Abstract
Four causative mutations (D1, D2, D3*, and D4) of chondrodysplastic dwarfism have been described in the equine aggrecan (ACAN) gene. Homozygotes for one of these mutations and heterozygotes for any combination of these mutations exhibit the disproportionate dwarfism phenotype. However, no case description of homozygotes for D4 (D4/D4) has been reported in the literature, to our knowledge. We report 2 Miniature horses with the genotype D4/D4 in the ACAN gene. Clinically, the 2 dwarfs had a domed head that was large compared to the rest of the body, mandibular prognathism, and short and bowed limbs, mainly in the proximal region of the metatarsal bones. Radiographic examination revealed contour irregularities of the subchondral bone in the long bones and confirmed mandibular prognathism; histopathology revealed irregular chondrocyte organization. To determine the genotypes of the horses, we performed DNA extraction from white blood cells, PCR, and Sanger sequencing. Genotyping demonstrated that these 2 animals had the D4/D4 genotype in the ACAN gene. The D4/D4 dwarfs were clinically similar to animals with the other ACAN genotypes reported for this disease. Identification of heterozygous animals makes mating selection possible and is the most important control measure to minimize economic losses and casualties.
Dwarfism, proportionate or disproportionate, is a form of abnormal development that is determined by genes contributing to small stature and may negatively interfere with the health and reproduction of the affected individual. 3 Aggrecan, encoded by the aggrecan (ACAN) gene, is a large proteoglycan that is essential for the proper functioning of articular cartilage. 8 Therefore, mutations in the ACAN gene are responsible for abnormalities in skeletal development in different species.1,2,4,5,7,12 In Miniature horses, dwarfism is autosomal recessive, and 4 independently segregating mutations (D1, D2, D3*, and D4) in the ACAN gene have been described as causative of chondrodysplastic dwarfism in Miniature horses.4,9 Genotypes involving D1 mutations cause fetal death, except in a single case, in which a full-term live dwarf was heterozygous for D1 and D4 (D1/D4). 4 In contrast, the other genotypes (D2/D2, D2/D3*, D2/D4, D3*/D3*, D3*/D4, and D4/D4) are theoretically expected to be involved in the birth of dwarf foals. 4 However, animals with the D4/D4 genotype have not been described in the literature, to our knowledge. We describe herein 2 Miniature horses with the D4/D4 genotype in the ACAN gene.
A Miniature horse neonatal foal with clinical signs of dwarfism was admitted alive to the Large Animal Internal Medicine Service of São Paulo State University (Unesp; Brazil) during the 2016/2017 foaling season, and a second foal was admitted during the 2017/2018 foaling season. The female (2016/2017) and the male (2017/2018) were both 1 d old and were from 2 different farms. All of the parents were normal in appearance, and mating between the same parents previously produced no dwarf births. In addition, no inbreeding was found between the parents. Blood samples were collected from the parents and affected animals for molecular analysis.
We performed our study in accordance with the policies of the Institutional Animal Care and Use Committee (0219/2016-CEUA) of Unesp. Epidemiologic, clinical, radiologic, and pathology investigations and ACAN genotyping tests were conducted on the 2 Miniature horses with clinical signs of dwarfism and 1 Miniature 1-d-old foal without clinical signs of this disease (control foal), who died as a result of complications of dystocia. The parents of the affected foals were also subjected to ACAN genotyping tests. In addition, sequencing of ACAN messenger RNA (mRNA) from these 2 affected animals was performed using primer pairs that amplified the exons of the ACAN gene, and sequencing of these regions in the parents was also attempted.
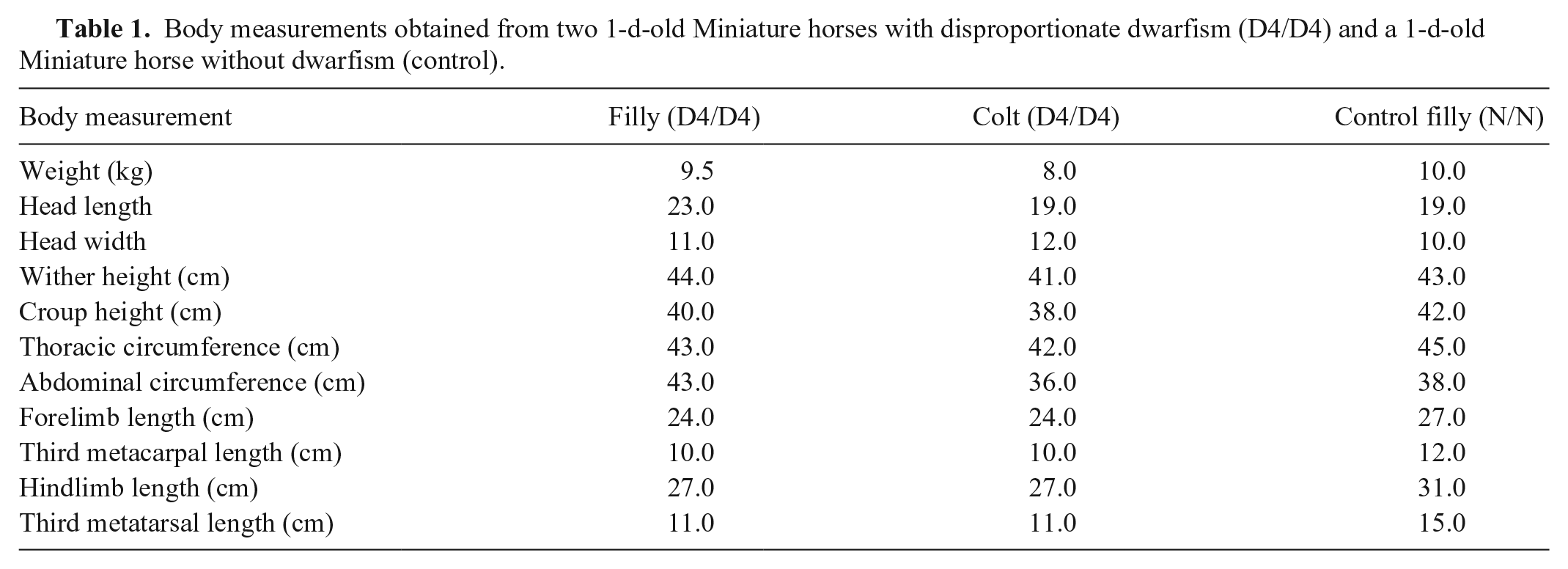
During the physical examination, the following body measurements were recorded for the 2 dwarf foals and the control foal: head length (distance from the base of the ear to the lips); head width (distance between the most lateral part of the eyes); height at the withers (vertical distance from the highest point of the thoracic vertebrae to the ground); height at the croup (vertical distance from the highest point of the croup to the ground); thoracic and abdominal circumferences; forelimb length (distance from the humeroradial joint to the metacarpophalangeal joint); hindlimb length (distance from the femorotibial joint to the metatarsophalangeal joint); and cannon (fore and hind) length. The limbs (tibia/fibula and radius/ulna) and head were radiographed. Upper airway endoscopy was also performed in one affected foal.
Unfortunately, the filly died during the first minutes of medical care because of complications of dwarfism; she could not follow her dam to nurse to maintain glucose concentrations and properly hydrate or maintain body temperature until being admitted to the hospital. The colt was euthanized on the second day of life, and the 2 foals were autopsied. Portions of all organs were collected at autopsy on the first and second day of life of the filly and the colt, respectively, and fixed in 10% neutral-buffered formalin for histopathology or frozen for genetic tests. For histologic examination, fixed tissues were routinely processed, stained with hematoxylin and eosin, and examined by light microscopy. The control foal was also subjected to histologic evaluation.
Genomic DNA was extracted from blood samples with a commercial kit (illustra blood genomicPrep mini spin kit; GE Healthcare Life Sciences, Opfikon, Switzerland) according to the manufacturer’s instructions. PCR was performed using specific primers designed with an online tool (PrimerQuest tool; Integrated DNA Technologies, Coralville, IA). The primers amplified 4 fragments (Supplementary Table 1) containing the mutations (D1, D2, D3*, and D4) that were previously described 4 in coding exons 2, 6, 7, and 14, respectively, in the ACAN gene. To search for new mutations, 42 primer pairs (Supplementary Table 2) were designed to sequence the 16 coding exons of the predicted ACAN mRNA. Using dwarf skin samples, total RNA purification, DNase treatment, and complementary DNA (cDNA) synthesis were performed using a previously described methodology. 10 PCR was standardized to a final volume of 25 μL (12.5 μL of PCR master mix–GoTaq Green PCR master mix; Promega, Madison, WI; 300 nM of each primer, 8.5 μL of nuclease-free water, and 2.5 μL of DNA or cDNA template) with the following conditions: initial denaturation at 95°C for 5 min, followed by 40 cycles of denaturation at 95°C for 30 s, annealing at 63°C for 1 min and extension at 72°C for 1 min, and a final extension at 72°C for 5 min. The PCR products were analyzed by 1.5% agarose gel electrophoresis. The products with the correct size were purified (Wizard SV gel and PCR clean-up system; Promega) according to the manufacturer’s instructions. To sequence the DNA, 10 μL of purified PCR product and 5 μL of forward primers were used with a sequencing kit (BigDye terminator cycle sequencing kit; Thermo Fisher Scientific, Waltham, MA). The sequences were determined (3500 series genetic analyzer; Thermo Fisher Scientific). The obtained sequences and the electropherograms were analyzed (Geneious v.10.0.9; Biomatters, Auckland, New Zealand) and compared with the normal equine ACAN gene sequence (RefSeq 100033876 and ENSECAG00000007493).
Physical examination revealed that the 2 dwarfs had a domed head that was large compared to the rest of the body, mandibular prognathism, and short and bowed limbs, mainly in the proximal region of the metatarsal bones (Figs. 1–4; Table 1).
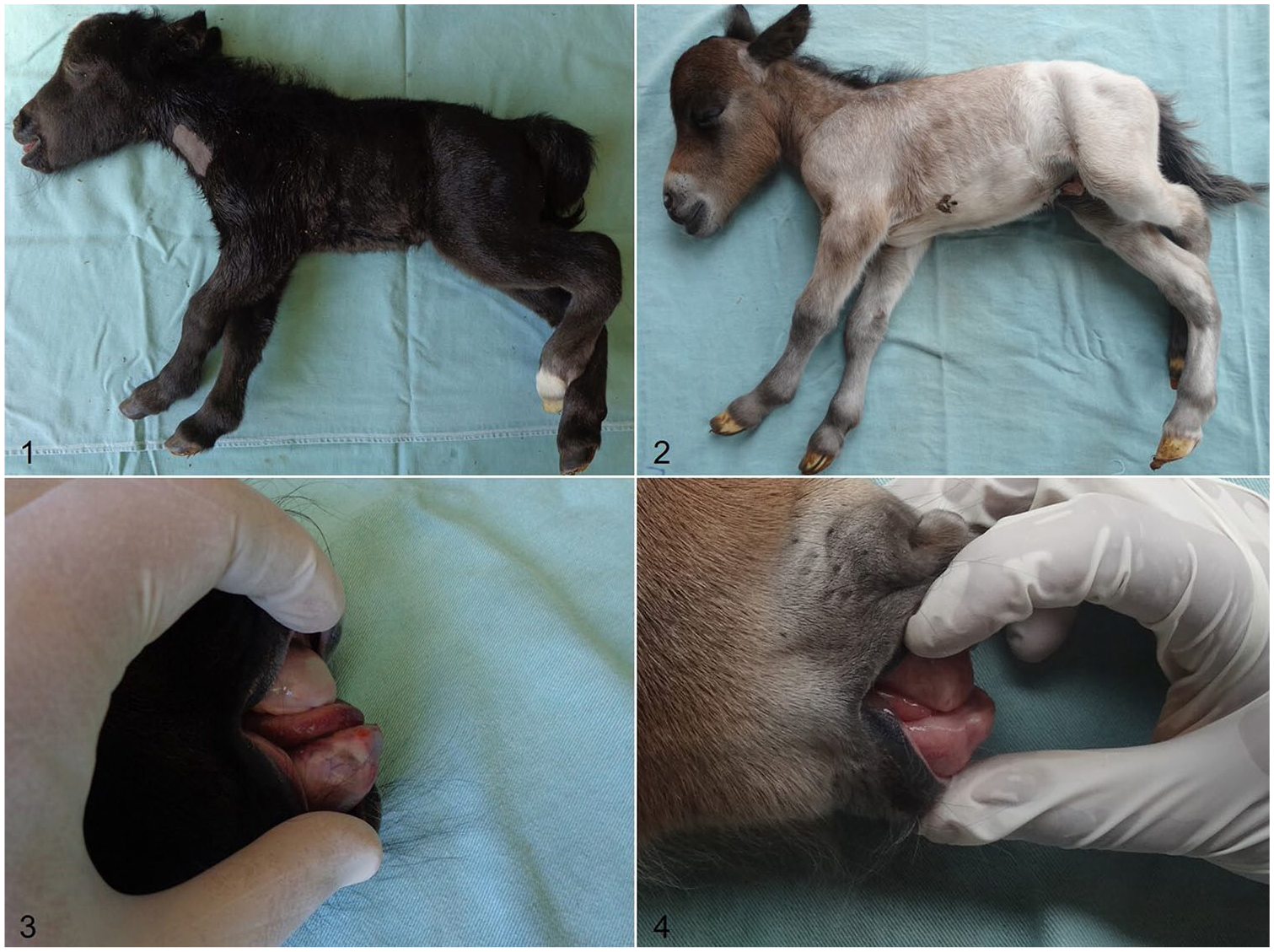
Clinical signs of dwarfism in Miniature horses.
Body measurements obtained from two 1-d-old Miniature horses with disproportionate dwarfism (D4/D4) and a 1-d-old Miniature horse without dwarfism (control).
In the radiographic examination of the 2 affected animals, irregularities of the subchondral bone in the long bones were observed in the proximal margin of the olecranon, in the distal region of the radius, in the dorsal aspect of the proximal metatarsal metaphysis, and in the femoral condyles. Angular deformities with deviations of the valgus and recurvatum types at the tarsal/metatarsal level and mandibular prognathism in the skull were also observed (Supplementary Figs. 1–4). Airway endoscopy of the affected colt revealed dorsal displacement of the soft palate, thickening of both arytenoid cartilages, and mild tracheal collapse (Supplementary Figs. 1–4).
No alteration was observed in the internal organs during autopsy, but microscopically, the proximal portion of the metatarsus (both in the articular cartilage and in the physis) showed mild and severe disorganization of the layers of chondrocytes in the affected filly and colt, respectively, and reduced extracellular matrix in both animals. The usual columnar alignment of hypertrophic cells was not present, and the demarcation between the epiphyseal plate growth zones was not apparent, mostly in the resting and proliferative zones. The ossifying trabeculae were also irregular. In addition, differentiated chondrocytes were absent from the articular cartilage in the colt, and the physis was thickened in the filly, but the same could not be stated about the colt given the intense disorganization of these cells (Supplementary Figs. 5–16).
The g.95257480_95257500del (D4) in the ACAN gene, which is responsible for dwarfism in Miniature horses, 4 was found to be homozygous (D4/D4) in the affected foals in our study. In addition, their parents were heterozygous at the same position (N/D4; Supplementary Figs. 17–20). All parents and the 2 affected foals were wild type (N/N) for the other 3 mutations (D1, D2, and D3*) in the ACAN gene that were described previously. 4 Two additional missense polymorphisms were found when sequencing exons of the ACAN gene (g.95271452C>T/p.A1875V and g.95270484A>G/p.R2198G, RefSeq 100033876). However, given that the stallions were also demonstrated to be homozygous for the same single-nucleotide polymorphisms and both mares were heterozygous for g.95271452C>T and homozygous for g.95270484A>G, these amino acid changes did not interfere with the functionality of the protein. Unfortunately, as reported in another study, 4 we were also unable to sequence the initial region of coding exon 11 (~1,000 bases).
The pathologic findings revealed chondrodysplasia-like characteristics, and genetic alterations confirmed the etiology of dwarfism in the affected foals. The foals were also evaluated radiographically (before confirmatory genetic test results for dwarfism), and the findings were not consistent with skeletal atavism because the horses did not have a complete fibula and ulna, as described previously. 11 In addition, the endoscopic findings were similar to those previously described in another foal with dwarfism. 6
Phenotypically, these two D4/D4 dwarfs were similar to the D2/D2, D2/D3*, D2/D4, D3*/D3*, and D3*/D4 genotypes described previously in dwarf foals.4,9 All of the dwarfs had a disproportionately short stature, a malformed skull, a shortened nasal bone, and mandibular prognathism,4,9 but the front and hind cannon length appeared to be unique to D4/D4 dwarfs. Compared with a control foal, these animals appeared to have shortened cannon bones and hindlimb deformity, with a plantarodorsal deviation in the region of the tarsometatarsal joint. The foals described in our study were neonates, and the adult phenotype of D4/D4 animals is still unknown. Possibly, in adult life, these animals could have a misshapen short barrel, similar to the other genotypes with D4 (D2/D4 and D3*/D4) that were described in a study conducted in the USA (Eberth JE. Chondrodysplasia-like dwarfism in the Miniature horse [master’s thesis]. Lexington, KY: University of Kentucky, 2013).
The D4 mutation is not an embryonic lethal mutation in heterozygotes given that the combinations D2/D4 and D3*/D4 have produced live dwarf foals previously. 4 However, D4/D4 animals have not been detected previously, possibly because of the small sample sizes of previous studies or because D4 is considered a rare mutation in the populations already studied and was therefore not examined. 4 We suggest that a prevalence study should be performed to better understand the occurrence of this disease, given that 2 D4/D4 Miniature foals were found within a short period of time, and homozygous D4 mutations have not been described previously.
The autosomal recessive inheritance pattern silently contributes to the spread of the disease in the population because the parents (heterozygotes) are normal in appearance. Therefore, the identification of heterozygous animals is the most important control measure to minimize economic losses and casualties of affected animals given that such identification makes mating selection possible.
Supplemental Material
Supplemental_material – Supplemental material for Description of the D4/D4 genotype in Miniature horses with dwarfism
Supplemental material, Supplemental_material for Description of the D4/D4 genotype in Miniature horses with dwarfism by Danilo G. A. Andrade, Roberta M. Basso, Maria C. R. Castiglioni, Jeana P. Silva, Vânia M. V. Machado, Renée Laufer-Amorim, Alexandre S. Borges and José P. Oliveira-Filho in Journal of Veterinary Diagnostic Investigation
Footnotes
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The Sao Paulo Research Foundation (FAPESP) provided financial support (2016/24767-7) and a scholarship for D.G.A. Andrade and R.M. Basso (2016/24025-0 and 2018/11365-3); National Council for Scientific and Technological Development (CNPq) research scholarship for J.P. Oliveira-Filho (307686/2018-3).
Supplementary material
Supplementary material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
