Abstract
A 7-year-old, spayed female, Wheaton terrier dog was icteric, lethargic, and anorexic with increased activity of hepatocellular and cholestatic liver enzymes and an extreme hyperbilirubinemia level of 609 μmol/L (reference interval: 1.0–4.0 μmol/L). Necropsy findings included profound icterus and red and yellow mottling of the liver. Yellow discoloration of the thalamic and subthalamic nuclei was detected on subgross examination of the formalin-fixed brain. Histologic examination of the brain revealed neuronal necrosis within the discolored nuclei, necrosis of Purkinje cells, and Alzheimer type II astrocytes in the cerebrocortical gray matter and in the nuclei, with gross discoloration. Histologic examination of the liver revealed extensive necrosis in a periacinar-to-bridging pattern and often extending to portal triads. A case of naturally occurring kernicterus in an adult dog secondary to extreme hyperbilirubinemia resulting from fulminant hepatic failure is reported. The few reports of this disease in domestic species involved neonates, namely 1 foal and 1 kitten.
Kernicterus or bilirubin encephalopathy is characterized by macroscopic yellow discoloration of the basal ganglia, subthalamic nuclei, hippocampus, and cerebellum, following penetration of the blood-brain barrier by unconjugated bilirubin (Bu).5 Neuronal necrosis is often present in the discolored areas, but why these areas are targeted is unknown.
Bilirubin, a product of heme catabolism, is found in the blood in a hydrophilic conjugated form and a lipophilic unconjugated form that is typically noncovalently bound to albumin. Intracellular bilirubin is toxic to cells, causing decreased protein synthesis, depressed cellular respiration, increased glycolysis, and uncoupled mitochondrial oxidative phosphorylation, ultimately leading to cell death.2 A key factor in the pathogenesis of kernicterus is that free Bu is lipophilic and can thus cross the blood-brain barrier. Increased Bu occurs with hyperbilirubinemia, which develops when bilirubin production exceeds excretion as a result of increased production, decreased conjugation, or decreased hepatocyte uptake.4 Albumin has the capacity to bind Bu in the plasma when the total bilirubin concentration is ≤340–430 μmol/L. Above this concentration, cellular neurotoxicity and kernicterus can occur. Hypoalbuminemia or decreased binding capacity due to competing molecules or acidosis will increase free Bu concentrations, allowing kernicterus to occur at lower total bilirubin levels.2
A 7-year-old, spayed female Wheaton terrier dog was referred to the Veterinary Teaching Hospital of the Western College of Veterinary Medicine with a 3-day history of icterus, lethargy, anorexia, and occasional vomiting. The dog had been spayed by the referring veterinarian 2 weeks previously. Significant serum chemistry parameters at the time of referral included bilirubin levels of 609 μmol/L (reference interval [RI]: 1.0–4.0 μmol/L), alkaline phosphatase level of 321 U/L (RI: 9–90 U/L), gamma glutamyltransferase level of 31 U/L (RI: 0–8 U/L), and alanine aminotransferase level of 998 U/L (RI: 19–59 U/L). The pH level of the blood was decreased at 7.29 (RI: 7.31–7.42), with increased lactate concentration of 9.32 mmol/L (RI: 0.46–2.31).
The following day, an exploratory laparotomy was performed, during which the liver was noted to be small and mottled in appearance and the gall bladder appeared mildly enlarged, but the bile duct was patent. A liver biopsy was collected by the guillotine method. Aside from profound jaundice, no other abnormalities were noted. By the next day, the dog was tachycardic and intermittently seizuring. In addition to similar biochemical changes as listed above, the dog had become pan-hypoproteinemic, with an albumin concentration of 18 g/L (RI: 28–38 g/L) and a globulin concentration of 11 g/L (RI: 23–37 g/L). Blood glucose level decreased over the course of the 3 days to 2.9 mmol/L (RI: 3.1–6.3 mmol/L), potentially indicating the onset of sepsis. The dog died later that day.
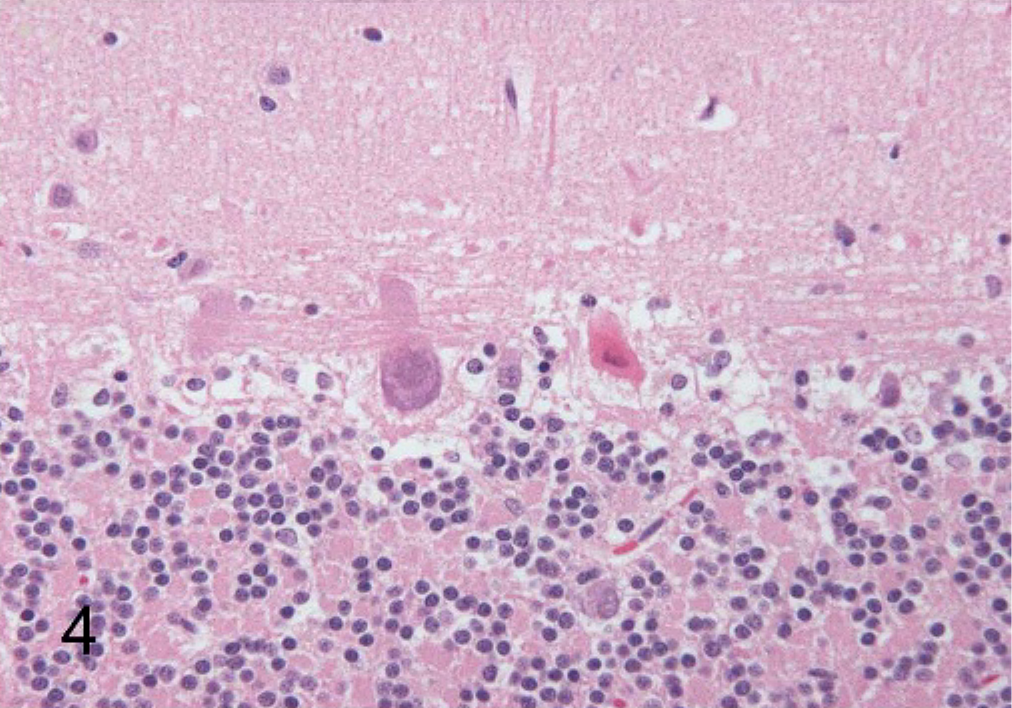
On necropsy, the dog was profoundly icteric with yellow-orange shading of the mucous membranes, ears, and body wall externally and striking yellow discoloration of all fat in the dog. The liver was small, appeared mottled red and yellow in a reticulated pattern, and had multifocal, 3- to 5-mm diameter sunken red areas over all lobes. The abdomen contained approximately 1 liter of frank blood that appeared to originate from the site of the liver biopsy, which was covered by a large blood clot (Fig. 1). Subgross examination of the formalin-fixed, sectioned brain revealed bilaterally symmetric yellow foci, which corresponded with the thalamic and subthalamic brainstem nuclei (Fig. 2). Histopathologic examination of the brain revealed contracted, angular neuron cell bodies in the subthalamic and thalamic nuclei and Purkinje cells in the cerebellum having condensed eosinophilic cytoplasm and shrunken pyknotic nuclei consistent with necrosis (Figs. 3, 4). Alzheimer type II astrocytic change was evident in cerebrocortical gray matter and in astrocytes in the discolored thalamic and subthalamic nuclei (Fig. 5). The liver had large, bridging-to-coalescing areas of pallor in which there were no recognizable hepatocytes (Fig. 6). A pattern of hepatocyte loss was difficult to appreciate but appeared to be periacinar to bridging, often extending to the portal triads. The remaining hepatocytes were undergoing ballooning degeneration punctuated by individual cell necrosis. With Hall's stain, globules of bilirubin were detectable in several bile canaliculi and larger bile ducts, indicating bile stasis.
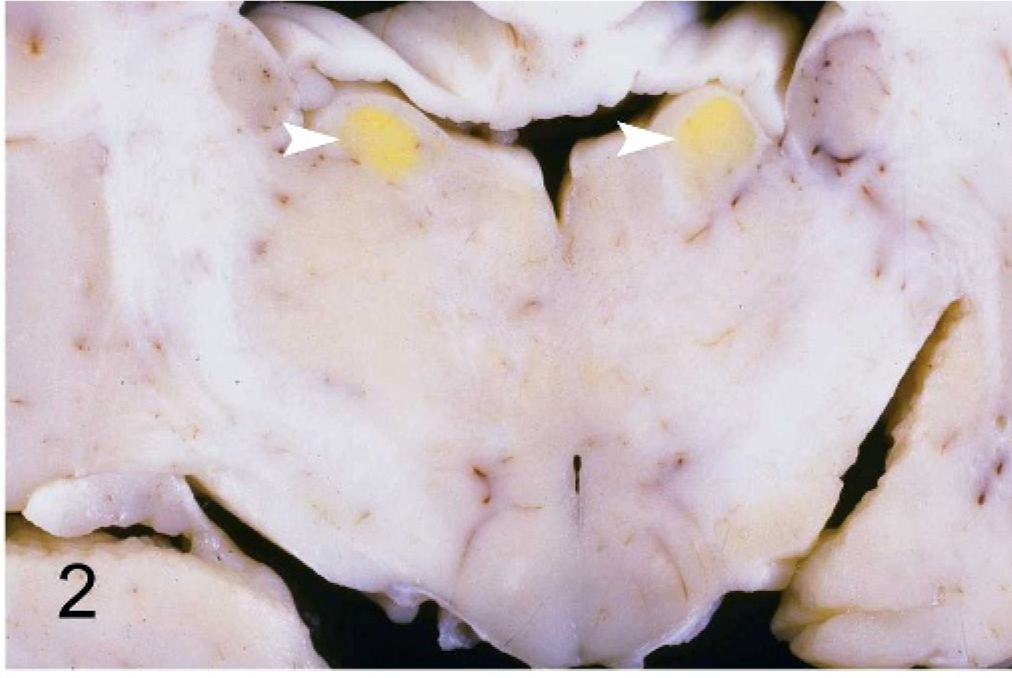
Thalamus; canine. Bilaterally symmetric yellow foci, which correspond with the thalamic (arrowheads, Fig. 2) and subthalamic (Fig. 3) brainstem nuclei.
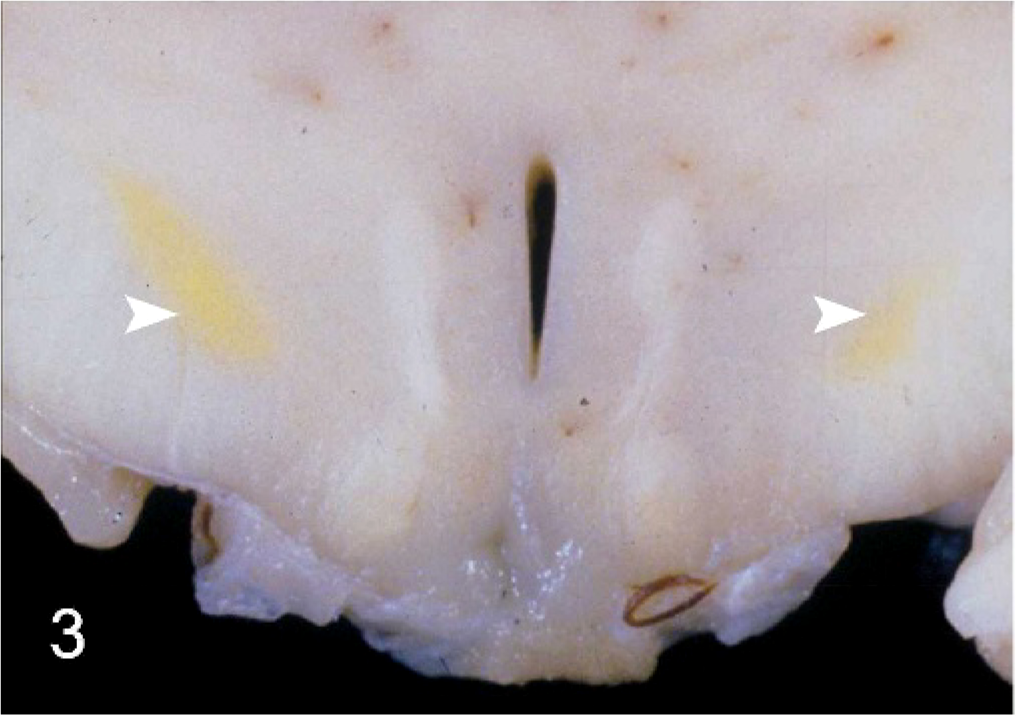
Thalamus; canine. Bilaterally symmetric yellow foci, which correspond with the thalamic (arrowheads, Fig. 2) and subthalamic (Fig. 3) brainstem nuclei.
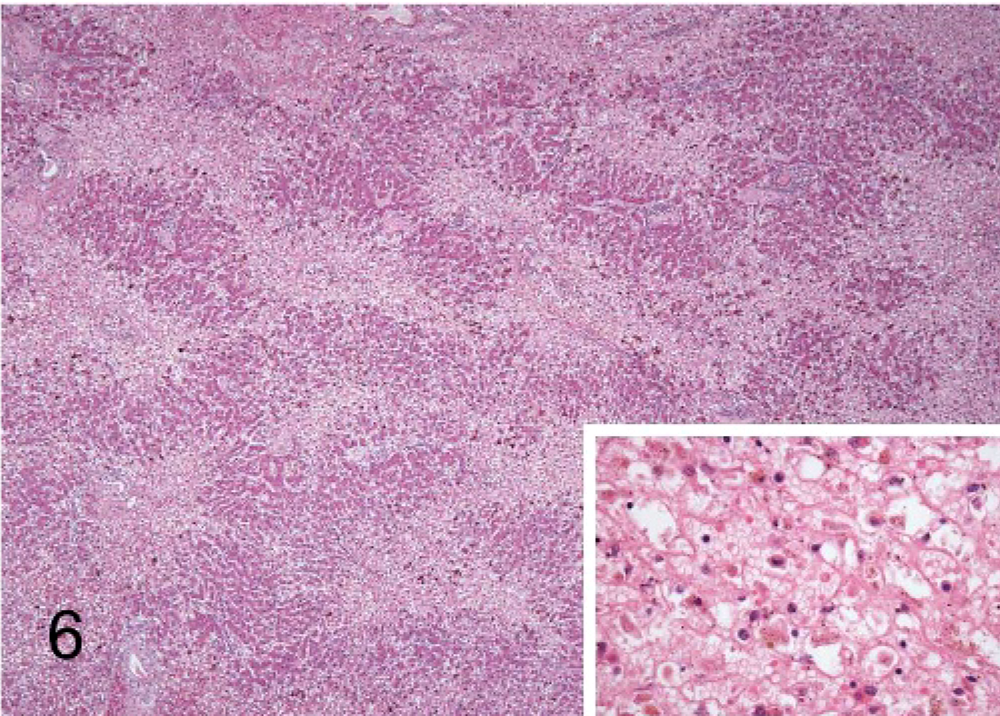
Liver; canine. Hepatocellular necrosis, periacinar to bridging, often extends to the portal triads. Remaining hepatic tissue is vacuolated and punctuated by individual cell necrosis. Inset provides a higher magnification of a necrotic area. HE 100× magnification; inset magnification is 500×.
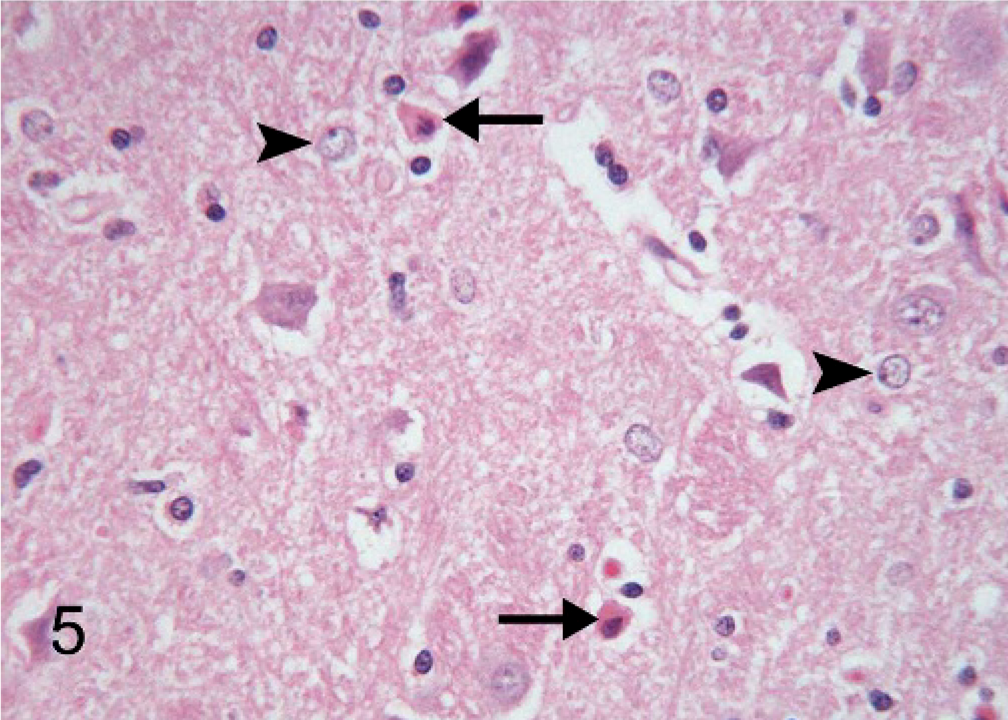
Thalamic nuclei; canine. Neuronal cell bodies are eosinophilic, shrunken, and angular, indicating necrosis (arrows). Alzheimer type II astrocytes are present (arrowheads). HE; 400× magnification.
Cerebellum; canine. Purkinje cells are shrunken and contain eosinophilic cytoplasm and contracted, angular nuclei, consistent with necrosis. HE; 500× magnification.
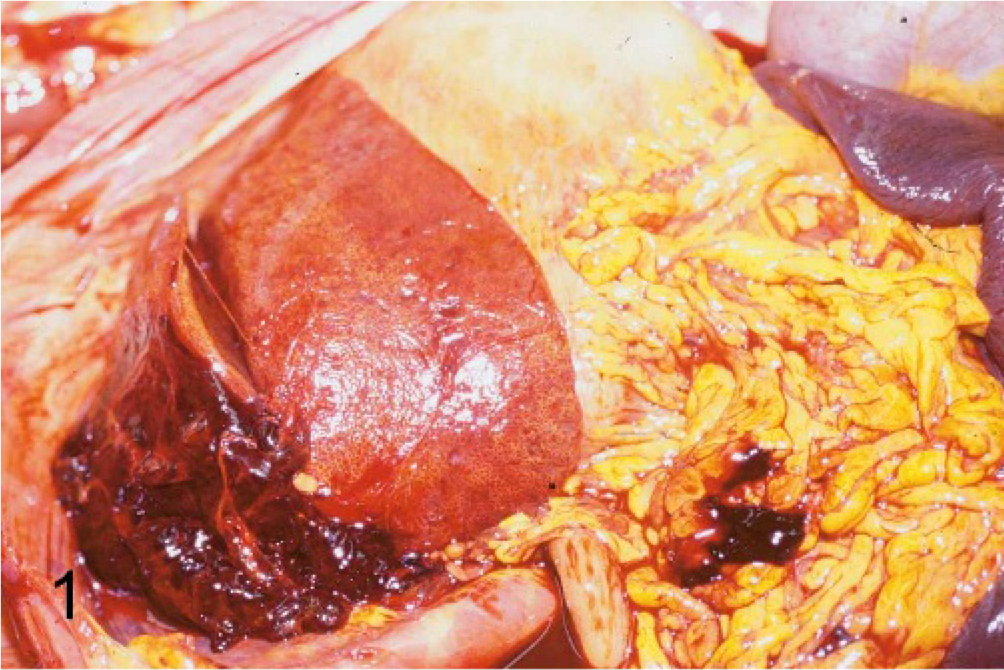
Abdominal cavity; canine. Profound icterus is evidenced by the striking yellow discoloration of all fat. The liver was small and mottled red and yellow. Note the large blood clot associated with the biopsy site.
Kernicterus or bilirubin encephalopathy is most commonly recognized in human infants. It results from a combination of both physiologic and pathologic factors, including 1) increased production of bilirubin as a result of hemolysis, 2) deficiency of hepatic uptake, 3) immaturity of the bilirubin conjugation system, and 4) increased bacterial deconjugation and resorption of bilirubin in the intestine.4 The Gunn rat is a model for this disease because of a recessive genetic mutation that results in low-to-absent hepatic uridine diphosphate (UDP)–glucuronyl transferase activity in animals homozygous for the trait. The reduced activity of this enzyme prevents conjugation of bilirubin.9
Aside from the Gunn rat, kernicterus appears to be a very rare condition in domestic animals. Published reports include that of a foal with neonatal isoerythrolysis3 and a kitten with no identifiable reason for the development of the disease but that had clinical signs very similar to those of human neonates with this condition.7 No reports of kernicterus occurring naturally in an adult animal could be found. In human adults, morphologically proven kernicterus is rare and is typically associated with an inherited UDP–glucuronyl transferase deficiency. Only 2 reports of kernicterus occurring secondary to liver disease in previously healthy non-neonate humans could be found.6,8
In this case, the etiology of the fulminant liver failure remains unknown. However, there is little doubt that this disorder caused hyperbilirubinemia of sufficient severity to overwhelm the binding capacity of albumin and allowed Bu to cross the blood-brain barrier, resulting in kernicterus. In addition to this extreme hyperbilirubinemia, a number of other factors occurring in this dog may have also contributed to the pathogenesis. Reduced production of albumin from decreased hepatic function and its role as a negative acute-phase protein resulted in hypoalbuminemia, decreasing the threshold amount of Bu that could be bound. The binding capacity of albumin was also further reduced by the state of lactic acidosis, which likely resulted from tissue hypoxia and decreased lactate utilization by the damaged liver. Finally, this dog was suffering from hepatic encephalopathy and potentially sepsis, both of which are thought to disrupt the blood-brain barrier.1,4
To the best of the authors' knowledge, this is the first report of naturally occurring kernicterus in an adult domestic animal. A well-known condition of human infants, kernicterus is also rare in human adults.
