Abstract
A 5-day-old quarter horse colt with a history of hypothermia, agonal breathing, and diarrhea was euthanized. At necropsy, numerous slightly raised, discrete, closely approximated submucosal nodules were observed in the colon and small intestine. Histologically, these nodules were composed of expanded submucosal mesenchyme that contained numerous neurons either individually or in ganglia. Thirty-two percent of these ganglia included 8 or more neurons, in contrast to 6% in an age-matched foal. Some nodules had necrosuppurative inflammation with vasculitis, thrombosis, and bacterial colonization. A few heterotopic neurons were randomly distributed in the mucosa and the muscularis mucosa. Histologic changes were most consistent with intestinal neuronal dysplasia, a disease of the submucosal plexus described in humans.
Intestinal neuronal dysplasia (IND), also known as neuronal intestinal dysplasia (NID), an idiopathic human intestinal motility disorder, is clinically manifested by constipation, abdominal distension, and overflow incontinence. The disease may involve the colon, the rectum, or the entire intestinal tract. IND is classified into 2 distinct entities: a less common type A (IND A), characterized by congenital aplasia or hypoplasia of the sympathetic innervation of the colon with functional bowel obstruction soon after birth, and a more common type B (IND B), classified as an innervation defect of the parasympathetic submucosal (Meissner’s) plexus. 10 Since its initial description, the diagnostic criteria for type B have been altered several times. Current criteria for IND B include (1) evaluation of a minimum of 25 submucosal ganglia, at least 20% of which must be giant ganglia containing 8 or more neuronal cell bodies, and (2) heterotopic distribution of neuronal cell bodies in the mucosa and muscularis mucosa. 11 The submucosal plexus appears early in development from primitive neuroblasts derived from the neural crest. The neuroblasts migrate by a poorly understood mechanism through the mesenchyme surrounding the developing gut. However, it is unclear whether these progenitor cells are programmed to migrate to the final site of maturation or are attracted by unknown substances. 1 The pathogenesis of IND B probably involves a defect in the migration or subsequent maturation of these ganglion progenitor cells. To our knowledge, IND B has not been described in nonhuman animals. Here, we describe a foal with a form of dysganglionosis very similar to human IND B.
Clinical History and Necropsy Findings
A 5-day-old quarter horse colt was presented with a history of lethargy, hypothermia, and diarrhea. Upon clinical examination, the foal had undetectable corneal or palpebral reflexes, erratic heart rate, and agonal breathing. Treatment was unsuccessful, and the foal was euthanized. At necropsy, numerous pale tan to red, firm nodules, 0.5 to 1.5 cm in diameter, were densely scattered through the submucosa of the right ventral colon, elevating the overlying mucosa (Fig. 1). Ventral and dorsal colonic mucosa was diffusely dark red to purple and edematous. The mucosa and serosa of the aborad jejunum were dark red to purple, and similar nodules were evident in jejunal submucosa. Subscapularly and extending into the hepatic parenchyma were multiple discrete, pale tan, firm, circular areas.
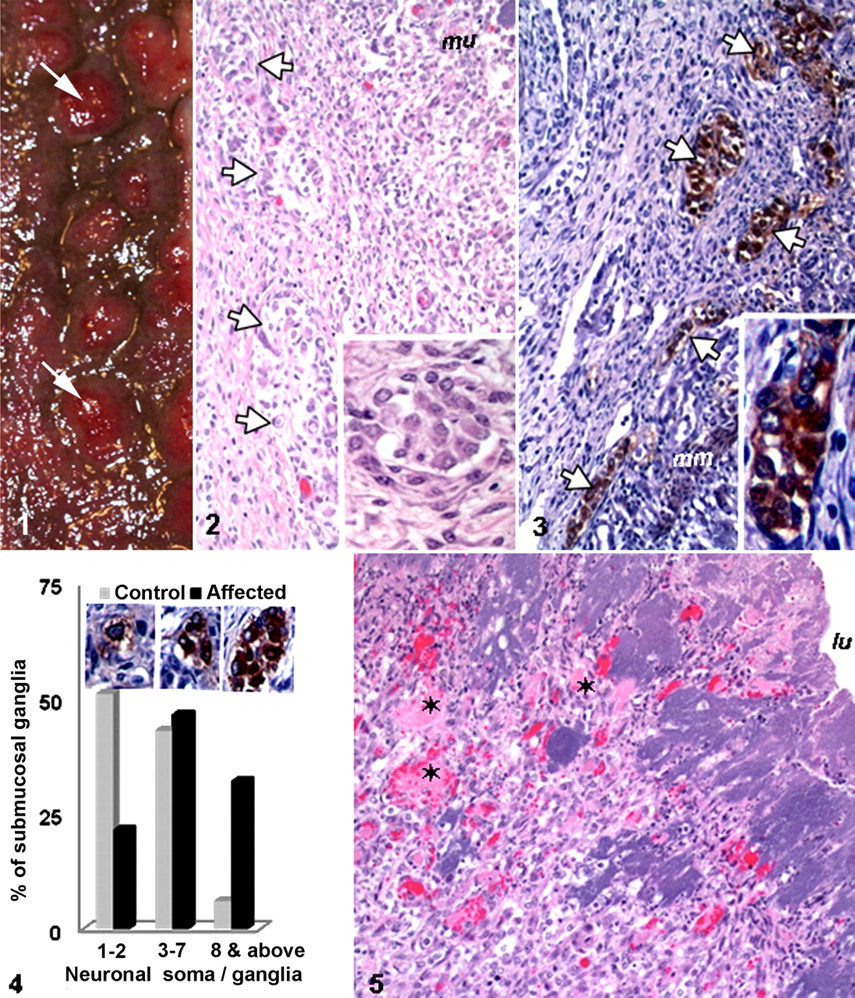
Histologic Examination
Tissue samples were fixed in 10% phosphate-buffered formalin, embedded in paraffin, sectioned at 4 to 5 μm, and stained with hematoxylin and eosin (HE). Immunohistochemistry on paraffin sections was performed with primary antibodies directed against neuron-specific enolase (NSE; DAKO, Carpinteria, CA), smooth muscle actin (SMA; DAKO), synaptophysin (DAKO), and nonphosphorylated neurofilament (Cell Marque Corporation, Rocklin, CA). The procedure used a labeled streptavidin-biotin system (LSAB2 System-HRP, DakoCytomation, Carpinteria, CA), modified as described. 6 Briefly, the sections were incubated with the primary antibody for 60 minutes at room temperature. The biotinylated secondary antibody was applied and incubated for 20 minutes. The antigen-antibody complexes were visualized using 3,3′-diaminobenzidine (DakoCytomation). Substitution of primary antibody with normal mouse immunoglobulin in a parallel section served as the negative control. Additional sections of representative tissue on the slides served as the positive control.
Histologically, the colon had abundant submucosal fibrovascular stroma organized into nodules that elevated the overlying mucosa. Numerous neuronal cell bodies arrayed singly or as ganglia within this fibrovascular stroma were surrounded by plump fibroblasts and a moderate number of lymphocytes, macrophages, and neutrophils (Fig. 2). Sections of ventral colon were evaluated immunohistochemically for neuron-specific enolase, nonphosphorylated neurofilament, and synaptophysin. These markers identified neuronal cell bodies in submucosal and myenteric plexuses and nerve fibers. Cytoplasmic labeling of NSE and neurofilaments was diffuse and homogeneous; that of synaptophysin was coarsely granular (Fig. 3). Two hundred neuronal cell bodies or ganglia were evaluated in 4 colonic sections with immunohistochemistry for NSE; colonic sections from an age-matched foal that died without intestinal disease were used for comparison and served as control (Fig. 4). The submucosal ganglia were composed of 2 to 69 neuronal cell bodies; 32% of the ganglia (vs 6% in the “control” foal) were classified as giant ganglia by the presence of 8 or more neuronal cell bodies. Most neuronal somata in the control foal were dispersed as individual cells or pairs. The myenteric plexuses were moderately hypercellular compared with those in the control foal. A few labeled neuronal cell bodies in the lamina propria were consistent with heterotopic distribution. Scattered neuronal somata in these areas also reacted with the antibody to synaptophysin.
Multifocally, the mucosa of the nodular and internodular areas was ulcerated and contained numerous degenerated neutrophils, cellular debris, fibrin, erythrocytes, and prominent colonies of coccobacilli (Fig. 5). Bacterial culture of intestine resulted in isolation of Streptococcus equi and Actinobacillus species. Many mucosal capillaries contained fibrin thrombi. A few submucosal blood vessels had mural fibrinoid change and contained fibrin thrombi. The hepatic lesions were acute, embolic necrosuppurative hepatitis with necrotizing vasculitis, thrombosis, and colonies of cocci. Diffusely, the hepatocytes had cytoplasmic lipid vacuoles.
Discussion
Intestinal neuronal dysplasia is a poorly understood colonic motility disorder of the enteric nervous system. 8 Most IND cases are classified as type B, which is characterized by the presence of numerous giant ganglia in the submucosa and intestinal dysmotility. The diagnosis is based on biopsy findings and the associated motility dysfunction. 12,15 An increased number of submucosal ganglia and the presence of numerous giant ganglia observed in this foal are similar to findings in human IND B. In addition to the increased size and number of submucosal ganglia, neurons were found in abnormal locations. Such neuronal heterotopia has been observed in IND B. 9 The myenteric plexuses were also hypercellular in this foal. Although myenteric plexus involvement is not requisite for a diagnosis of IND B, hypercellularity of the myenteric plexus was described in a recent human case. 7
The lesions in this foal must be differentiated from ganglioneuromatosis, which is an overgrowth of neural elements arising from autonomic ganglia. 3 –5,13 Ganglioneuromas contain all the components of the enteric nervous system, including neuronal cell bodies, neuronal processes, enteric glial cells, and Schwann cells, and may be best regarded as hamartomatous rather than neoplastic. The primary lesion in this foal was an increased number of neuronal somata plus some fibroblasts and leukocytes but not glial cells or nerve fibers. Therefore, the lesion does not resemble ganglioneuromatosis. The changes in the colonic plexuses of this foal are considered dysplastic; thus, the term dysganglionosis was chosen. Unfortunately, the jejunum and other parts of the gastrointestinal tract were not examined microscopically. However, the gross lesions in the aborad jejunum closely resembled those of the colon.
It is believed that IND B is related to premature expression of laminin A during embryogenesis, which results in premature differentiation of nerve cells in the myenteric and submucosal plexuses and intercepts neuroblast colonization in the rectum. 9 To understand the mechanism of neuronal hyperplasia, Shirasawa et al 14 deleted Enx (Hox11L1), a homeobox gene, which resulted in neuronal hyperplasia in the myenteric plexus, increased number of neuronal cell bodies in ganglia, and massive distension of the colon (megacolon), suggesting that Enx may play a role in IND. The SPRY2 or SPROUTY2 gene encodes a protein that belongs to a conserved family of inducible inhibitors of tyrosine kinase signaling. 2,16 A recent study to define the physiological function of mammalian SPROUTY2 in mice through deletion resulted in enteric nerve hyperplasia that led to esophageal achalasia and intestinal pseudoobstruction. 16 This observation suggests a possible role of SPROUTY2 in IND as well as in other intestinal motility disorders. However, mutational analysis of the SPROUTY2 gene in IND patients has failed to demonstrate its role in the pathogenesis of the disease. 2
It is unclear in this case whether the enteric dysganglionosis was an incidental lesion or a predisposing factor to bacterial colitis. Considering that dysganglionosis could have altered intestinal motility, a key factor in intestinal health, it is tempting to speculate that enteric nervous system dysregulation could have predisposed the foal to intestinal infection.
In conclusion, this report documents an equine case of IND B-like malformation of the enteric nervous system that may have altered intestinal motility, leading to bacterial colitis. Although seemingly rare, this disorder should be considered when evaluating foals with enteric disease.
Footnotes
Acknowledgements
We thank Ms Sarah Jones and her group for the preparation and staining of the tissue sections, Mr John Roths for photographic help, and Dr M. Libal and her group for help with bacteriology.
The author(s) declared no conflicts of interest with respect to the authorship and/or publication of this article.
The author(s) received no financial support for the research and/or authorship of this article.
