Abstract
Outbreaks of chondrodysplasia in calves occur sporadically every 10–15 years, particularly following prolonged drought conditions, throughout Northeastern Victoria and the Southern Tablelands of New South Wales, Australia. An outbreak spanning 2 calving seasons (2003–2004) involving numerous losses through stillbirth, perinatal loss, and poor growth was investigated. Investigations of 4 representative cases are presented here with a definition of the gross and histopathologic defects and an overview of epidemiologic data gathered from affected farms. Calves showed variable disproportionate dwarfism without arthrogryposis. Long bones were shortened and showed axial rotation. Articular surfaces were distorted with misshapen weight-bearing surfaces associated with variable thickness of articular cartilage. Physes were distorted and variable in thickness with occasional foci of complete closure. The major histologic abnormality in the physes was disorderly development of the zones of cartilage hypertrophy, with reduced number and irregular arrangement of hypertrophic chondrocytes; similar less severe changes were present in the zones of cartilage proliferation. Histochemical staining of the cartilage matrix was variable in intensity, and there was evidence of abnormal resorption of cartilage matrix at the level of the primary spongiosa. Osteoid formation and subsequent bone remodelling seemed unaffected, and diaphyseal cortical bone appeared normal at the gross and light microscopic level. No infectious agents were identified, and other known causes for chondrodysplasia in calves were excluded. The most likely cause for the syndrome was considered to be congenital manganese deficiency. Further surveys of tissue and blood manganese levels from cows and calves with and without clinical signs from the region are planned.
Sporadic congenital deformities are expected in the bovine population. Outbreaks of higher incidence of congenital disease such as that described here may be associated with considerable losses of animals and consequent financial loss. Across Northeastern Victoria and the Southern Tablelands of New South Wales (NSW), Australia, outbreaks of chondrodysplastic disease have been reported, particularly in years following prolonged drought conditions. Outbreaks occurring in 2 consecutive spring calving seasons (July to November, 2003 and 2004) and involving numerous losses through stillbirth, perinatal loss, and poor growth were investigated to exclude potential infectious disease and to define the gross and histopathologic defects. The cause of the syndrome is still unresolved, although congenital manganese deficiency is suspected; despite marked improvement in the climatic conditions in this area, a lesser number of farms saw deformities among calves in a third consecutive calving season (2005). Investigations are ongoing.
Congenital disease may result from genetic abnormality, infectious disease, or other in utero insults. “Chondrodysplasia” is a general term for abnormalities of cartilage, usually caused by defective nutrition or metabolism of cartilage, which results in disproportionate dwarfism.19
Genetic chondrodysplastic syndromes are reported in Holstein, Dexter, and several other breeds of cattle. Beef breeds selected for short stocky phenotype have an increased prevalence of brachycephalic calves, with longitudinal compression of the vertebrae and shortening of the distal long bones, believed to be hypochondroplasia rather than a dysfunction of endochondral ossification.19
There are many examples of toxic and nutritional causes of musculoskeletal deformity. Hypervitaminosis A causes vertebral abnormalities and may be compounded by protein-energy malnutrition.5 “Crooked calves” are caused by ingestion of certain Lupinus sp. containing toxic concentrations of the quinolizidine alkaloid, anagyrine, by the dam, particularly between 40 and 70 days of gestation.23 This leads to arthrogryposis, spinal deformities, and disordered long bone growth, possibly due to reduced intrauterine motility either through tonic contraction of the uterus or suppressed motility and apparent sedation of the fetus,3 leading to a functional arthrogryposis without identifiable histologic or histochemical lesions in the bone or muscle.1 Many other plant-associated toxins are implicated in skeletal disease, for example teratogenic piperidine alkaloids in Astragalus sp., Oxytropis sp., and Nicotiana glauca, which may act in a similar manner.3 Fungal toxins have been presumptively associated with outbreaks of congenital spinal stenosis and disproportionate dwarfism, leading to myelomalacia and posterior paralysis in 3 outbreaks in Western Canada, where heavy growth of Penicillium sp. and Fusarium sp. in wet straw bales was also causally associated with pruritus and mortality in dams.20 “Acorn calves,” described in western regions of North America and also recorded in New Zealand and South Africa, were associated in the original reports with ingestion of acorns.2,19 Affected calves showed joint laxity, varus, and valgus deformities, and disproportionate dwarfism. More recent work has suggested that some of these cases may be consistent with a deficiency in dietary intake of manganese, as supported by small feeding trials in cattle.6,7,10,22
Infectious causes of skeletal deformities seen in cattle in Australia include bovine viral diarrhea virus (BVDV), Akabane and Aino viruses. These tend to cause arthrogrypotic deformity of the skeleton rather than chondrodysplasia.
Consideration and, to a large part, elimination of these differential diagnoses combined with evaluation of the anatomic pathology of several cases led investigators to propose that deficiency of manganese could be responsible for the abnormalities in this outbreak. Manganese deficiency was suspected, although not conclusively proven, in several previous field outbreaks of skeletal disease in calves.24,28
This article presents the gross and histopathologic features of 2 outbreaks of congenital disease in calves, defines the disease, and discusses the potential relationship with manganese deficiency.
Methods
Veterinary field staff and epidemiologists performed clinical investigations with visits to numerous affected farms and epidemiologic data collection through analysis of questionnaires completed by farmers. The case definition formulated among the investigative group was as follows: “Birth of abnormally short, stunted, or deformed calves with a spectrum of axial and appendicular skeletal defects predominantly of reduced long bone/diaphysis length, angular rotations, irregular growth plates, and articular cartilages, but without significant abnormality of cortical bone, born to dams without clinical signs in the area of Northeast Victoria and Southwest New South Wales.”
Portions of vertebrae and long bones from calf No. 1, a stillborn calf from a farm in Northern Victoria, were received fixed in 10% neutral buffered formalin (NBF) and examined grossly and histopathologically. Calf No. 2, one live 21-day-old calf from a second farm, and calves Nos. 3 and 4, entire stillborn calves from a third farm, underwent full postmortem and histologic examination using standard methods. Carcasses of calves Nos. 2–4 were weighed, and skeletal components of calves Nos. 3 and 4 were recorded using standard measuring tapes. Radiographic analysis of intact demuscled limb bones and sections through vertebrae were prepared using a band saw, after fixation but before decalcification, on calves Nos. 3 and 4 (not presented).
Five-millimeter sections were taken from a wide range of axial and appendicular skeletal sites and fixed in 10% NBF for 24–48 hours before decalcification, using a standard aqueous solution of 18% formic acid and 3.5% sodium formate for approximately 1 week at room temperature with continuous slow agitation. Following standard paraffin embedding, 4-μm sections were cut and stained with hematoxylin and eosin (HE). Histochemical stains used to evaluate cartilage and bone included Giemsa and Acid Giemsa, periodic acid–Schiff (PAS), Alcian blue, Alcian blue–PAS, toluidine blue, safranin O/fast green,15 and Verhoeff elastic stain.
For comparison of bone measurements and histopathology, 20 femora of grossly normal young calves randomly collected by staff at an abattoir in an unaffected region were weighed and measured, and histologic sections were prepared from the distal physis of each. Age and sex matching was not possible in this investigation.
Fresh frozen liver was collected from calves Nos. 3 and 4. Glutathione peroxidase was analyzed using an enzymatic method according to the Standing Committee on Agriculture, Commonwealth Scientific and Industrial Research Organ, Australia, 1993. Vitamin B12 was analyzed spectrophotometrically with competitive binding assay, and copper was analyzed by atomic absorption spectrophotometry. Manganese concentration was determined following wet combustion in perchloric/nitric acid solution and dilution in dilute hydrochloric acid to correct volume by flame atomic absorption spectrophotometry at 279.5 nm on an AAnalayst 300 Spectrometer (PerkinElmer, Waltham, MA). The reference ranges available for concentration of each of these elements are based on adult cattle, and no reference range for neonates was available at the start of the study.
Agar gel immunodiffusion was performed on serum samples from calves Nos. 2–4 and several dams and live calves from affected farms to detect BVDV and Akabane virus antigen. Virus neutralization was performed to detect Aino virus antigen. The National Arbovirus Monitoring Program of Animal Health Australia was consulted to assess presence of insect vectors and serologic evidence of arboviruses in the region during the periods of gestation (www.namp.com.au/reports/report_0203.pdf, 2005).
Results
Many farms across Northeastern Victoria experienced births of deformed calves during the spring (July to November) of 2003 and 2004, common calving periods for this region. Although some farms reported births of marginally increased numbers of mildly chondrodysplastic calves in preceding years, exceeding the expected number of sporadic cases, the outbreaks in 2003–2004 were widespread with significant portions (≤90%) of cohort groups affected. The spectrum of severity ranged from stillbirths and perinatal deaths (due to inability to stand and respiratory compromise) to calves deformed but able to suckle and follow the dam. Less-affected calves were vigorous and seemed to show some clinical reduction of deformities over time. Total reported losses due to high morbidity and mortality numbered into hundreds of calves; given economic conditions in this region that had suffered severe drought for several years, unreported losses were expected to approximate 1,000 calves.
Dams of all age groups produced deformed calves. Affected calves appeared to be clustered in management cohort groups rather than showing a pattern of spread expected with an infectious agent. The groups of dams giving birth to affected calves had predominantly been at pasture on hilly granitic country that had been extremely dry over the majority of each gestation period, although not all dams were in poor body condition. No toxic plants were found on repeated examination of the grazing areas or reported during careful questioning of farmers. Some farms had been blanketed in heavy bushfire smoke during the first gestation period (2003), but there did not appear to be an association between density of smoke and the affected cohort groups.
Common supplemental food, medication, or management procedures, and genetic links were not identified during clinical investigation and epidemiologic study. The calves affected were a range of beef breeds and crosses, including Murray Grey, Hereford, and Angus. Numerous sires were involved, and cows giving birth to deformed calves had previously produced normal calves and, in many cases, went on to produce normal calves in the following season. One mildly affected heifer calf was observed to produce a normal calf at her first calving.
Gross pathology
External examination of the calves revealed similar deformities in all calves presented intact. All had low body weight (Table 1) and short stature, with moderate-to-marked limb-trunk disproportion (Fig. 1). The heads appeared slightly domed in calves Nos. 2–4 (Fig. 1). There was variable brachygnathism, and calves Nos. 3 and 4 showed protrusion of the tongue with oversize relative to the oral cavity.

Carcass, calf No. 4. Disproportionate dwarfism, kyphosis, domed skull, and supinatory rotation of distal limbs are seen.
Selected individual and average measurements of skeletal elements of calves Nos. 2–4.



– = not available.
Limb deformities varied in severity, with the live 21-day-old calf (calf No. 2) showing a bilateral valgus deformity of the forelimbs, over-extension of the fetlocks, disproportionate shortening (length-to-epiphyseal diameter) predominately of proximal limb bones, and reduced range of movement of most joints. Calves Nos. 3 and 4, periparturient mortalities, showed more severe deformities with moderate-to-marked disproportionate shortening of proximal limb bones and supinatory rotation of the distal forelimbs and, to a lesser extent, the hind limbs (Fig. 2). This appeared to result from distortion of the diaphyseal shafts of the long bones rather than from abnormal direction of joint movement. The joints did not show significant laxity or fixation (arthrogryposis), and muscle mass appeared normal relative to the size of the calves.

Midsagittal section of forelimb; calf No. 4. Distortion of proximal limb bones and irregular growth plates are noted.
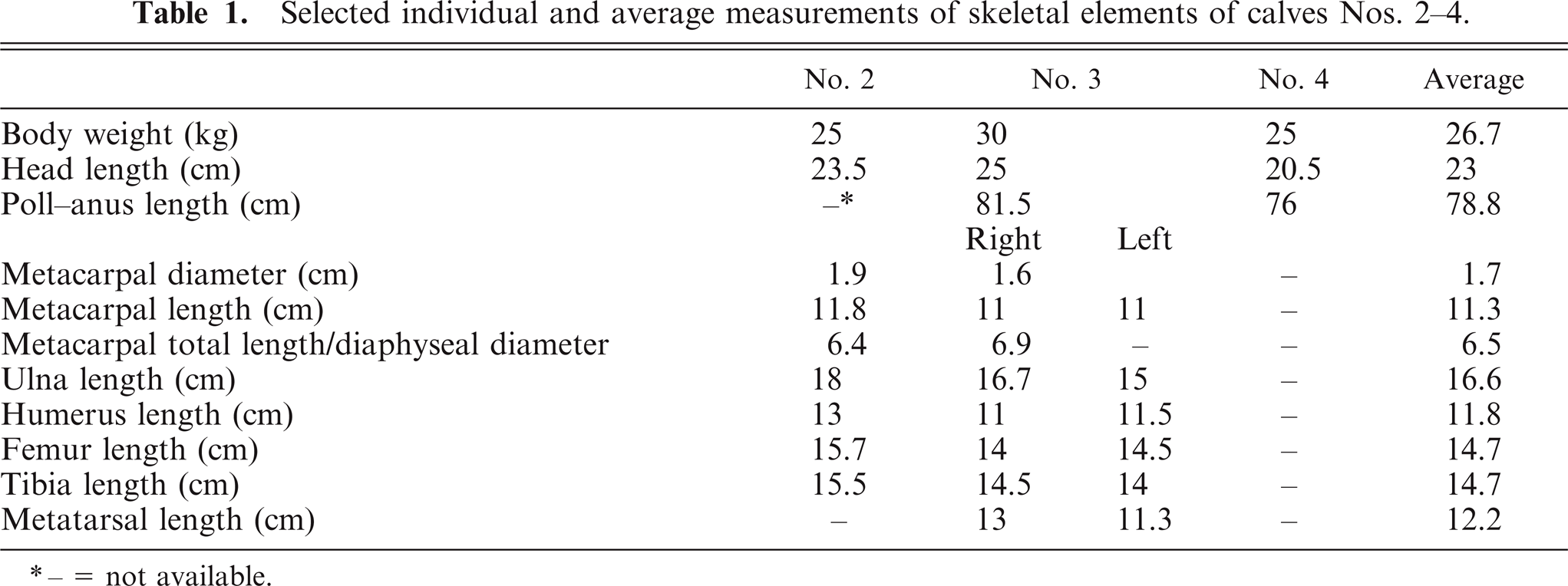
The vertebral columns showed variable kyphosis and lordosis. The atlas was irregularly distorted with increased concavity of the cranial and caudal articular surfaces. Growth plates of the vertebrae appeared distorted and showed variable thickness. Irregular interlacing trabeculae of cartilage extending between the cranial and caudal vertebral growth plates traversing the vertebral bodies were noted in all calves with variable frequency and extent along the vertebral column (Fig. 3). The significance of these changes is uncertain since the authors have not identified studies characterizing morphology of the bovine neonate spinal column. The shape of several of the vertebral bodies appeared distorted in calves Nos. 2–4, with cranial concavity and caudal convexity of articular surfaces and loss of the central waist normally seen in the vertebral body (Fig. 3). Compression of the spinal cord was not evident except in calf No. 1, where the spinal canal was virtually occluded in the single cervical vertebra (atlas) presented for examination. Changes similar to those of the vertebrae were also identified in basicranial bones; distortion of basicranial bones appeared to lead to shortening of the ventral cranial vault and thus skull doming. Calf No. 4 showed a depression fracture of the cranial vault, fracture and displacement of the basicranial bones with hemorrhage before death, considered consistent with trauma shortly after birth (e.g., trampling by the dam).
Midsagittal section of cervicothoracic vertebral column; calf No. 4. D-shaped distorted vertebral bodies are seen.
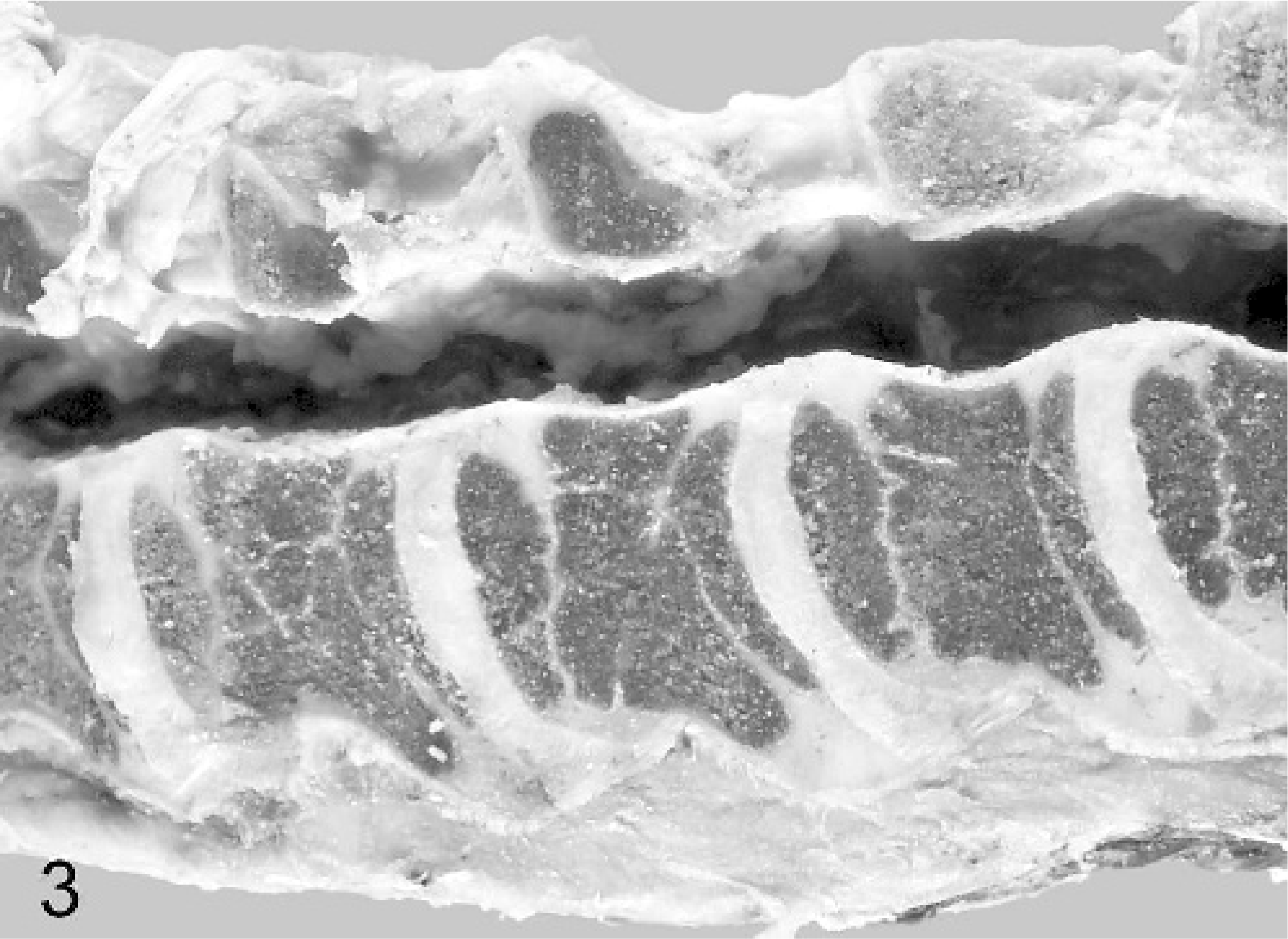
Examination of the limbs revealed longitudinal shortening of the long bones, most marked in the proximal limbs, with the distal limbs below the metacarpus and the metatarsus relatively less affected. The scapulae were normal in shape and proportionate to body size. On longitudinal section, numerous long bones revealed undulating irregularities of the physes. These were thin and irregularly wavy, with several stepped graduations in thickness across the growth plate (Figs. 4, 5). In calves Nos. 2–4, variably prominent growth-arrest lines were visible in the metaphyses and epiphyses (Fig. 5).
Midsagittal section of humerus; calf No. 3. Marked longitudinal shortening and growth-arrest lines in the metaphyses and proximal epiphysis (arrow) are depicted.
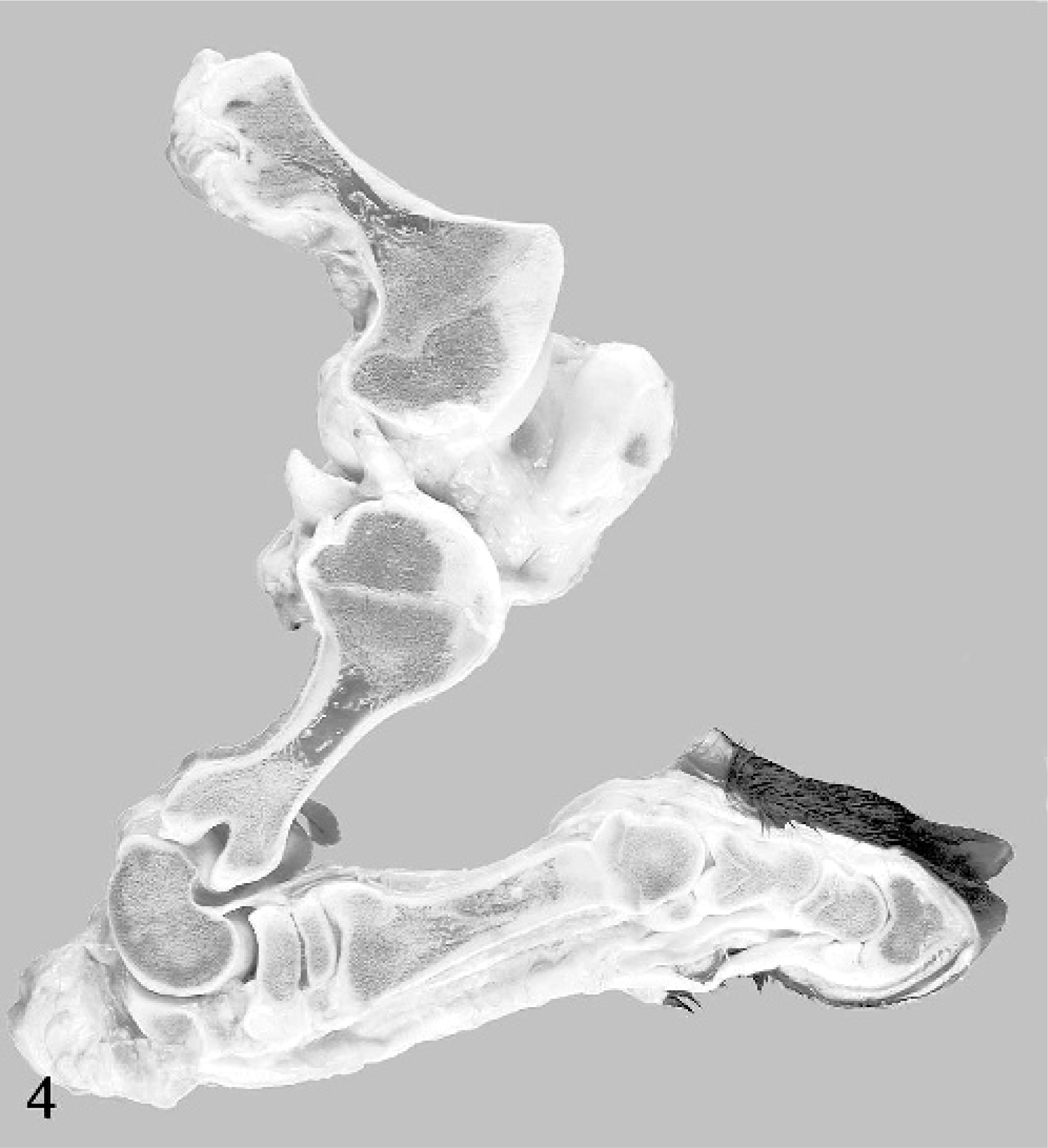
Midsagittal section of hindlimb; calf No. 4. Note prominent distortion with irregularity of growth plates of the femur and tibia.
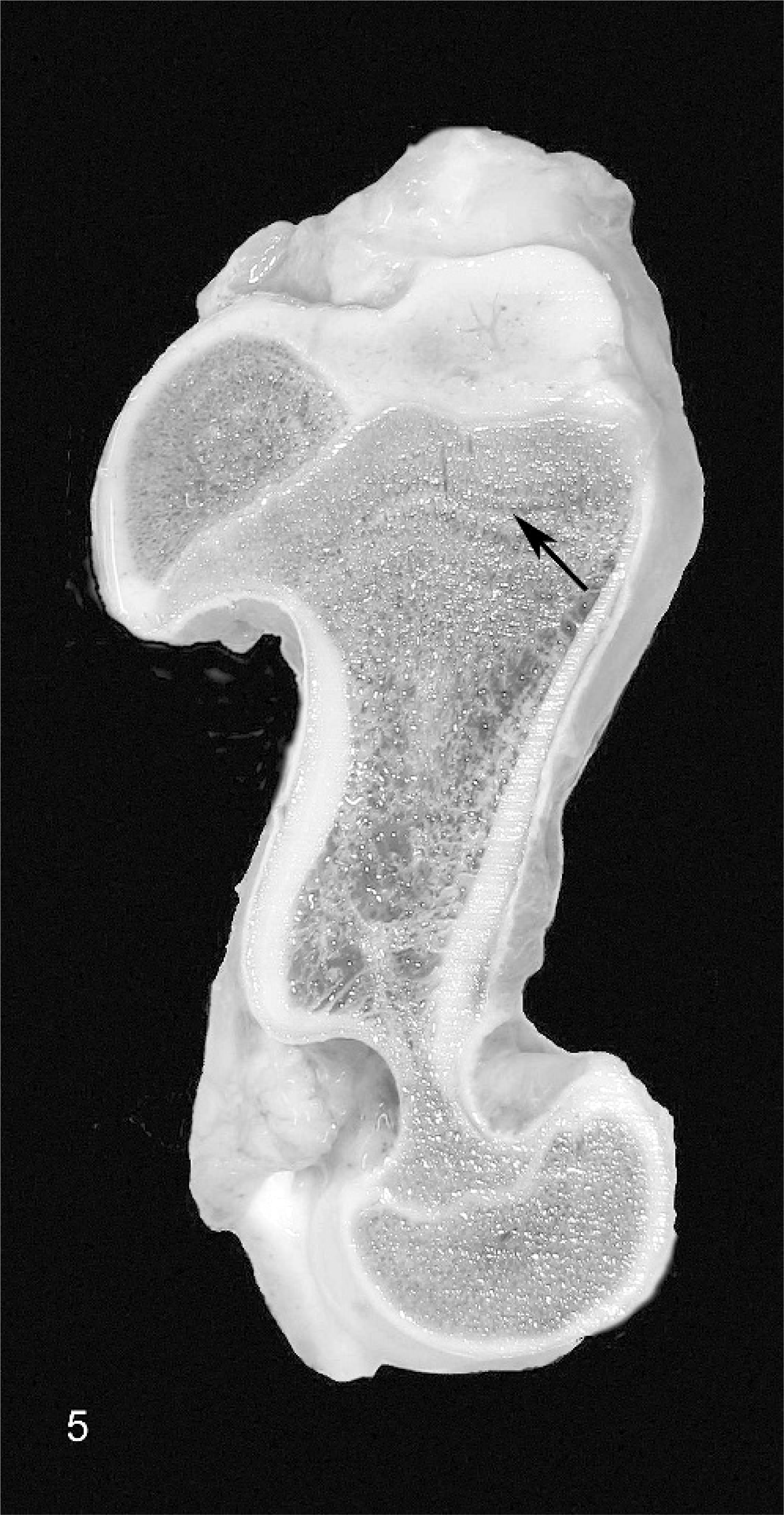
The humeri showed marked bilaterally symmetric diaphyseal shortening with mild outward (supinatory) rotation of the distal epiphysis. The elbows showed prominent lateral ridges and lateral displacement of the axial ridges of the radial articular surfaces, the radius and ulna were bowed, and the distal articulation was markedly irregular with numerous articular facets corresponding with the markedly curving distorted proximal carpal bones. All carpal bones were abnormal, misshapen, and difficult to classify (Fig. 6).
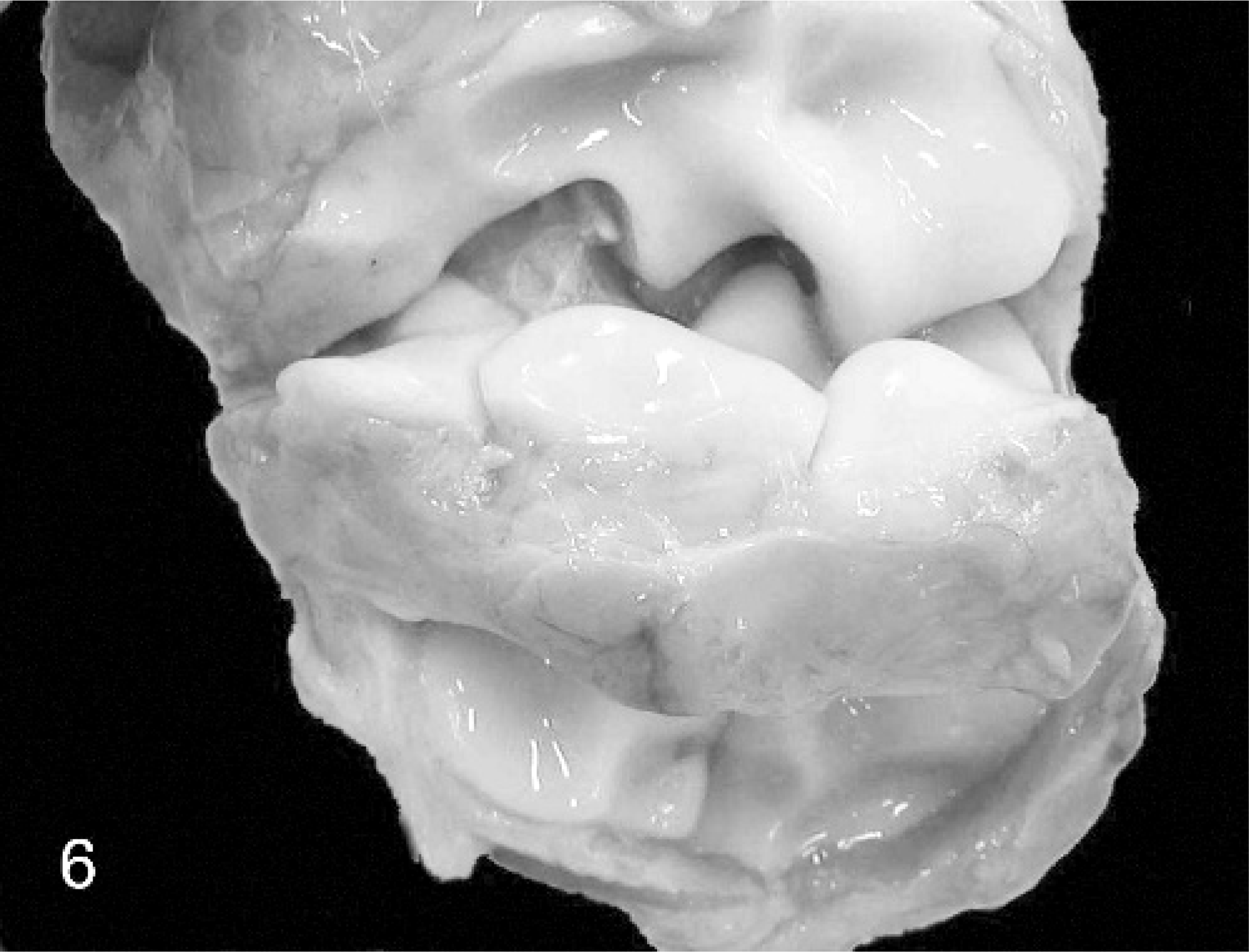
Carpal joint; calf No. 3. The carpus is opened to reveal markedly distorted and irregular articular facets leading to abnormally reduced range of movement without arthrogryposis.
Hind limbs were similarly deformed, with the femoral and tibial bones showing markedly decreased diaphyseal length and proportionally enlarged epiphyses similar to that of the forelimbs. The femora showed shortening and marked distal outward rotational deformity, with flattened trochlear ridges leading to laterally displaced patellae in the severely affected calf No. 2 and laterally placed patellae remaining within the trochlear groove in calf No. 4. The distal femoral articular surfaces were asymmetrically distorted. The tibial crest was spirally rotated from the proximal surface, with medial elevation of the tibial plateau. The diaphysis of the tibia was medially bowed, and the distal epiphysis was rotated inwardly (pronated), partially reversing the effect of femoral supination on the limb as a whole in calves Nos. 3 and 4.
The articular cartilages of all cases showed irregular thickness, with some areas of erosion and prominent subchondral vessels. There was variable multifocal lifting and separation of the articular cartilages in calf No. 2, with suppurative polyarthritis and osteomyelitis of the proximal tibia.
Only skeletal elements were examined in calf No. 1; abnormalities of nonskeletal elements were limited to hypoplasia and lateral flattening of the tracheal cartilage rings and extensive primary atelectasis, with only small areas of inflation of the lungs in calves Nos. 3 and 4.
Morphometric analysis
Measurements of skeletal components of calves Nos. 3 and 4 are presented (Table 1) and compared with measurements of the femora of young but not age-matched calves from an abattoir (Table 2). Previously measured skeletal elements of calves are compared (Table 3).
Average length of 20 femora sourced from normal calves at an abattoir in a nonaffected region of Victoria.
= from Mee 199518
= from Tyler et al. 196125
The Student's t-test was used to compare the mean length of the femora from affected animals (group A) with that of the femora from normal animals (group N). There was a difference between the mean length of the femora from group A (14.7 cm) and the mean length of femora from group N (20.6 cm) (P < .001). The assumption of independence of observations may not be satisfied because femora were randomly selected after boning and may represent femora from between 10 and 20 animals in the N group. Poll–anus length (81.5 cm) and eye–anus length (90 cm) for calf No. 3 (closest to the smallest body weight group in the referenced data) are less than those of average calves in this source (90.3 and 100.8 cm, respectively).18,25 However, variables including breed, sex and regional variations in average neonatal calf weight limit interpretation.
Histopathologic findings
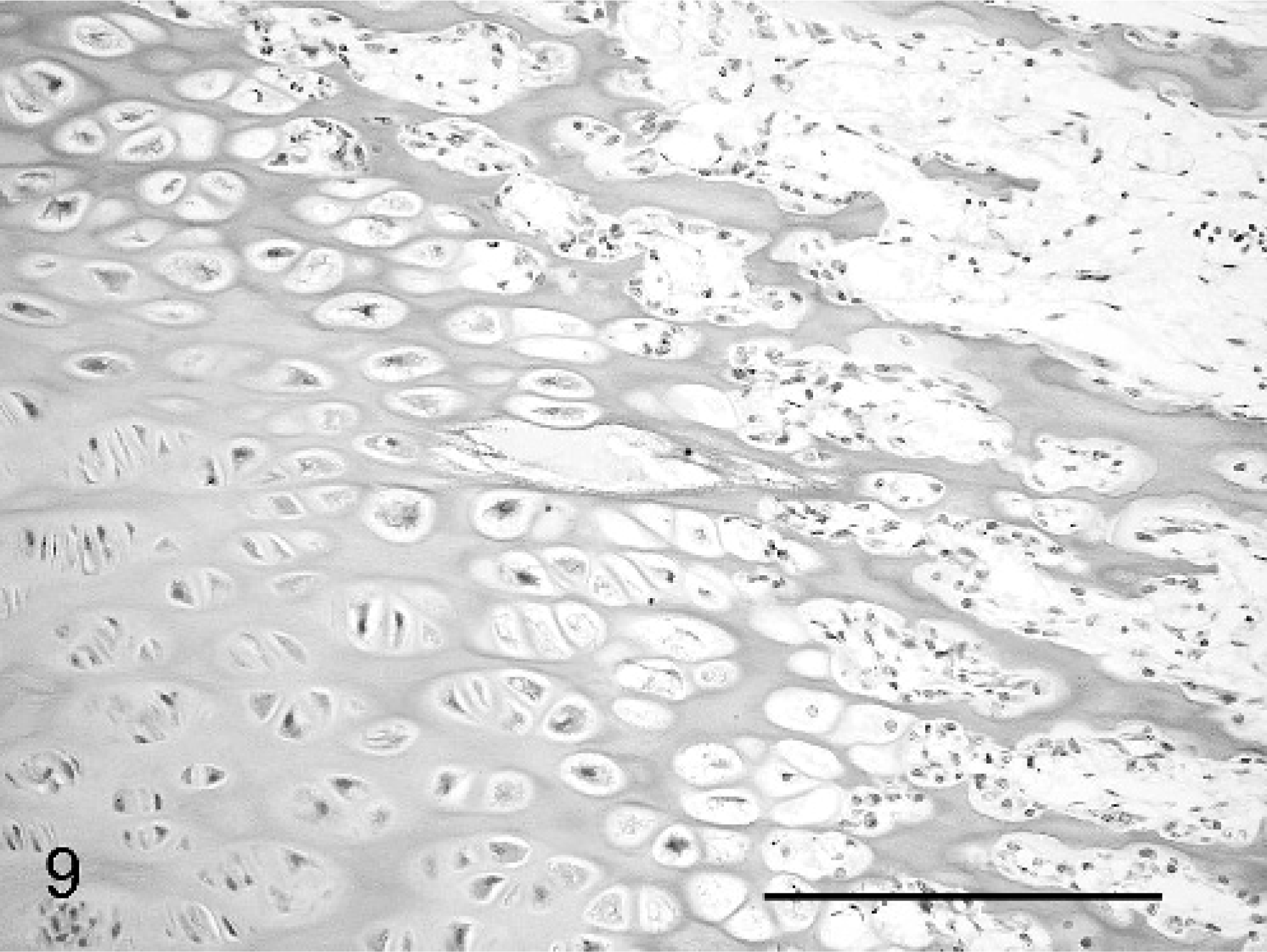
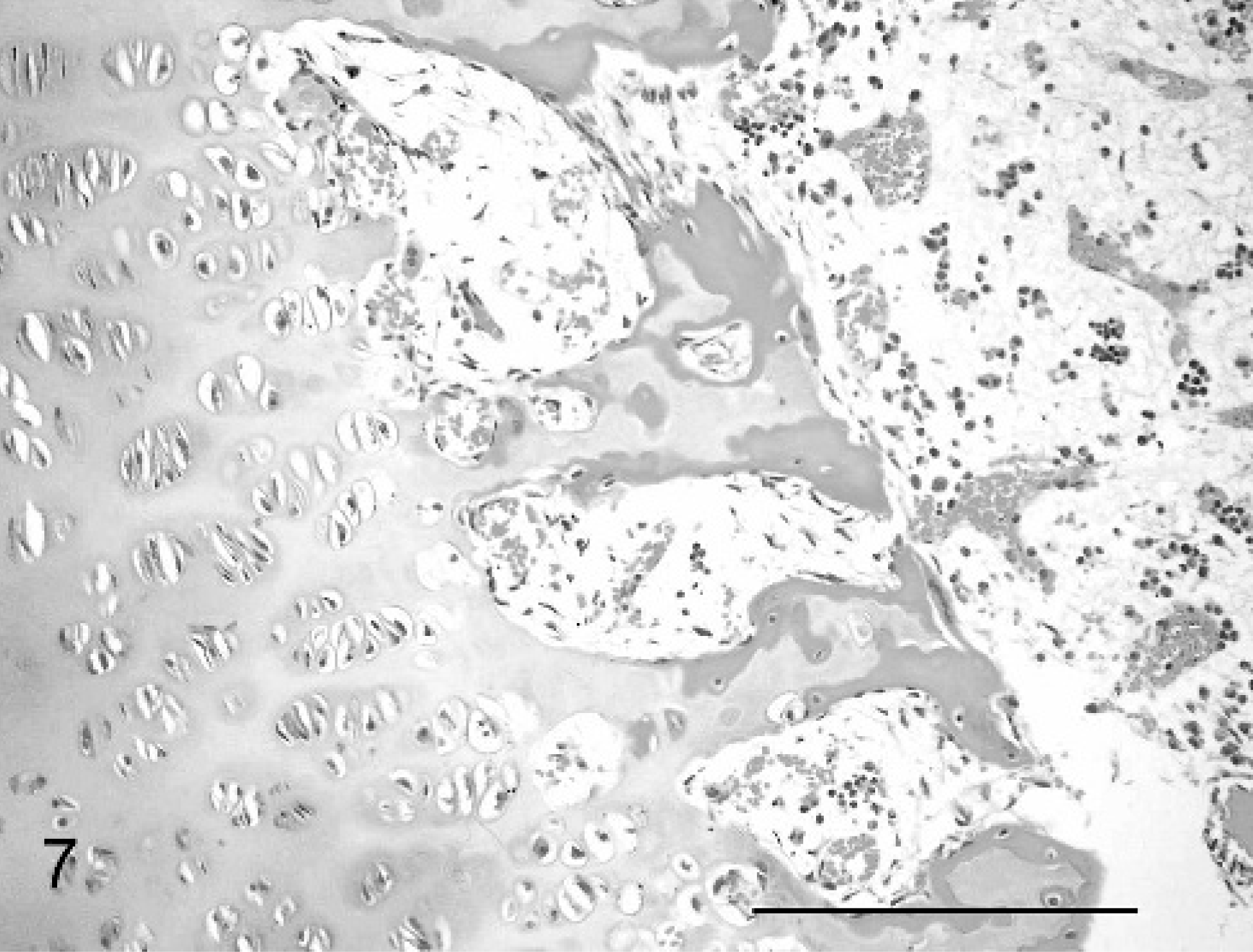
The cases all showed widespread physeal and metaphyseal abnormalities of varying degrees of severity. At the physis, the zone of cartilage hypertrophy was irregularly reduced, consisting of disorderly columns varying markedly in number and alignment along the growth plate, often with only 3–6 cells (Fig. 7), compared with neat columns of 10–20 cells in control animals (Fig. 8). The zones of resting and proliferating cartilage appeared relatively normal, although small multifocal nodules of increased numbers of proliferative chondrocytes were seen in each case. Multifocal cystic spaces were present both within the zone of hypertrophy and interspersed between the degenerate/calcifying chondrocytes of the zone of mineralization, suggestive of failure of formation of matrix or premature loss of chondrocytes and matrix. These cysts showed basophilic granular-to-fibrillar staining with toluidine blue and PAS, suggesting abnormal cartilage matrix filling the spaces (Fig. 9). A high rate of osteoclastic activity was indicated in these areas by the presence of abundant osteoclasts. Trabeculae within the primary spongiosa were short and irregular; a low proportion showed evidence of fracture close to the physis, with osteoclasts present at the blunt distal ends of trabeculae (Fig. 7).
Metaphyseal aspect of physis, femur; calf No. 2. Irregular mineralization of truncated spongiosa, cystic space filled with faintly granular mucinous material, and irregular granularity of chondrocyte cytoplasm are shown. Periodic acid–Schiff. Bar = 100 μm.
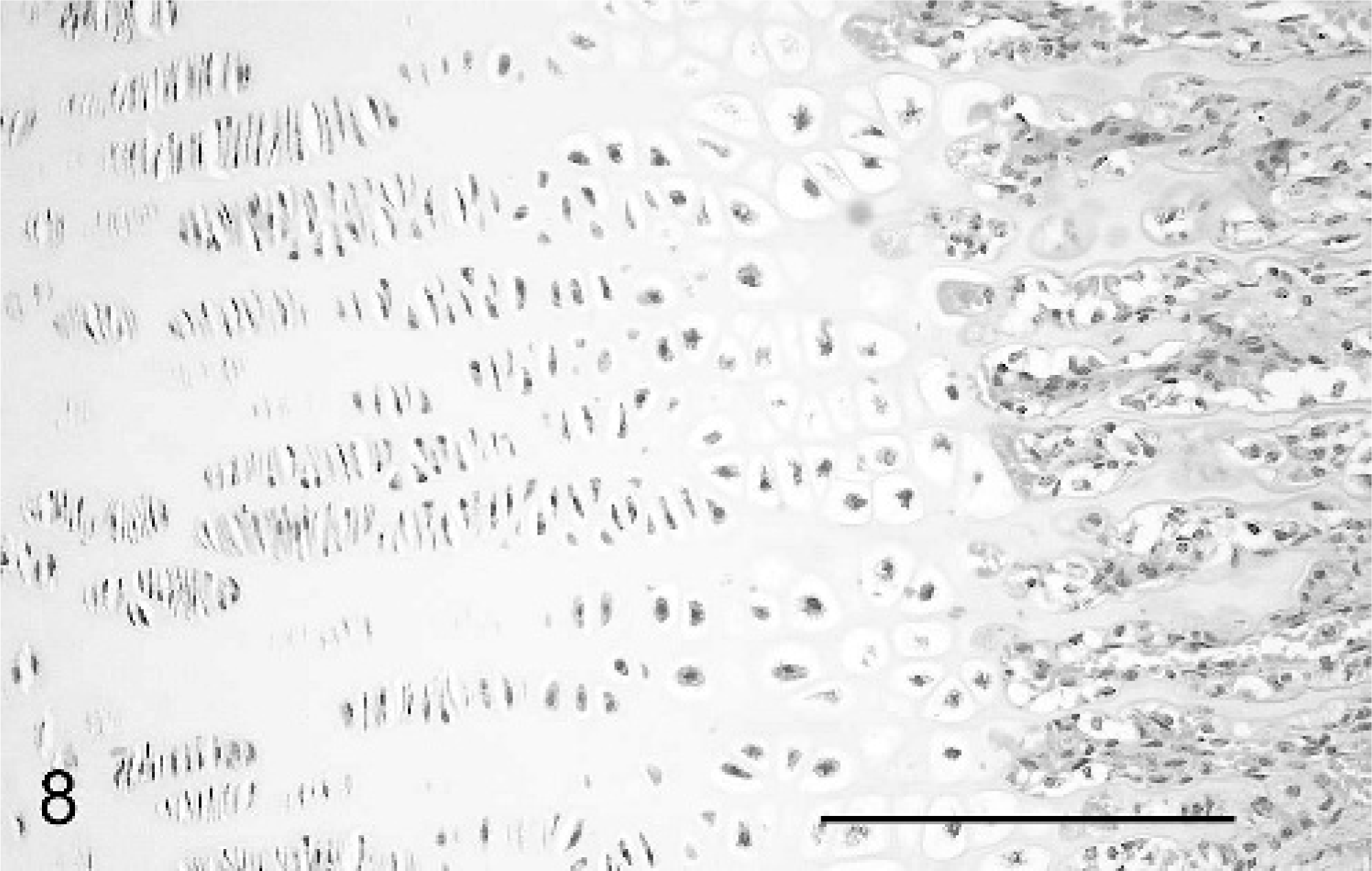
Metaphyseal aspect of physis, femur; neonatal calf control. Neatly aligned columns of hypertrophic chondrocytes and long primary spongiosa are compared with Fig. 7. HE. Bar = 100 μm.
Metaphyseal aspect of physis, femur; calf No. 2. Reduced number and irregular arrangement of hypertrophic chondrocytes, presence of osteoclasts at abnormally truncated primary spongiosa, and irregular resorption of cartilage are seen. HE. Bar = 100 μm.
The abnormalities of the cartilage matrix were most clearly demonstrated by variable granular Alcian blue staining, compared with that of controls (Figs. 10, 11).
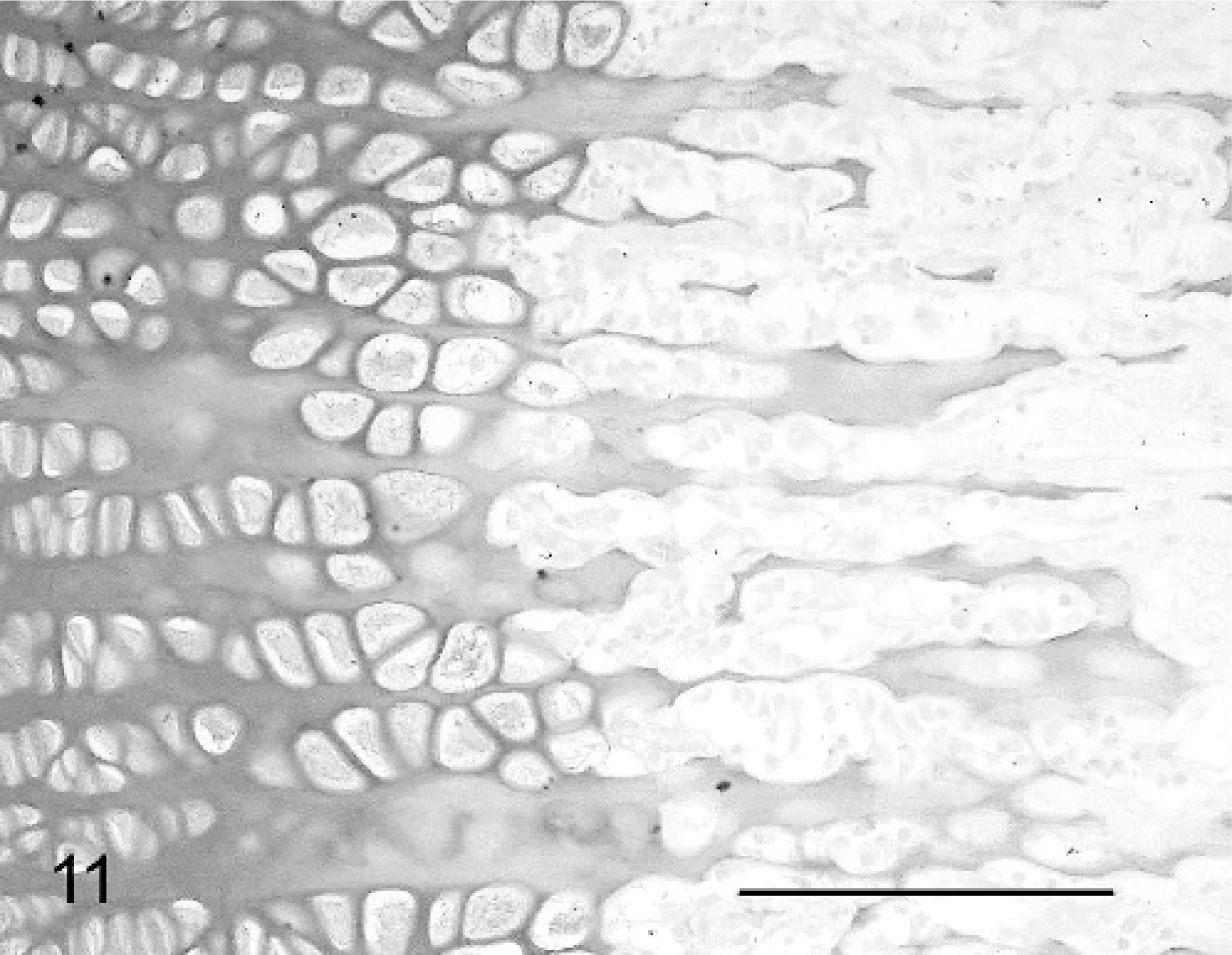
Physis, femur; neonatal calf control. Compare regularity of staining and physeal structure with Fig. 10. Alcian blue. Bar = 100 μm.
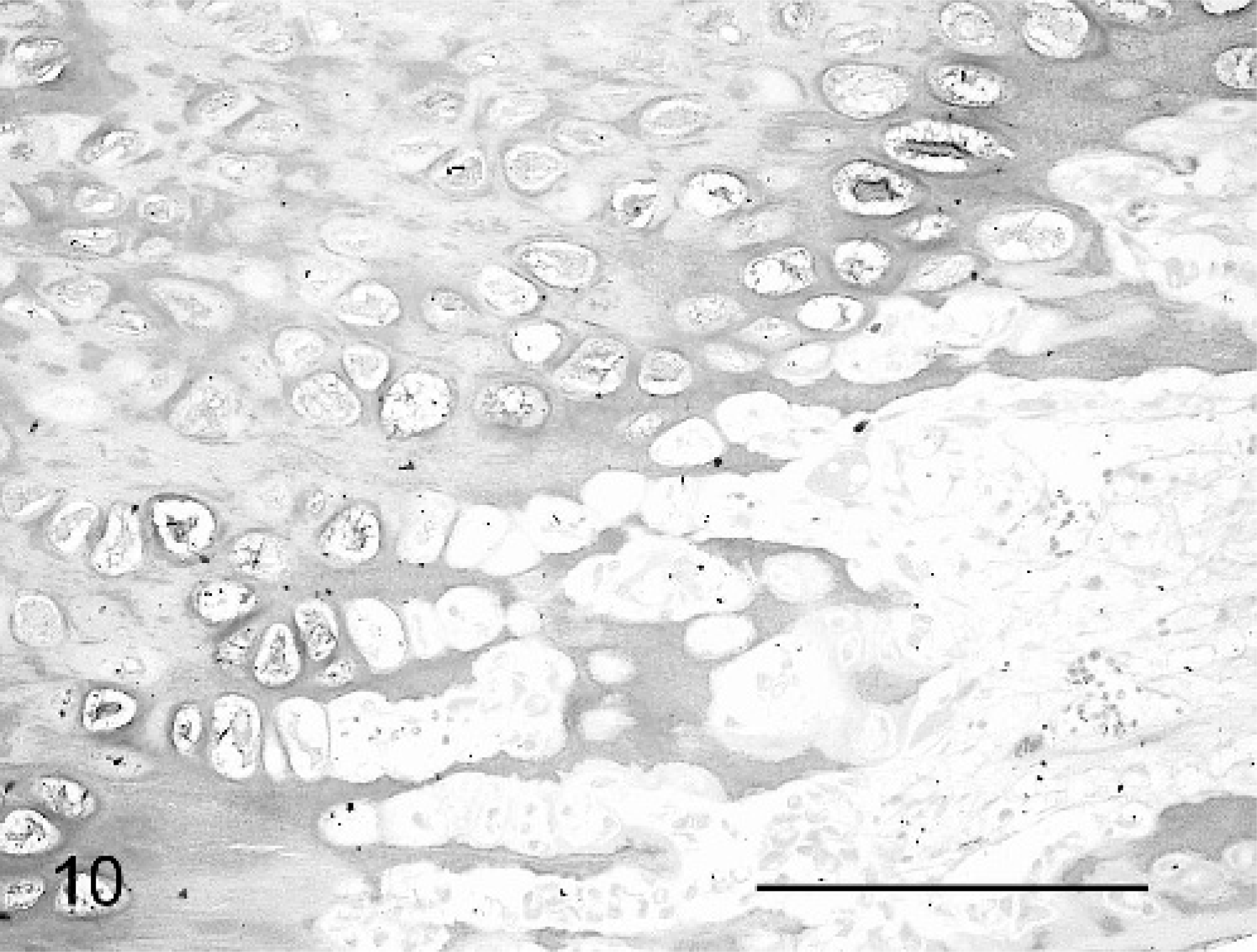
Physis, femur; calf No. 4. Variably dense and granular staining of physis is seen. Alcian blue. Bar = 100 μm.
The epiphyseal surface of the physis of the long bones examined in calf No. 1 showed development of a band of mature bone horizontal and adjacent to the physis (terminal plate), indicating closure of the growth plate (Fig. 12). The fused epiphyseal and articular cartilages were apparently less affected. Horizontal metaphyseal trabeculae of bone (growth-arrest lines suggesting periods of reduced dam nutritional status) were found running parallel to and at a short distance from the physes of some bones in all cases (Fig. 5).
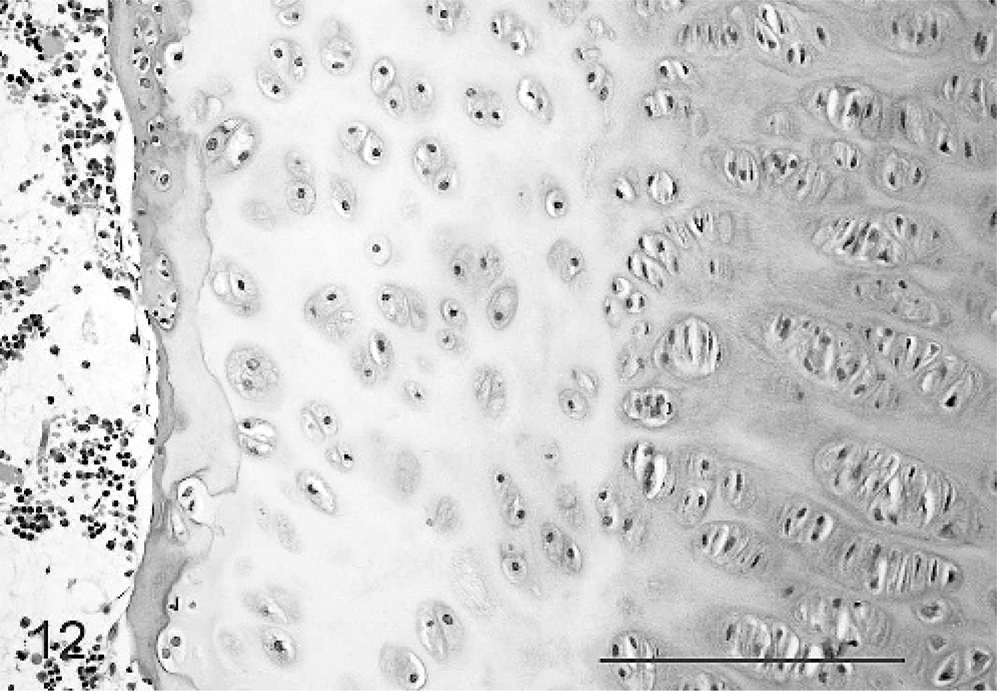
Epiphyseal aspect of physis, femur; calf No. 1. Formation of a terminal plate of osteoid indicates closure of the epiphyseal aspect of the physis. HE. Bar = 100 μm.
Sections of many long bones examined from all cases showed variable osteopenia, with a reduced quantity of trabecular bone in the epiphysis and medulla. Cortical lamellar bone was normal in histologic appearance as was the perichondrial collar, a lamellar plate of bone surrounding the physis.
Sections of vertebrae examined from each animal showed irregular outlines of the cranial and caudal physes due to variation in depth of both cranial and caudal aspects of the zones of hypertrophy and mineralization (Fig. 13).
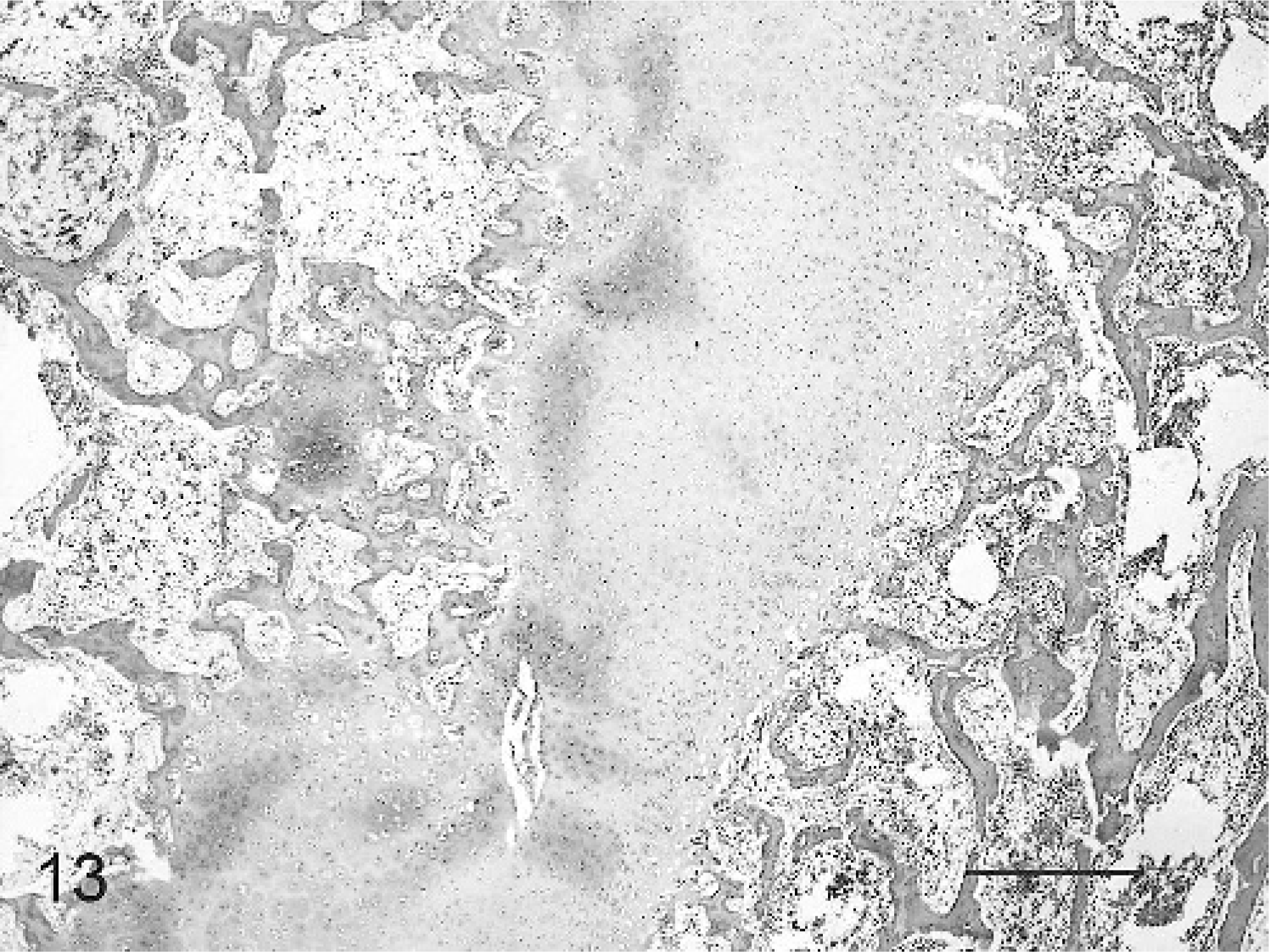
Vertebral end plate physis; calf No. 2. Irregularity of the physis and cartilage cores are present within the vertebral body. HE. Bar = 1,000 μm.
Sections of mandible demonstrated decreased medullary trabecular bone, but lamellar cortical bone was within normal limits. The bones lining the nasal cavity and turbinates were composed of fine irregular spicules of bone showing high cellularity, numerous irregular growth-arrest lines, and irregular scalloped surfaces lined with reduced osteoblast populations.
In calf No. 1, the atlas showed a pathologic in utero fracture of a focus of ectopic cartilage, disorderly proliferation of new bone, and cartilage with altered staining characteristics and increased cellularity and proliferation of the periosteum. This caused distortion and compression of the spinal canal and cord.
The proximal tibial physis of calf No. 2 had bacterial osteomyelitis. A necrotic cartilage sequestrum was surrounded by clusters of degenerate neutrophils grouped around colonies of small coccobacilli and associated with dense infiltrates of lymphocytes, plasma cells, and macrophages and a thick rim of fibroblastic connective tissue bridging the physis, thus causing pathologic fracture. In this case, lymph nodes taken from mesenteric and prescapular regions all showed a mild hypertrophy of follicles, consistent with excessive immune stimulation; however, viscera and neurologic tissues showed no significant changes. Microbial cultures were not performed.
Mineral analysis
In calves Nos. 3 and 4, portions of fresh frozen liver were submitted for mineral analysis (Table 4). The manganese concentrations detected in these unsuckled neonates were below the normal reference range; however, since this range was determined for adult cattle, further analysis is required to assess the range for neonatal calves before reliable interpretation can be made.
Liver manganese, copper, cobalt, and selenium analysis from calves Nos. 3 and 4.
μmol/kg wet weight.
mmol/kg wet weight.
U/g wet weight.
Virology
Serologic analysis for BVDV, Akabane and Aino viruses performed on these calves was negative as was that from numerous other animals from herds affected during the outbreak (data not presented). The National Arbovirus Monitoring Program did not report any seropositive animal or insect vector detection in the affected regions during the gestation periods.
Discussion
These cases showed sufficient gross and histologic similarity and distinction from other reported chondrodysplastic syndromes to constitute a recognizable clinical and pathologic entity. Disorder of the zone of cartilage hypertrophy with abnormal growth and resorption of cartilage, particularly at the physes of long bones and vertebrae, the basicranial and turbinate bones, but excluding facial and mandibular bones, was observed. Gross and histologic features suggest a chondrodysplastic disorder confined to bones of mesodermal and sclerotomal origin and not affecting bones derived from the neural crest. The presence of abnormalities of the physis not affecting the perichondrial collar and diaphyseal shaft suggests the disorder is limited to bone formed through interstitial growth from cartilage and by endochondral ossification but not by appositional growth, consistent with chondrodysplasia.
The common causes of chondrodysplasia including genetic defects, most toxic and nutritional causes, and infectious agents were excluded sequentially by epidemiologic and pathologic investigation.
In the affected herds in this outbreak, no familial link could be established, and the range of breeds and crosses affected was felt to preclude any possibility of a genetic etiology.
Vitamin A supplementation was rarely performed; therefore, hypervitaminosis A was excluded. Many farms with diverse management practices were affected; therefore, disease related to management procedures or therapeutic product administration was considered unlikely.
Pastures had been bare for much of the gestation period in these outbreaks. While there was no known access to any toxic plants, under conditions of undernutrition, cattle will graze plants normally rejected as unpalatable. The presence of toxic plants during the lengthy period of development and growth of bone and cartilage (from 40 days onward), and not during investigation several months later, cannot be excluded. The skeletal changes within individual affected calves and the range of severity seen among cohort groups suggest a prolonged period of exposure extending throughout the period of skeletal mineralization and growth, increasing the likelihood of detection of toxic plants. In this region of Australia, lupins are not common, and oak trees are rare; aflatoxicosis was unlikely since there was little rain and silage feeding was uncommon. No epidemiologic association could be made between bushfire smoke intensity and the congenital abnormalities.
Infection with teratogenic viruses was unlikely since serology was negative and the lesions were dissimilar to those expected with infectious deformities. Viral culture and immunoglobulin G analysis was performed in other cases without evidence of viral disease or in utero immune challenge (data not presented). Viral agents known to cause congenital defects (e.g., BVDV, Akabane and Aino viruses) tend to cause neurologic defects and arthrogryposis rather than deformity at the growth plate. No histologic lesions of the nervous system were detected. Nationwide surveillance programs showed that insect vectors responsible for transmission of arboviral disease were excluded from these areas throughout the period of gestation; thus, Akabane and Aino viruses were considered unlikely.
Anecdotal and unpublished data on previous Australian outbreaks similar to this case have suggested a possible association with long periods of drought, deformed calves tending to present following prolonged dry conditions or in the seasons following early onset of more normal rain patterns, particularly in the southeastern regions of NSW. Extensive investigations have always failed to ascertain a definite cause (A. Philbey, T. Ross, personal communication).
Sequential elimination of reported causes of deformities in this investigation led the authors to suspect involvement of manganese deficiency in the pathogenesis. Histopathologic descriptions of experimental manganese deficiency–induced deformities have been sparse to date, although the gross pathology described is similar to that in affected cases presented here. In a field investigation of suspected manganese deficiency, narrowed irregular growth plates with reduced zone of cartilage hypertrophy were described in three 3-month-old calves, but the authors did not describe histologic changes in neonates.24 Growth-plate irregularity with reduced and disorderly proliferating chondrocytes and absence of hypertrophic chondrocytes, with residual necrotic remnants among fibrillar matrix, was described in another field outbreak putatively associated with manganese deficiency.28 Respiratory difficulty has been a common factor in this and in other investigations18,24 and may represent abnormalities of the tracheal cartilage rings leading to reduced luminal diameter, distorted nasal passages, or a combination of these and other factors. Calves not so severely affected as to die in the perinatal period in outbreaks of postulated manganese deficiency have reportedly shown limited-to-good clinical improvement with time, if assistance is given to feed and stand, with reduction in deformation but residual stunting, as noted in this outbreak.28
Evaluation of manganese concentration
Normal reference ranges for manganese concentration are not available in neonates and work to formulate these is still in progress, hampered by the difficulty in obtaining fresh tissues from normal calves in the affected areas. This has limited the interpretation of manganese concentrations in affected calves.
Previous soil and pasture mineral analyses suffer similar difficulty in interpretation of results due to regional variation in soil composition and numerous other compounding factors in manganese absorption (e.g., variable manganese uptake by plants, combination of pasture species, and interference from other elements).
Our review of the literature suggests liver concentration of manganese is most appropriate for analysis in these neonatal calves, although the element can be routinely detected in bone ash, blood, and hair samples.8,9,14 Liver samples are easily obtained by farmers or veterinarians. They are simple to process, and liver concentrations of manganese are relatively high compared with manganese concentrations in other tissues, which may allow more accurate analysis. However, some authors suggest that liver manganese concentration is less well correlated to deficiency states than manganese concentrations in other tissues, including plasma and heart muscle, at least in growing animals.26 Reports of alteration in hair content of manganese with environmental factors unrelated to total body concentration and with hair pigmentation have been identified, so this simple test was not used.9
Manganese in skeletal disease
Manganese is an essential trace element involved as a cofactor in several critical enzymatic pathways of glycosaminoglycan and collagen synthesis and as a component of metalloenzymes, including superoxide dismutase (SOD), among other functions. Deficiency of SOD may be implicated in damage to membranes of mitochondria, endoplasmic reticulum, and golgi bodies and thus lead to loss of integrity, potentially leading to inability to synthesize mucopolysaccharides.14 Manganese is also essential in incorporating carbohydrates into mucopolysaccharides at the epiphyseal plate through manganese-dependant activation of some glycosyltransferases, which transport trisaccharides to link polysaccharide to protein in bone.8,16 Unlike many other metal-activated enzymes, similar metals such as magnesium or iron are less effective than manganese at activation of these particular enzymes.27
The literature on manganese metabolism and deficiency has been thoroughly reviewed.9,14,26,27 Deficiency of manganese has been associated experimentally and clinically with reduced reproductive performance7,22 and skeletal abnormalities7,10,22,24,28 in calves and laboratory animals and with ataxia associated with abnormal development of the otoliths in laboratory rodents but not cattle.13
Manganese bioavailability
Concentrations of manganese in plants vary. Grasses are richer than legumes, and straw and corn silage have particularly low concentrations.9 Uptake of bivalent manganese by plants is more efficient than that of the trivalent or tetravalent forms present in highest concentrations in soil. Low pH soils have a relatively high proportion of bivalent manganese.
Discussion with nutritionists in this investigation led to a suggestion that rumen pH may have a similar effect on bioavailability of manganese. At the time of writing, investigations were being undertaken to review the current knowledge of this interaction and determine whether manipulation of rumen pH rather than direct supplementation of manganese may aid in improving manganese absorption (T. Parker, personal communication).
Overall bioavailability from ingested plant matter is low, with <1% intestinal absorption in cattle. It appears from some studies that high dietary levels of iron, copper, zinc, sulphur, and, to a lesser extent, calcium and phosphorus lead to further reduction in manganese bioavailability.8,9 Although the soil in the affected area is high in granite, which usually has a high level of manganese, the bioavailability of the manganese may be reduced by high dietary intake of other elements in soil such as copper, cobalt, calcium, phosphorus, and iron, which show significant interference with manganese.26 Under severe drought conditions, cows may ingest a high volume of soil and dust, leading to altered dietary mineral balance.
Manganese has been shown in rats and sheep to be preferentially sequestered by the placenta in conditions of manganese deficiency of the dam, but this sequestration is insufficient to return the fetal manganese concentration to normal.11 Unlike neonates, fetuses are not able to preferentially concentrate manganese in the liver.14 It is believed that manganese concentrations in the liver of neonatal presuckled calves represent the manganese status of the dam.26
Retention of absorbed manganese varies with age; calves may apparently alter the percentage of dietary manganese retained after intestinal absorption according to the concentration of the element in the food and in response to deficiency.12 Manganese concentration in tissues has been shown to change rapidly after birth and onset of suckling in calves if there is high manganese concentration in the diet, as indicated by radioactive isotope studies of manganese metabolism.4 Young calves are able to rapidly incorporate supplemental manganese from oral and intravenous administration into tissues, and the percentage retained varies according to the dietary and whole body concentrations of the element at the time of administration of manganese.4 Once suckling commences, it is probable that the manganese concentration in liver rises rapidly and more markedly than in other tissues. This is attributed to the liver's role in manganese homeostasis through storage in mitochondria and excretion in bile. Higher manganese concentrations were found more often in suckled affected calves of a few days old (data not presented) than in neonatal unsuckled affected calves in this outbreak. Results suggested that within a few days, the liver concentration of manganese in affected calves approached normal levels despite the low concentration of manganese found in milk.
The percentage of dietary manganese absorbed in adults is relatively constant. Manganese is largely removed from first-pass portal circulation by the liver, and peripheral blood has a very low concentration of manganese bound to α-2-macroglobulin.
The apparent difference in manganese absorption may reflect the incorporation of the majority of dietary manganese into ruminal micro-organisms in adults, as evidenced by more efficient cellulose digestion by micro-organisms in the presence of supplemental manganese.17 The reason adult cows are unable to enhance retention of manganese if deficient is unclear.
Feeding of silage alone through winter in northern latitudes has been identified as a risk factor for a condition known as congenital joint laxity and dwarfism, which has some similarities to experimental manganese deficiency.21 It is postulated by some authors that this may relate in part to manganese deficiency indirectly through reduced bioavailability of manganese present in silage.10
In this outbreak, drought conditions may have led to rumen content with a high percentage of dry matter and thus a relatively high pH. Under these conditions, it is postulated that manganese is present within the rumen mostly as poorly bioavailable forms and thus absorption may be reduced. One potential avenue of investigation at present is analysis of the effects of rumen pH on manganese absorption; alterations of rumen pH may be more readily achieved through the use of supplemental grain feeding or dietary cation-anion–deficit rations without the need to supplement manganese directly.
Conclusion
The outbreak was characterized by disproportionate dwarfism, with reduced long bone/diaphysis length secondary to abnormality of the hyaline cartilage of physes and epiphyses evident on gross and histologic examination. Changes affected all bones formed by endochondral ossification; thus, the vertebral column was shortened and the skull domed due to reduced basicranial bone length. Tracheal rings were distorted, indicating a generalized abnormality of hyaline cartilage.
Manganese deficiency remains the main postulated cause at the time of writing. Confirmation of manganese deficiency in calves is difficult and time-consuming. Further studies are ongoing, and veterinary nutritional specialists are assessing advice to be given to farmers in the area. Ideally, supplementation will be planned to provide a controlled experiment of the effect of dietary manganese on the birth of deformed calves in this region, provided sufficient resources are available to gather the epidemiologic data and for manganese concentration analysis and other ongoing exclusions. One review has recommended the addition of 0.5 g/day per head of manganese as sulphate, the most soluble form, or as oxide, carbonate, or methionine salts to cattle.8 Avoidance of management procedures that increase the pH of soil such as liming may also aid in disease prevention.26
Footnotes
Acknowledgements
We thank Professor K. V. F. Jubb for his support and advice regarding pathologic investigations of submitted cases and Associate Professor John Glastonbury for critical review of the manuscript. We acknowledge Josie Wilson, Dennis Miller, Paul Benham, Garry Anderson, and Dayle Tyrrell of the Faculty of Veterinary Science, the University of Melbourne, for assistance with preparation of histology sections, postmortem examinations, photography, statistical analysis, and radiologic interpretation, respectively. The many Department of Primary Industries Victoria field staff, Stephen J. Whittaker of the Rural Lands Protection Board, and Alex Stephens and Luzia Rast of NSW Department of Primary Industries in particular must be acknowledged for their field investigation work. Tony Parker is acknowledged for his investigations into the effect of rumen pH on bioavailability of manganese, and his assistance in planning therapeutic strategies. Staff at Primary Industries Research Victoria for further pathologic investigations, including mineral analysis and virology studies; Gribbles Veterinary Pathology for serological and mineral analysis; and Regional Laboratory Services Benalla for mineral analysis are gratefully acknowledged.
