Abstract
A 20-year-old female horse showed a nodular, firm, focal ulcerated mast cell tumor at the right dorsobuccal face of the tongue. Histologically, the nonencapsulated tumor consisted of dense, infiltrating aggregates of well-differentiated, Cresyl violet-positive mast cells accompanied by numerous eosinophils. Furthermore, they exhibited a strong, diffuse, intracytoplasmatic immunohistochemical signal for tryptase and a faint membrane-associated and perinuclear signal for tyrosine kinase receptor KIT. Confocal laser scanning microscopy confirmed an aberrant spatial colocalization of KIT in the Golgi apparatus, which may be the result of a defective protein processing within the tumor cells. The tumor was not associated with a poor prognosis.
Mast cell tumors (MCTs) are frequently observed tumors of the skin of domestic and laboratory animals but have also been described in other organs and tissues, including mucosa of gastrointestinal tract, salivary glands, eyes, testes, and spleen.3,11 In horses, they are most commonly found in the skin and rarely in the eye, the nose, or at multiple sites in cases of systemic mastocytosis.5,7–10,12 Equine cutaneous MCTs show usually a well-differentiated histological pattern associated with a benign biologic behavior and a low rate of recurrence.5 Furthermore, they often show prominent foci of necrosis, collagenolysis, fibrosis, and dystrophic mineralization, commonly associated with a high number of eosinophilic granulocytes.3,5,12
The transmembranous tyrosine kinase receptor protein KIT is encoded by the proto-oncogene c-kit. It is expressed in numerous tissues and cells, including mast cells, hematopoetic precursor cells, melanocytes, and interstitial cells of Cajal in the intestine.1,2,6,13 In human and dogs, genetic alterations in the c-kit proto-oncogene have been implicated in the development and progression of mast cell neoplasias and gastrointestinal stromal tumors.13 Furthermore, an aberrant immunohistochemical expression pattern of KIT protein in canine MCTs has been associated with different tumor grades and poor prognosis.4
In the present study, we describe for the first time a MCT of the tongue with an atypical distribution of the stem cell receptor protein KIT.
A 20-year-old female horse showed a nodular, firm, focal ulcerated lesion at the right dorsobuccal face of the tongue (Fig. 1). The analysis of several serologically and blood chemically parameters, including urea nitrogen, creatinine, chloride, potassium, sodium, calcium, phosphorus, iron, triglycerides, serum glucose, total protein, total bilirubin, alkaline phosphatase, alanine transferase, aspartate aminotransferase, gamma-glutamyltranspeptidase, glutamate dehydrogenase, lactate dehydrogenase, alpha-amylase, serum cholesterine, and creatinine kinase, showed a low increase of phosphorus and serum glucose and normal ranges otherwise. The red and white blood cell counts showed a moderate decrease of leucocytes, thrombocytes, neutrophils, and lymphocytes. A 16 × 10 × 12 mm irregular, patchy white mass with indistinct borders was surgically excised. The tissue was fixed in 10%, neutral-buffered formaldehyde and submitted for histological examination. The tissue was routinely processed, embedded in paraffin wax, sectioned at 4 μm, stained with HE and Cresyl violet, and analyzed by light microscopy. Chymase detection was performed with a Naphtol AS-D Chloroacetate Esterase Kit (Sigma) according to the manufacturer's instructions. Additionally, immunohistochemistry was performed using antibodies against human mast cell tryptase (1 in 40, mouse monoclonal; Dako) and human KIT (1 in 30, rabbit polyclonal; Callbiochem) as previously described.2,4
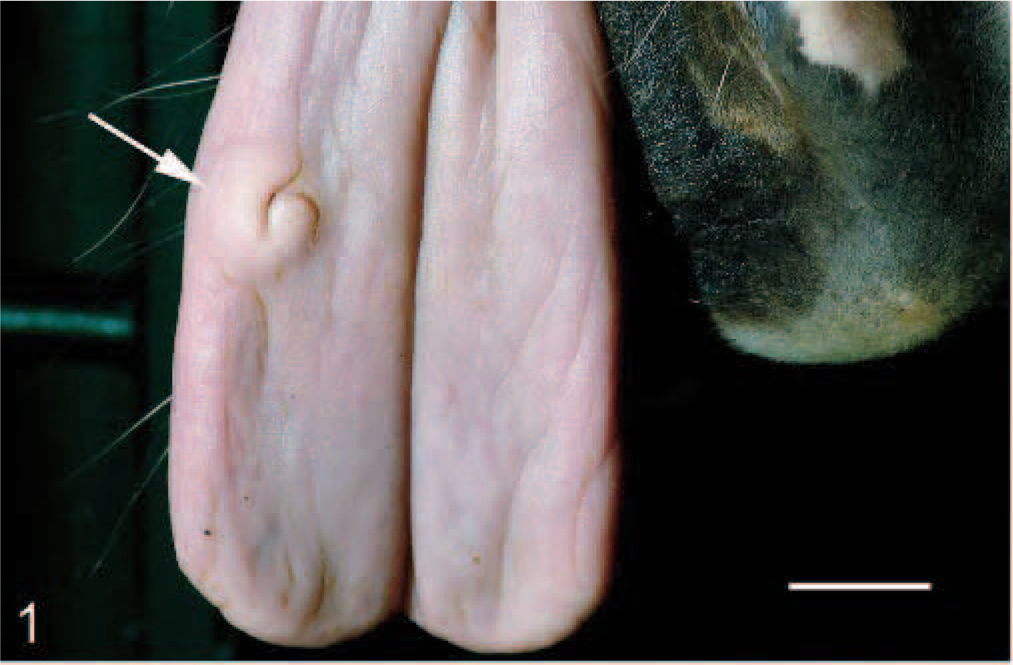
Tongue, tumor, horse. Focal, partial ulcerated tumor with indistinct borders on the right dorsobuccal face of the tongue (arrow). Bar = 15 mm.
The spatial localization of KIT protein in the cytoplasm of tumor cells was studied by means of confocal laser scanning microscopy (Fluoview 1000, Olympus). Double immunofluorescence was performed with antibodies against human KIT protein (1 in 30, rabbit polyclonal; Callbiochem) and human Golgi apparatus protein GM130 (GM130, mouse monoclonal; BD Bioscience). A positive binding reaction of primary antibody was visualized by Alexa Fluor 488−tagged goat-anti-mouse (Molecular Probes) and Alexa Fluor 568−tagged goat-anti-rabbit (Molecular Probes) secondary antibody, respectively, in a consecutive matter. For the excitation of Alexa Fluor 488, the 488-nm line of a multiline Argon laser was used, and for the excitation of Alexa Fluor 568, a 543-nm helium-neon laser was used. Each laser line was scanned separately for the individual excitation of the dyes to exclude crosstalk, and data sets were finally merged. Equine jejunum and bone marrow (KIT) and rat and horse cerebrum (GM130) were used as positive controls for specifity of primary antibodies. To estimate the tissue-specific background, fluorescence/immunofluorescence reactions were performed with the exclusion of 1 or both primary antibodies. The 2D data sets were analyzed with the colocalization module of FV10-ASW® 1.2 Fluoview software (Olympus) and shown as a scatterplot diagram.
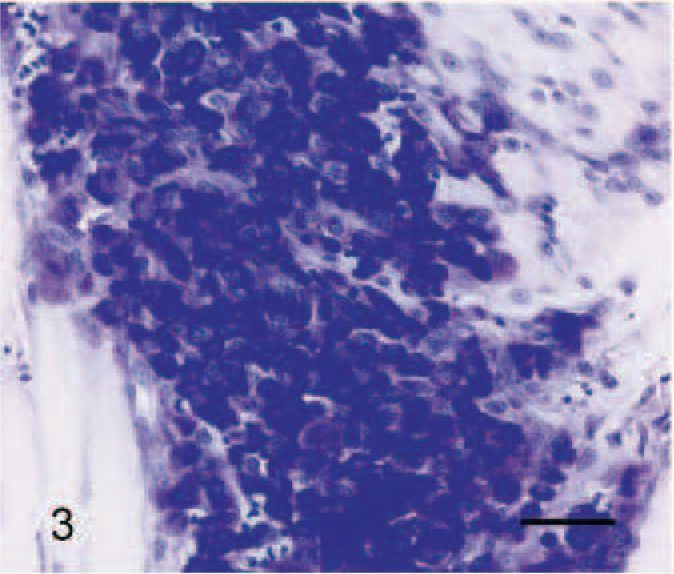
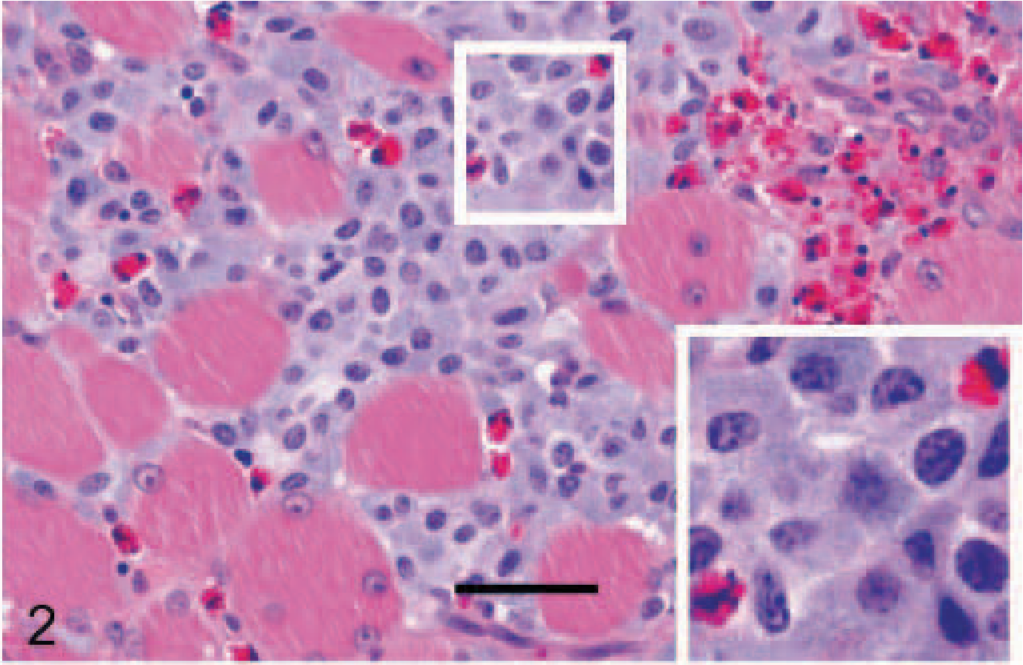
Histologically, the nonencapsulated tumor consisted of dense aggregates of well-differentiated mast cells that were infiltrating the peripheral tissue in nests or strands (Fig. 2). The tumor cells were separated by a thin fibrovascular stroma. Multifocally, confluent areas of liquefaction necrosis and collagenolysis were evident within the center of the tumor. A number of eosinophils were intermingled within the tumor cell clusters and were also present in the adjacent tissue. The cytoplasm of tumor cells had a basophilic appearance with distinct borders and a dense amount of moderately basophilic, fine granules. Most cells had round to ovoid, slightly pleomorphic nuclei with a prominent nucleolus and a finely granular heterochromatin (Fig. 2). The mitotic index was less than 1 mitosis per high-power field. Nearly all tumor cells were positive for Cresyl violet staining (Fig. 3). Only a few cells showed positive signals for chymase (data not shown).
Tongue, tumor, horse. Most tumor cells showed a strong cytoplasmic, granular, blue reaction. Cresy violet staining. Bar = 40 μm.
Tongue, tumor, horse. Numerous tumors cells with distinct cell borders accompanied by eosinophils were infiltrating skeletal muscle fibers of the tongue. Rectangular area: Tumor cells showing fine granular, basophilic cytoplasm, and moderate anisokaryosis (inlet). HE staining. Bar = 40 μm.
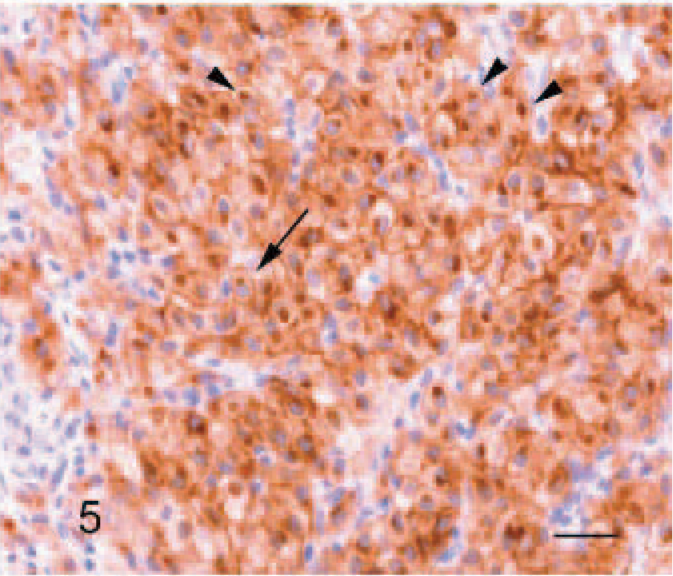
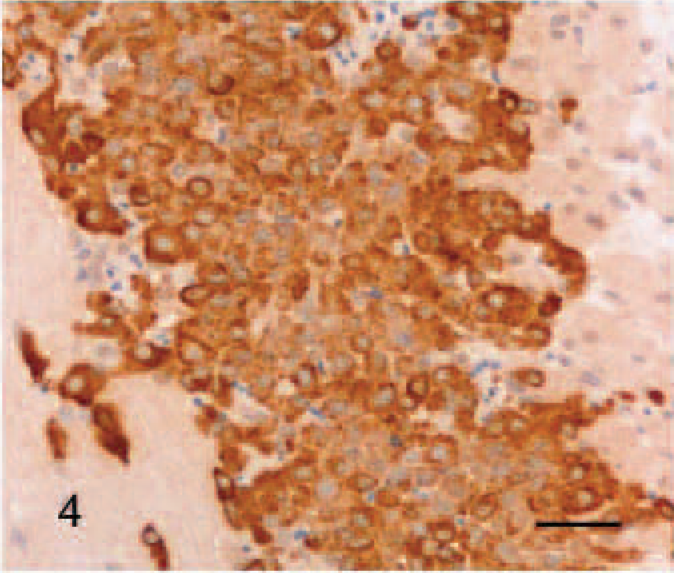
Immunohistochemically, the tumor cells exhibited a strong, diffuse, intracytoplasmatic signal for tryptase (Fig. 4). The antibody reaction to KIT showed a faint membraneous signal with a strong focal clustering near the nuclear membrane (Fig. 5). Double immunohistochemistry and confocal analysis for Golgi apparatus–specific GM130 protein (channel 1, green) and mast cell−specific KIT protein (channel 2, red) revealed clearly distinct spectral signals with minimal crossover for each detection channel. Colocalization analysis of KIT protein demonstrated a prominent signal within Golgi apparatus and cytoplasmic membrane in 2D-scanning mode (Fig. 6). 3D analyses of the specimen confirmed the colocalization in the z direction (Fig. 7). In control tissues, normal mast cells of the propria or the submucosa in the jejunum and hematopoetic precursor cells in the bone marrow showed a faint, strongly membrane-associated signal but never an intracytoplasmic signal for KIT. There was no colocalization with GM130 protein of Golgi apparatus visible (data not shown).
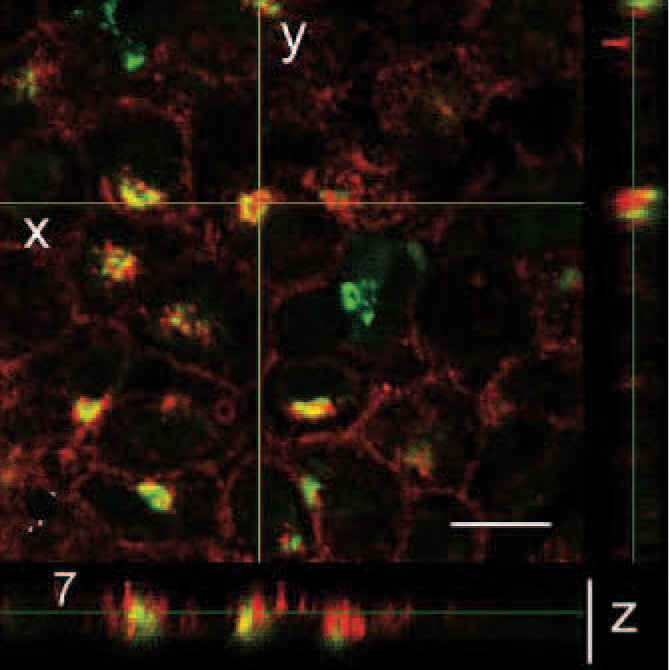
3D data analysis of Fig. 6d. 3D analysis of the tumor confirm colocalization of Golgi-specific protein GM130 and KIT in the z direction. Image stacks were collected with a Plan S Apo × 60 oil emersion objective (N.A. 1,35) with 0,65-μm thick optical slices. Bar = 20 μm.
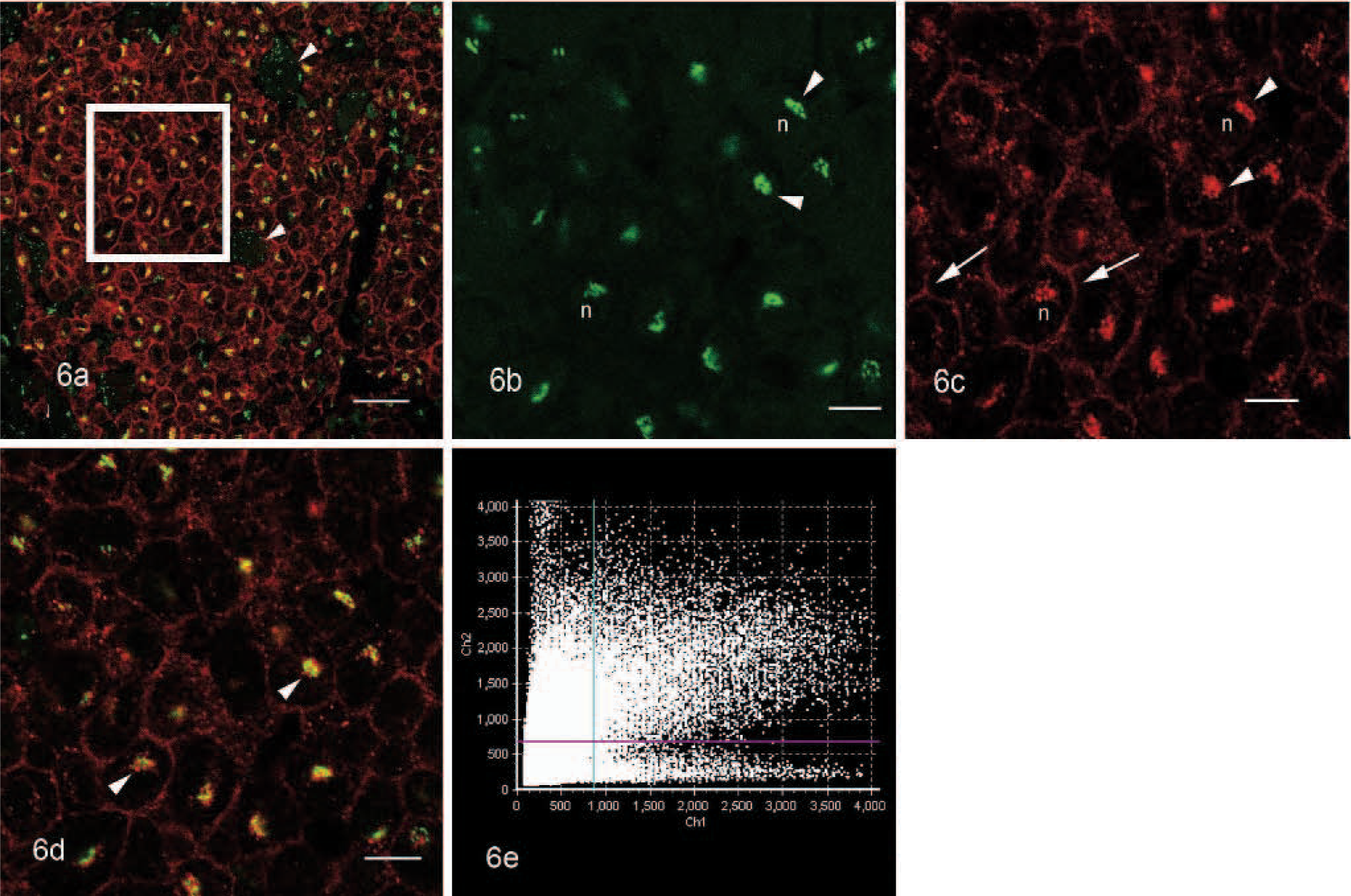
Tongue, tumor, horse. Confocal images showing fluorescence signals and colocalization of Golgi-specific protein GM130 and KIT.
Tongue, tumor, horse. Tumor cells react with antibodies to KIT. Note the faint membrane-associated (arrow) and strong perinuclear staining (arrowheads). Streptavidin-biotin-alkaline phosphatase method, Mayer's hematoxylin counterstain. Bar = 60 μm.
Tongue, tumor, horse. Antibodies to tryptase stain most tumor cells with a diffuse, cytoplasmic granularity. Streptavidin-biotin-alkaline phosphatase method, Mayer's hematoxylin counterstain. Bar = 40 μm.
MCTs of the gastrointestinal tract, including the oral cavity, have occasionally been described in several domestic animal species, including dogs and cattle.3 This study describes the occurrence of an equine MCT in the tongue. It was associated with a prominent accumulation of KIT in the Golgi apparatus of the tumor cells. In canine cutaneous MCTs, a stippled membrane−associated and diffuse, cytoplasmic staining of KIT protein was correlated with a poor prognostic outcome.4,13 In the case described here, only tumor mast cells but not mast cells in control tissues showed a strong intracytoplasmic, perinuclear and a faint membrane-associated KIT staining in conventional or fluorescence immunohistochemistry. Numerous mutations in c-KIT proto-oncogen have been implicated in the progression of neoplastic mast cell disease in humans and dogs and were significantly associated with aberrant KIT localization.13 The reasons for aberrant KIT localization are ambiguous but were thought to be associated with changes in transcriptional regulation or cellular alterations in protein trafficking or golgi processing.13
During a follow-up period of 8 months after surgery, there were no observations of recurrences or metastases. A variable survival rate after treatment of canine oral MCTs has been reported, and the affected dogs may exceed a disease-free period of 12 months or more.3 In the case present here, changes in KIT localization seem not to be associated with a poor prognosis.
In summary, the equine oral MCT described in this report represents the first case of this tumor in the tongue. Tumor cells were uniformly immunopositive for tryptase and KIT and indicated that these antigens are excellent immunohistochemical markers for equine normal and neoplastic mast cells. Furthermore, the aberrant spatial localization of KIT in the Golgi apparatus of the tumor cells may be the result of defective protein processing within the tumor cells but was not associated with a poor prognosis.
Footnotes
Acknowledgements
We thank Petra Grüning and Robert Alberding for excellent technical support. We are grateful to Dr. Helge Schmidt and Hitoshi Hatana (Olympus Europe, Hamburg, Germany) for technical support and assistance with the confocal laser scanning microscope.
