Abstract
Immunohistochemical techniques were used to investigate the origin of a spindle cell tumor in the anterior uveal tract of dogs and the influence of ultraviolet radiation on the development of this tumor. Thirteen tumors were identified from the 4,007 canine ocular samples examined at the Comparative Ocular Pathology Laboratory of Wisconsin between 1978 and 2005. Siberian Husky and Siberian Husky mix dogs were overrepresented (10/13 dogs, overall median age 10 years). Light microscopic evaluation (all dogs) and electron microscopy (2 dogs) were performed. Immunohistochemical staining included alpha-smooth muscle actin (SMA), vimentin, S-100, desmin, glial fibrillary acidic protein (GFAP), Melan A, microphthalmic transcription factor (MITF-1), protein gene product 9.5 (PGP 9.5), laminin, gadd45, p53, proliferating cell nuclear antigen (PCNA), anti-UVssDNA (antibody for detection of (6–4)-dipyrimidine photoproducts), and telomerase reverse transcriptase (TERT). All tumors occurred in the iris with or without ciliary body involvement and were composed of spindle cells arranged in fascicles and whorls (variable Antoni A and B behavior). All tumors were positive when immunostained for vimentin and S-100. Nine of 13 tumors exhibited GFAP immunopositivity. All tumors were negative for SMA, desmin, Melan A, and MITF-1. Tumors were variably positive for PGP 9.5, laminin, gadd45, p53, PCNA, anti-UVssDNA, and TERT. Electron microscopy revealed intermittent basal laminae between cells. These tumors are morphologically and immunohistochemically most consistent with schwannoma. The relationship between spindle cell tumors of the anterior uvea of dogs, altered neural crest, blue iris color, and ultraviolet radiation has not yet been fully elucidated.
Intraocular spindle cell tumors of dogs are rare and include melanocytic tumors,11 peripheral nerve sheath tumors,43 and metastatic sarcomas. The most common metastatic spindle cell tumors of the canine eye at the Comparative Ocular Pathology Laboratory of Wisconsin (COPLOW) are hemangiosarcoma, osteosarcoma, metastatic malignant melanoma, and anaplastic mesenchymal tumors of undetermined origin. At the COPLOW, a morphologically distinct spindle cell tumor has been identified that is exclusively associated with partial or complete blue uveal color.28,29 The association between blue iridal color and tumors has been most extensively studied in humans in association with intraocular melanoma20,30 and patients with neurocristopathies who exhibit both pigment abnormalities and peripheral nerve sheath tumors.42 In blue eyes, there is virtually no histopathologically visible melanin within the iris stroma; it is present only in the posterior iridal epithelium. Melanin absorbs ultraviolet (UV) radiation;2,22 UV radiation has been implicated in the pathogenesis of human uveal melanoma2,22,44,46 and skin cancer.39
The purpose of this study was to describe a morphologically distinct uveal tumor of blue-eyed dogs and to perform immunohistochemical staining to investigate both the cell of origin and the cause of malignant transformation relative to (blue) uveal color. Immunohistochemical stains were used to rule out melanoma (Melan A, MITF-1), to rule in a tumor of peripheral nerve sheath origin (vimentin, S-100, glial fibrillary acidic protein [GFAP], protein gene product 9.5 [PGP 9.5], laminin), and to investigate the role of UV damage in the pathogenesis of this tumor (p53, anti-UVssDNA [antibody for detection of (6-4)-dipyrimidine photoproducts], proliferating cell nuclear antigen [PCNA], gadd45, and TERT).
Materials and Methods
Thirteen canine anterior uveal spindle cell tumors from the COPLOW collection were included in this study based solely on their morphologic features. All tumors in this study involved the anterior uvea (Fig. 1) of blue-eyed dogs. All tissues were formalin fixed and paraffin embedded. All stains performed on the 13 tumors included are reported. Immunohistochemistry for these dogs was performed based on tissue availability. Immunohistochemical staining results are reported in Table 1 as follows: negative = < 10% of neoplastic cells stained positively; + = 10–25% positive; ++ = 25–50% positive; and +++ = > 50% positive.
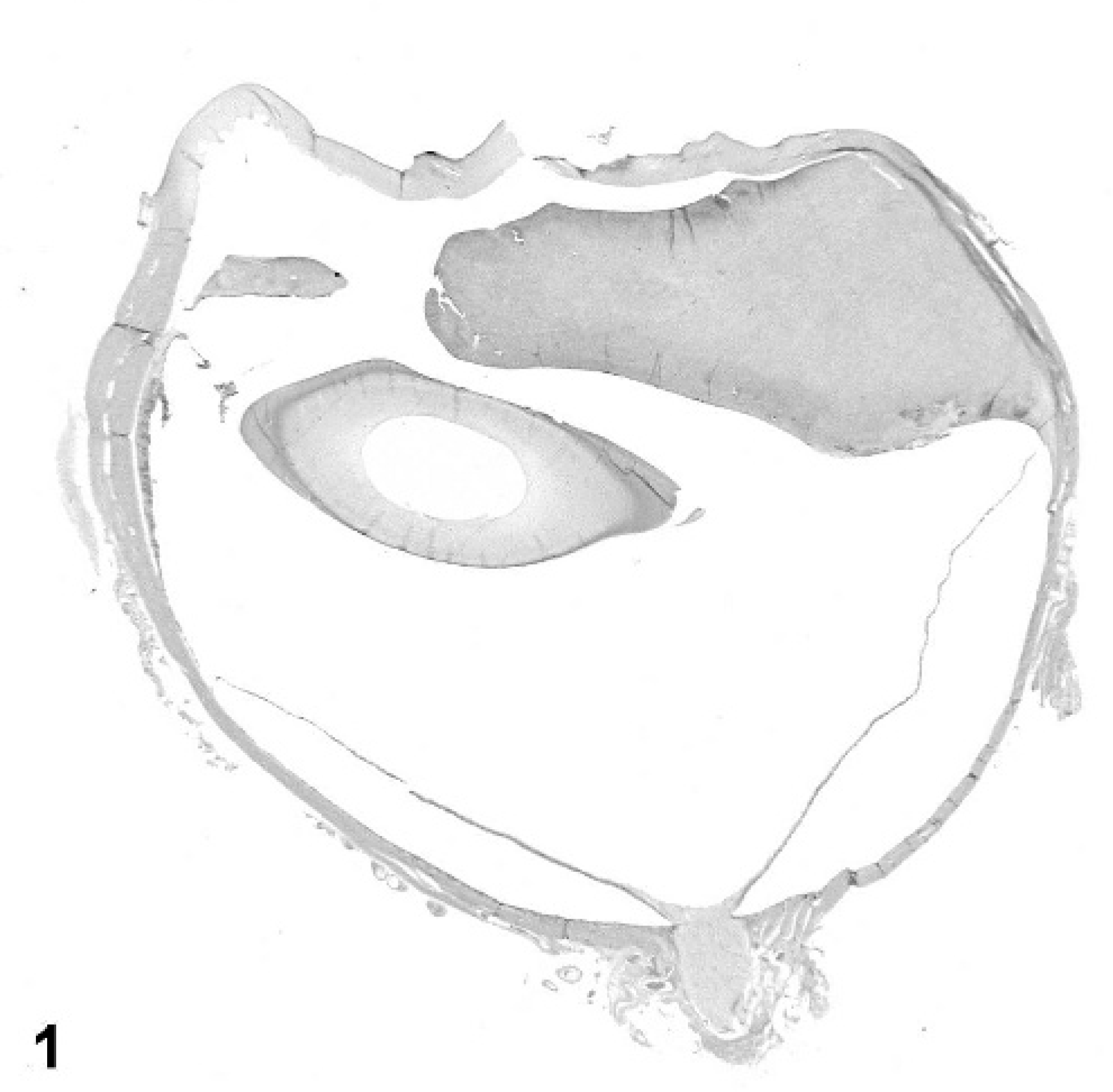
Subgross photo of an eye (anterior at top, optic nerve at bottom); dog No. 13. The image shows an asymmetric expansion of the anterior uvea by spindle-cell tumor. Retinal detachment and corneal defects are artifactual. HE stain.
The following stains were performed at the COPLOW: vimentin (Clone V9, No. M 0725, Dako Corp., Carpinteria, CA); GFAP (No. Z 0334, Dako); smooth muscle actin (Clone 1A4, No. M 0851, Dako); and desmin (Clone D33, No. M 0760, Dako). For these stains, 5-μm sections were mounted on slides and allowed to dry overnight at 56°C. Slides were placed in warm xylene, deparaffinized with 3 changes of xylene for 5 minutes each, and then hydrated in water. Samples were blocked for endogenous peroxidase with 3% H2O2 for 5 minutes. Samples were rinsed in distilled water before digestion with pronase for 5 minutes at room temperature or with 5% trypsin at 37°C for 8–20 minutes depending on antigen. Samples were cooled in distilled water followed by Tris-buffered saline (TBS, 0.05 M) for 2 minutes. Samples were drained and covered with 1–2 drops of Biogenex blocking serum for 30 minutes. Slides were drained and covered with 1–2 drops of primary antibody (rabbit or mouse origin) for 30 minutes or overnight. Sections were rinsed twice in 0.05 M TBS pH 7.4 for 2 minutes. Slides were drained and covered with secondary antibody for 15–30 minutes at room temperature. Slides were rinsed 3 times with 0.05 M TBS pH 7.4 for 2 minutes. Sections were developed in 3,3-diaminobenzidine solution (DAB), prepared by combining 2 ml of Tris, 50 μL of 3% H2O2, and 500 μl of stock DAB solution. Sections were developed for 30 seconds to 5 minutes, in parallel with positive controls. Sections were rinsed in TBS and distilled water. Slides were counterstained with Harris hematoxylin for 10–20 seconds, decolorized blue in L1CO3, and rinsed in tap water for 1 minute. Sections were dehydrated in 70%, 95%, 100% xylene and cover-slipped using Permount. Appropriate positive and negative controls were used in all cases. Immunohistochemical interpretation was carried out by the authors R. R. Dubielzig, G. Klauss, and M. K. Zarfoss.
The following immunohistochemical stains were performed at the Michigan State University Department of Pathobiology and Diagnostic Investigation: MITF-1, Melan A, S-100, PGP 9.5, laminin. Deparaffinization, antigen retrieval, and counterstaining were performed with an automated immunostainer (Ventana Medical Systems, Inc., Tucson, AZ). Paraffin-embedded tissue sections (5-μm thickness) were deparaffinized with EZ Prep (Ventana Medical Systems, Inc.), washed with Sodium Chloride Sodium Cirrate buffer (SSC) solution (Ventana Medical Systems, Inc.) and incubated with reaction buffer (Ventana Medical Systems, Inc.) at 37°C for 2 minutes. Slides were incubated at 37°C for 10 minutes with a mouse monoclonal anti-MITF-1 (Abcam, Cambridge, UK) and a mouse monoclonal anti-Melan A (DakoCytomation, Carpinteria, CA) at a concentration of 1:100 and 1:20, respectively. A biotinylated anti-mouse and anti-rabbit secondary antibody conjugated with alkaline phosphatase-streptavidin (enhanced alkaline phosphatase red detection kit, Ventana Medical Systems, Inc.) was applied to locate and visualize the bound primary antibody. After that, sections were incubated at 37°C for 32 minutes, counterstained with hematoxylin, followed with a bluing reagent to change the hue of hematoxylin to blue color (Ventana Medical Systems, Inc.). To remove the liquid cover slip, reagent sections were washed with a detergent and rinsed with warm tap water. Finally, sections were rinsed with 100% ethanol, followed by a 1:1 mixture of 50% ethanol and 50% xylene, then 100% xylene, and then cover-slipped. Immunohistochemical staining for PGP 9.5 (R&D Systems, Minneapolis, MN), laminin and S-100 (both Dako Corp.) was performed on a Dako autostainer as previously described21 with a labeled streptavidin-biotin-peroxidase complex system to visualize the immune reactions. Immunohistochemical positivity was observed and recorded by the authors M. Kiupel and Y. Jones.
The following antibodies were used in immunohistochemical staining at The Ohio State University College of Veterinary Medicine: p53 (Zymed 13-4100, 0.5 mg/ml, detects mutant p53, Zymed Laboratories, Inc., San Francisco, CA), PCNA (Clone PC10, No. M 0879, Dako Corp.), gadd45 (Santa Cruz; C-20, sc-792, Santa Cruz Biotechnology, Inc., Santa Cruz, CA), anti-UVssDNA (Trevigen Cat No. 4350-MC-100, Trevigen, Inc., Gaithersberg, MD), and TERT (Santa Cruz; L-20, sc7214, Santa Cruz Biotechnology, Inc., Santa Cruz, CA). Protocol for anti-UVssDNA was as follows: 5 μm sections were deparaffinized on slides, incubated in 0.05 N HCl for 5 minutes on ice, and washed 3 times in 1× phosphate-buffered saline (PBS). Slides were incubated in 100 μg/ml RNase in 150 mM NaCl, 15 mM sodium citrate for 1 hour at 37°C, and then washed sequentially in 1× PBS, 50% ethanol, and 70% ethanol for 2 minutes each. DNA was denatured in situ with 0.15 N NaOH in 70% ethanol for 4 minutes at room temperature. Samples were washed sequentially in 70% ethanol, 50% ethanol, and 1× PBS for 2 minutes each, and then incubated in 5 μg/ml proteinase K in 20 mM Tris, 1 mM ethylenediamine tetraacetic acid (EDTA), pH 7.5 for 10 minutes at 37°C. Samples were then washed in 1× PBS. The anti-UVssDNA antibody (primary) was diluted to 6 μg/ml in 1× PBS, 0.01% Tween 20, on ice. Slides were incubated in diluted primary antibody for 1 hour at 37°C (or overnight at 4°C, or at room temperature for 3–4 hours), and then washed 3 times in 1× PBS for 5 minutes each. Slides were incubated in biotinylated anti-mouse antibodies (secondary) for 45 minutes at 37°C, and then washed 10 times in 1× PBS. Slides were incubated in peroxidase-conjugated streptavidin for 30 minutes at 37°C, and then washed 10 times in 1× PBS. Slides were then incubated in Vector NovaRED (chromogen, Vector Laboratories, Burlingame, CA) for 5 minutes at room temperature, and then washed 10 times in tap water. Slides were counterstained in Mayer's hematoxylin counterstain for 3 minutes at room temperature. Slides were then washed 10 times in tap water, dehydrated, mounted, and cover-slipped. Samples were visualized by light microscopy (Olympus BX-50, Olympus America Inc., Center Valley, PA) and digitally photographed (Olympus DP12 digital camera). Positivity was determined by CMHC and KN.
Protocol for PCNA: tissues were deparaffinized, hydrated, and then pretreated with Dako Target Retrieval Solution (catalog No. S1699, Dako Corp.). Retrieval solution was preheated to 90°C. Slides were incubated 20 minutes in steamer, and then cooled for 20 minutes. Slides were placed in Dako wash buffer (TBS/Tween 20, catalog No. S3006, Dako Corp.) for 5 minutes. Procedure on Dako autostainer was as follows: slides were rinsed once with water (rw) × 1, blocked for 5 minutes in peroxidase block in 3% H2O2, rw × 1, and rinsed once with wash buffer (rwb) × 1. Slides were then blocked with Dako serum-free protein block (catalog No. X0909, Dako Corp.) for 10 minutes. Samples were subsequently incubated for 30 minutes in primary anti-PCNA antibody, diluted 1:200 in Dako antibody diluent with background reducing components (catalog No. S3022, Dako Corp.), rwb × 2. Slides were incubated for 30 minutes in secondary antibody (Vector biotinylated horse anti-mouse, catalog No. BA-2000, diluted 1:200, Vector Labs.) in serum-free protein block, rwb × 2 and placed for 30 minutes in ABC (Vector R.T.U. Vectastain elite ABC, catalog No. PK-7100, Vector Labs.), rwb × 2, rw × 1, incubated for 5 minutes in Dako liquid DAB substrate (catalog No. K3466, Dako Corp.), rw × 1, rwb × 1. Slides were counterstained for 2 minutes in Richard-Allen hematoxylin 2 (Richard-Allen Scientific, Kalamazoo, MI) catalog No. 7231, rw × 2, dehydrated, and cover-slipped.
Protocol for TERT (1:350), p53, gadd45 (1:250) was as follows: samples were deparaffinized, rehydrated, and blocked with 3% peroxidase for 10 minutes at 37°C. Power Block (1×, catalog No. BS-1310-25, Biogenex, San Ramon, CA) was used for 10 minutes at 37°C. The sample was incubated with primary antibody for 1 hour at 37°C, and then washed. The sample was incubated in secondary antibody (super-sensitive label, peroxidase-conjugated streptavidin) for 30 minutes at 37°C, and then washed. Positivity was detected using Vector NovaRED for 5 minutes before washing. Samples were counterstained with Mayer's hematoxylin for 3 minutes at room temperature, washed, dehydrated, and cover-slipped. Positive controls for gadd45 and TERT were cataractous lens epithelial cells.8 Negative controls, where primary of secondary antibodies were omitted, were performed in all cases.
For electron microscopy, 1 mm3 of 2 formalin-fixed tumors were harvested and postfixed in gluteraldehyde. This tissue was postfixed a second time in osmium tetroxide and embedded in acrylic resin. Thin sections (approximately 90 nm) were cut, mounted on 200-mesh copper grids, stained with 5% methanolic uranyl acetate and Reynolds' lead citrate, and examined and photographed on a Philips 410 transmission electron microscope (Philips Medical Systems, Andover, MA).
Results
For signalment, follow-up, morphologic characteristics, and immunohistochemical results, see Table 1. While there was no sex predilection, Siberian Husky purebred or mix dogs were overrepresented (10/13 cases). The median age was 10 years. Histologically, all tumors asymmetrically expanded the iris and ciliary body and consisted entirely of spindle-shaped mesenchymal cells (Figs. 1– 4). Tumor cells formed solid sheets of interwoven bundles, streams or whorls, often with palisading nuclei. Tumor morphology, nuclear features, necrosis, and mitotic index were highly variable. Antoni A and/or Antoni B behavior were exhibited by all tumors, consistent with human schwannomas (Figs. 3, 4).32 Tumors were graded as 1, 2, or 3 as previously described.34 All cases exhibited some degree of lymphoplasmacytic uveitis. Most cases (except for cases 2, 4, 11, and 13) exhibited either clinical or histopathologic evidence of glaucoma (historical ocular hypertension, loss of retinal ganglion cells, optic nerve cupping/gliosis). Other secondary findings within the globes included preiridal fibrovascular membranes, intraocular hemorrhage, peripheral corneal stromal neovascularization and inflammatory infiltrate, and cataract.
Signalment, morphology, immunopositivity, and follow-up for 13 uveal spindle-cell tumors of blue-eyed dogs.∗

GFAP = glial fibrillary acidic protein; PGP 9.5 = protein gene product 9.5; PCNA, proliferating cell nuclear antigen; anti-UVssDNA = antibody for detection of (6-4)-dipyrimidine photoproducts; TERT = telomerase reverse transcriptase; FS = female spayed; SH = Siberian Husky; MC = male castrated; + = 10–25% positive; ++ = 25–50% positive; +++ = >50% positive; − = <10% positive.
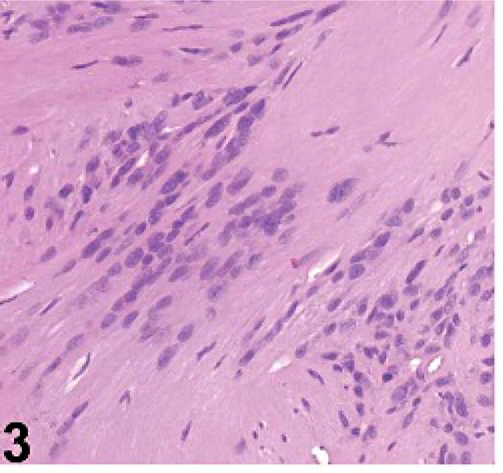
Uveal spindle-cell tumor; dog No. 1. Antoni B behavior is shown. HE stain.
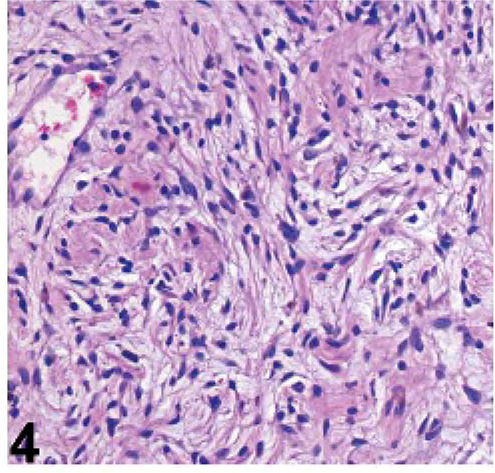
Uveal spindle-cell tumor; dog No. 3. Antoni A behavior is shown. HE stain.
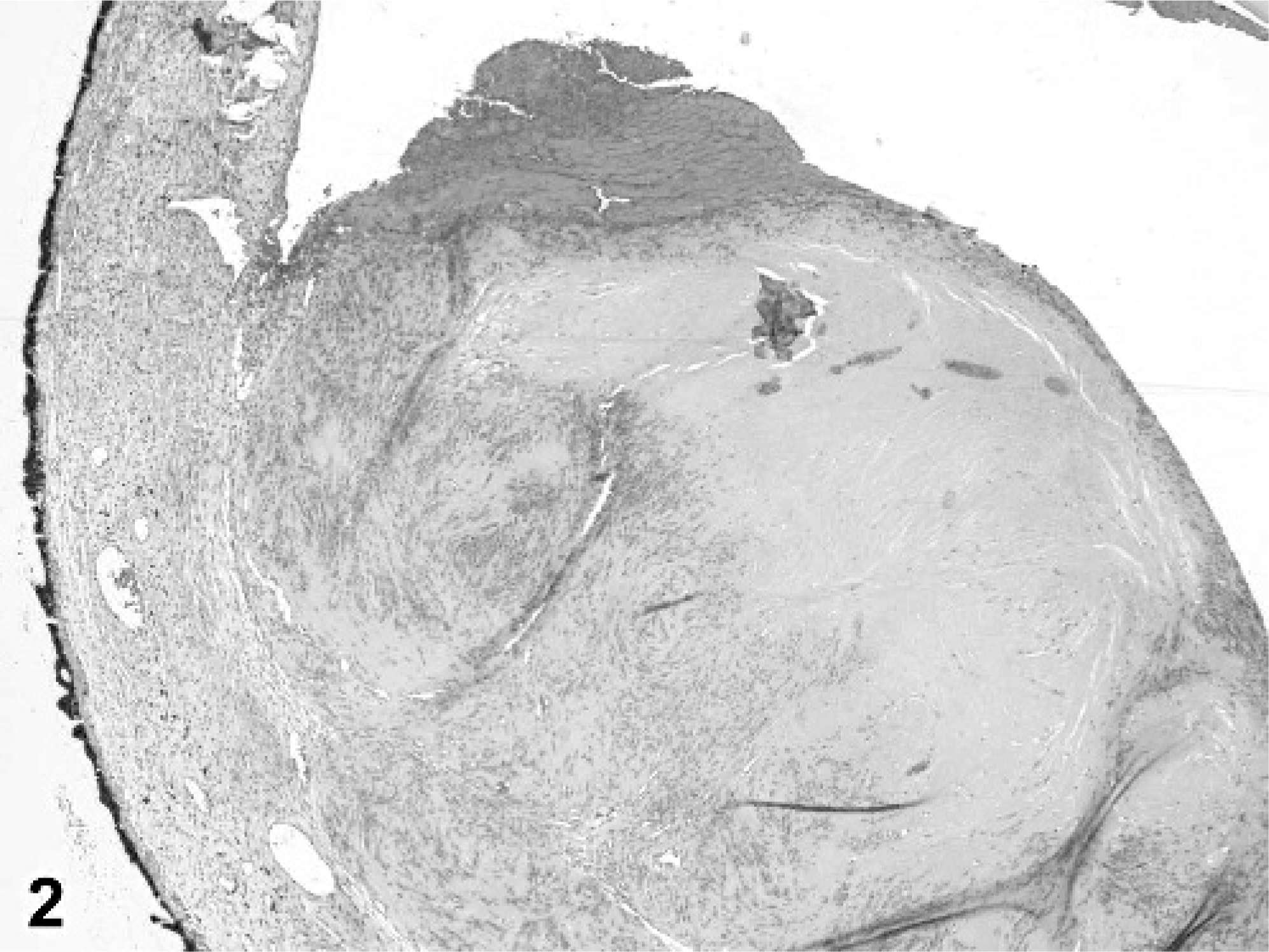
Iris; dog No. 3. Along the left, relatively normal iris stroma is bordered by posterior iridal epithelium. To the right, there is a round mass of mesenchymal cells lined anteriorly by hemorrhage. Within the center of the image, the neoplastic cells exhibit Antoni A behavior. HE stain.
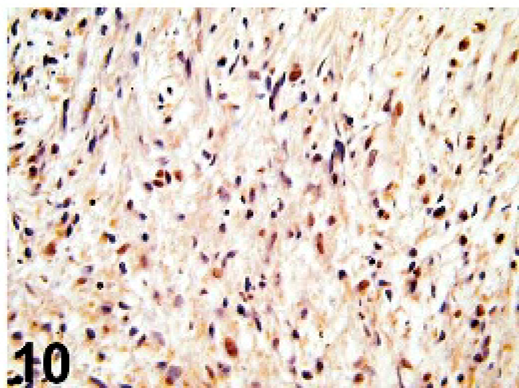
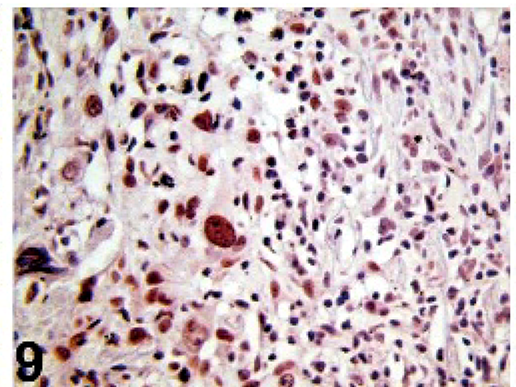
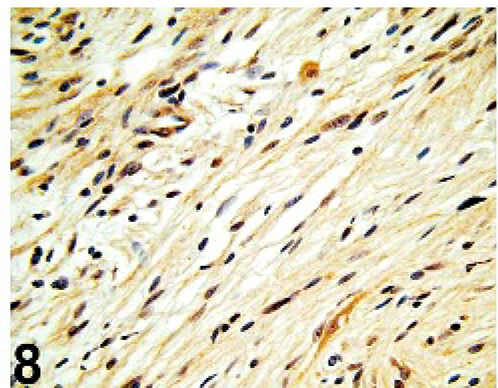
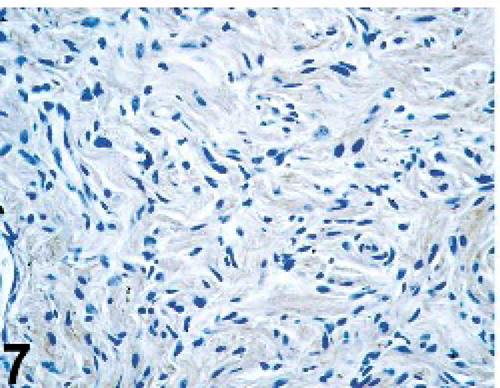
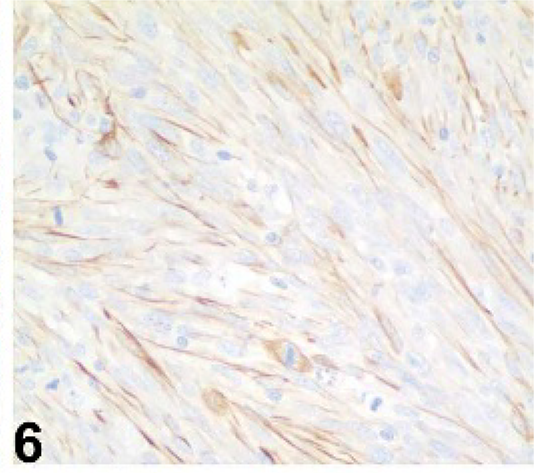
All dogs tested were negative for SMA (6/6), desmin (6/6), Melan A (7/7), and MITF-1 (7/7) and positive for vimentin (10/10) and S-100 (7/7). The tumors were positive for GFAP in 9/13 dogs, positive for PGP 9.5 in 7/9 dogs, and positive for laminin in 2/9 dogs (Figs. 5 –7). Results for gadd45, p53, PCNA, anti-UVssDNA, and TERT were variable and are shown in Table 1 and Figs. 8 –10.
Uveal spindle-cell tumor; dog No. 6. Nuclear and cytoplasmic immunopositivity is shown. Telomerase reverse transcriptase (TERT) immunohistochemistry.
Uveal spindle-cell tumor; dog No. 8. Nuclear immunopositivity is shown. UV single strand DNA (UVssDNA) immunohistochemistry.
Uveal spindle-cell tumor; dog No. 11. Nuclear and cytoplasmic immunopositivity is shown. Growth arrest and DNA damage45 (Gadd45) immunohistochemistry.
Uveal spindle-cell tumor; dog No. 7. Extracellular immunopositivity is shown. Laminin immunohistochemistry.
Uveal spindle-cell tumor; dog No. 13. Intracytoplasmic immunopositivity is shown. Glial fibrillary acidic protein (GFAP) immunohistochemistry.
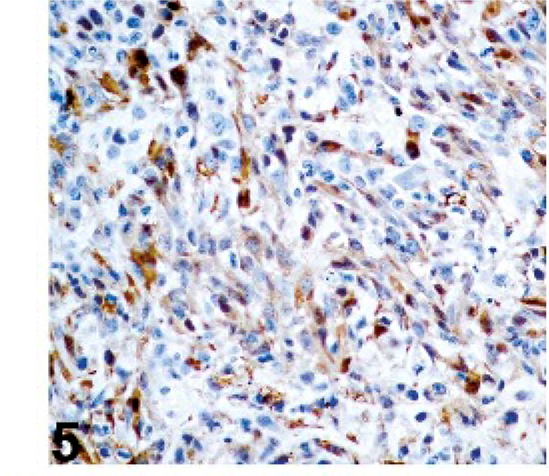
Uveal spindle-cell tumor; dog No. 8. Intracytoplasmic immunopositivity is shown. S-100 immunohistochemistry.
Follow-up ranged from 0–28 months (see Table 1). While several patients died with clinical signs consistent with thoracic and abdominal metastasis, no necropsies were performed. Furthermore, dogs with possible metastases did not have any obvious morphologic or immunohistochemical similarities that might relate to prognosis.
Electron microscopic findings revealed that tumor cells had long interdigitating cytoplasmic processes and intermittent basal laminae at the plasma membrane (Fig. 11). The extracellular matrix consisted of collagen bundles.
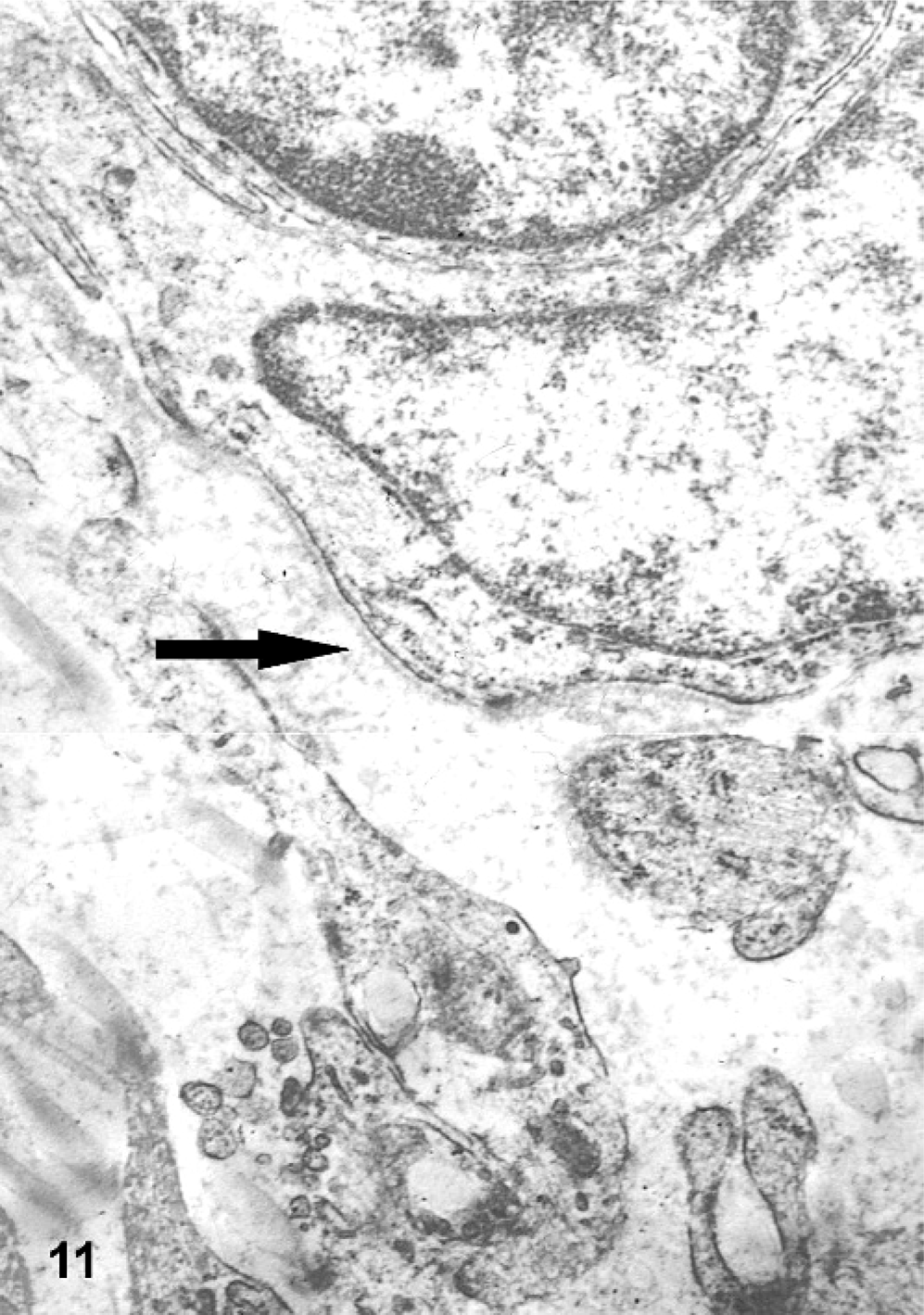
Electron microscopic image of spindle-cell tumor; dog No. 10. The arrow is directed at the basal lamina.
Discussion
Immunohistochemical stains were initially performed to rule out nonneural tumors. Vimentin immunoreactivity suggests that this tumor is of mesenchymal origin.13 Leiomyosarcoma or rhabdomyosarcoma are unlikely because the tumors did not stain for SMA and desmin.5 MITF-1 and Melan A immunonegativity suggest that this tumor is not of melanocyte origin.6,16 Immunohistochemical staining for c-kit was also performed on nine tumors (CD117, results not shown); all tumor cells were immunonegative. The GFAP immunopositivity makes a diagnosis of fibrosarcoma unlikely.38
Glial fibrillary acidic protein staining, in 9/13 dogs, suggests central nervous system/astrocyte origin; however, GFAP positivity has also been associated with both nonmyelinating Schwann cells52 and normal rodent uvea.4 Electron microscopy was also consistent with a tumor of peripheral nerve sheath origin due to the interrupted basal laminae between cells.32 Protein gene product 9.5 is a relatively neuron-specific protein similar to neuron-specific enolase.51 Laminin is found in basement membranes of endothelial cells, smooth muscle cells, and Schwann cells.5 Laminin staining was positive in only 2/9 cases. Formalin fixation may have interfered with stain uptake, or laminin expression may have been variable. This tumor is immunohistochemically and morphologically most consistent with a peripheral nerve sheath tumor, which arises from Schwann cells, neurofibroblasts, or other perineural cells.32 Such tumors in animals are generally classified as (benign) schwannomas or malignant peripheral nerve sheath tumors. The presence of Antoni A and B patterns in these tumors is also consistent with neural origin.26,32 While electron microscopy, light microscopy, and GFAP immunohistochemistry are most consistent with a schwannoma, these findings cannot reliably differentiate between a benign/malignant schwannoma, neurofibroma, neurofibrosarcoma, or other tumor of nerve origin in dogs.25,52
To the authors' knowledge, no intraocular peripheral nerve sheath tumors have been reported in the dog except for a recent case report by Sato et al.43 While morphologically similar to the tumors in this study, those authors clearly stated in a personal correspondence (Sato) that there were no identifiably blue areas in the affected (or unaffected) eye of their patient, a Shetland Sheepdog. This tumor immunohistochemically stained positively for S-100 and vimentin. At the COPLOW, no similar uveal tumors have been diagnosed in any other species or in brown-eyed dogs. Relative to brown-eyed dogs, blue-eyed dogs are at much higher risk for this tumor. Other studies have shown extraocular, benign, canine schwannomas to be vimentin positive (most consistent feature), desmin negative, variably GFAP positive, and often S-100 positive.32,38
Why do these tumors only occur in dogs with blue irides? To answer this question, the embryology and anatomy of the canine iris must be considered. While recent studies suggest that mammalian uveal stroma is formed from a mixture of neural crest and mesoderm, neural crest is believed to give rise to melanocytes within the iris.10,15,24 The color of the iris is normally dictated by heritable factors and determined by the pigment granules within the posterior iris epithelium, Rayleigh scattering of light by the stromal extracellular matrix, type of melanin, and the number and area of melanin granules within the iris stromal melanocytes.24,50 Aside from polygenic inheritance affecting melanin content of the iris, blue iridal color can result from a tyrosinase defect preventing melanin production in stromal melanocytes (heterochromic laboratory beagles,45 albinism, Siamese cats48), as well as abnormal neural crest migration into the uvea causing an absence of stromal melanocytes (white cats, associated with deafness3). At the COPLOW, two goniodysgenic canine irides (lacking tumor) were stained with GFAP. These irides showed increased linear GFAP positivity, consistent with nonmyelinated nerves, within the iris stroma compared with a similarly stained, bleached brown iris. Melan A immunohistochemistry on the same 2 goniodysgenic canine blue eyes found no convincingly positive melanocytes within the iridal stroma (results not shown). Lack of melanocytes within the uveal stroma would corroborate that this is not a primary intraocular melanocytic tumor; furthermore, it suggests a defect in neural crest. However, these findings cannot necessarily be extrapolated to all blue-eyed dogs; furthermore, the coat color of the patients with uveal spindle cell tumors was generally not recorded.
In addition to iridal melanocytes, neural crest is also responsible for the development of peripheral nerve tissue, including Schwann cells.35 In human neural crest–related diseases, genetic mutations in PAX 3 gene (Waardenburg syndrome) and NF1 gene (neurofibromatosis) are associated with hypopigmented irides or light brown iridal café au lait spots, respectively.23,42 While iris color is not necessarily blue in humans with neurofibromatosis, iridal lesions include pigmented hamartomas that are considered to be of neural crest origin (Lisch nodules).23 The NF1 gene is a tumor suppressor gene and neurofibromas and schwannomas are a feature of neurofibromatosis. Peripheral nerve sheath tumors related to human neurofibromatosis 1 are usually benign but can undergo malignant transformation.14,37 More research needs to be performed to determine whether or not a genetic defect in neural crest development contributes to oncogenesis in this tumor (particularly in Siberian Huskies).7
Ultraviolet radiation has been shown to contribute to oncogenesis in many tumors, including uveal melanoma.20,22,44,46 Schwannomas have been reported in humans in association with radiation therapy (but not UV damage).41 Positive immunohistochemical staining for p53, anti-UVssDNA, PCNA, gadd45, and TERT have all been associated with UV damage and oncogenesis. Specifically, research suggests that gadd45 and PCNA facilitate repair after DNA undergoes UV-related damage.1,47 Telomerase refers to a ribonucleoprotein complex that has 2 main components: an RNA subunit (TR) and a catalytic subunit (TERT). All cells express TR, but TERT is only expressed in cells that have telomerase activity. Telomerase activity facilitates repeated cell division and is therefore necessary for tumor proliferation.17,18 Up-regulation of TERT is strongly associated with exposure to the sun in normal human skin samples and exposure to UV radiation results in increased telomerase activity in lens epithelial cells, cultured splenocytes, and fibroblasts, suggesting a role in UV-induced oxidative stress.9,18,19,49 In one study, 100% of tumor samples with UV-specific p53 mutations also exhibited TERT activity; both telomerase activity and p53 mutations have been implicated in oncogenesis.49 The p53 gene is commonly mutated in human cancers and is integral in DNA repair after UV damage;47 it controls cell cycle arrest, apoptosis, and senescence, among other antitumor functions.12 Mutations in p53 cause increased p53 expression because the mutated form has a much longer half-life than the wild-type form.27 The UVssDNA antibody is specific for proteins that contain the (6-4)-dipyrimidine photoproduct, a product of UV damage.53 With this in mind, increased expression of gadd45, PCNA, TERT, p53, and anti-UVssDNA should all be up-regulated in tumors influenced by UV damage.
The results of this study were inconsistent for markers of UV-induced tissue damage and do not clearly support a role for UV damage in the development of this tumor. However, these results do not exclude UV damage as a factor because UV markers are in some cases only temporarily up-regulated after UV damage.36 There are other unmeasured effects of UV radiation36 and other UV-related antibodies that were not available for use in this study.31 Technically, inconsistent immunohistochemical results may be due to (delayed) fixation,13,52 anaplasia, or multiple cellular origins. Uveal spindle-cell tumors of blue-eyed dogs may arise as a result of a multifactorial process including a genetic neural crest defect and UV damage.40 Other melanin-related or genetic mechanisms may also be in play. Melanin has been shown to be associated with intraocular zinc storage (an antioxidant).33
We present 13 cases of a uveal spindle-cell tumor of blue-eyed dogs with morphologic and immunohistochemical features most consistent with a schwannoma. Morphologic features of malignancy were variable. While several of the dogs have died with clinical signs consistent with metastatic disease, no necropsies have been performed. The cell of origin, its relationship to neural crest, and the contribution of UV damage in the pathogenesis of uveal tumors in blue-eyed dogs have yet to be fully elucidated. Until that time, we will continue to refer to these tumors as uveal spindle-cell tumors of blue-eyed dogs. These tumors continue to accumulate at the COPLOW. We will continue to examine these cases and seek follow-up to better assess their behavioral malignancy. This tumor should be considered as a differential diagnosis in all intraocular masses arising in the blue canine uvea.
Footnotes
Acknowledgements
The authors would like to thank Drs. Steven Weisbrode and Donna Kusewitt for expert consultation. The authors would also like to thank the referring veterinarians and owners of these dogs for providing follow-up information.
