Abstract
Ganglioneuromas are complex tumors that arise in peripheral ganglia and are composed of well-differentiated neurons, nerve processes, Schwann cells, and enteric glial cells. The term ganglioneuromatosis (GN) denotes a regional or segmental proliferation of ganglioneuromatous tissue. This report describes an 8-year-old mixed breed horse with GN in a 25-cm segment of small colon. Grossly, the lesion consisted of numerous sessile to pedunculated nodules extending from the serosal surface. Histologic examination revealed the nodules to consist of fascicles of spindle-shaped cells consistent with Schwann cells, clusters of neurons, supporting enteric glial cells, and thick bands of perineurial collagen. Most of the nodules coincided with the location of the myenteric plexus and extended through the outer layer of the tunica muscularis to the serosal surface. Neuronal processes were demonstrated within the lesion with electron microscopy. With immunohistochemistry neurons were positive for neuron specific enolase (NSE) and S-100 and the Schwann cells and enteric glial cells were positive for S-100 and glial fibrillary acidic protein (GFAP). The pathogenesis of GN is poorly understood. GN, although rare, should be included in the differential diagnosis of gastrointestinal tumors in the horse.
Tumors derived from autonomic ganglia include ganglioneuromas, ganglioneuroblastomas, and neuroblastomas.6,14 Ganglioneuromas are benign tumors that are composed of a mixture of well-differentiated neurons and abundant stroma containing nerve processes and Schwann cells. Neuroblastomas are malignant neoplasms with nests of poorly differentiated neuroblastic cells and minimal stroma. Tumors intermediate in differentiation and behavior between ganglioneuromas and neuroblastomas are called ganglioneuroblastomas. Ganglioneuromas arising from enteric ganglia have been reported in humans and a variety of domestic animal species, including dogs,5,10,11,16 cats,9 pigs,15 and cattle.2 The literature includes a single report of an intestinal ganglioneuroma in the horse.1 This case involved a solitary tumor in the small intestine that caused a physical obstruction. The term ganglioneuromatosis has been used to describe enteric ganglioneuromas that are poorly demarcated, affect a segmental area of bowel, and appear to arise from more than one site simultaneously.2,5,6 Here, we describe a case of a horse with ganglioneuromatosis in the small colon.
An 8-year-old, mixed breed gelding with a history of chronic upward fixation of the patella was donated to the Texas A&M University Veterinary Medical Teaching Hospital. The patellar problem was deemed inoperable, and the horse was euthanized and necropsied. At necropsy, numerous sessile to pedunculated, firm nodules were found on the antimesenteric wall of a 35-cm segment of small colon, 40 cm from the anus (Fig. 1). The nodules were multifocally adherent to the mesocolon. They ranged in size from 3 mm to 1.5 cm in diameter. Most of the smaller nodules were white, and most of the larger nodules were purple externally and dark red internally. The tumors appeared to be arising within the tunica muscularis (Fig. 2). The bowel wall adjacent to some of the nodules was thickened. The mucosa subjacent to the nodules had 2–4-mm depressions. No other masses were found in the horse.
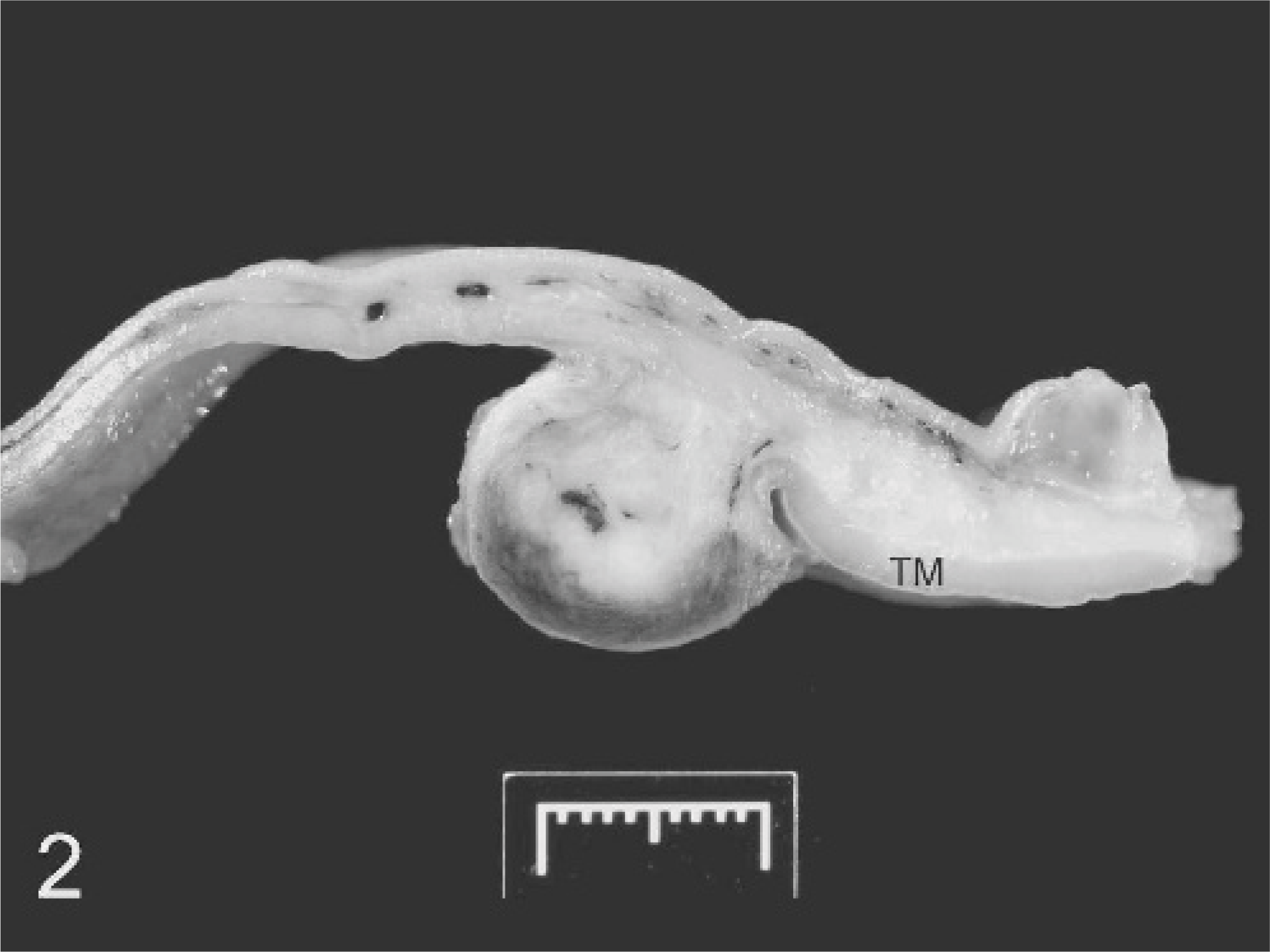
Small colon; horse. Cross-section of fixed small colon showing nodule arising in the areas of the myenteric plexus and extending through the outer layer of the tunica muscularis (TM). The thickened area of bowel wall adjacent to the tumor is caused by small islands of neoplastic tissue infiltrating the submucosa and inner layer of the tunica muscularis.
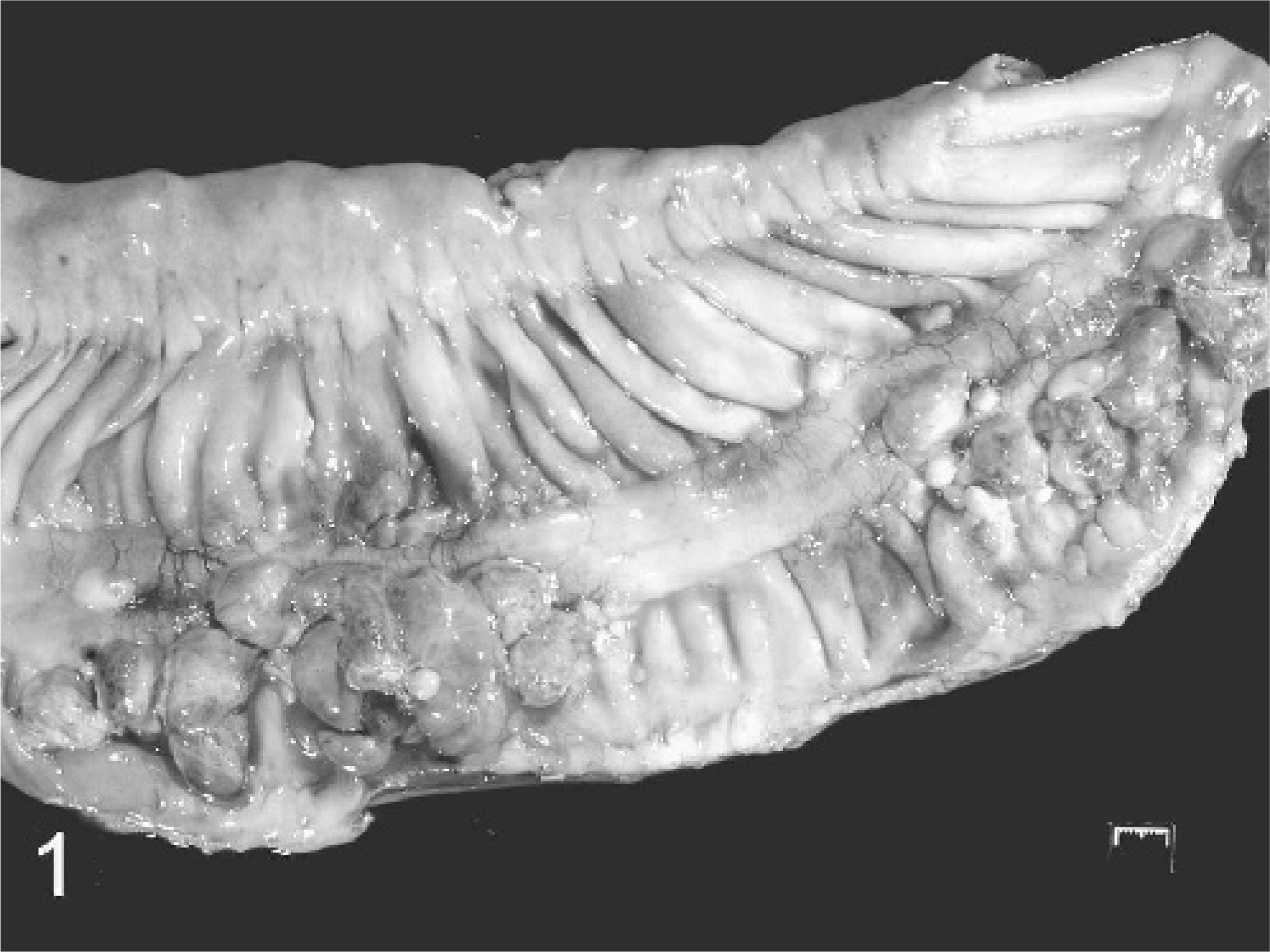
Small colon; horse. Multilobular nodules are extending from the serosal surface within a 35-cm segment of the small colon.
Samples of small colon were immersed in 10% formalin and processed for routine histopathology. In addition to hematoxylin and eosin staining, sections of the colonic nodules were stained with Masson trichrome, Holmes, Bielschowsky, and Luxol fast blue stains. Selected sections were stained with the strepavidin-biotin-immunoperoxidase method using the following primary antibodies: S-100 protein (Dako Corp., Carpinteria, CA; rabbit polyclonal), neuron specific enolase (Dako; mouse monoclonal), and glial fibrillary acidic protein (Dako; rabbit polyclonal). All 3 antibodies were used at a 1:1,000 dilution. Brain tissue was used for the positive control, and the primary antibodies were excluded for the negative control. Samples were processed for transmission electron microscopy according to standard protocols. Briefly, small segments (1–2 mm3) of the formalin-fixed tissue were fixed for an additional 4 hours in 2% glutaraldehyde, postfixed in 1% osmium tetroxide with 0.5% potassium ferrocyanide in 0.1M cacodylate buffer for 1 hour, dehydrated in a graded alcohol series, and embedded in Epon/Araldite resin mixture. Ultrathin sections were cut, poststained with uranyl acetate and lead citrate, and examined with an electron microscope.
Histologic examination of the small colon revealed multiple poorly demarcated nodules that appeared to arise in the area of the myenteric plexus and extend through the outer layer of the tunica muscularis. In some areas, the overlying serosa remained intact. The nodules were composed of prominent interweaving fascicles of spindle-shaped mesenchymal cells with elongated, thin, wavy nuclei, morphologically consistent with Schwann cells (Fig. 3). The cells had minimal cytoplasm and nuclei with finely stippled chromatin. Occasional clusters of well-differentiated neuronal cell bodies were present within the nodules (Fig. 4). The neuronal cell bodies were 10–20 μm in diameter, had a moderate amount of cytoplasm containing Nissl substance, and had eccentric nuclei. The neurons were surrounded by scattered supporting cells with inconspicuous cytoplasm and small hyperchromatic nuclei. Based on morphology and immunohistochemical findings, these cells were interpreted as enteric glial cells. Mitotic figures were not evident among the spindle cell, neuron, or enteric glial cell populations. Trichrome staining revealed thick bands of collagen with spindle-shaped fibroblasts in both the tunica muscularis and submucosa, separating the tumor fascicles and neuronal cell bodies. Thin strands of collagen were also present within many of the tumor fascicles. Isolated from the grossly visible nodules, smaller islets of proliferating nerve tissue were present in both layers of the tunica muscularis and to a lesser extent in the submucosa. The mucosa was normal in appearance. Holmes and Bielschowsky stains failed to reveal argyrophilic filaments (axons) within the nerve fascicles, and Luxol fast blue staining failed to reveal the presence of myelin. With immunohistochemistry, Schwann cells, enteric glial cells, and neuronal cell bodies stained positive for S-100 protein. Neuron specific enolase (NSE) consistently stained neuronal cell bodies, and glial fibrillary acidic protein (GFAP) stained some Schwann cells and most enteric glial cells. Electron microscopy of the tumor confirmed the presence of nerve processes interspersed among bundles of collagen fibers (Fig. 5). These processes were unmyelinated and contained large numbers of intermediate filaments (neurofilaments) and a few microtubule profiles. The processes also contained numerous membrane-bound structures consistent with synaptic vesicles.

Small colon; horse. Ultrastructural examination of one of the nodules reveals neuronal processes (outlined by arrowheads) and accumulations of collagen fibrils (arrow). Electron micrograph. Bar = 1 μm.
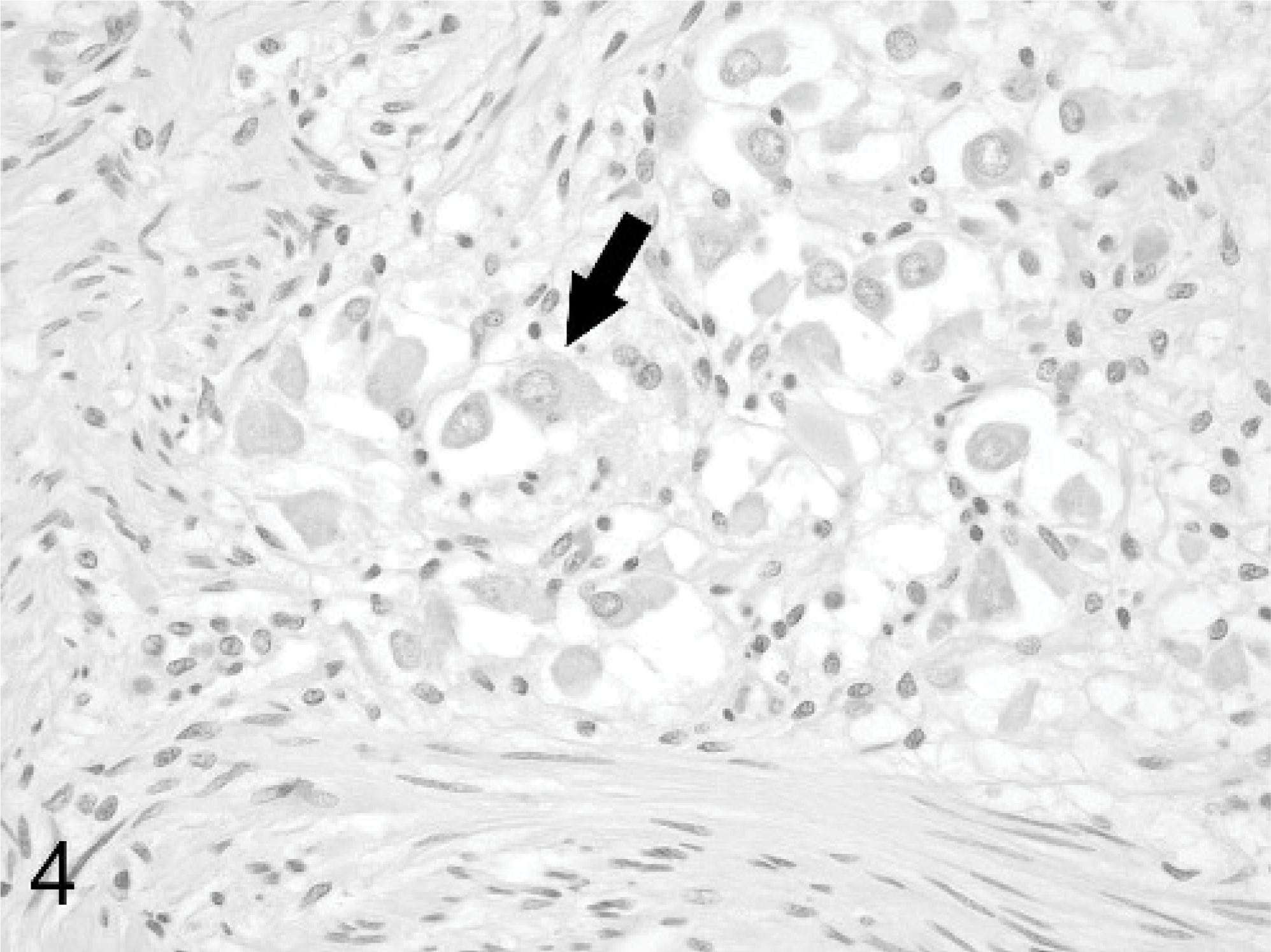
Small colon; horse. Occasional clusters of well-differentiated neuronal cell bodies (arrow) are present within the nodules. HE stain.
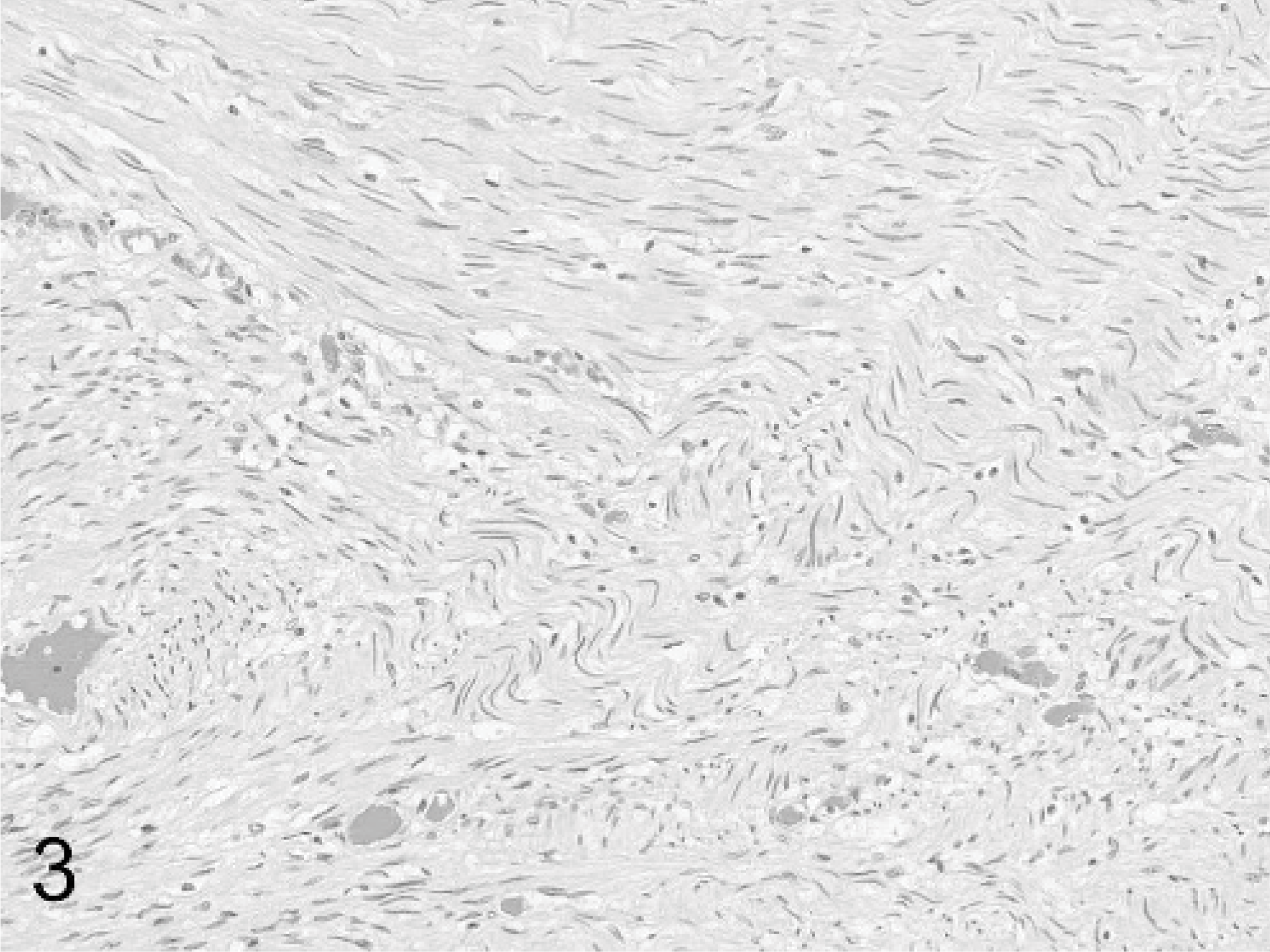
Small colon; horse. The bulk of the tumors consist of interweaving fascicles of spindle-shaped cells with elongated nuclei morphologically consistent with Schwann cells. HE stain.
Although human ganglioneuromas can arise anywhere within the autonomic nervous system, they are most commonly found retroperitoneally, in the posterior mediastinum, and in the adrenal gland.17 Ganglioneuromas of the gastrointestinal tract are rare and can be either single or multiple.6 The lesions are located in the mucosa and/or submucosa, are not associated with systemic disease, and have a good prognosis.6,13 Diffuse or intestinal ganglioneuromatosis (GN) is considered a separate entity, characterized by infiltration of the bowel wall by poorly demarcated, proliferative ganglioneuromatous tissue.6 Such lesions result in a stricture or nodular mass that affects bowel segments up to 20 cm in length. The lesions are often centered on the myenteric plexuses but can become transmural and often involve the mucosa.13 GN is often associated with multiple tumor syndromes such as multiple endocrine neoplasia (MEN) type IIb, von Recklinghausen's disease (neurofibromatosis 1), or Cowden's disease and results in clinical signs such as abdominal distension, constipation, megacolon, and diarrhea.3,12,13 MEN IIb patients with GN often develop mucosal neuromas of the lips and tongue, medullary thyroid carcinoma, and pheochromocytoma. The GN often precedes these lesions.12,13
Findings in the horse of the current case support designating the lesions as GN. The proliferative tissue was located in a segmental area of the small colon, was poorly demarcated, and appeared to be arising at multiple independent sites. Differences between this case and GN in humans include a lack of mucosal involvement, no history of gastrointestinal disturbance, and no clinical or gross evidence of a multiple tumor syndrome such as MEN. Endocrine disease cannot be ruled out conclusively, because histopathology of endocrine organs was not performed. The immunohistochemical staining in this case showed similarities to that described in human cases of GN, with neuronal cell bodies staining with NSE and Schwann cells staining for S-100 and GFAP.3,13 Both Schwann cells and enteric glial cells have been shown to express GFAP;7 therefore, the GFAP positivity of these cell types within the lesion would be expected.
The other report of ganglioneuroma in a horse described a solitary lesion in the jejunum that reduced the luminal diameter and resulted in a strangulating lesion. Histologically, the tumor was partially encapsulated and primarily involved the tunica muscularis.1 At least 2 of the ganglioneuromatous lesions described in the veterinary literature are consistent with GN, i.e., segmental lesions with numerous, poorly circumscribed growths. One report describes a 12-month-old Lhasa Apso dog with GN in a 15-cm segment of the distal colon.5 The other report is of a 7-month-old Hereford-Angus steer with a 30-cm segment of affected descending colon.2 The lesion distribution and histologic appearance described in both cases closely resemble this equine case.
The pathogenesis of ganglioneuromas and GN is unclear. All of the components of the enteric nervous system (neurons, neuronal processes, Schwann cells, and enteric glial cells) are present in the lesions. For this reason, solitary ganglioneuromas may be best regarded as hamartomas, or when located in the mucosa, as choristomas.13 With its more diffuse nature, GN may represent hyperplasia of enteric nervous system elements in response to overproduction of a nerve growth factor.4 An immunohistochemical and electron microscopic study of human cases suggested that GN is a complex hyperplasia of several peptidergic, cholinergic, and adrenergic nerve fibers, not overgrowth of a single fiber type.3 Genetic mutations have been documented in the multiple tumor syndromes with which GN has been associated. MEN IIb, for example, is caused by activating mutations in the RET proto-oncogene, which is important in signaling for cell growth and differentiation of tissues derived from neural crest cells.8 Whether similar mutations are involved in animal cases of GN remains to be determined. It is notable that none of the reported cases of gastrointestinal ganglioneuroma or GN in animals describe tumors in other organs.
The GN in this horse was considered an incidental finding, because the horse was not showing clinical evidence of gastrointestinal disease. With time, the lesion might have progressed and eventually resulted in clinical signs. Although a rare condition, ganglioneuromatosis should be included in the differential diagnosis for nodular intestinal lesions in the horse.
Footnotes
Acknowledgements
We thank Rosemary Volmer for help with histochemical stains and Dr. Andy Ambrus for assistance with immunohistochemistry.
