Abstract
The objectives of this study were to describe the anatomy, histology, and ultrastructure of the equine filum terminale (FT) and to describe the FT in hereditary equine regional dermal asthenia (HERDA), a model of human Ehlers-Danlos syndromes (EDS). Those humans suffer from tethered cord syndrome (TCS) caused by an abnormally structured FT wherein its attachment at the base of the vertebral column leads to long-term stretch-induced injury to the spinal cord. The pathophysiology of TCS in EDS is poorly understood, and there is a need for an animal model of the condition. Histopathologic and ultrastructural examinations were performed on FT from HERDA (n = 4) and control horses (n = 5) and were compared to FT from human TCS patients with and without EDS. Adipose, fibrous tissue, and neuronal elements were assessed. CD3 and CD20 immunohistochemistry was performed to clarify cell types (HERDA n = 2; control n = 5). Collagen fibrils were assessed in cross-section for fibril diameter and shape, and in longitudinal section for fibril disorganization, swelling, and fragmentation. The equine and human FT were similar, with both containing fibrous tissue, ependyma, neuropil, and nerve twigs. Hypervascularity was observed in both HERDA horses and human EDS-TCS patients and was not observed in equine or human controls. Moderate to severe abnormalities in collagen fibril orientation and architecture were observed in all HERDA horses and were similar to those observed in human EDS-TCS patients.
Keywords
Ehlers-Danlos syndromes (EDS) are a group of hereditary disorders of connective tissue causing variable degrees of joint hypermobility, skin hyperextensibility and fragility, and vascular and visceral fragility. More recently, a wide spectrum of neurological manifestations associated with EDS were described, including some associated with a high degree of chronic pain and disability. 11 One such manifestation is tethered cord syndrome, a condition in which the filum terminale (FT) is abnormally structured and leads to a clinical presentation associated with a constellation of neurological, neurocutaneous, orthopedic, and urological and/or gastrointestinal signs and symptoms. 1,29,31,33 Prior studies have hypothesized that clinical signs result from chronic mechanical tension secondary to FT tethering or inelastic tissue within the FT, ultimately leading to spinal cord stretching with concomitant decrease in local blood flow and impaired mitochondrial oxidative metabolism at the level of the spinal cord. 33 In the EDS population, it is theorized that tethering occurs due to an abnormal arrangement of collagen within the FT, leading to reduced elasticity, as well as hypermobility of the spine. 11,18 Unfortunately, there is a gap in knowledge about the pathophysiology of TCS in EDS and a need for an animal model of this condition. To identify the impact of heritable disorders of connective tissue on the FT, we examined horses with hereditary equine regional dermal asthenia (HERDA), an EDS-like syndrome resulting from a mutation in peptidyl-prolyl cis-trans-isomerase-B (PPIB) encoding for cyclophilin B. 20,27 The histopathology, immunohistochemistry (IHC), and ultrastructure of the FT was studied in both HERDA and control horses and compared to the FT of humans with TCS with and without EDS. The objectives of this study were to characterize the normal equine FT and evaluate the impact of HERDA on the FT.
In humans, the FT is a fibrous band of connective tissue spanning from the conus medullaris to the coccyx. 21 Drawings of the equine FT are included in some veterinary anatomy texts, along with brief anatomic descriptions; however, detailed accounts of the anatomy, histology, and ultrastructure are lacking. The equine FT is usually depicted as intradural, originating at the S2 vertebra and terminating at the S4 vertebra. 3 The human FT has been described as having an intradural segment, the filum terminale internum, which runs from the conus medullaris to the termination of the thecal sac, while the extradural filum terminale externum runs from the termination of the thecal sac to the coccyx. The FT is formed from embryologic spinal cord following retrogressive differentiation and is theorized to act as a spinal cord stabilizer, working in concert with the denticulate ligaments. 4,21,28
Materials and Methods
Animals
Nine horses (4 HERDA; 5 controls) were evaluated. The HERDA group consisted of 4 Quarter Horses, while the control group consisted of 3 Quarter Horses, 1 Arabian, and 1 Morgan. The HERDA group mean age was 8 years (range 2–21 years) and comprised 2 mares and 2 geldings. The control group mean age was 6 years (range 2–18 years) and comprised 4 mares and 1 gelding. Control horses were donated due to nonneurological illnesses, which consisted of orthopedic and ophthalmic disease. Genetic testing and physical examination were required for HERDA horse enrollment. All HERDA horses were confirmed homozygous positive, HRD/HRD, for the PPIB mutation and displayed a phenotype consistent with the disorder. Control Quarter Horses were confirmed homozygous negative, N/N, for the PPIB mutation and were phenotypically normal. Experimental protocols were approved by the guidelines and regulations set forth by the Michigan State University Institutional Animal Care and Use Committee (IACUC Protocol# 02/18/020-00).
Horses were humanely euthanized to allow anatomic examination of the FT. A 15 cm segment of the FT was transected cranially at the termination of the conus medullaris and caudally at the point at which it became confluent with the cauda equina. Samples were collected within one hour of euthanasia in all but one control animal.
Humans
Example specimens were obtained from EDS-TCS (n = 4) and TCS (n = 2) patients during surgical detethering of the FT (Rhode Island Hospital IRB 787945 and 1107867) for the purpose of histopathologic and ultrastructural comparison with equine specimens. The 4 EDS-TCS patients included one 4-year-old male and 3 females ranging in age from 18 years to 39 years. The included EDS patients were formally diagnosed according to the criteria proposed by the Ehlers-Danlos Society. 15 The TCS group included one 14-year-old male and one 16-year-old male. All patients presented with clinical signs of TCS as well as abnormal urodynamic testing supporting the diagnosis. The imaging showed the conus medullaris at a level of L2 or L2/3 with a variably thickened FT (>2 mm), and/or a fat signal in the FT on standard axial and sagittal T1 and T2 lumbar non-fat suppressed MRI images.
Histology
Equine specimens 1.5 cm in length were obtained from 3 cm caudal to the termination of the conus medullaris and submitted for histopathologic analysis. Specimens (HERDA n = 4; control n = 5) were fixed for a minimum of 24 hours in 10% neutral buffered formalin and were routinely processed for histopathologic examination, embedded in paraffin, and sectioned at 5 μm. Resulting sections were stained with hematoxylin and eosin (HE) and Masson’s trichrome. One HE-stained section was evaluated per animal. Hypervascularity was defined as the presence of a single large dilated vessel within the FT or numerous small vessels. The presence of adipose tissue was scored on a 0 to 3 scale, with 0 = no adipose present, 1 = adipose present in less than 10% of the specimen, 2 = adipose present in 10% to 50% of the specimen, and 3 = adipose present in greater than 50% of the specimen. IHC for CD3 and CD20 (T and B lymphocytes, respectively) was performed on 2 HERDA cases and 5 controls. Rabbit monoclonal anti-CD20 antibody (RB = 9013-P; Thermo Scientific) and rabbit monoclonal anti-CD3 antibody (A0452; DAKO) were used. 5,25 For immunohistochemistry, cellularity was scored as mild (<20 lymphocytes/hpf), moderate (20–100 lymphocytes/hpf), or severe (>100 lymphocytes/hpf). Specimens were photographed in sagittal section.
Human FT specimens (EDS-TCS n = 2; TCS n = 1) were fixed in formalin at the time of surgery. Samples were processed for histopathologic examination according to standard procedure, embedded in paraffin, and sectioned at 4 μm. Slides were stained with HE and Masson’s trichrome, and immunolabeling for the common leukocyte marker CD45 (EDS-TCS n = 2) was performed using prediluted mouse anti-human monoclonal antibody to CD45 (PD7/26 and 2B1; Dako; cat# GA751). Specimens were photographed in sagittal section.
Ultrastructure
Equine specimens of 1.5 cm in length were submitted for ultrastructural analysis and were obtained from 4.5 cm caudal to the termination of the conus medullaris. Specimens (HERDA n = 4; control n = 5) were formalin-fixed for 24 hours prior to transfer to glutaraldehyde in preparation for transmission electron microscopy (TEM) processing. After primary fixation in glutaraldehyde, samples were washed with 0.1 M cacodylate buffer. Samples were postfixed with 1% osmium tetroxide in 0.1 M cacodylate buffer, dehydrated in a gradient series of acetone, infiltrated, and embedded in Spurr. 24 A Power Tome Ultramicrotome (RMC, Boeckeler Instruments) was used to cut 70 nm thin sections and post stained with uranyl acetate and lead citrate. The FT was imaged using a JEOL 100CX Transmission Electron Microscope (Japan Electron Optics Laboratory).
Human FT specimens (EDS-TCS n = 2; TCS n = 1) were fixed in glutaraldehyde prior to being washed in upgraded alcohols for dehydration followed by 4 changes of propylene oxide, infiltrated and embedded in epon-araldite. 17 Ultra-thin sections were cut using Reichert-Jung Ultracut E (Leica Microsystems) ultramicrotome at 80 nm and post stained with uranyl acetate and lead citrate. Imaging was performed with a Philips CM10 electron microscope (Philips).
Results
Gross and Histologic Findings
The intradural FT, similar to the human filum terminale internum, was identified in all 9 horses (Fig. 1) and originated at the S2 vertebra and terminated at approximately S4.

Normal control, conus medullaris, horse. The conus medullaris (black arrow) transitions to the intradural filum terminale (black star) at the second sacral vertebra.
Histologic features of the equine FT (Table 1) included a central canal surrounded by neuropil and more laterally by a dense layer of fibrous tissue. Small nerve twigs were present throughout the FT. Mineralized collagen was occasionally observed. The presence of neuropil, ependyma, and nerve twigs did not differ between HERDA horses and controls (Figs. 2a, b, 3).
Histopathologic and Ultrastructural Findings in the Filum Terminale of 4 Horses With Hereditary Equine Regional Dermal Asthenia (HERDA) and 5 Controls.
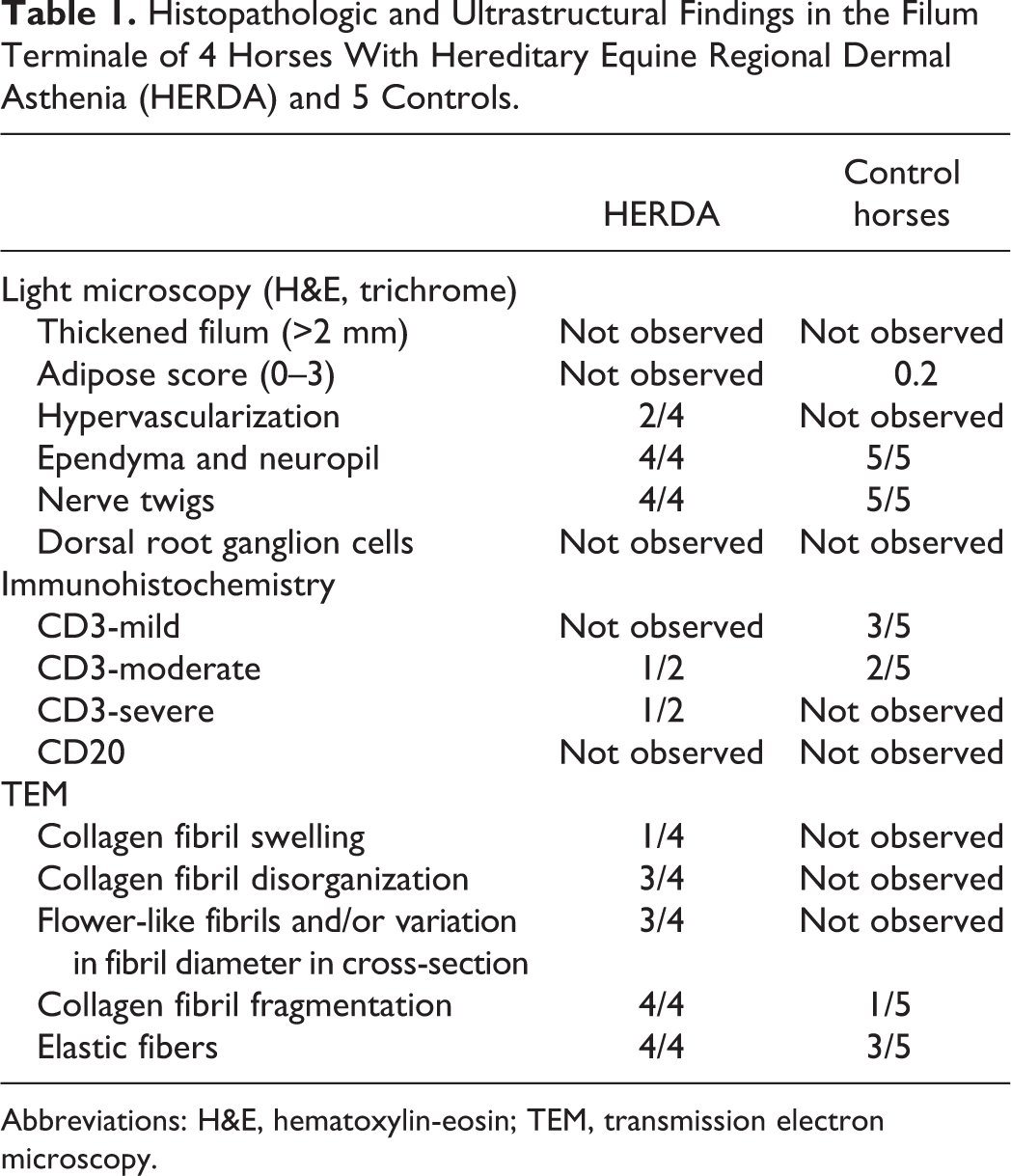
Abbreviations: H&E, hematoxylin-eosin; TEM, transmission electron microscopy.
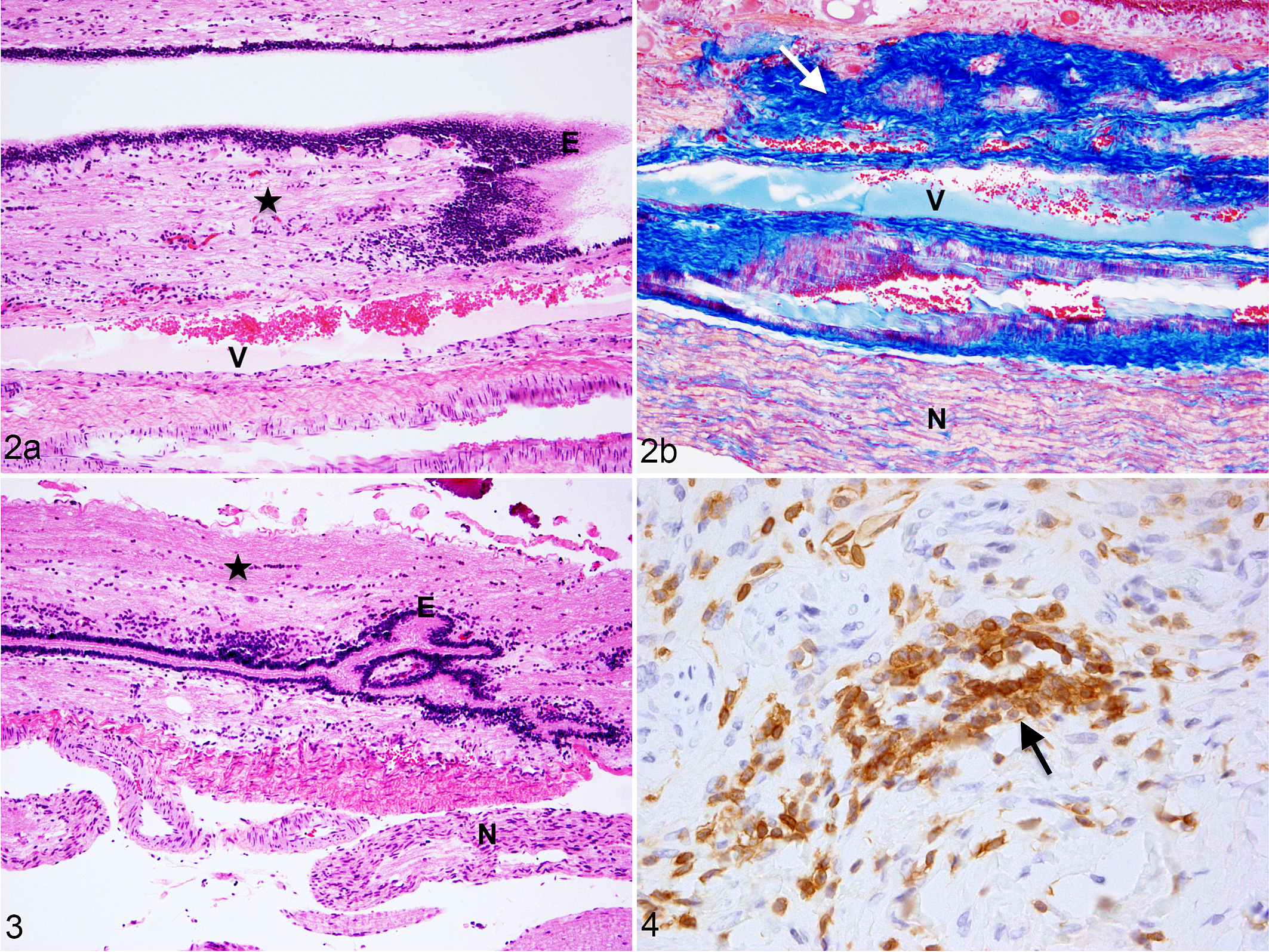
Hereditary equine regional dermal asthenia (HERDA), filum terminale, horse. (a, b) The filum terminale contains ependyma (E), neuropil (black star), fibrous tissue (white arrow), hypervascularization (V), and nerve twigs (N). (a) Hematoxylin and eosin (HE); (b) Masson’s trichrome.
Two of 4 HERDA horses displayed hypervascularity (Fig. 2a, b). Hypervascularity was not demonstrated in any of the 5 control horses. Fibrous tissue was observed in all horses in sections stained with Masson’s trichrome (Fig. 2b). Adipose tissue was rarely observed in the control horse FT (mean adipose score = 0.2) and was not observed in the FT of horses with HERDA. Immunolabeling for CD3 was observed in the FT of control horses and of HERDA horses (Table 1, Fig. 4).
Hypervascularity was observed in both EDS-TCS specimens and was not observed in the TCS specimen (Fig. 5a, b). Adipose tissue was not observed in either EDS-TCS specimen but was present in the TCS specimen (adipose score = 3; Fig. 6). Ependyma, neuropil, and nerve twigs were observed in both EDS-TCS specimens while only nerve twigs were observed in the TCS specimen (Figs. 5a, b, 6). Additionally, dorsal root ganglion cells were observed in 1 of 2 EDS-TCS specimens but were not observed in the TCS specimen. CD45+ leukocytes were observed in both human EDS-TCS specimens (Fig. 7). The TCS specimen did not undergo CD45 immunolabeling.
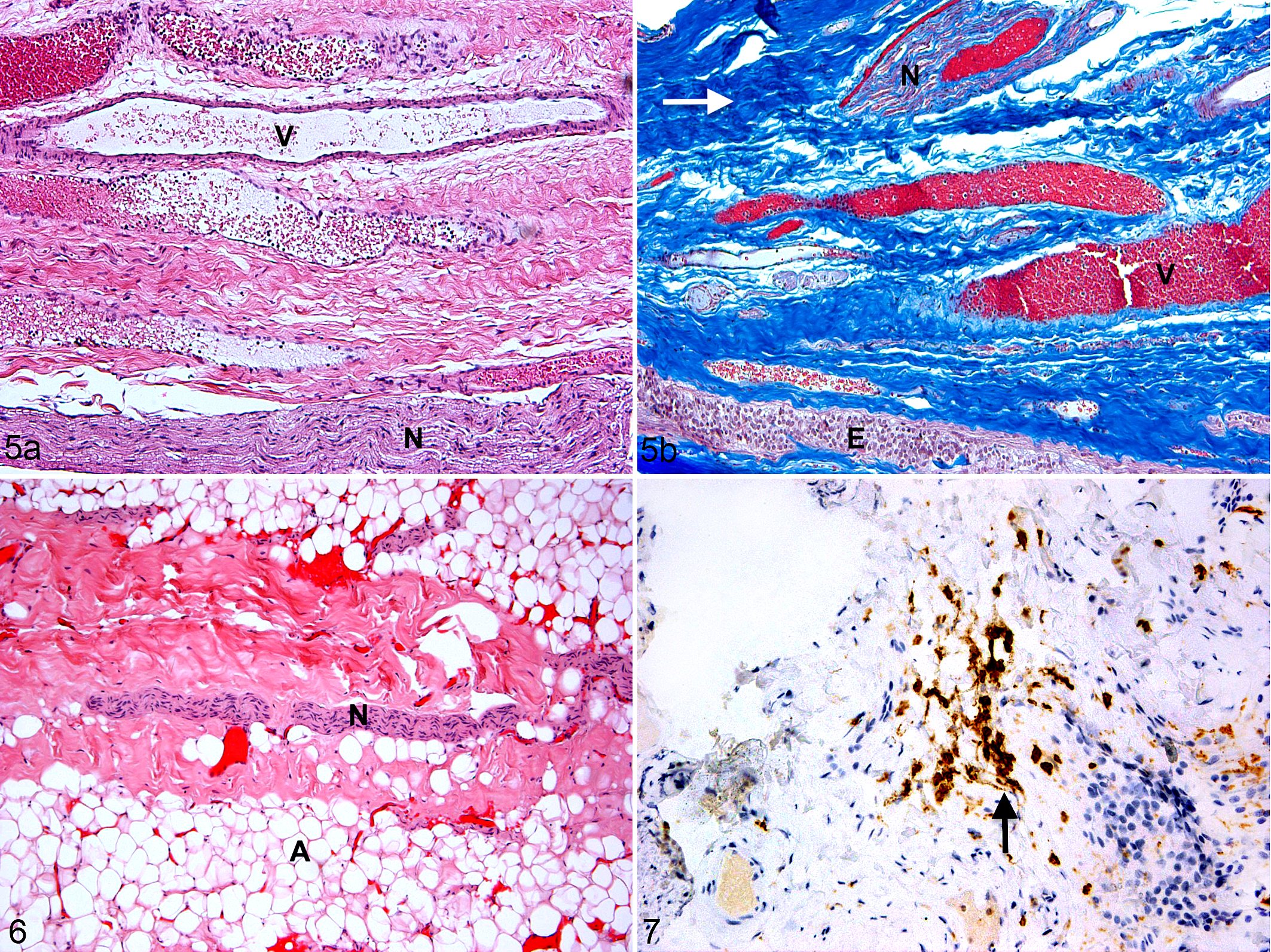
Ehlers-Danlos syndrome and tethered cord syndrome (EDS-TCS), filum terminale, human. (a) The filum terminale contains ependyma (E), fibrous tissue (white arrow), hypervascularization (V), and nerve twigs (N). (a) Hematoxylin and eosin (HE); (b) Masson’s trichrome.
Filum Terminale Ultrastructure
Collagen fibrils and elastic fibers were evaluated in longitudinal and cross-section (Table 1). Three of 4 HERDA horses had fibril disorganization with abnormalities including whirling fibrils and hook-shaped fibrils. In contrast, collagen from the FT of 5 control horses was well-organized (Figs. 8a, 9a). Flower-like collagen and/or variation in fibril diameter in cross-section were observed in 3 of 4 HERDA horses and none of the 5 controls (Figs. 8b, 9b). Fragmented collagen fibrils were observed in 4 of 4 HERDA horses and 1 of 5 controls (Fig. 8a). Fibril swelling was observed in longitudinal section in 1 of 4 HERDA horses and 0 of 5 controls (Fig. 8a). Overall, with regard to all collagen abnormalities, HERDA horses had more ultrastructural abnormalities compared with control horses. Elastic fibers were present in 4 of 4 HERDA horses and 3 of 5 control horses.
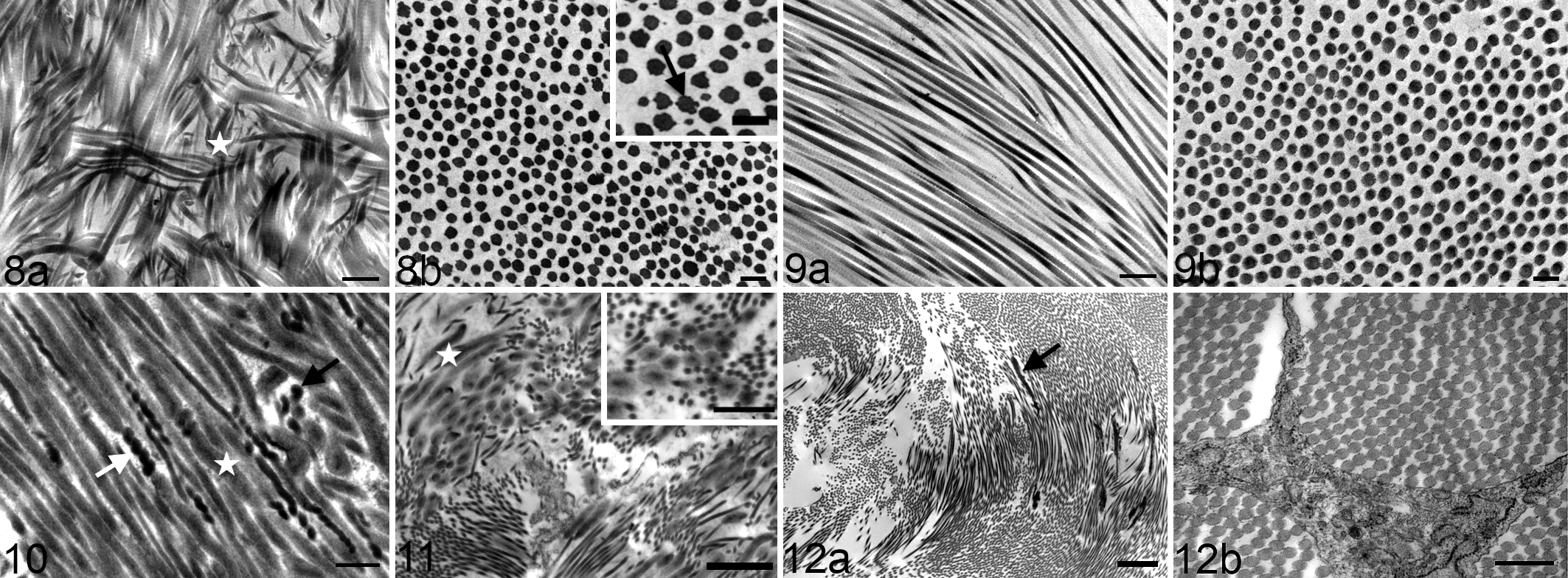
Hereditary equine regional dermal asthenia, filum terminale, horse. Transmission electron microscopy (TEM). (a) Severe collagen fibril disorganization, mild fibril swelling, and fibril fragmentation (white star). Bar = 0.5 μm. (b) Flower-like fibrils (black arrow) and variation in fibril diameter. Bar = 0.2 μm. Inset: higher magnification. Bar = 200 nm.
Corkscrewing and beading of the collagen fibrils, a finding not observed in the equine population, was present in 2 EDS-TCS specimens but was not observed in the TCS specimen (Fig. 10). Fibril disorganization was observed in 2 EDS-TCS FT specimens but was not present in the TCS specimen (Figs. 11, 12a). Variation in fibril size in cross-section was present in 2 EDS-TCS FT samples but was not observed in the TCS specimen (Figs. 11, 12b). Fibril fragmentation was present in 2 EDS-TCS specimens but was not observed in the TCS specimen. Fibril swelling was observed in 2 EDS-TCS samples but was not observed in the TCS specimen (Figs. 10, 11). Elastic fibers were present in 2 EDS-TCS and in the TCS specimen (Fig. 12a).
Discussion
The results of this study support that the anatomy, histology, and ultrastructure of the equine FT are similar to that of the human FT. Our findings in both the HERDA horse FT and human EDS-TCS FT demonstrate that connective tissue disorders are associated with disorganization of the collagenous elements of the FT and are a potential pathogenetic mechanism for abnormal development.
To the authors’ knowledge, the histology of the normal equine FT has not previously been evaluated; our results suggest that the equine and human FT are similar histologically. The histology of the human FT was first described as an extension of the pia mater containing fibrous tissue, ependyma, glial tissue, corpora amylacea, myelinated axons, and dorsal root ganglion cells. 10 Small amounts of adipose tissue are hypothesized to be a normal finding in the FT of up to 5.8% of healthy humans. 6 Similar to past human studies, the control horse FT contained fibrous tissue, adipose tissue, neuropil, ependyma, and nerve twigs. Dorsal root ganglion cells were not observed in the horse. This may be a species difference or the result of a sectioning artifact.
Histopathological findings in the FT in humans with TCS included hypervascularization, increased amounts of connective tissue with dense collagen fibers, areas of hyalinization, nerve twigs, and occasional adipose tissue. 23 These results are similar to our finding of hypervascularization in HERDA horses and EDS-TCS patients. The increased vascularization may result from increased mechanical stress similar to that which is observed in human tendinopathy. 2,13,22,26 Variable numbers of lymphocytes were present in HERDA and control horses and were also noted in the human EDS-TCS samples. Given the presence of lymphocytes in the control horse group, this may be a normal finding or could indicate an inflammatory lesion.
While the ultrastructure of the normal equine FT has not previously been described, prior studies described the normal ultrastructure of the human FT using SEM and of the frog FT using TEM. 7,8 In a scanning electron microscopy study of the FT in adult human cadavers, longitudinal bundles of type I collagen were connected by type III transversal collagen fibers alluding to the significance of the collagenous elements in the FT. 7 Scanning electron microscopic findings in 31 pediatric human cases of TCS suggested that these patients had altered elasticity secondary to a reduced elastin to collagen ratio as well as an abnormal 3D structure of collagen fibrils. 14
Past studies have demonstrated ultrastructural abnormalities of collagen in horses with HERDA in tissues including the skin and cornea. 16,32 Similar to findings in other body tissues and what was observed in the human EDS-TCS patients, 12 some HERDA horses had collagen fibril swelling, disorganization, flower-like fibrils and/or variation in fibril size in cross-section, and fragmented collagen fibrils. These abnormalities were not commonly observed in the control horse population. Corkscrewing and beading of collagen fibrils, a finding observed in human EDS-TCS, was not observed in the HERDA or control horse population. Stretch-induced corkscrewing and beading of the collagen fibrils occurs following mechanical rupture of bovine tail tendon. 30 The lack of corkscrewing and beading in HERDA may reflect an abnormal FT secondary to genetic mutation rather than mechanical tethering.
Limitations of this study include the study’s small sample size; however, horses with HERDA are a difficult population to obtain and maintain and the sample size is in line with other recent FT TEM studies. 7 At this time, we are limited in our ability to detect and diagnose TCS in the horse. Clinical neurological abnormalities were not observed in our HERDA population and have not previously been described. Additionally, lumbosacral magnetic resonance imaging and urodynamic testing, 9,11,19 which are routinely used to diagnose TCS in humans, are not routinely available in horses. Furthermore, HERDA horses are often euthanized due to their illness and therefore some affected horses may not live long enough to display neurologic signs associated with their condition. Humans with EDS often do not develop clinical signs of tethering until their late teens or early 20s, and HERDA horses are often euthanized prior to 3 years of age.
Conclusions
These observations shed new light on the understanding of the anatomy, histopathology, and ultrastructure of the equine FT terminale. Additionally, the findings suggest connective tissue disorders such as HERDA and EDS may act as pathogenetic risk factors for the development of TCS through severe derangements in collagen fibril architecture within the FT. These derangements, in conjunction with a lifetime of spinal trauma secondary to hypermobility, may predispose to tethering. More work is needed to evaluate the impact of FT abnormalities in the HERDA population, including correlation with clinical signs.
Footnotes
Acknowledgements
Thanks to Dr. Alicia Withrow, Michigan State University Center for Advanced Microscopy, and to Grant Jolly, Department of Pathology and Laboratory Medicine at Rhode Island Hospital, for their assistance with transmission electron microscopy. Thanks to Dr. Kirstin Bubeck at the Cummings School of Veterinary Medicine at Tufts University for her assistance with necropsy examination in one control horse. Thanks to Dr. Jon Patterson of the Michigan State University Veterinary Diagnostic Laboratory, and Dr. Edward Stopa of the Rhode Island Hospital, Department of Pathology, for their assistance with equine and human neuropathology.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: Funding for this study was received from the Norman Prince Neurosciences Institute.
