Abstract
Hepatoblastoma was diagnosed in 3 Thoroughbreds at the University of Kentucky Livestock Disease Diagnostic Center (LDDC) since 1997. Case #1 involved a fetus with a well-demarcated, multilobulated, solitary mass that extended from the left liver lobe. Case #2 was observed in a neonate with a primary hepatic mass and multiple metastases in the skin, brain, meninges, and stylohyoid bone. Case #3 was a solitary hepatic mass incidentally discovered in a neonate at necropsy. Microscopically, the masses were similarly composed of sheets and cords of fetal and embryonal epithelial cells that frequently formed sinusoid-like structures. Intermixed with the neoplastic epithelial cells were variable amounts of hemorrhage, necrosis, osteoid, and bone. Immunohistochemically, the epithelial cells stained variably positive for alpha- fetoprotein, frequently positive for vimentin, and occasionally positive for cytokeratin. All 3 cases were diagnosed as mixed hepatoblastoma with teratoid features.
Hepatoblastomas are malignant hepatic neoplasms of young children and animals. These tumors occur with an incidence of 0.5–1.5 cases per million children and account for approximately 25% of all hepatic tumors and 50% of the metastatic hepatic tumors diagnosed in children.1,10 Hepatoblastomas are rarely diagnosed in animals, but reports have been described in mice, horses, sheep, pigs, a dog, a bull, and an alpaca.3,6,9,13,15
A human classification scheme has been utilized to describe hepatoblastomas in animals.1,3,13,15 Hepatoblastomas are categorized as either epithelial or mixed (epithelial/mesenchymal) variants. The recognized epithelial cell types of hepatoblastoma include fetal, embryonal and fetal, macrotrabecular, or anaplastic small cell types. Fetal epithelial cells are polygonal, appear similar to hepatocytes, contain variable amounts of glycogen and fat, and can be arranged into irregular plates that may form canaliculi and sinusoids. Embryonal epithelial cells are small, elongated, contain scant cytoplasm, have hyperchromatic nuclei, and may be arranged in rosettes, cords, or ribbons. Macrotrabecular types appear similar to hepatocellular carcinomas and can include glandular elements and areas of squamous metaplasia. Anaplastic small cells are frequently arranged in sheets and are indistinguishable from other blastic precursor cells. Mesenchymal variants are composed of undifferentiated spindle mesenchyme, osteoid, and feature variably anaplastic epithelial cells. Mixed tumors may have teratoid features, which can include cartilage, bone, striated muscle, and neural tissue.1
Equine hepatoblastomas have been previously reported in 2 adults, a 10-month-old foal, and a fetus.3,5,8,11 Hepatic tumors are extremely rare in equine fetuses; only 2 cases have been reported, which include a mixed hamartoma and a hepatoblastoma.8,12 In this report, we describe the gross and microscopic features of 3 cases of equine hepatoblastoma. Cases #1 and #3 were diagnosed as solitary mixed hepatoblastomas with teratoid features. Case #2 involved an equine neonate with a primary hepatic mixed hepatoblastoma with teratoid features and multiple epithelial type metastases to the skin, brain, meninges, and stylohyoid bone. To the authors' knowledge, these cases are the first to describe mixed hepatoblastomas with teratoid features in an equine fetus (case #1) and 2 neonates (cases #2 and #3). Additionally, case #2 is the first report of hepatoblastoma with brain, bone, and skin metastases in any animal species.
In April of 1997, February of 2006, and April of 2006, a Thoroughbred full-term fetus (case #1), 2-hour-old neonate (case #2), and <1-hour-old neonate (case #3), respectively, were submitted to the LDDC necropsy service. Both, the fetus (case #1) and 2-hour-old neonate (case #2) were submitted with limited clinical histories of dystocia. Case #3 was a neonate, <1 hour old, submitted after euthanasia due to a congenital deviated misshapen nose (wry nose). Necropsy of the fetus (case #1) revealed a large amount of blood in the thoracic and peritoneal cavities. A 19 × 16 × 9-cm reddish-tan multilobulated mass extended from the left liver lobe. On cut surface, the mass had cavitated necrohemorrhagic foci that were separated by connective tissue trabeculae. Fractures were present in the fifth through eighth ribs on the left side. The neonate, in case #2, had multiple 0.5 × 0.5 × 1-cm well-demarcated raised, soft, pink cutaneous masses located over the withers, dorsum, and flank. The second through 11th ribs on the left side were fractured, and the peritoneal cavity contained a small amount of translucent yellow fluid. A multilobulated well-demarcated 20 × 16 × 11-cm tan mass extended from the left liver lobe (Fig. 1). On cut surface, the mass was necrotic and contained small to moderate amounts of serosanguinous fluid and hemorrhage. Within the brain, the lateral ventricles were distended by a small to moderate amount of yellow fluid. The cerebellum was cavitated and contained a 1 cm in diameter well-demarcated pink mass surrounded by yellow gelatinous material (Fig. 2). Additional 0.3–2 cm in diameter reddish-tan mottled masses were in the choroid plexus, cerebral meninges, frontal sinuses, and nasal septum. A red 4 cm in diameter well-demarcated region was observed in the left stylohyoid bone. Necropsy of the maldeveloped neonate (case #3) revealed a round, well-demarcated, and encapsulated 8.5 cm in diameter mass that bulged from the left liver lobe. On cut surface the mass was discolored yellow and had a gritty texture.
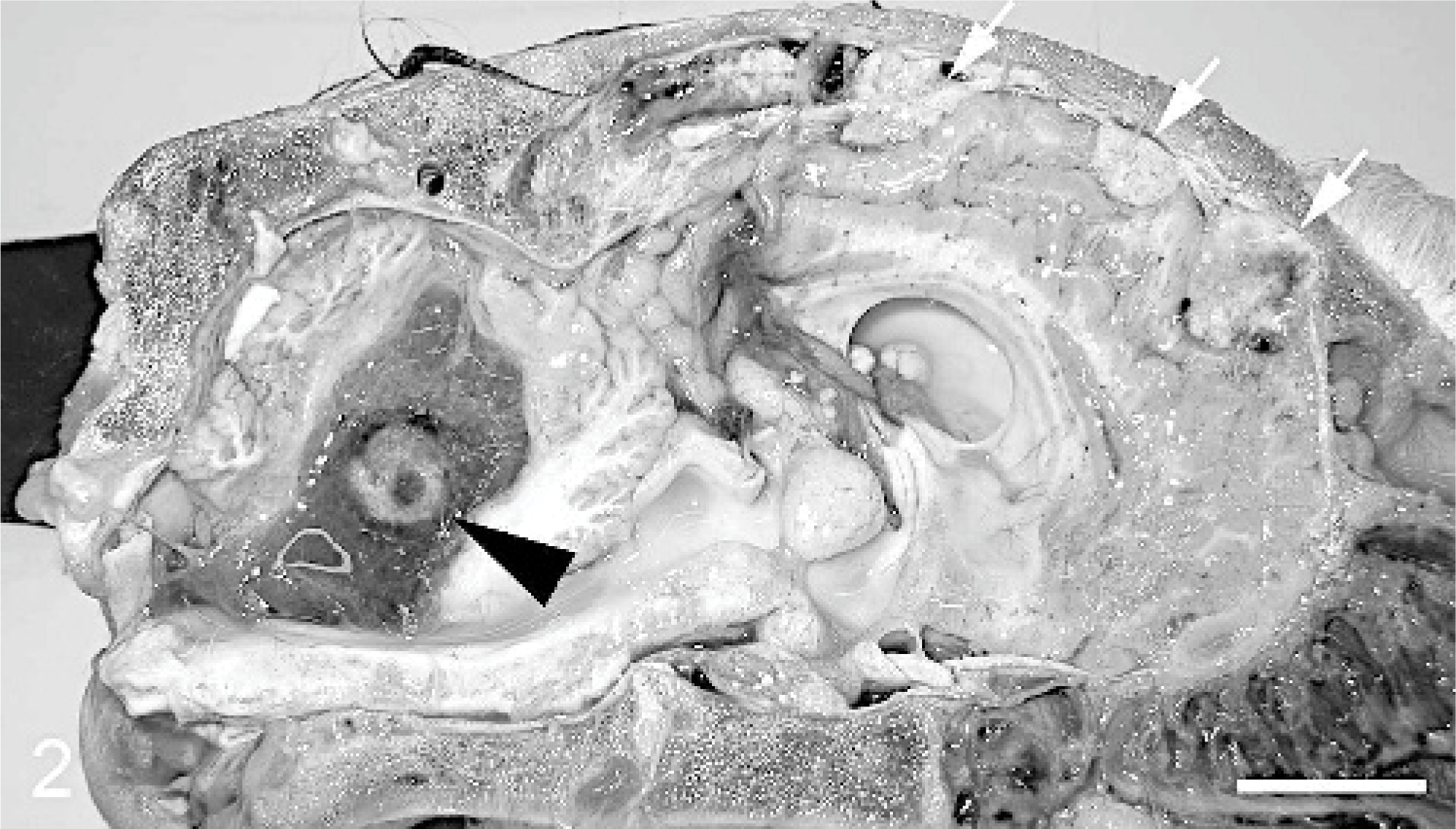
Cerebellar and meningeal metastases, Thoroughbred neonate, case #2. The cerebellum is cavitated and contains a 1 cm in diameter well-demarcated pink mass (arrowhead) that is surrounded by yellow gelatinous material. Additional 0.3–1.25 cm in diameter reddish-tan mottled masses (arrows) are evident in the cerebral meninges. Bar = 2 cm.
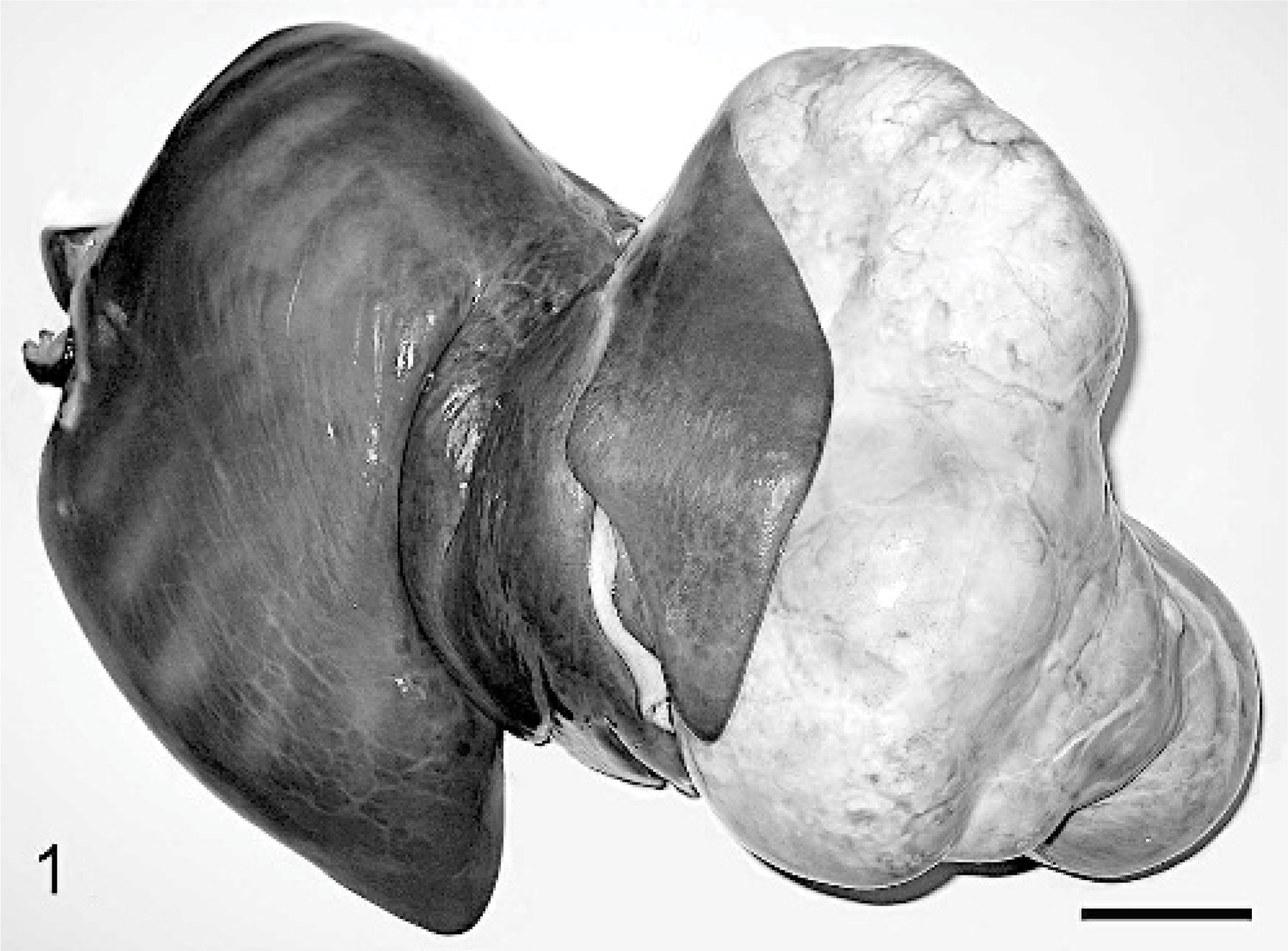
Hepatic mass, Thoroughbred neonate, case #2. A multilobulated, well-demarcated 20 × 16 × 11-cm tan mass extends from the left liver lobe. Bar = 5 cm.
Appropriate samples were taken from each case and placed in phosphate buffered 10% formalin for histopathologic examination. The tissues fixed for 24 hours, were routinely processed, and were stained with hematoxylin and eosin for microscopic evaluation. Oil-red-o stains (ORO) (cases #2 and #3) and periodic acid–Schiff (PAS) stains with and without diastase treatment (cases #1, #2, and #3) were performed on formalin-fixed sections of the hepatic masses and metastases. Formalin-fixed sections of the hepatic mass were not available for ORO staining of the equine fetus (case #1). Immunohistochemical staining techniques were performed with various antisera: Alpha-1 fetoprotein (AFP) (DakoCytomation, Glostrup, Denmark, rabbit antihuman polyclonal), vimentin (DakoCytomation, mouse antiswine monoclonal, clone: V9), and cytokeratin (CK) (DakoCytomation, mouse antihuman monoclonal, clone: AE1/AE3). The EnVision + dual link peroxidase system (DakoCytomation) was utilized for marker visualization.
Microscopic features of the primary hepatic tumors from all 3 cases were similar. Each hepatic mass was expansile, well demarcated, and separated from the normal congested parenchyma by a dense capsule. The capsule contained moderate amounts of fibrous connective tissue, haphazardly arranged bile ducts, and low numbers of blood vessels and lymphatics. Each hepatic mass was composed of sheets and cords of 10–35-μm polygonal hepatocyte-like epithelial cells (Fig. 3) that frequently formed haphazardly arranged hepatic cords and sinusoid-like spaces. The polygonal cells had variably distinct cell borders, moderate amounts of acidophilic cytoplasm that contained 1 to multiple 3–10-μm clear vacuoles, and a single 5–12-μm round to oval nucleus that had finely stippled to vacuolated and peripheralized chromatin. These polygonal cells were consistent with fetal epithelial cells. There was mild to moderate anisocytosis, anisokaryosis, but mitotic figures were rarely identified. Individual to small clusters of 3–7-μm irregularly arranged polygonal to fusiform cells were mixed within the population of fetal epithelial cells. These cells had distinct cellular borders, scant amounts of acidophilic cytoplasm, and a single round to oval 3–5-μm nucleus that had coarsely clumped chromatin. These polygonal to fusiform cells were consistent with embryonal epithelial cells. Exclusive to case #2 were loosely arranged regions composed of individual 10–15 × 5–7-μm well-differentiated spindle cells and lesser numbers of polygonal cells that were supported by a mesenchymal stroma (Fig. 4). The spindle cells had variably distinct cell borders, scant amounts of acidophilic cytoplasm, and a single elongated and flattened 5–10 × 3–5-μm hyperchromatic nucleus. Intermixed throughout the epithelial cells were foci of necrosis, hemorrhage, mineral, osteoid, rare spicules of bone (Fig. 5), cartilage (case #1 only), and extramedullary hematopoiesis. Unlike cases #1 and #2, the hepatic mass from case #3 was partially encapsulated and frequently extended into the normal adjacent hepatic parenchyma. Additional differences included moderate numbers of bone spicules, frequent bile duct cholestasis, minimal amounts of extramedullary hematopoiesis, less frequent intracytoplasmic vacuoles, and a less pronounced fibrous stroma.
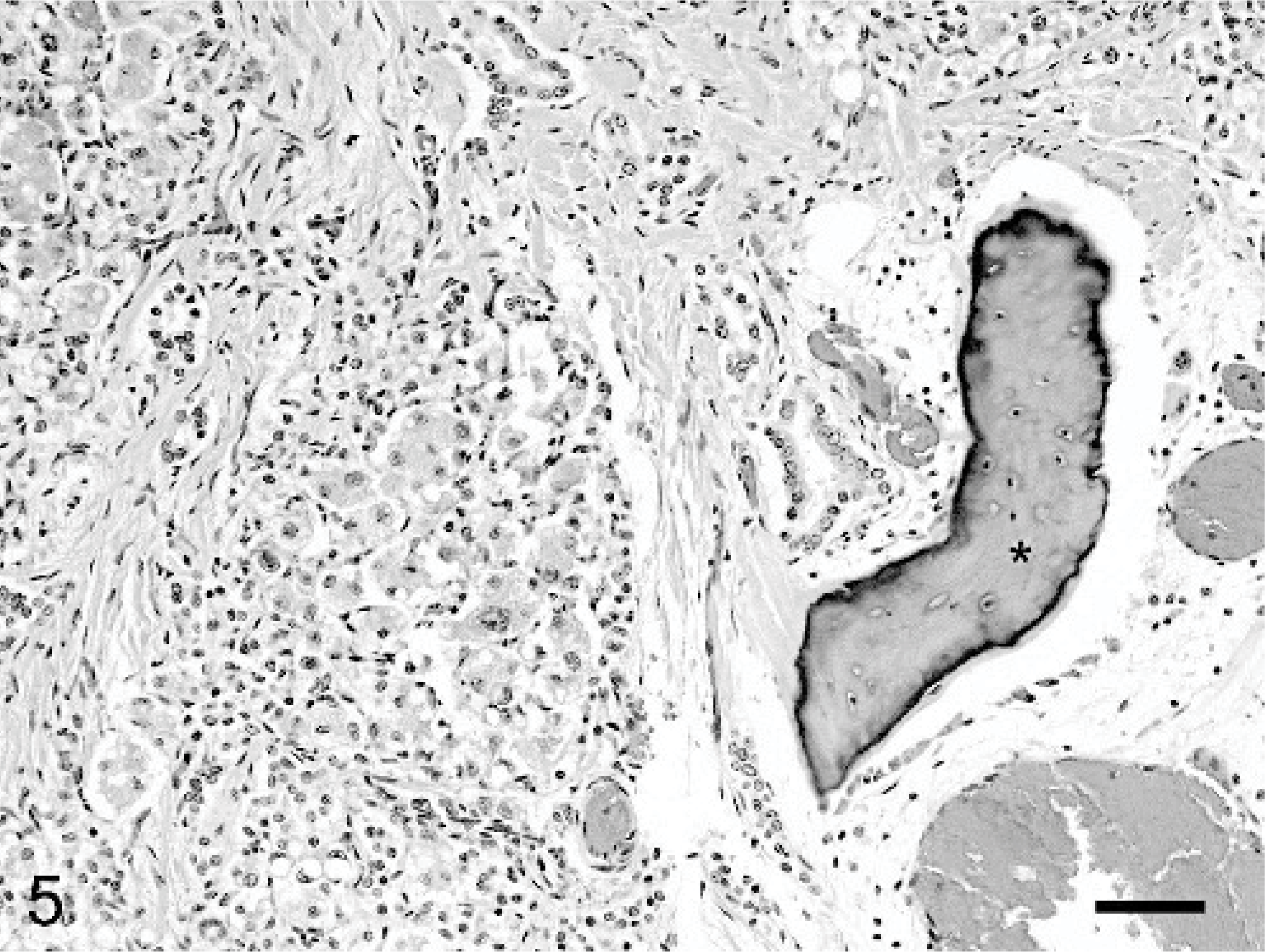
Hepatic mass, Thoroughbred neonate, case #3. A piece of bone (∗) is surrounded by fibrous connective tissue. Fetal epithelial cells abut the connective tissue and attempt to form hepatic cords. HE. Bar = 70 μm.
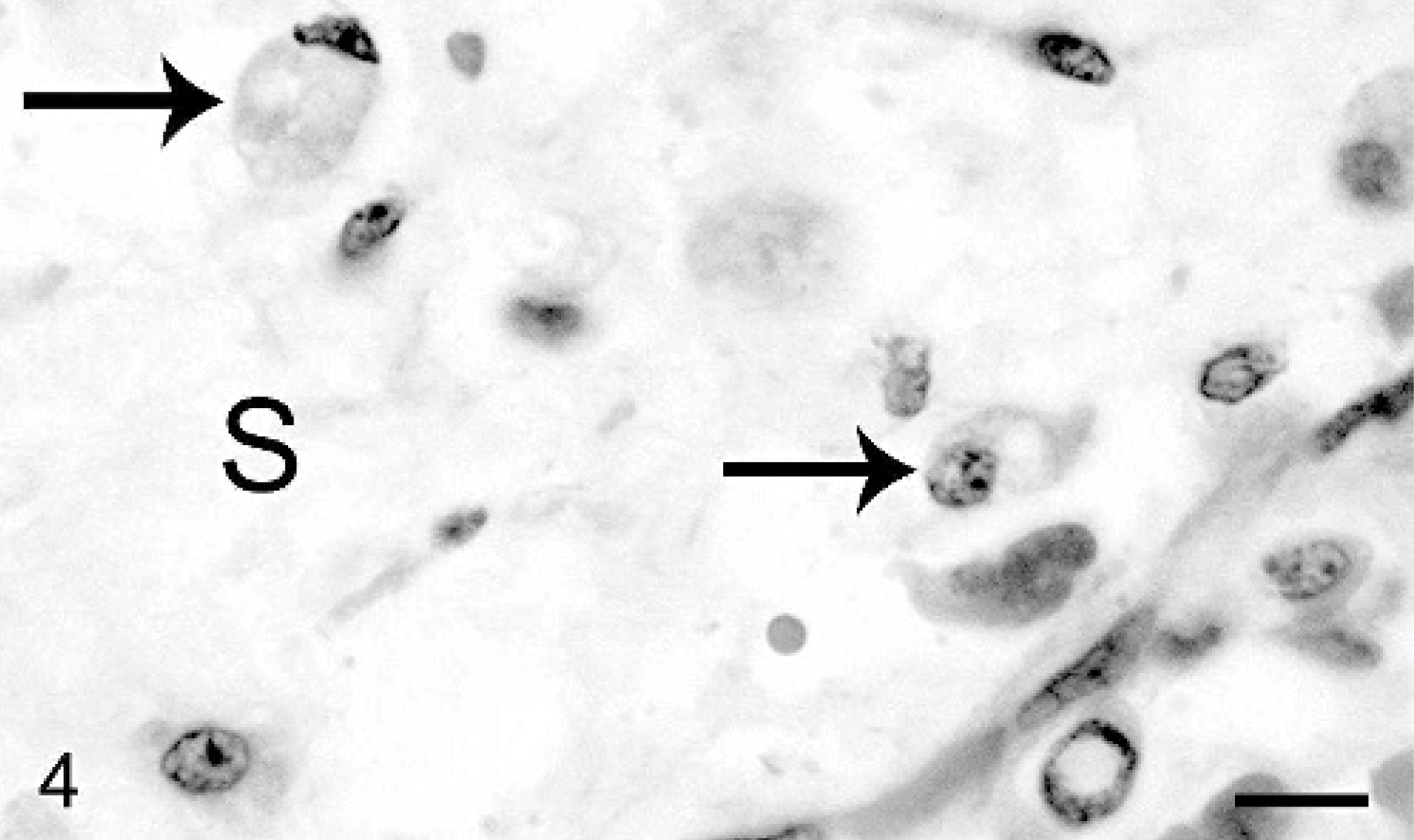
Hepatic mass, Thoroughbred neonate, case #2. Well-demarcated mesenchymal regions contain low numbers of vacuolated neoplastic epithelial cells (arrows) that are supported by a loosely arranged stroma (S). HE. Bar = 20 μm.
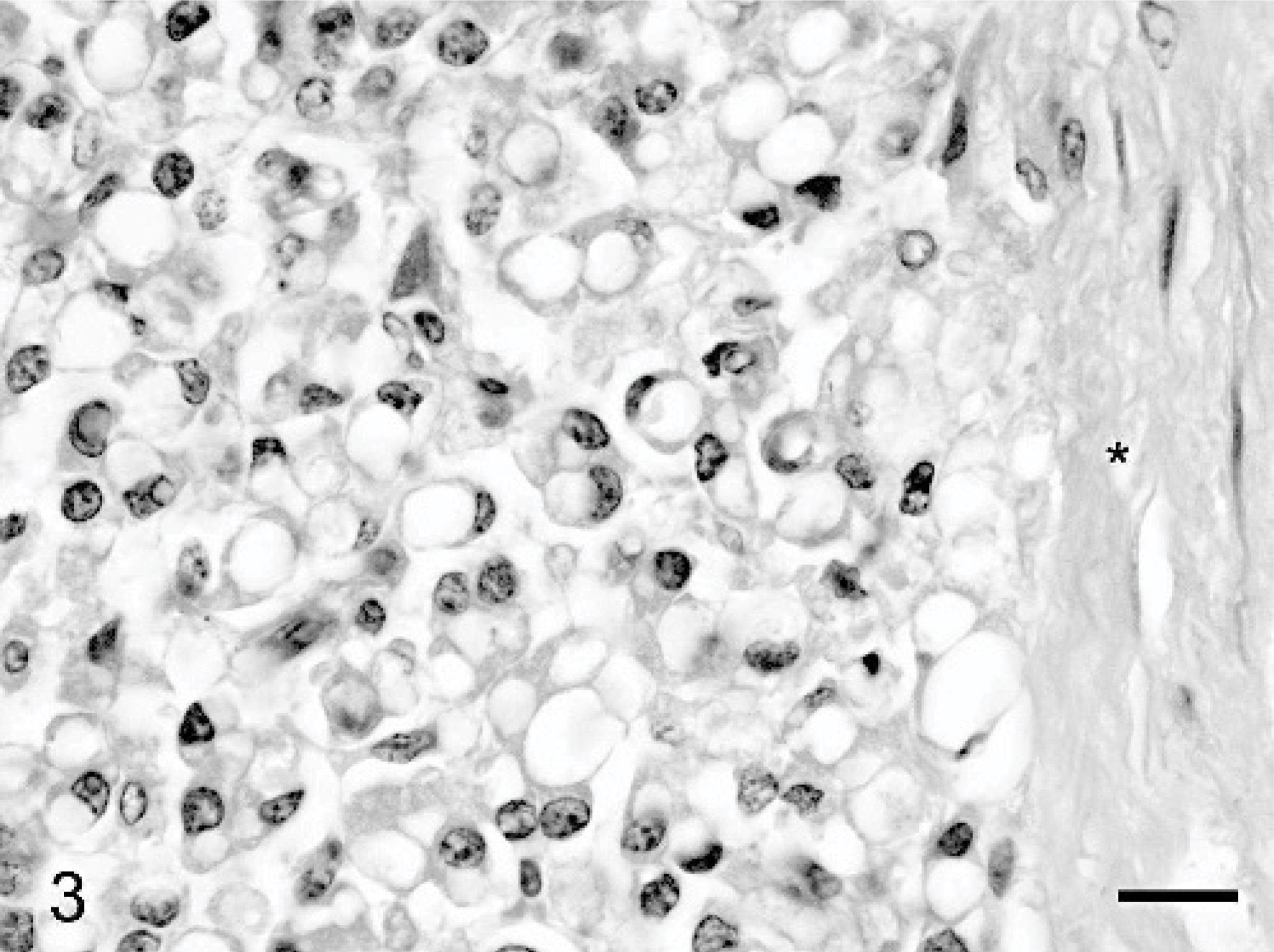
Hepatic mass, Thoroughbred neonate, case #2. Vacuolated neoplastic epithelial cells are partially encapsulated by a dense fibrous connective tissue capsule (∗). HE. Bar = 20 μm.
Tumor metastases (case #2) were observed in the skin (Figs. 6, 7), meninges, brain, and bone. The liver, skin, and meningeal masses were expansile and compressive tumors, while brain and bone masses were infiltrative and destructive. All of the metastases were composed of sheets and cords of embryonal and fetal epithelial cells that were supported by a scant fibrovascular stroma.
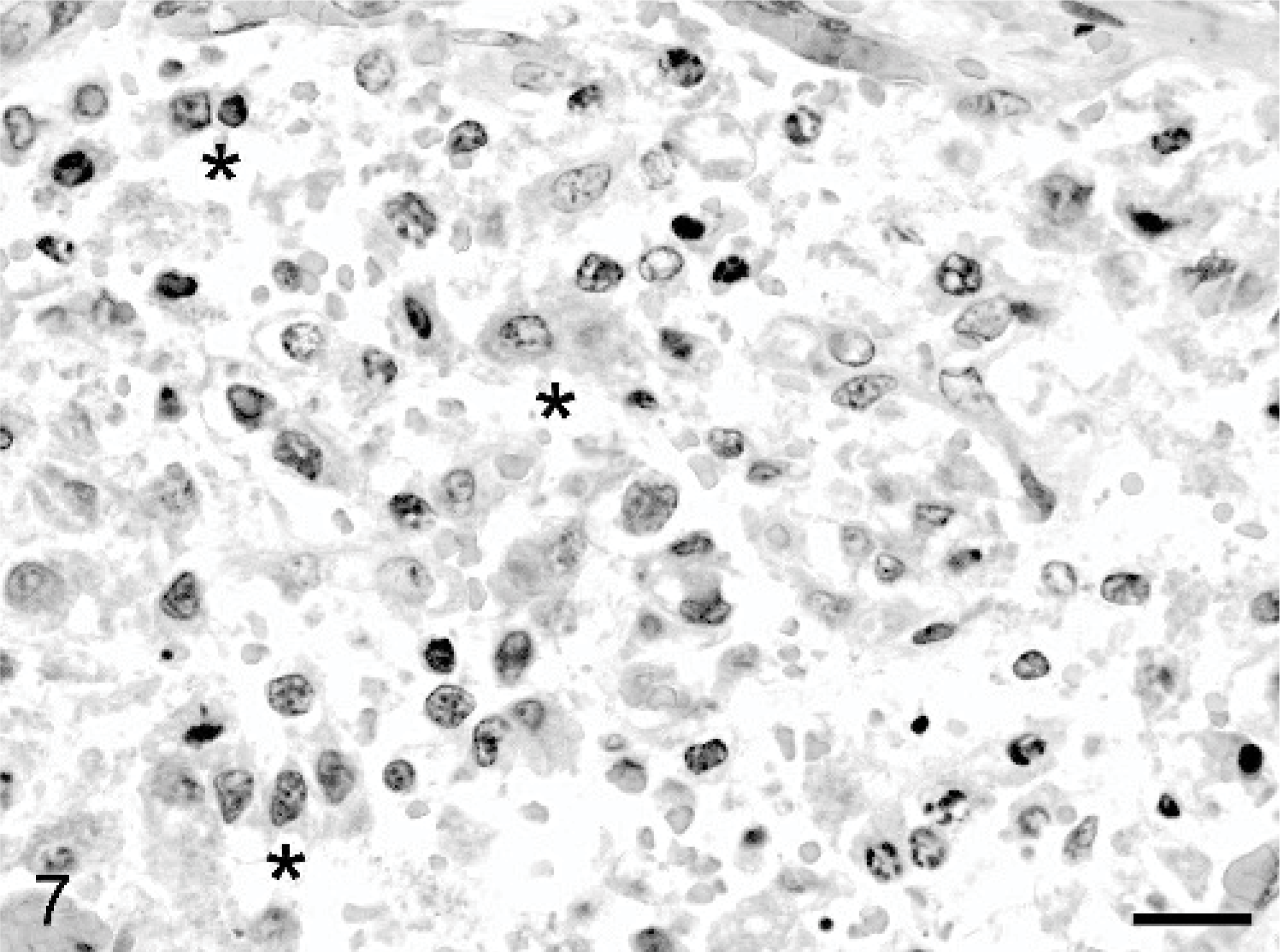
Dermal mass, Thoroughbred neonate, case #2. The mass is composed of haphazardly arranged cords and individual fetal and embryonal epithelial cells. The neoplastic epithelial cells occasionally form sinusoid-like spaces (∗) that contain variable numbers of erythrocytes. HE. Bar = 20 μm.
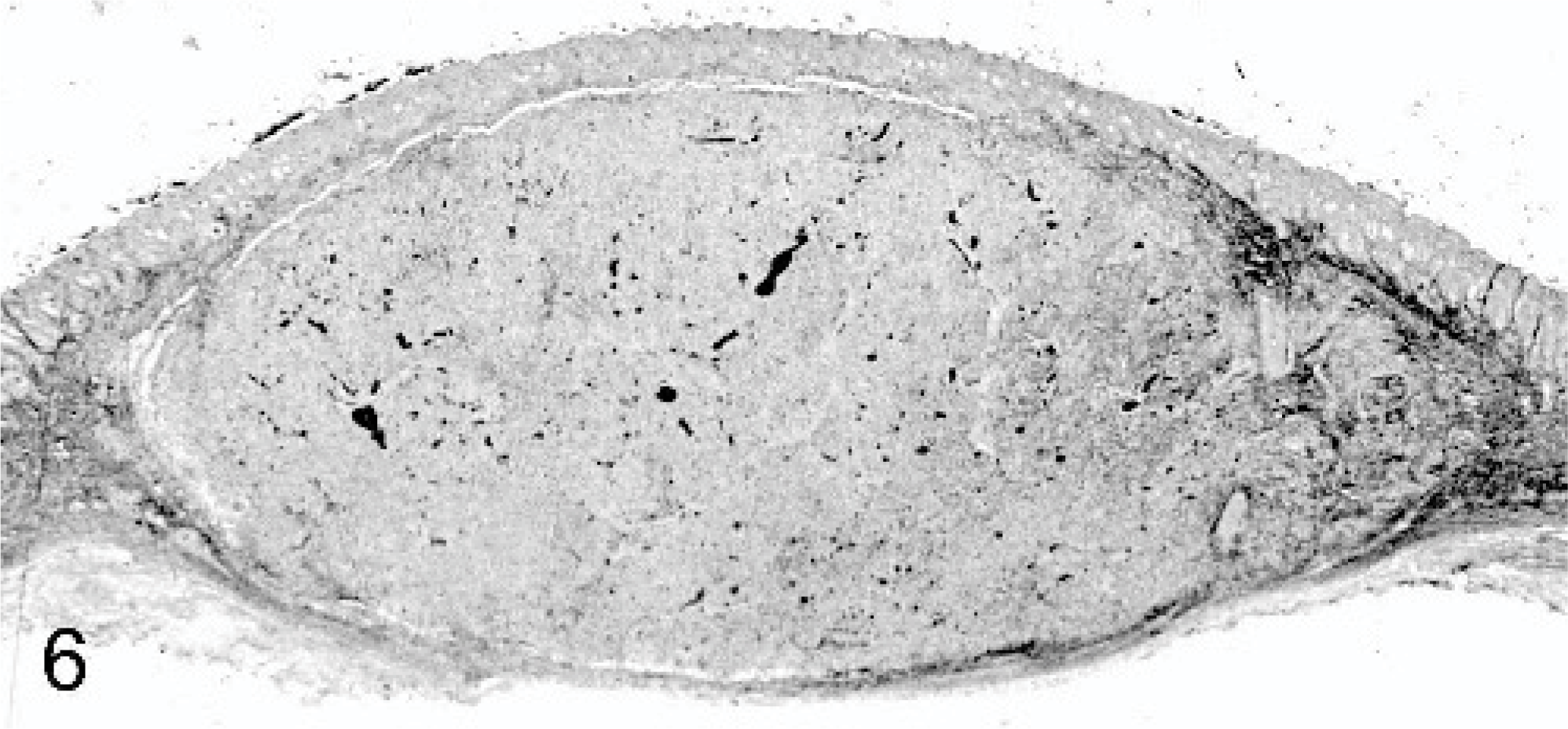
Dermal mass, Thoroughbred neonate, case #2. An encapsulated, well-demarcated, and exophytic mass abuts the epidermis and extends into the panniculus. HE.
Immunohistochemically, the neoplastic cells from cases #1, #2, and #3 stained similarly for AFP, CK, and vimentin. The cytoplasm of the neoplastic cells stained variably positive for AFP. Stromal spindle cells, mesenchymal spindle cells, and fetal epithelial cells stained variably positive for vimentin. Rows and cords of bile duct–like epithelial cells stained positive for CK and were located in the capsule, dense fibrous stroma and intermixed amongst the neoplastic cells. PAS staining, with and without diastase treatment, was similar in neoplastic cells of cases #1 and #2. Small intracytoplasmic vacuoles (cases #1 and #2) stained PAS positive without diastase treatment and negative with diastase treatment, suggesting the presence of glycogen. The neoplastic cells in case #3 stained negative for PAS. Intracytoplasmic vacuoles within neoplastic cells stained variably positive (case #2) or exclusively positive for lipid (case #3) with ORO.
Hepatoblastomas are potentially fatal malignant hepatic tumors of children. Both epithelial and mixed variants have been previously reported in horses.3,5,8,11 Although rare, the metastatic potential of hepatoblastomas have been described in both humans and animals.2,4,7,8,11 Human metastases occur most frequently to the lungs and rarely to the brain, skin, choroid, and iris.2,4,7,14 Metastasis in animals have been reported in the lung, tracheobronchial lymph node, mesenteric lymph nodes, omentum, cerebral blood vessels, and myocardial blood vessels.8,11 Metastases were not evident in cases #1 and #3, but case #2 had previously unreported metastatic lesions in brain, bone, and skin.
The fetal hepatic mass (case #1) was diagnosed as a solid mixed hepatoblastoma with teratoid features. The mass had 2 epithelial cell populations consisting of both fetal and embryonal epithelial cell types. Intermixed within the epithelial cells were minimal amounts of mineral, osteoid, cartilage, and bone formation, and large amounts of hemorrhage, extramedullary hematopoiesis, and necrosis. Only a single case of hepatoblastoma has been previously reported in an equine fetus.8 Unlike the fetal case reported by Neu in 1993, the tumor in case #1 had teratoid features, was solitary, lacked metastases, and had a minimal population of embryonal epithelial cells.
To date, only a single case of neonatal hepatoblastoma has been described in animals, which involved an alpaca cria.15 Cases #2 and #3 described mixed hepatoblastomas with teratoid features in equine neonates. Case #2 had multiple metastases to the bone, brain, meninges, and skin. Unlike the hepatic mass, metastases were composed only of fetal and embryonal epithelial cells and varied in invasiveness dependant on their location of metastasis. Case #3 described a mixed hepatoblastoma with teratoid features in a neonatal equine, which was euthanized for an unrelated congenital condition. Excluding the horse's age, case #3 was similar to that reported by Cantile et al. in 2000, which described a mixed hepatoblastoma with mesenchymal components in a foal.3
Immunohistochemistry and other special stains have been utilized to better characterize hepatoblastomas in both humans and animals.1,3,5,13,15 However, results can be inconsistent. These staining inconsistencies may be caused by variable epithelial, mesenchymal, and teratoid cell populations, variable intracytoplasmic constituents, such as lipid and glycogen, or cellular anaplasia.1,3,5,9,13,15 AFP is traditionally the most consistent marker found in hepatoblastomas.1,3,5,13,15 The staining characteristics reported here were consistent with those previously reported for hepatoblastomas. The primary hepatic tumors (cases #1, #2, and #3) and metastases (case #2) stained variably positive for AFP, CK, vimentin, and demonstrated variable amounts of lipid (cases #2 and #3) and glycogen (cases #1 and #2).
This report describes the gross and histopathologic findings of 3 rare cases of equine hepatoblastoma with teratoid features. Theses mixed hepatoblastomas are the first to report teratoid features in an equine fetus (case #1) or neonate (cases #2 and #3). Additionally, case #2 was the first report in animals to describe a hepatoblastoma with brain, bone, and skin metastases.
Footnotes
Acknowledgements
We thank Drs. Lenn Harrison and Neil Williams for reviewing the manuscript. This report was published (06-14-064) with the permission of the Dean and Director of the Kentucky Agriculture Experiment Station and College of Agriculture, University of Kentucky, Lexington.
