Abstract
A 7-month-old female domestic shorthair cat was diagnosed with chronic regenerative hemolytic anemia characterized by increased osmotic fragility of unknown etiology. At 13 months of age, the cat was evaluated for acute collapse. The cat was icteric with severe hyperbilirubinemia but no hematocrit changes. Severe obtundation and lateral recumbency progressed to tetraparesis and loss of proprioception in all 4 limbs, and a cerebellar or brainstem lesion was suspected. Postmortem examination revealed suppurative cholangiohepatitis and acute neuronal necrosis in the nuclei of the brainstem and cerebellum, consistent with bilirubin encephalopathy. This is the first known occurrence of cholangiohepatitis and bilirubin encephalopathy in an adult cat with chronic hemolytic anemia. Although rare, bilirubin encephalopathy should be considered a possible sequela to hyperbilirubinemia in adult patients. It remains unknown whether increased osmotic fragility was related to the cholangiohepatopathy.
A 7-month-old female spayed domestic shorthair cat was referred to the Colorado State University Veterinary Teaching Hospital for evaluation of persistent regenerative anemia. The owners reported that the cat always slept more than the other cats in the household and had a calmer demeanor. Physical examination was unremarkable except for pale mucous membranes. A complete blood count revealed regenerative anemia with a hematocrit of 19% (reference interval [RI]: 28%–43%), a decreased mean corpuscular hemoglobin concentration of 31 g/dl (RI: 33–36 g/dl), an increased mean corpuscular volume of 66 fl (RI: 37–51 fl), reticulocytosis of 244 000/µl (RI: 0–50 000/µl), and moderate thrombocytosis of 674 000/µl (RI: 200 000–500 000/µl). Blood smear examinations revealed macrocytosis, anisocytosis, polychromasia, and numerous Howell Jolly bodies and eccentrocytes. However, the blood smear results were negative for hemoparasites and autoagglutination, and a direct Coombs test at 37°C was negative (IDEXX Laboratories, Westbrook, ME). A chemistry panel revealed a mild hyperbilirubinemia of 0.3 mg/dl (RI: 0–0.1 mg/dl) and a mildly elevated alanine aminotransferase (ALT) activity of 157 IU/l (RI: 30–140 IU/l). Serum albumin concentration and leukogram levels were normal. Abdominal ultrasound showed mild hepatosplenomegaly.
Prior to having infectious disease test results back, cefovecin sodium (subcutaneously once), a 3-week course of doxycycline, and a 5-day course of fenbendazole were empirically administered. All infectious disease–screening results were negative, including tests for feline leukemia virus antigen, feline immunodeficiency virus antibody, and feline corona virus antibody (IDEXX Laboratories, Westbrook, ME). Polymerase chain reaction results for Mycoplasma haemofelis and Mycoplasma haemominutum (previously Candidatus haemominutum) and for multiple Bartonella spp (B. henselae, B. bacilliformis, B. clarridgeiae, B. elizabethae, B. quintana, and B. vinsonii subsp berkhoffii) were also negative.
A hereditary erythrocyte defect was suspected as the cause of the regenerative hemolytic anemia. Genetic testing for erythrocytic pyruvate kinase (PK) deficiency was negative (PennGen, University of Pennsylvania, Philadelphia, PA). However, the erythrocyte osmotic fragility (OF) test result was abnormal, with the proband’s erythrocytes lysing at higher saline concentrations, indicating severe erythrocytic fragility (PennGen; Fig. 1).
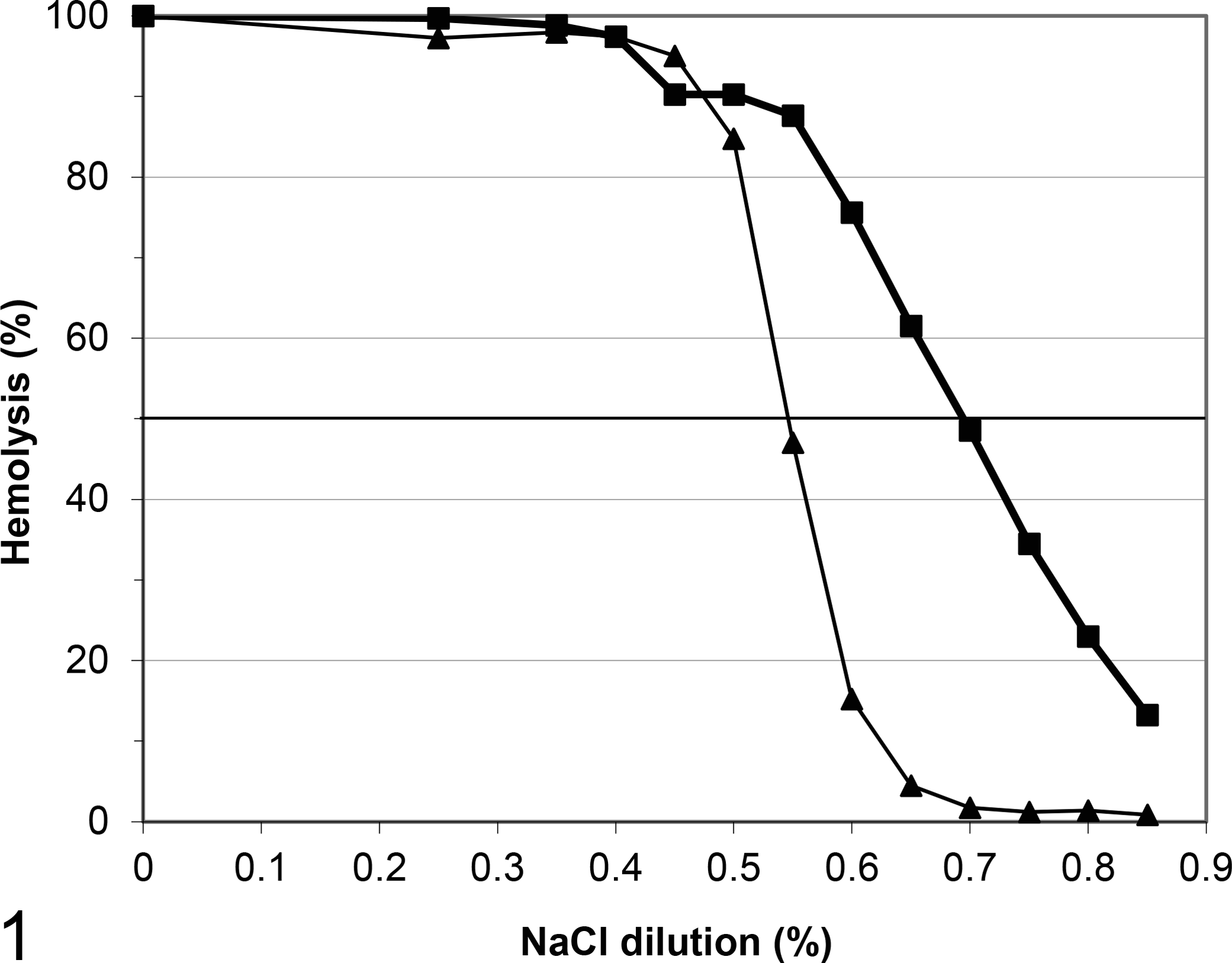
Erythrocytic osmotic fragility curve: domestic shorthair cat with chronic regenerative anemia (▪; 50% hemolysis at 0.70%) and a simultaneously tested healthy control cat (▴; 50% hemolysis at 0.55%).
At the 1-, 5-, and 6-month rechecks, physical examination, clinical history, complete blood count, and OF test results remained virtually unchanged, although the plasma at the last visit was mildly icteric. No additional diagnostics were elected. Six days after the 6-month recheck, the cat was presented to Colorado State University for evaluation of acute collapse, unresponsiveness, and severe icterus. Her pupils were mydriatic with occasional vertical nystagmus. A complete blood count revealed macrocytic regenerative anemia as seen on prior examinations (hematocrit, 21%; mean corpuscular volume, 62 fl; mean corpuscular hemoglobin concentration, 31 g/dl; reticulocytes, 191 000/µl; moderate numbers of Howell Jolly bodies). The leukogram was normal. Serum chemistry screen revealed severe hyperbilirubinemia (20.5 mg/dl) and a mild elevation of ALT activity (199 IU/l); albumin concentration was normal. An abdominal ultrasound showed again hepatosplenomegaly. The patient was hospitalized overnight and given supportive care, including intravenously administered fluids, N-acetylcysteine, and clindamycin and supplemental oxygen.
The next day, the neurologic signs progressed to obtundation and tetraparesis with intention tremors, loss of proprioception in all 4 limbs (normal reflexes in the pelvic and reduced in the thoracic limbs), and opisthotonos. Positional nystagmus, absent menace, and sluggish pupillary light responses were also noted. Serum bilirubin concentration (25.5 mg/dl) and ALT activity also increased slightly (225 IU/l), indicating a hepatopathy. The cat was alkalotic (pH 7.46, RI: 7.33–7.44) with bicarbonate of 22.7 mm/l (RI: 16.0–22.0 mm/l), pCO2 of 32.3 mm Hg (RI: 35.0–42.0 mm Hg), and anion gap of 16 (RI: 18–27 mEq/l). The cat was euthanized due to poor prognosis in the face of progressive neuropathy (cerebellar or brainstem lesion) and hepatopathy.
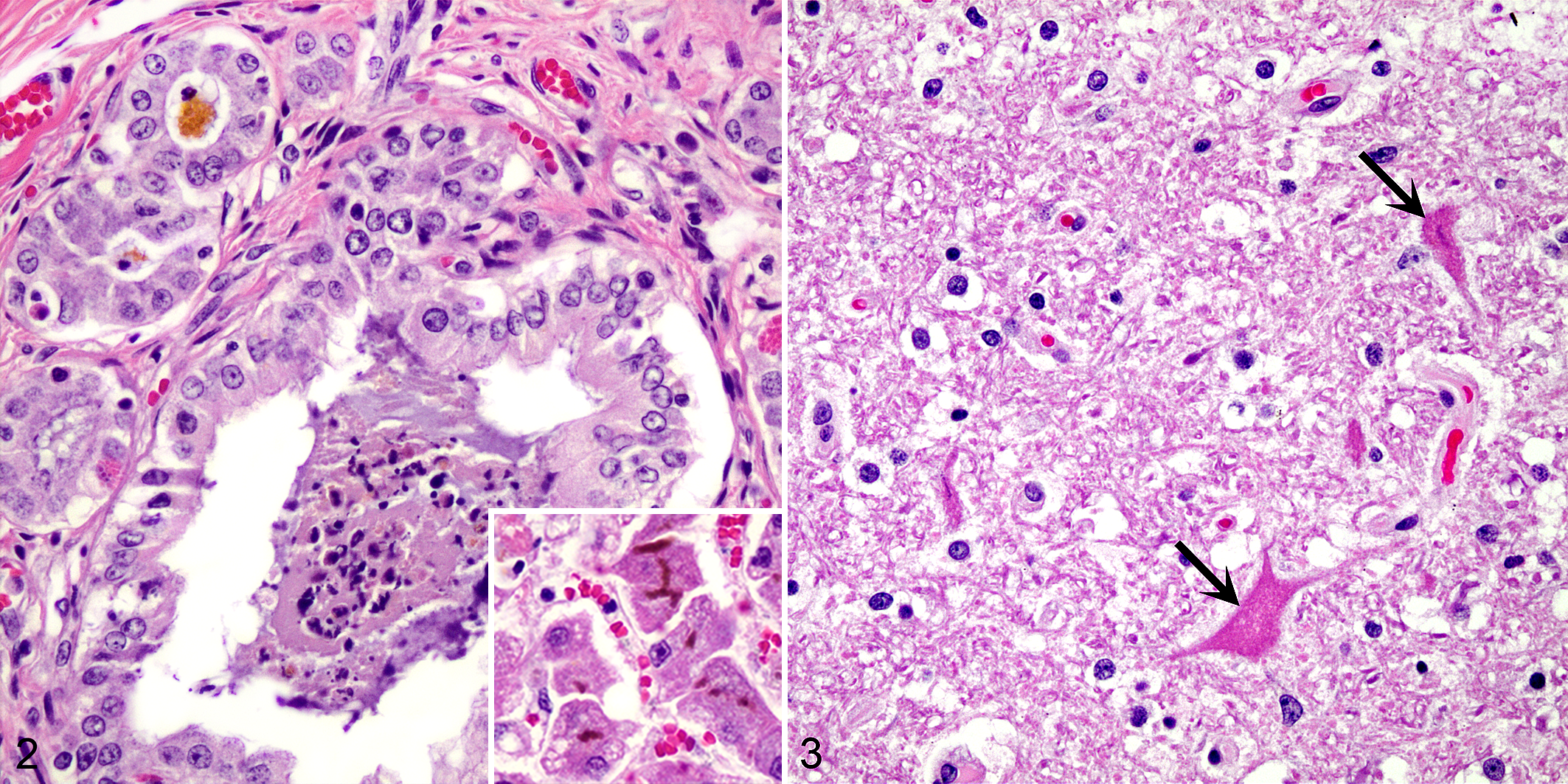
On postmortem examination, severe icterus was present. The liver was mildly enlarged with an enhanced reticular pattern, which correlated histologically with a marked suppurative, subacute cholangiohepatitis and severe cholestasis (Fig. 2). The bone marrow was diffusely dark red, and severe erythroid hyperplasia was present histologically; splenic erythroid hematopoiesis was also noted. Macroscopically, the brainstem and cerebellum contained small bilaterally symmetric foci of yellow discoloration, which corresponded histologically to foci of acute neuronal necrosis in the deep cerebellar nuclei (Fig. 3). Affected neurons were shrunken with hypereosinophilic cytoplasm and pyknotic or lost nuclei. Small extracellular deposits of bilirubin pigment were present multifocally in the splenic sinusoids, bone marrow, as well as infrequently within the lymphatics draining to mesenteric lymph nodes. There were no other noteworthy histologic lesions in sections of brain or other parenchymal tissues.
This represents the first report of bilirubin encephalopathy associated with chronic hemolytic anemia and cholangiohepatitis in an adult cat. Bilirubin encephalopathy is the result of toxic effects of unconjugated bilirubin within the brain, which results in necrosis of susceptible neurons. 9,13 Although unconjugated bilirubin was not measured in this case, the combination of (1) severe diffuse icterus with (2) grossly evident, bilaterally symmetric yellow foci within brainstem nuclei and (3) histologic evidence of neuronal necrosis in these sites is consistent with this condition. Though rare in small animals, bilirubin encephalopathy is a well-known complication in hyperbilirubinemic foals with neonatal isoerythrolysis secondary to blood incompatibilities. Approximately 8% of foals with neonatal isoerythrolysis developed bilirubin encephalopathy in 1 retrospective study. 6 A case of bilirubin encephalopathy in a kitten has been reported, although the underlying cause was not clear and a blood incompatibility issue was not ruled out. 12 There are only 2 reported cases in dogs—1 in an adult dog with massive hepatic necrosis and 1 in a dog with severe immune-mediated hemolytic anemia. 1,8 This is the first known report of bilirubin encephalopathy in an adult cat. The term kernicterus—historically used to describe the macroscopic yellowing of nuclei in human neonates—has been extended to encompass the occurrence of bilirubin encephalopathy in neonatal domestic animals.
It is not clear why bilirubin encephalopathy develops in some cases of hyperbilirubinemia and not others. The immaturity of the neonatal blood-brain barrier and the bilirubin conjugation system has been proposed as a potential factor in predisposing young patients. 9 The underlying cause and severity of hyperbilirubinemia, the rate of progression, and other comorbid conditions (eg, concurrent sepsis, inflammation, acidosis, or hypoalbuminemia) may affect whether neurologic signs occur. 5,14 Treatment with sulfonamides has also been implicated as a potential risk factor, but no sulfonamide anitibiotics were administered to this patient. 7 While sepsis secondary to severe cholangiohepatitis cannot be completely ruled out, the patient did not have a leukocytosis, hypoglycemia, or acidosis during crisis. The patient was mildly alkalotic and had normal serum albumin concentration at the time of final presentation.
The molecular mechanisms of bilirubin-induced neurotoxicity have been extensively studied but remain only partially understood. 13 Disruption of plasma, mitochondrial, and/or endoplasmic reticulum membranes of neurons is implicated in the initiation of neuronal cell injury. 13 The loss of membrane integrity serves as a catalyst for further cellular perturbations, including ongoing mitochondrial damage, calcium release into the cytoplasm, and excitotoxicity. Such processes likely occur with some degree of spatial and temporal overlap, with activation of apoptotic and necrotic pathways triggered by irreversible membrane damage and increased intracellular calcium. 2,13 Neuronal necrosis results in gross yellow discoloration of the brain, chiefly within susceptible nuclei, including the basal ganglia and deep cerebellar, thalamic, and subthalamic nuclei. Neurologic signs of bilirubin encephalopathy include opisthotonos, coma, and seizures. 9
The OF of erythrocytes is an index of the erythrocyte’s surface:volume ratio and ability to resist lysis in hypotonic solutions. 3,4,10,11 Increased OF is common with many forms of acquired membrane injury—such as immune-mediated hemolytic anemia, Heinz body anemia, and infection with hemoparasites—but in the cases where no cause was identified, a hereditary red cell defect has been suspected. 3,4,10,11 Chronic anemia with increased OF has only recently been described in domestic shorthair and purebred cats, particularly Abyssinian and Somali cats. 3,4,11 These cats frequently present with lethargy, pallor, inappetence or pica, and splenomegaly and typically chronic to intermittent anemia with variable regeneration. No specific cause has been identified; hence, an intrinsic membrane defect is suspected. PK deficiency has a very similar clinical picture to that of cats with primary OF; however, cats with PK deficiency typically have mean erythrocytic OF values within or near the reference range. 3,4
Chronic hemolytic anemia and cholangiohepatitis with severe cholestasis likely contributed to hyperbilirubinemia that progressed to bilirubin encephalopathy in this case. The relationship of chronic hemolytic anemia to hepatic disease and ultimate neurologic presentation is unclear. This case illustrates that bilirubin encephalopathy, a condition typically considered in neonates, can rarely occur in adult cats with severe unconjugated hyperbilirubinemia. It also indicates the need for clinicians to be aware of the differential diagnosis of idiopathic OF in young domestic shorthair cats with persistent or recurrent anemia. 3,11
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: Studies at the University of Pennsylvania were supported in part by NIH #OD 010939.
