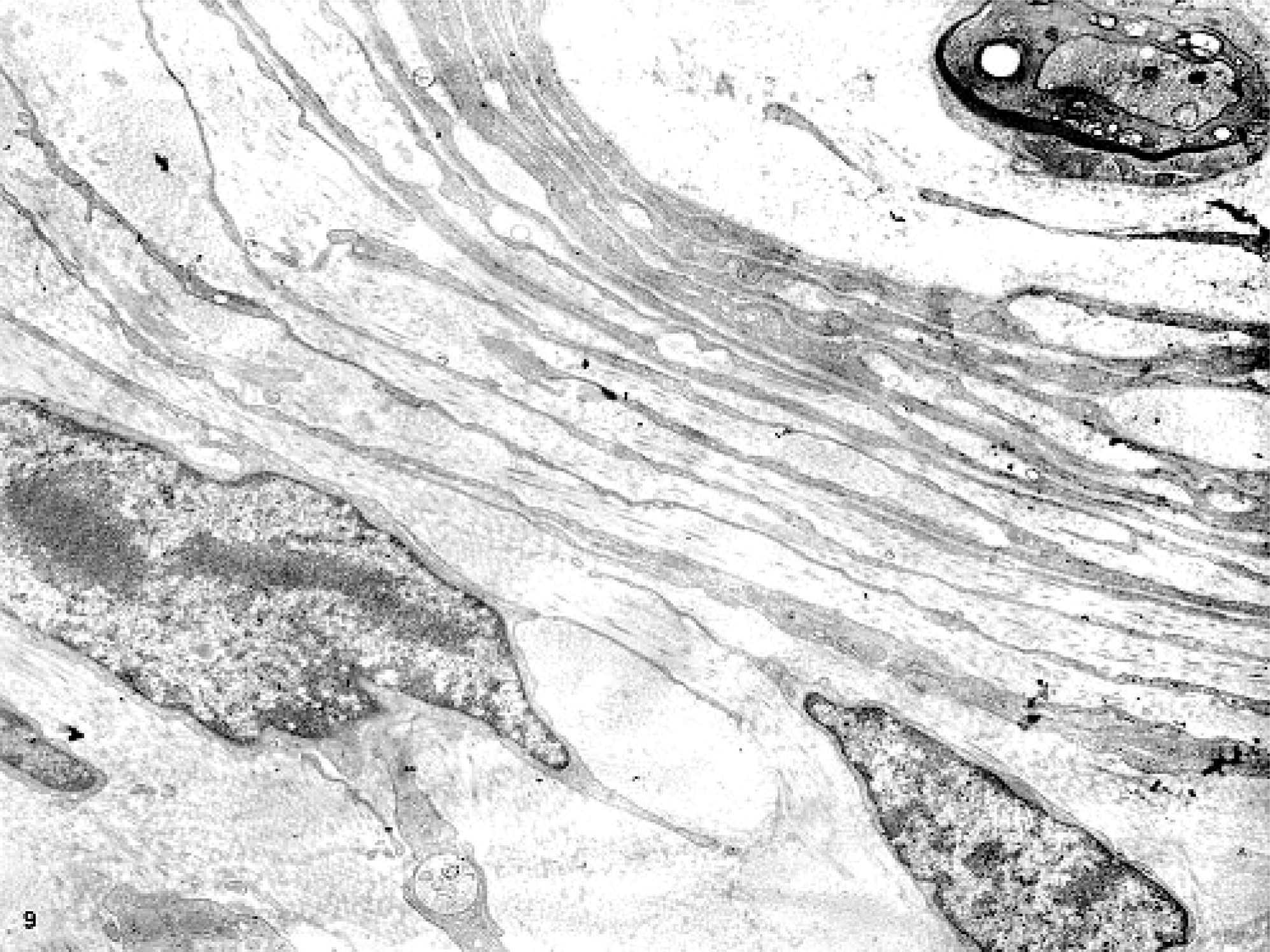
Glossal submucosa; horse. Closely apposed slender processes of cells surrounding a myelinated nerve. Transmission electron micrograph.
When taking together the light microscopic, immunohistochemical, and ultrastructural findings, the diagnosis of an atypical perineurial proliferative disorder was made. The horse was reexamined twice by one author (R. Rock), once in December 2005 for excising fresh tissue for an ultrastructural evaluation and then later in June 2006 as a follow-up. In his opinion, the lesions appeared to be progressive. The masses at the tip of the tongue were larger, with a palpable mass estimated to be 3 cm in diameter at the site of the original surgery left of the midpoint of the tongue. Notably, all masses were still on the left side of tongue and were larger on the dorsal surface. The progression of the mass lesions from a point on the tongue around the level of the commissure of the lips (Fig. 1) to the rostral aspect of the tongue (Fig. 2) could point to a neoplastic disorder. Microscopically, cells that formed the whorls appeared orderly and bland, and did not exhibit overtly neoplastic features. Hence, it is uncertain whether this atypical proliferation was neoplastic or hyperplastic. When considering all the information till the follow-up in June 2006, in Dr. Fletcher's opinion, this was a unique example of multifocal perineurial hyperplasia or hamartoma. One author (B. Summers) favored the interpretation that the disorder was neoplastic, somewhat reminiscent of but not identical to intraneural perineurioma in humans, which interestingly, was viewed as a localized hypertrophic neuropathy for many years.
In humans, perineuriomas are classified into 2 major categories.10
Intraneural perineuriomas, which commonly present as an elongated swelling of a peripheral nerve. Histologically, multiple individual axons within the nerve are encircled by proliferative perineurial cells. Now accepted as a neoplasm, for a long time this was thought to be a non-neoplastic proliferative disorder and was reported as localized hypertrophic neuropathy. Rare examples have been reported in dogs 5, 9, 12 and in chicken.15
Soft tissue (extraneural) perineuriomas, of which there are a few patterns recognized in humans.10
Some cases of perineuriomas in humans express epithelial membrane antigen (EMA) and/or claudin-1.10 The lack of detection of EMA expression when using available (human) antibodies in control animal/bird tissues was also mentioned in the reports of perineuriomas in veterinary species.9,12,15 The presence of perineurial cells alone in the whorls differentiated this lesion from schwannoma and neurofibroma (solitary form of von Recklinghausen's disease).4,7,10,14 Benign schwannomas do not show the concentric whorled arrangement around nerves seen in this horse. Rather, they grow as compact masses in histologic patterns referred to as Antoni A and B areas, contain nuclear palisading, and feature distinct structures known as Verocay bodies. Dermal Schwannomas in horses are consistently S-100 positive (Summers, personal observations). Ultrastructurally, they share similarities with perineurial cells, but they have a continuous external lamina and fewer pinocytotic vesicles. In neurofibromas, the proliferating cells are a combination of all elements of peripheral nerves, i.e., axons, Schwann cells, fibroblasts, and perineurial cells. Morphologically, this case also bears some similarity to what was recently described in a human case as a sclerosing Pacinian-like perineurioma.2
The presentation of this progressive nodular lesion in the tongue of this horse is unusual. Knowledge of perineurial cell proliferation in the oral cavity of veterinary species will help veterinary diagnosticians and clinicians detect and differentiate these from other spindle-cell proliferations. It is hoped that, with time, the advent of further useful immunohistochemical markers for veterinary species will facilitate characterization of such lesions. An improved classification may also help us analyze differences, if any, in clinical outcomes in different species.
Footnotes
Acknowledgements
We thank Dr. Christopher D. M. Fletcher (professor and director of surgical pathology, Brigham and Women's Hospital, Boston, MA) for helping us analyze this case. Several veterinary/human pathologists and dentists provided their opinions on this case. They include Dr. Beth Valentine (Oregon State University); Dr. E. J. Ehrhart (Colorado State University); Dr. Richard R. Dubielzig (University of Wisconsin); Drs. Fredrickson, Lichtensteiger, and Valli (University of Illinois, Urbana Champaign [UIUC]); Drs. John F. Van Vleet and Margaret Miller (Purdue University); Dr. Dean K. White (human oral and maxillofacial pathologist, University of Kentucky College of Dentistry); Dr. Matti Kiupel (Michigan State University); Dr. Steven Weisbrode (The Ohio State University); Dr. Tom Allen (equine veterinarian); and Dr. David Klugh (equine veterinarian). We thank all these people for their valuable time. We appreciate the help provided by Lou Ann Miller (Electron Microscopy unit, UIUC), and the Histology laboratory at the Veterinary Diagnostic Laboratory, UIUC. We also thank Dr. Dee Ann Kuster for providing follow-up information on this case. The work done on this case was possible with laboratory support provided by the Veterinary Diagnostic Laboratory, UIUC.
