Abstract
Malignant fibrous histiocytoma (MFH) is regarded as soft tissue-derived undifferentiated pleomorphic sarcoma, of which the histogenesis remains to be proven. To investigate the cellular characteristics, a homotransplantable tumor line (KJ) was established from a spontaneous MFH that developed in the subcutis of an aged F344 rat. KJ tumors have been produced in syngeneic rats by serial subcutaneous implantation of tissue fragments. The original and KJ tumors consisted of oval and fusiform cells arranged in interlacing bundles with fibrous stroma. Occasional giant cells with bizarre nuclei were observed. Enzyme/immunohistochemically, neoplastic cells reacted to ED1 and ED2 (antibodies specific for rat histiocytes/macrophages), and showed a positive reaction to vimentin and lysosomal enzyme markers such as acid phosphatase (ACP) and nonspecific esterase (Non-SE). Electron microscopically, neoplastic cells possessed lysosomal granules in cytoplasm. A cloned cell line (KJ-A) was isolated from a KJ tumor. KJ-A cells showed positive reactions to ED1, ED2, ACP, and Non-SE, and had cytoplasmic lysosomal granules. Tumors induced by KJ-A cells exhibited histologic and enzyme/immunohistochemical findings similar to those of KJ tumors. Lipopolysaccharide (LPS) treatment increased the number of ED1-positive cells and the expression of tumor necrosis factor-α mRNA by reverse transcription-polymerase chain reaction. Collectively, it is likely that rat MFH cells originally possess histiocyte/macrophage-like features that may be enhanced by LPS. Because tumor lines are useful for in vivo and in vitro studies concerning different characteristics of the original neoplasms. KJ and KJ-A should prove useful for studies concerning the morphogenesis of MFH.
Keywords
The term malignant fibrous histiocytoma (MFH) refers to a group of soft tissue tumors consisting of a dual population of histiocytic and fibroblastic cells arranged in interlacing bundles or a storiform growth pattern.6,7,35 Although MFHs exhibit a predominantly fibroblastic appearance, it has been postulated that MFH might be derived from cells with a histiocytic nature that could assume the appearance and function of fibroblasts (facultative fibroblasts).7,15,35 However, there is another concept that MFH merely represents a morphologic pattern shared by a wide variety of poorly differentiated pleomorphic sarcomas;8,32 thus, the histogenesis and cellular nature of this tumor type remain to be proven.
In humans, MFHs have been regarded as the most common soft tissue sarcoma of late adult life.8,19,35 On the other hand, spontaneously occurring MFHs have rarely been detected in rats, dogs, cattle, and pigs,6,11,18,22,24,25,30 although chemically induced MFH-like sarcomas have been reported in rats.16,20,26 Cutaneous soft-tissue neoplasms in rats show heterogeneous histology, and it has therefore been difficult to characterize and interpret their cellular nature. F344 rats have been widely used as an experimental animal in carcinogenicity studies. Toxicologic pathologists who perform the evaluations in these studies require detailed knowledge concerning the characteristics of spontaneously occurring neoplastic lesions. Because F344 rats are an inbred strain with a genetically homogeneous nature, we have utilized this strain to establish tumor lines (homotransplantable and cultured cell lines) from spontaneous tumors,33,37–40 in order to clarify the biologic behavior, morphogenesis, and paraneoplastic syndromes associated with these tumors. In the present study, we succeeded in establishing a homotransplantable tumor line (KJ) in syngeneic rats from a spontaneous MFH found in the subcutaneous tissue of an aged male F344 rat. Furthermore, we established an in vitro cell line (KJ-P) from a KJ tumor, and then isolated a cloned cell line (KJ-A) from KJ-P. Histologically, the primary tumor was first regarded as a fibrosarcoma, because the tumor exhibited histology of interlacing bundles supported by collagenous stroma. However, the electron microscopic and enzyme/immunohistochemical analyses for the primary tumor and the derivatives (KJ, KJ-P, and KJ-A) revealed that neoplastic cells possessed histiocytic characteristics. This report describes morphologic and cellular characteristics of the original tumor, KJ tumors, and cultured cells of KJ-P and KJ-A.
Materials and Methods
Homotransplantable tumor line KJ in vivo and in vitro cell lines KJ-P and KJ-A
Immediately after the removal of the original tumor, a tissue fragment, 2 mm in diameter, was cut and implanted subcutaneously into syngeneic F344 rats (Charles River Japan, Shiga, Japan) through a trocar under light anesthesia with ether. Serial transplantations then were made at 4- to 5-week intervals until the fourth generation by implanting a tumor tissue fragment.37,38,40 In each passage, 2 or 3 F344 rats were used. The homotransplantable tumor line was named KJ.
Tissue fragments from KJ tumors at the fourth passage were aseptically dispersed and incubated in tissue culture flasks at 37°C in a humidified 5% CO2 atmosphere. The growth medium was Eagle's minimum essential medium (MEM; Nissui, Tokyo, Japan) containing 10% fetal bovine serum (Bioserum, UBL, Japan), 0.03% L-glutamine (Nissui), penicillin (100 U/ml), and streptomycin (100 μg/ml). A cell line established from a KJ tumor was named KJ-P, and was subcultured until the 40th generation. The KJ-P at the 20th generation was cloned twice using a limiting dilution technique,33,40 and a cloned cell line (named KJ-A) was isolated. To examine tumorigenicity, 7 × 106 cells of KJ-P or KJ-A were inoculated subcutaneously into syngeneic F344 rats.
The experiment was in compliance with institutional guidelines for animal care. All rats used in this study were euthanatized by exsanguination under ether anesthesia.
In vivo observations
The primary tumor and KJ tumors, as well as tumors induced in syngeneic rats by KJ-P or KJ-A cells, were fixed in 10% neutral buffered formalin and Zamboni's solution (0.21% picric acid, 2% paraformaldehyde, and 130 mM phosphate buffer, pH 7.4).38,40 Tissue sections were stained by hematoxylin and eosin (HE), periodic acid–Schiff (PAS), Watanabe's silver impregnation, azan-Mallory, alcian blue (pH 2.5) and phosphotungustic acid hematoxylin (PTAH) methods. Frozen sections from the formalin-fixed tissues were stained with oil red O.
The immunohistochemical analyses were performed by the avidin-biotin complex (ABC) method (Vectastain ABC Kit; Vector Laboratories, Inc., Burlingame, CA, USA) with primary antibodies described as follows (Table 1). Formalin-fixed, deparaffinized sections were used for cytokeratin (Dako Corp., Carpinteria, CA, USA; monoclonal antibody; predilution), vimentin (Dako; monoclonal antibody; × 200), desmin (Dako; monoclonal antibody; predilution), α-smooth muscle actin (α-SMA; Dako; monoclonal antibody; × 100), S-100 protein (Dako; polyclonal antibody; × 200), and ED1 (Chemicon International Inc., Temecule, CA, USA; × 500). Zamboni's solution–fixed, deparaffinized sections were used for ED2 (Serotec Ltd., Oxford, UK; monoclonal antibody; × 500) and OX6 (Serotec; monoclonal antibody; × 500). After deparaffinization, sections for α-SMA, S-100 protein, ED1, ED2, OX6, and lysozyme immunohistochemistry were treated with 0.1% trypsin for 15 minutes, and those for cytokeratin, vimentin, and desmin immunohistochemistry were treated by microwave for 5 minutes; these sections were then incubated with 3% H2O2 for 10 minutes to quench endogenous peroxidase. Thereafter, the sections were treated with 1.5% nonfat milk for 45 minutes at room temperature, and then incubated with each primary antibody for 14 hours at 4°C. A 30-minute incubation with biotinylated goat anti-mouse antibody (for monoclonal antibodies) or with biotinylated goat anti-rabbit antibody (for polyclonal antibody) followed at room temperature. Final incubation was carried out for 30 minutes with an avidin–horseradish peroxidase complex, and positive reactions were visualized with 3,3′-diaminobenzidine. Sections were counterstained lightly with hematoxylin. Nonimmunized mouse or rabbit serum was used for negative controls. Rat normal epidermis (for cytokeratin), subcutaneous vascular smooth muscles (for vimentin, α-SMA, and desmin), sciatic nerve (for S-100 protein), and splenic macrophages (for ED1, ED2, and OX6) were used as positive controls. In rat lesions, ED1 is used for detection of exudate macrophages, and ED2 labels histocytes (tissue macrophages).3,5,12,36 OX6 stains dendritic cells expressing rat major histocompatibility complex (MHC) class II (Ia) antigens.36,38 For enzyme histochemistry, frozen sections from fresh tumor tissues were fixed in cold acetone, and then stained for acid phosphatase (ACP) by the Gomori's method (pH 5.0), nonspecific esterase (Non-SE) by the α-naphthyl acetate method (pH 7.4), and alkaline phosphatase (ALP) by the naphthol AS method (pH 9.0); these methods have been described previously.33,38,39 For electron microscopy, small blocks from tumors were fixed in 2.5% buffered glutaradehyde and postfixed in 1% buffered osmium tetroxide. The ultrathin sections from epoxy resin–embedded samples were stained with uranyl acetate and lead citrate, and examined under an electron microscope (Hitachi, H-7500, Tokyo, Japan) at 80 kV.
Enzyme/immunohistochemical staining results in the original tumor and the homotransplantable tumor (KJ), as well as in KJ-P and KJ-A cells and their induced tumors.∗
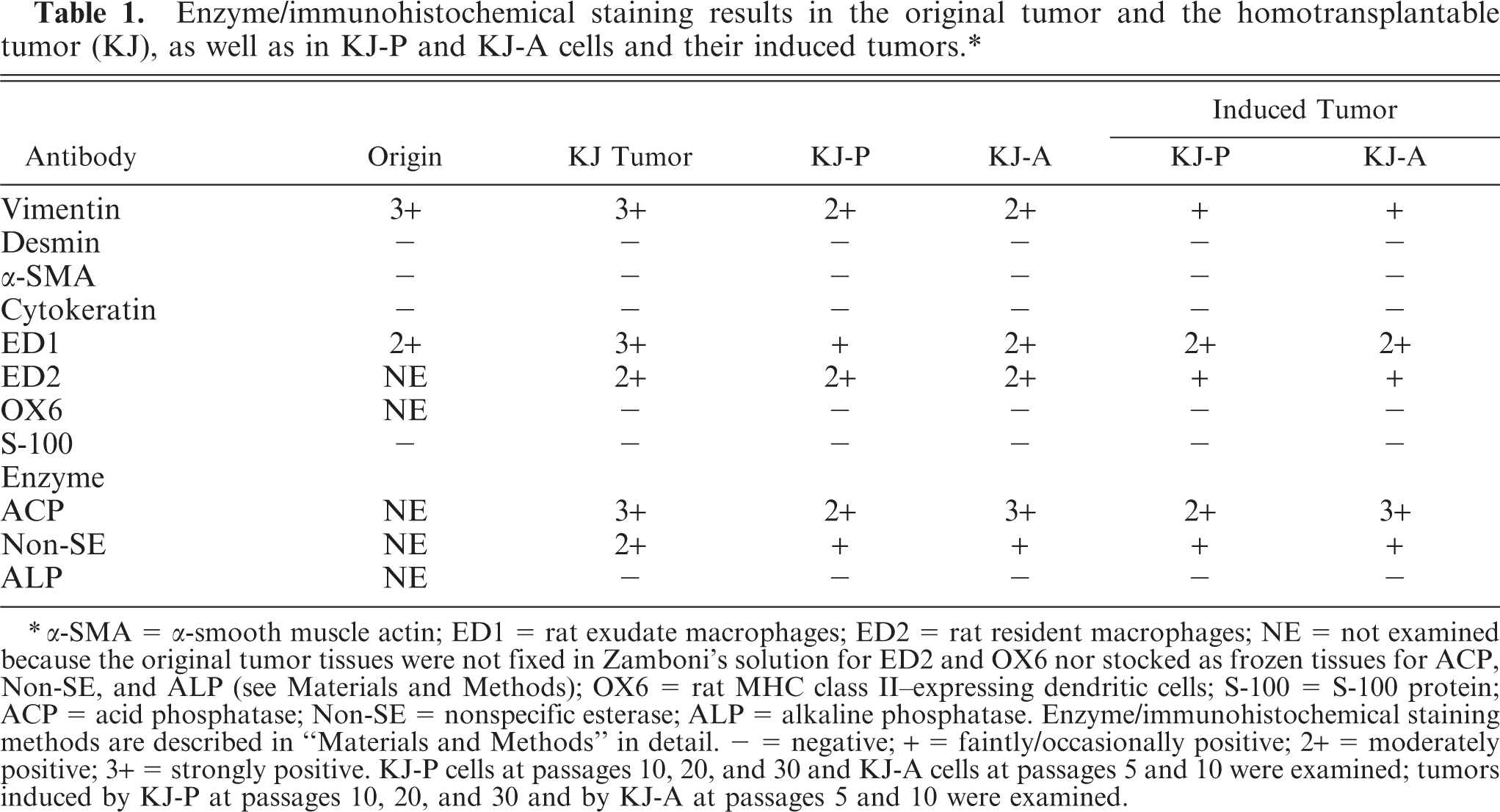
α-SMA = α-smooth muscle actin; ED1 = rat exudate macrophages; ED2 = rat resident macrophages; NE = not examined because the original tumor tissues were not fixed in Zamboni's solution for ED2 and OX6 nor stocked as frozen tissues for ACP, Non-SE, and ALP (see Materials and Methods); OX6 = rat MHC class II–expressing dendritic cells; S-100 = S-100 protein; ACP = acid phosphatase; Non-SE = nonspecific esterase; ALP = alkaline phosphatase. Enzyme/immunohistochemical staining methods are described in “Materials and Methods” in detail. − = negative; + = faintly/occasionally positive; 2+ = moderately positive; 3+ = strongly positive. KJ-P cells at passages 10, 20, and 30 and KJ-A cells at passages 5 and 10 were examined; tumors induced by KJ-P at passages 10, 20, and 30 and by KJ-A at passages 5 and 10 were examined.
In vitro observations
The following in vitro observations were conducted using KJ-P at the 10th, 20th, and 30th generations and KJ-A at the 5th and 15th generations. Doubling time was determined from viable cell numbers counted at 3 and 5 days after seeding 0.5 × 104 cells.33,37 Chromosome numbers were counted in 100 cells by methods described elsewhere.33,37 Cells grown on tissue culture glass slides were fixed in cold acetone for 5 minutes and stained for ACP, Non-SE, and ALP; the acetone-fixed cells were also stained immunocytochemically by the ABC method with the primary antibodies described above (Table 1). Cultured cells fixed in Bouin's solution were stained with HE for morphology. For electron microscopy, pelleted cells collected from KJ-P and KJ-A cultures were processed and examined using the same methods described above.
Effects of lipopolysaccharide (LPS) on KJ-A cells
The effects of LPS on KJ-A cells were investigated with regard to immunoexpression of macrophage markers (ED1 and ED2) and mRNA expression of tumor necrosis factor-α (TNF-α). After washing in phosphate buffered solution, KJ-A cells grown in slide chambers were incubated in serum-free medium for 24 hours. Subsequently, LPS (0, 10, or 50 μg/ml; Escherichia coli, 0127; Bacto, Detroit, MI, USA) diluted in 1% serum-containing MEM was added to the cultures. After 24- or 48-hour incubation, the cells were fixed in acetone and stained immunocytochemically with ED1 and ED2 using the ABC method. The expression of TNF-α mRNA was examined by means of reverse transcription–polymerase chain reaction (RT-PCR). From KJ-A cells incubated with LPS at various concentrations (0, 10, or 50 μg/ml) for 24 or 48 hours, total RNA was isolated by Trizol reagent (GIBCO BRL, Grand Island, NY, USA).33,38,40 RT-PCR was conducted as described previously.33,38,40 From 5 μg of total RNA, the single-strand cDNA was synthesized with Super Script Preamplification System (GIBCO BRL), and cDNAs were amplified by PCR with Taq DNA polymerase (Takara Shuzou, Kyoto, Japan) and each of the specific primers (sense and antisense) for TNF-α or β-actin (control). The following conditions were used for the amplification for TNF-α: 32 cycles of 2 minutes of denaturation at 94°C, 1 minute of annealing at 55°C, and 0.5 minutes of synthesis at 42°C.39 The oligonucleotides used for PCR were as follows: TNF-α sense primer 5′-TGTCTACTGAACTTGGGGGTG-3′ and antisense primer 5′-GAGGCTGACTTTCTCCTGGTA-3′; β-actin sense primer 5′-TAAAGACCTCTATGCCAACAC-3′ and antisense primer 5′-CTCCTGCTTGCTGATCCACAT-3′.39 The PCR products were electrophoresed in 2% agarose gels. DNA was stained with ethidium bromide on the gel. The intensity of bands of TNF-α was evaluated semiquantitatively with image analysis software (NIH Image 1.61, Bethesda, MD, USA) relative to that of β-actin.33
Statistical evaluation
Paired samples were evaluated using Student's t-test. Significance was accepted at P < .05.
Results
The primary tumor
An original tumor, 2.5 × 2 × 2 cm, developed in the subcutaneous tissue of the thorax area of a 24-month-old male F344 rat. The tumor was well demarcated by thin fibrous tissues (Fig. 1). No infiltrative growth or metastatic lesions were seen in the rat. The tumor consisted of oval or fusiform cells arranged in fascicles or interlacing bundles (Fig. 2); there were various amount of collagen fibers, stained blue by the azan-Mallory stain, between neoplastic cells. Giant cells with large nuclei were occasionally seen throughout tumor tissues (Fig. 2). Immunohistochemically, all neoplastic cells gave a positive reaction to vimentin (Fig. 3) and ED1-reacting cells were frequently seen; however, no cells reacting to desmin, α-SMA, or cytokeratin were detected (Table 1). Electron microscopically, neoplastic cells showed round or polygonal configuration, and had variously sized lysosomal granules in the cytoplasm (Fig. 4). The original tumor tissues were not fixed in Zamboni's solution or stocked as frozen tissues, so that ED2 and OX6 immunostainings, as well as ACP, Non-SE, and ALP enzyme stainings, could not be conducted (Table 1).
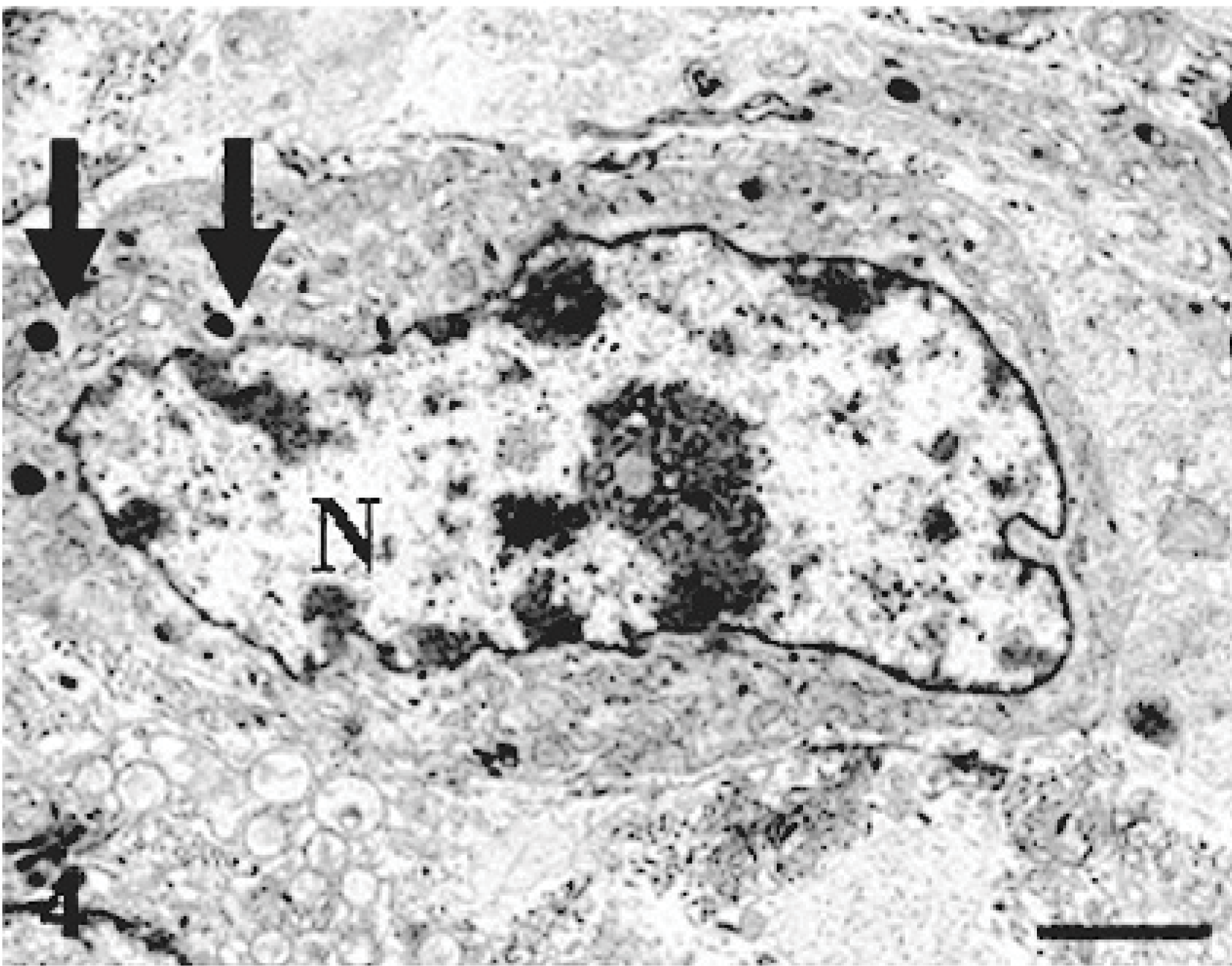
MFH; rat. Electron micrograph of a neoplastic cell in the primary tumor possessing lysosomal granules in the cytoplasm (arrows). N = nucleus. Uranyl acetate and lead citrate. Bar = 5 μm.

MFH; rat. The primary tumor comprised of oval or fusiform cells reacting to vimentin (arrows). Immunohistochemistry. Bar = 40 μm.
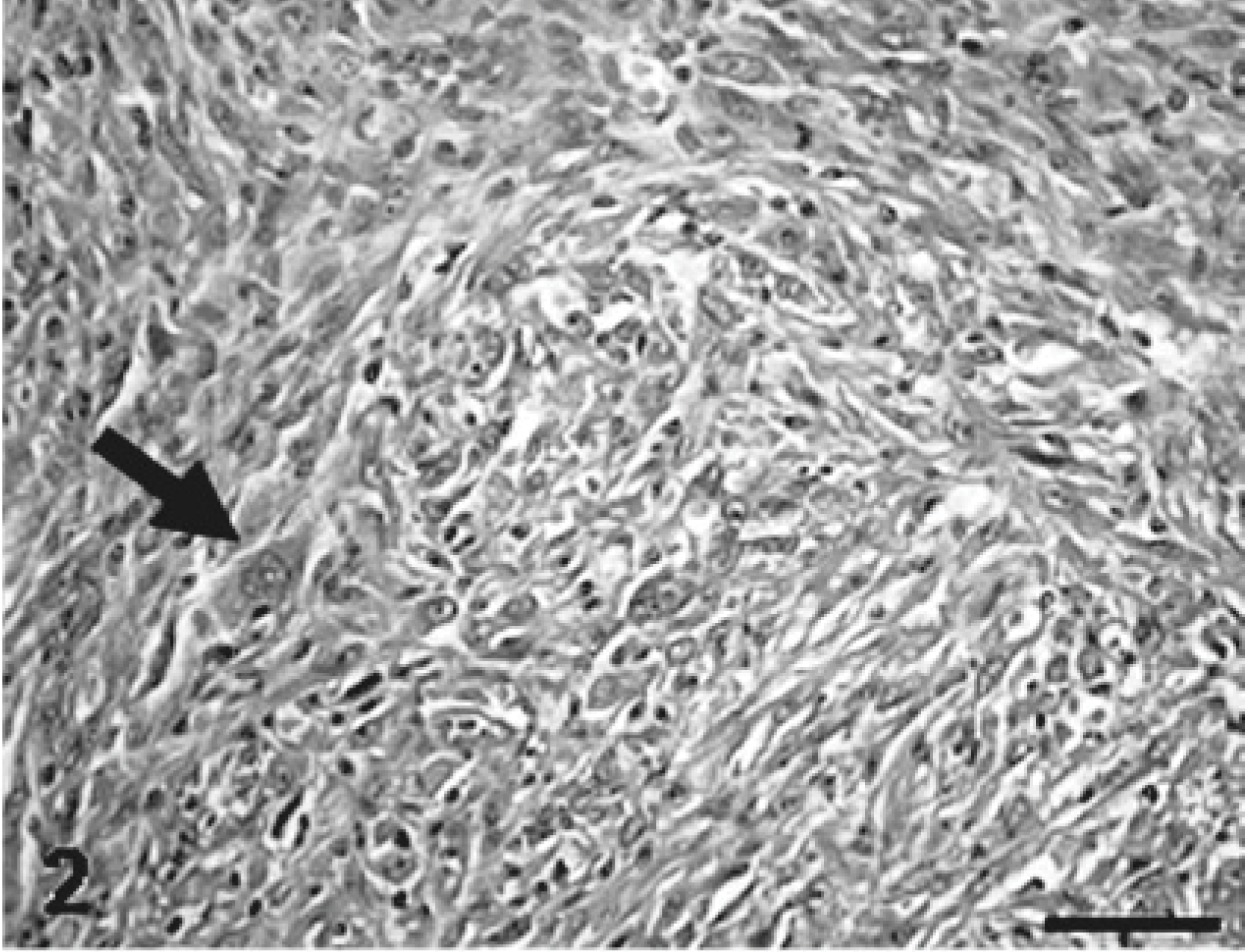
MFH; rat. The primary tumor consisting of oval or fusiform cells arranged in an interlacing bundle. Giant cells with large nuclei are present (arrow). HE. Bar = 50 μm.
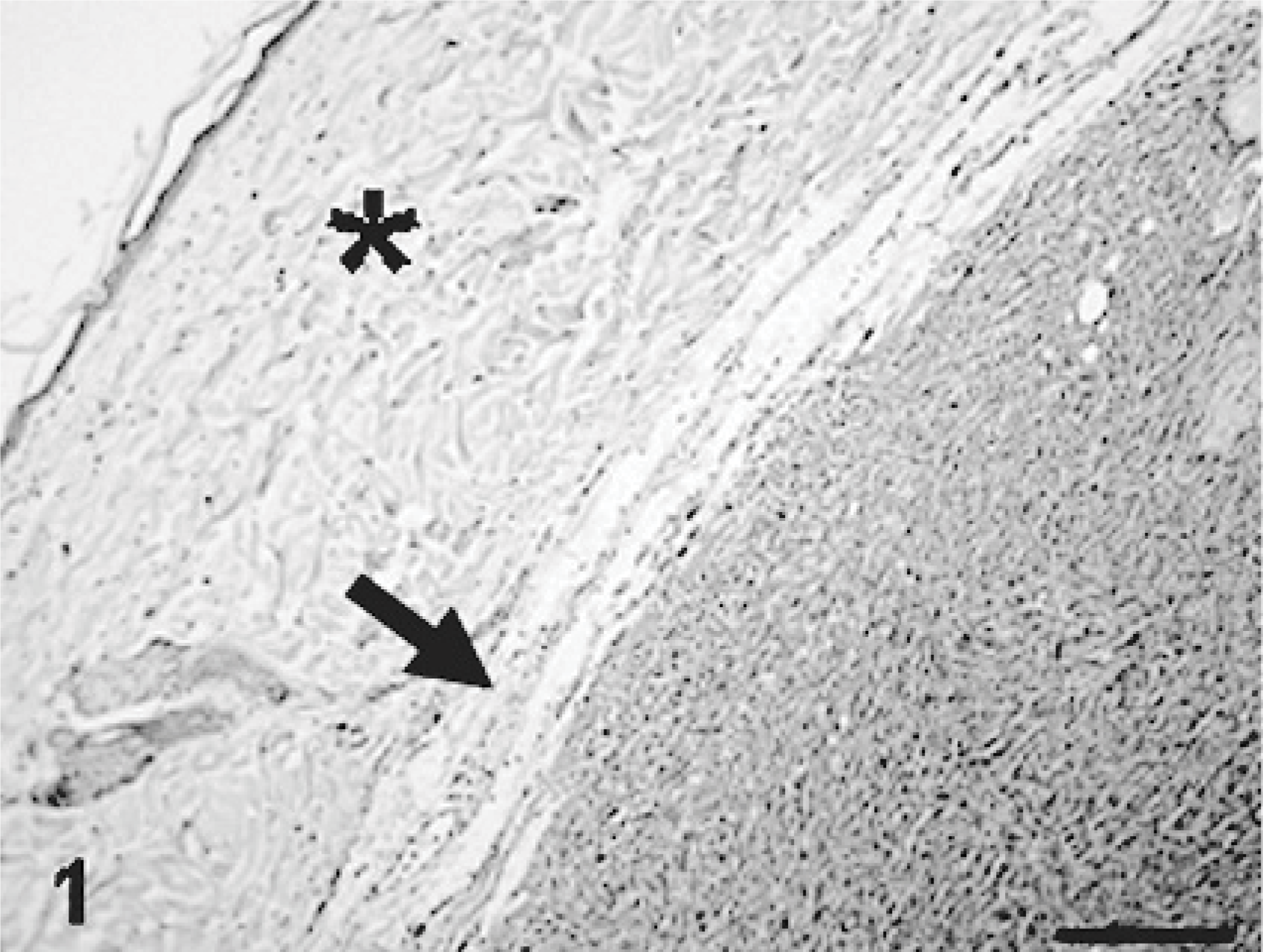
MFH; rat. The primary tumor arising in the subcutis of an aged F344 male rat. The tumor is surrounded by thin fibrous tissues (arrow). ∗ = epidermis. Bar = 100 μm.
KJ tumors
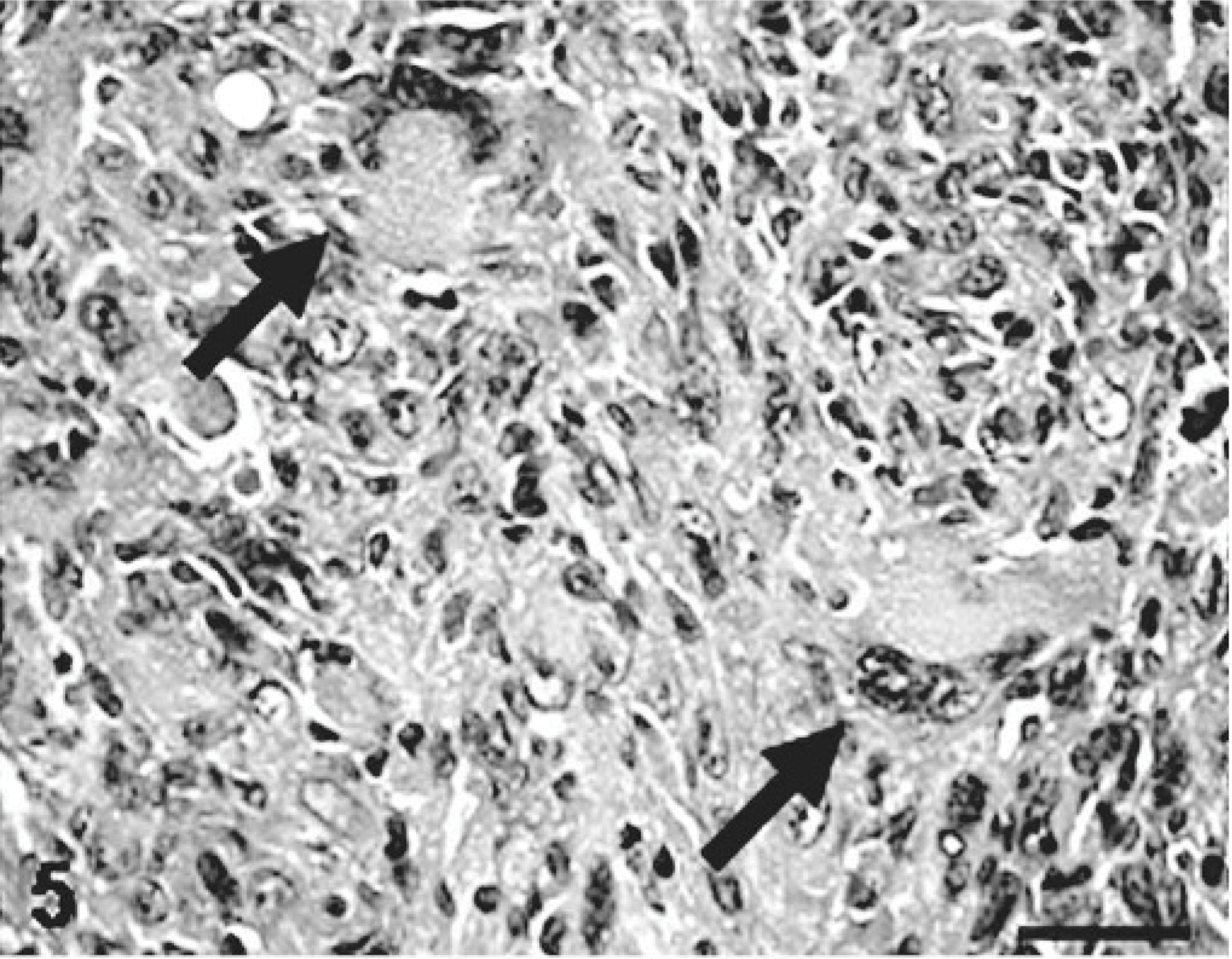
Homotransplantable KJ tumors were established in 100% of rats receiving transplanted tissues in each generation. Tissue fragments, 2 mm in diameter, developed into nodules of 3–5 cm in diameter at 4 or 5 weeks after the subcutaneous implantation. In KJ tumors, neoplastic infiltration into surrounding tissues was often observed, but no metastatic lesions were detected in KJ tumor–bearing rats. Histologically, KJ tumors were generally similar to the original tumor, showing growth patterns of interlacing bundles; however, nuclear atypia became more prominent, and giant cells with peripherally located bizarre nuclei and abundant cytoplasm were more frequently present in the KJ tumors (Fig. 5). In addition, there were areas consisting of small, round histiocytic cells arranged haphazardly without any growth pattern; cellular borders were indistinct (Fig. 6). Mitotic figures were frequently observed throughout tumor tissues. The interstitial matrix was faintly positive for the Alcian blue stain and negative for PAS reaction. A small amount of collagen fibers, stained blue by the azan-Mallory, were present among neoplastic cells, and reticular fibers, demonstrable by the Watanabe's silver impregnation, were also seen among neoplastic cells in various degrees. Oil red O–positive staining was not seen in neoplastic cells, and PTAH stains failed to demonstrate intracytoplasmic cross or longitudinal striations, indicative of derivation of muscle cells. Ultrastructural findings of neoplastic cells in KJ tumors corresponded to those in the primary tumor; neoplastic cells characteristically had variously sized cytoplasmic lysosomal granules.
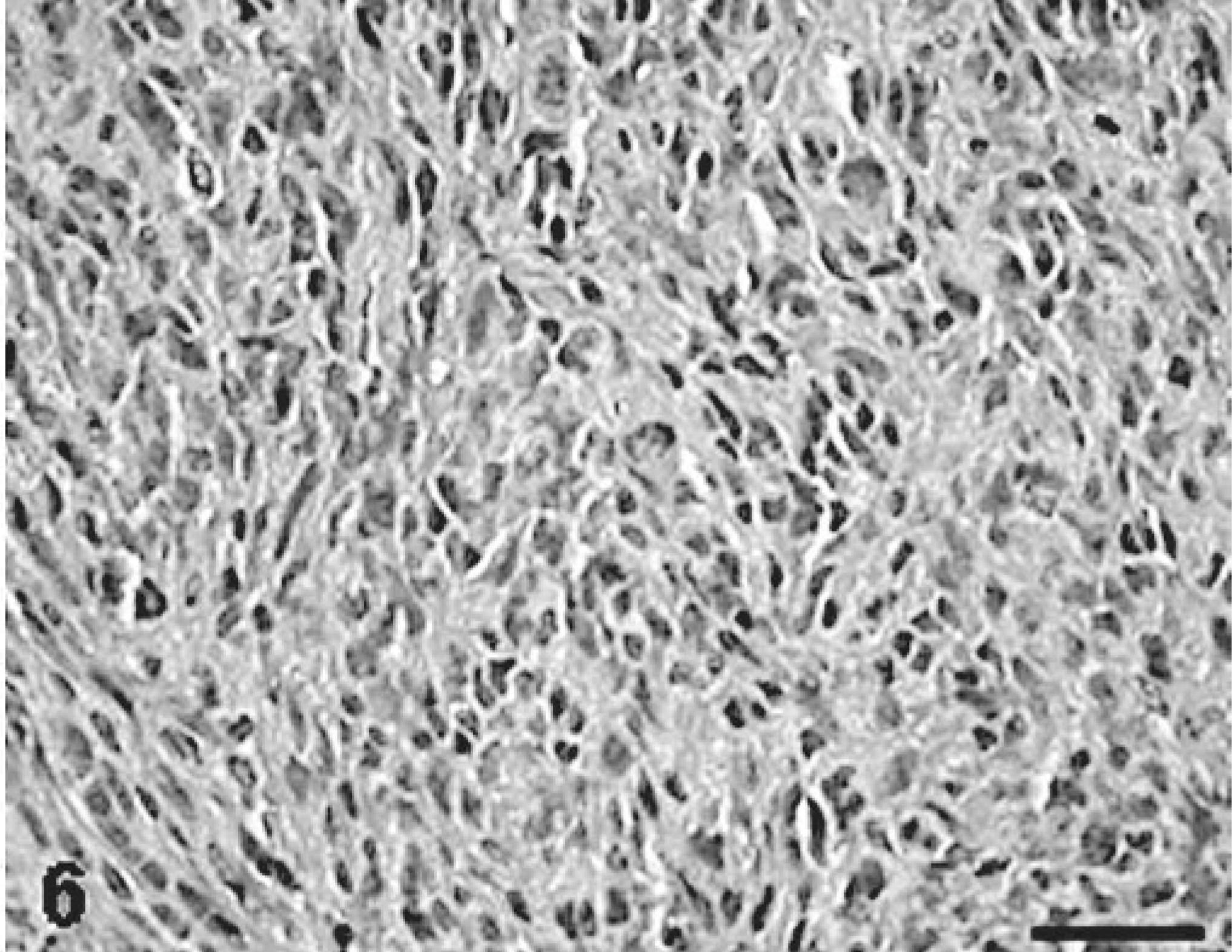
MFH-derived KJ tumor; rat. An area consisting of histiocytic small round cells arranged haphazardly; cellular borders are indistinct. HE. Bar = 40 μm.
Homotransplantable KJ tumor established from an MFH; rat. An area consisting of large, giant cells with peripherally located bizarre nuclei and abundant cytoplasm (arrows). HE. Bar = 40 μm.
KJ-P and KJ-A cells
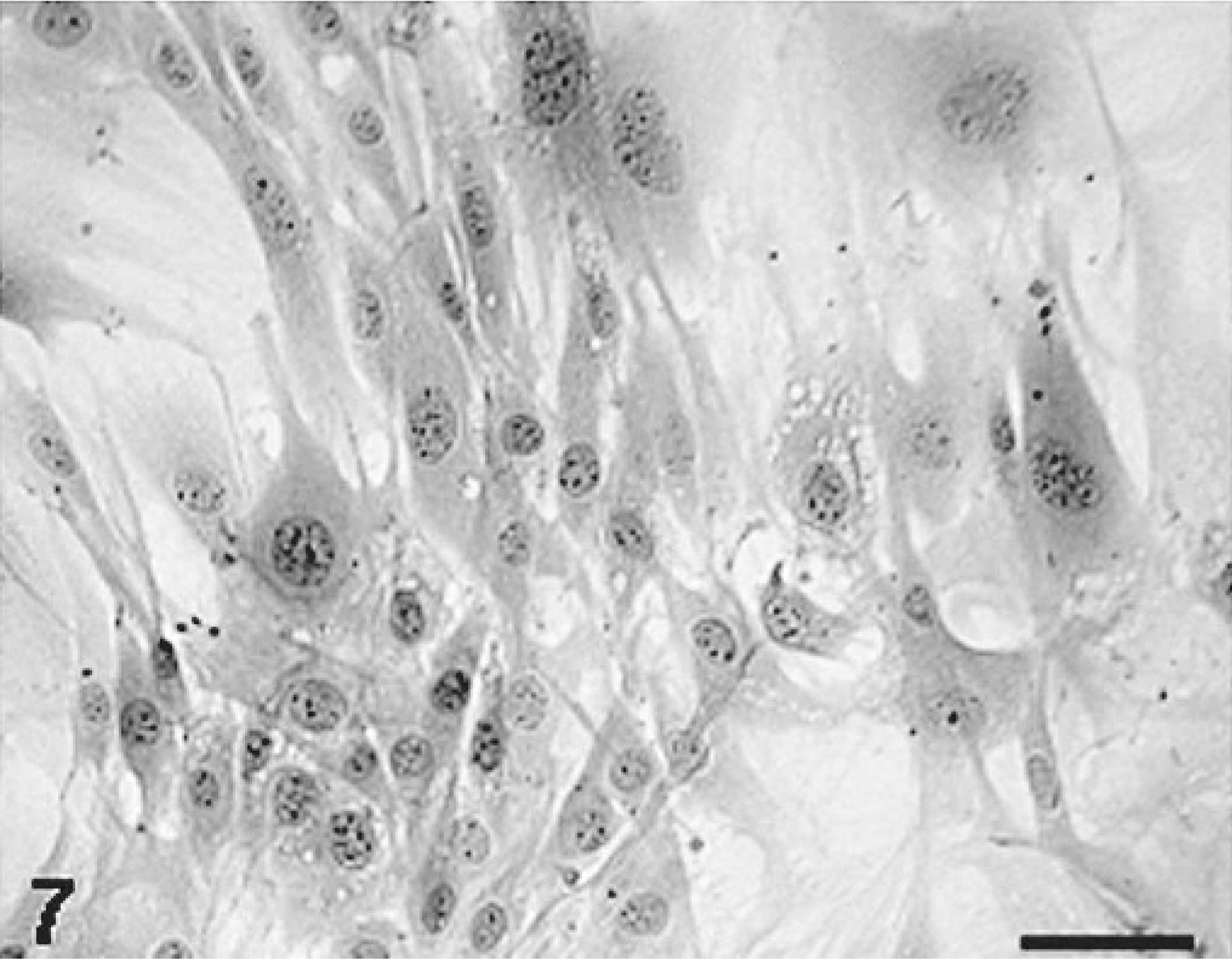
Mean doubling times of KJ-P (examined at passages 10, 20, and 30) and of KJ-A (examined at passages 5 and 10) were 26.5 and 19.9 hours, respectively. The modal chromosomal number of KJ-P (examined at passages 10, 20, and 30) and KJ-A (examined at passages 5 and 10) was distributed mainly between 65 and 69 and between 60 and 64, respectively, indicating numeric aberration, in contrast to 42 in normal rat cells.33 Cellular morphology of KJ-P and KJ-A was similar; their cultures consisted of round, polygonal, or spindle cells with round or fusiform nuclei arranged in an ambiguous interlacing pattern (Fig. 7). Occasional giant cells were present in the cultures. Ultrastructurally, cells of both KJ-P and KJ-A possessed some cytoplasmic projections and a number of lysosomal granules in the cytoplasm (Fig. 8).
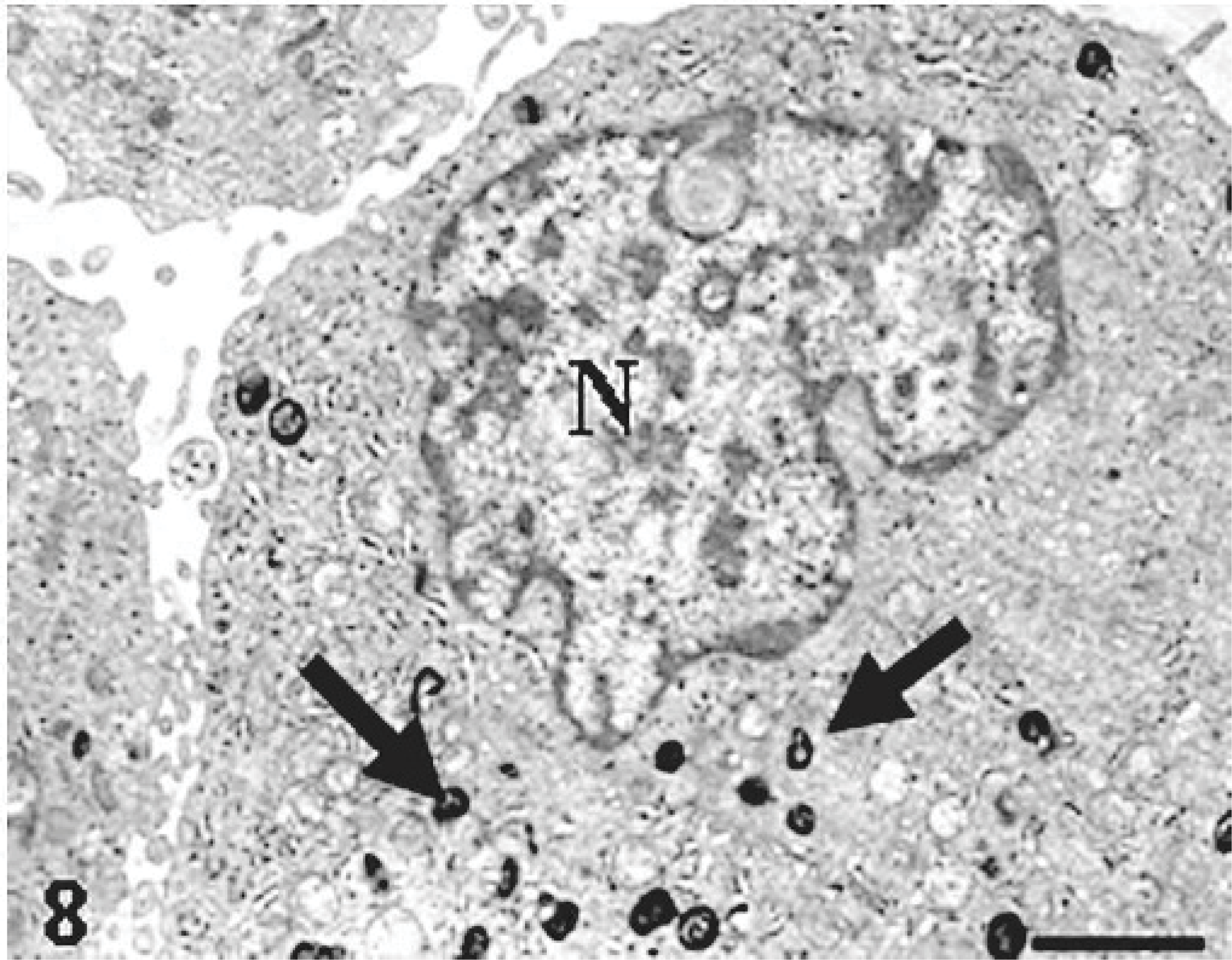
MFH-derived KJ-A; electron micrograph of a neoplastic cell in KJ-A characteristically possessing variously sized cytoplasmic lysosomal granules (arrows). Uranyl acetate and lead citrate. N = nucleus. Bar = 4 μm.
A cloned cell line (KJ-A) isolated from a MFH-derived KJ tumor; the culture consists of spindle and polygonal cells with round or fusiform nuclei arranged in an ambiguous interlacing pattern. HE. Bar = 20 μm.
All rats inoculated subcutaneously with KJ-P or KJ-A cells developed tumors at the inoculation site; the tumors grew as nodules, 3–4 cm in diameter, 5 or 6 weeks after the inoculation. Histologic and ultrastructural findings in the induced tumors bore resemblances to those of the primary tumor and KJ tumors.
Enzyme/immunohistochemical findings in KJ tumors, KJ-P and KJ-A cells, and tumors induced by KJ-P and KJ-A cells (Table 1)
Immunohistochemically, neoplastic cells in KJ tumors and tumors induced by KJ-P and KJ-A cells reacted to vimentin, corresponding to the original tumor. There were no cells reacting to desmin, α-SMA, cytokeratin, or S-100 protein. Interestingly, many neoplastic cells reacted to ED1 (Fig. 9) and ACP, and neoplastic cells reacting to ED2 (Fig. 10) and Non-SE were often observed. There were no cells reacting to OX6.
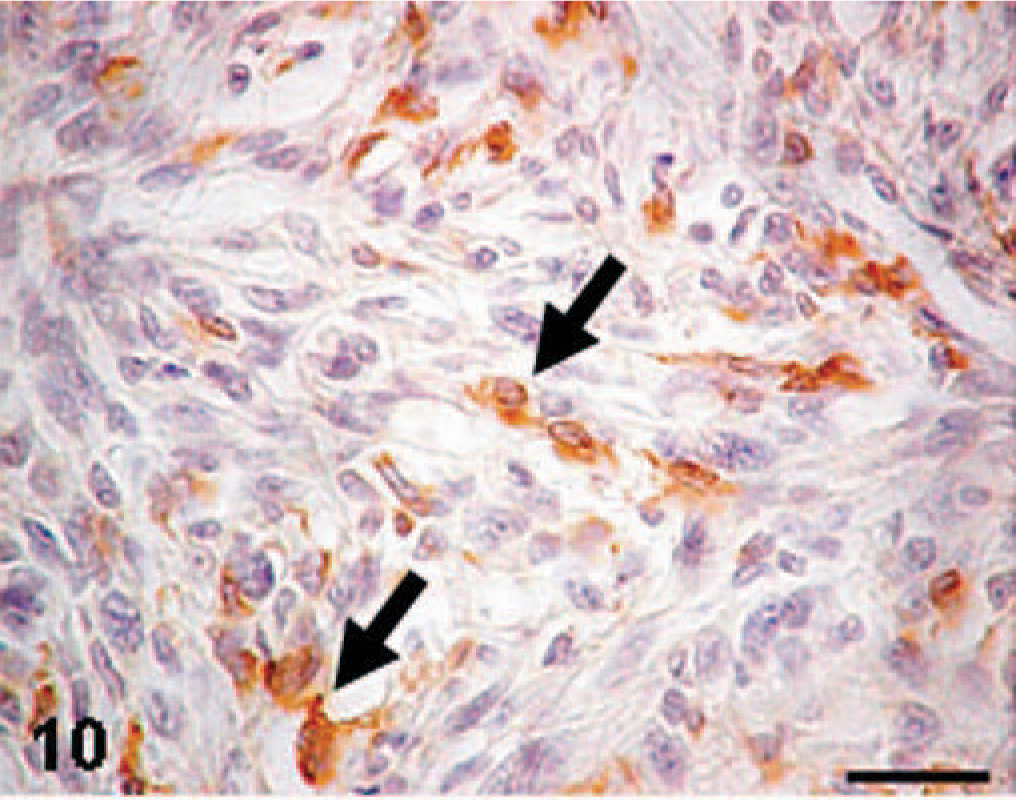
MFH-derived KJ tumor; rat. A KJ tumor consisting of round or fusiform cells (arrows) reacting to ED2 (an antibody specific for rat tissue macrophages). Immunohistochemistry. Bar = 40 μm.
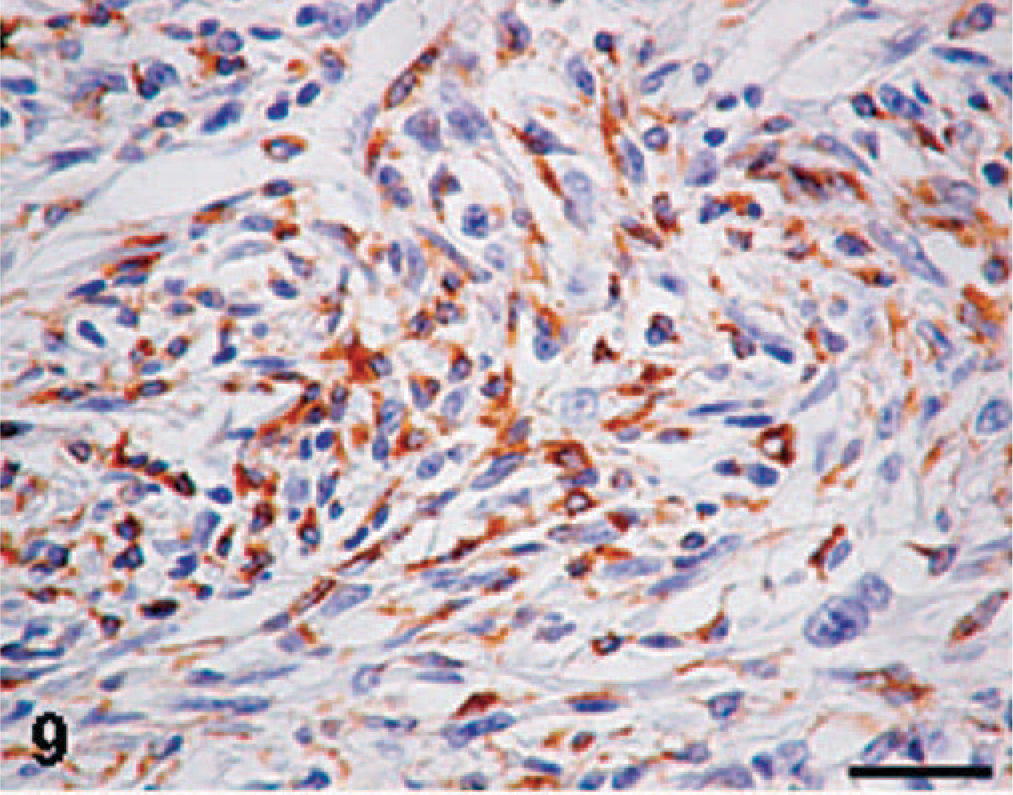
MFH-derived KJ tumor; rat. A KJ tumor consisting of round or fusiform cells arranged in an interlacing bundle; the majority of neoplastic cells show a positive reaction to ED1 (an antibody specific for rat exudate macrophages). Immunohistochemistry. Bar = 40 μm.
Enzyme/immunohistochemical findings of cultured cells of KJ-P and KJ-A were similar to each other, and these findings were in agreement with those in KJ tumors and tumors induced by KJ-P and KJ-A; cultured cells reacted to vimentin (Fig. 11), ED1 (Fig. 12), ED2 (Fig. 13), ACP (Fig. 14), and Non-SE in varying degrees. The immunopositive products to ED1 appeared granular in the cytoplasm (Fig. 12), presumably corresponding to cytoplasmic lysosomes seen by the electron microscopy, because ED1-recognizing antigens are on lysosomal membranes.3 Cultured cells of KJ-P and KJ-A did not react to desmin, α-SMA, cytokeratin, or ALP.
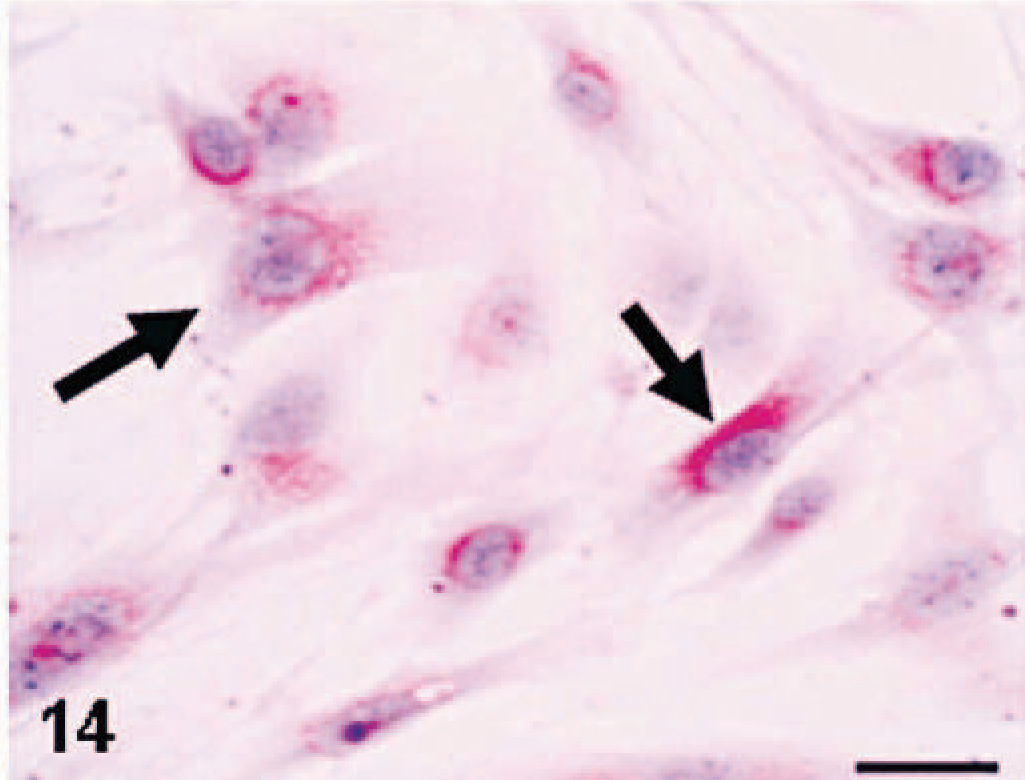
MFH-derived KJ-A; the culture consists of fusiform or polygonal cells; neoplastic cells show a positive reaction to acid phosphatase (a lysosomal enzyme marker) (arrows). Enzyme histochemistry. Bar = 20 μm.
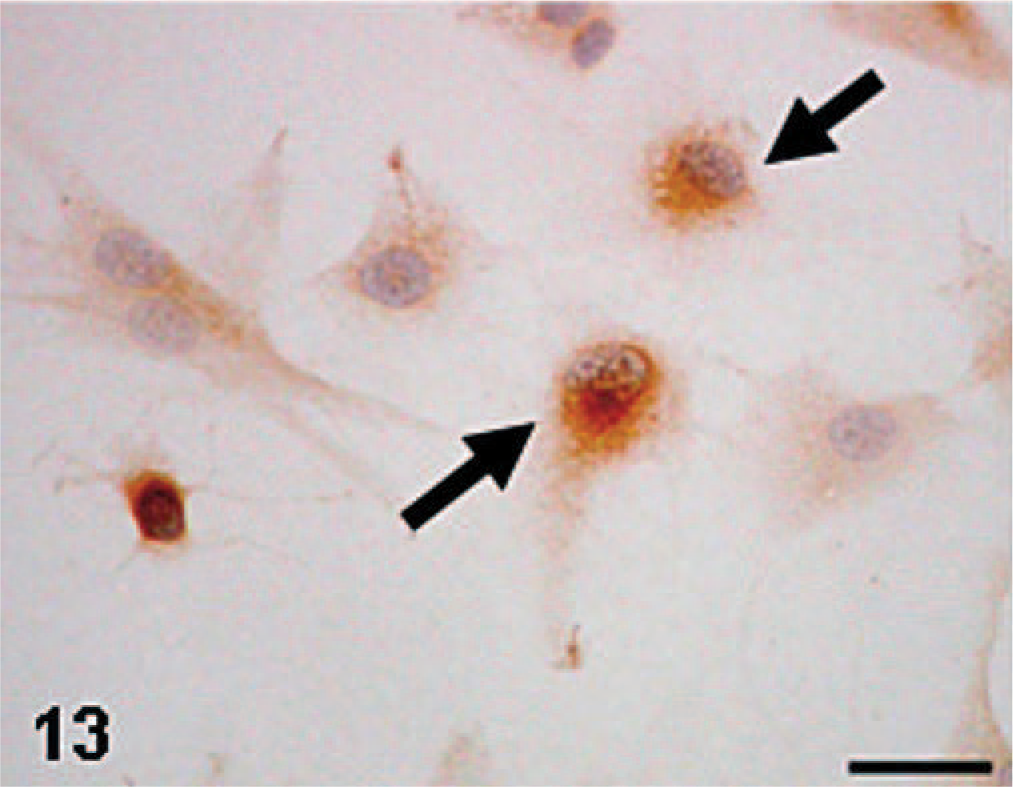
MFH-derived KJ-A; the culture consists of round or polygonal cells; cultured cells show a positive reaction to ED2 (arrows). Immunohistochemistry. Bar = 15 μm.
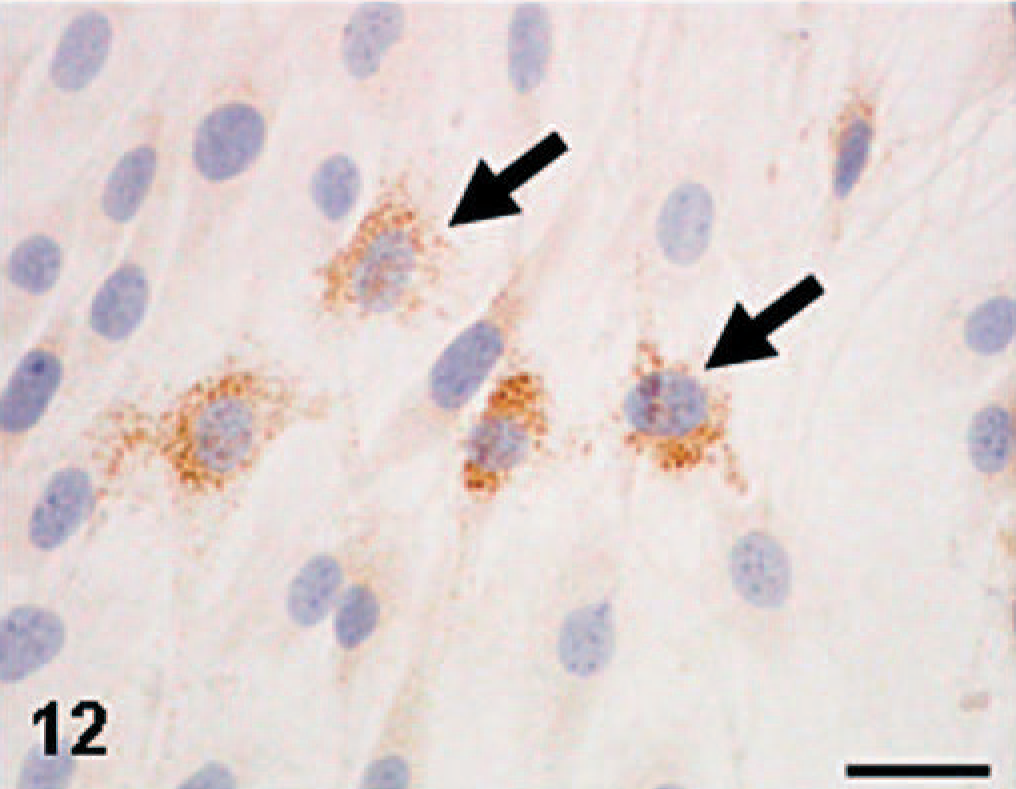
MFH-derived KJ-A; the culture consists of round or polygonal cells; cultured cells show a positive reaction (granular in appearance) to ED1 (arrows). Immunohistochemistry. Bar = 15 μm.
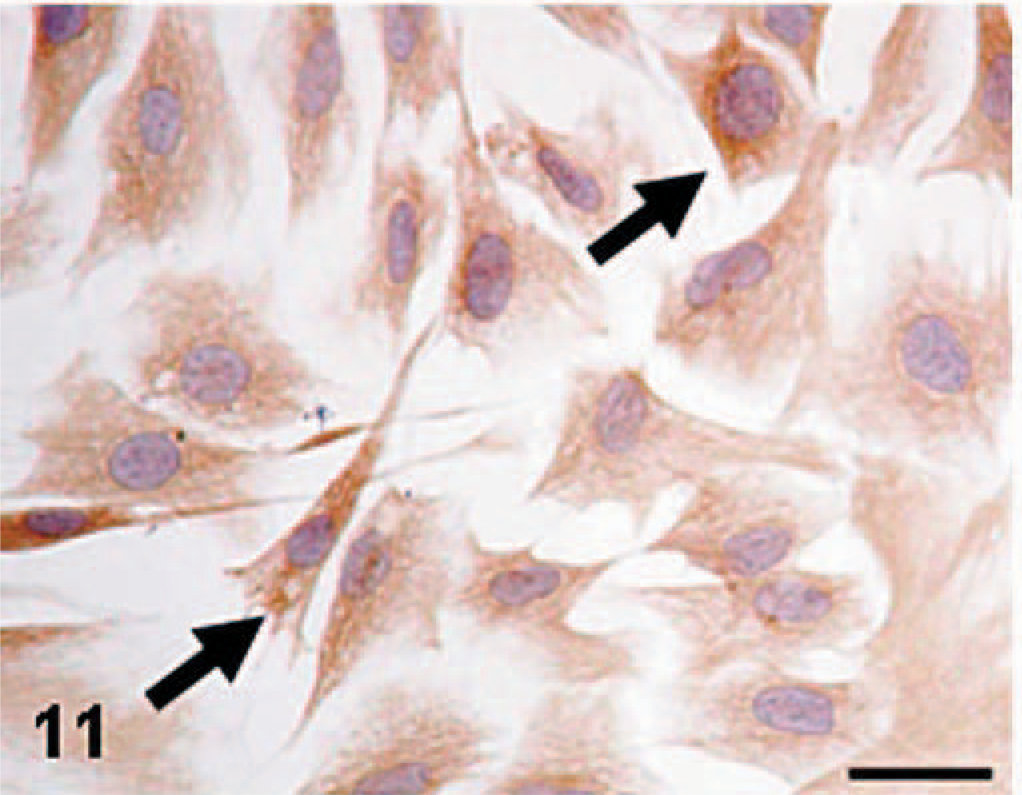
MFH-derived KJ-A; the culture consists of fusiform or polygonal cells reacting to vimentin (arrows). Immunohistochemistry. Bar = 20 μm.
Effects of LPS on immunoexpression to ED1 and ED2, and TNF-α mRNA expression in KJ-A cells
The 24-hour incubation of KJ-A cells with LPS increased the number of ED1-positive cells, with a significant difference at 50 μg/ml in contrast to 0 μg/ml; 48 hours after LPS addition, the positive cell number showed a significant increase at 10 and 50 μg/ml (Fig. 15). On the other hand, the number of ED2-positive cells did not alter following the addition of LPS (Fig. 15). Twenty-four hours after LPS addition, TNF-α mRNA expression slightly increased at 10 and 50 μg/ml; TNF-α mRNA expression was markedly increased at 10 and 50 μg/ml after 48 hours' incubation of LPS (Fig. 16). These results showed that the ED1-positive cell number and mRNA expression of TNF-α were increased dose-dependently following LPS addition.
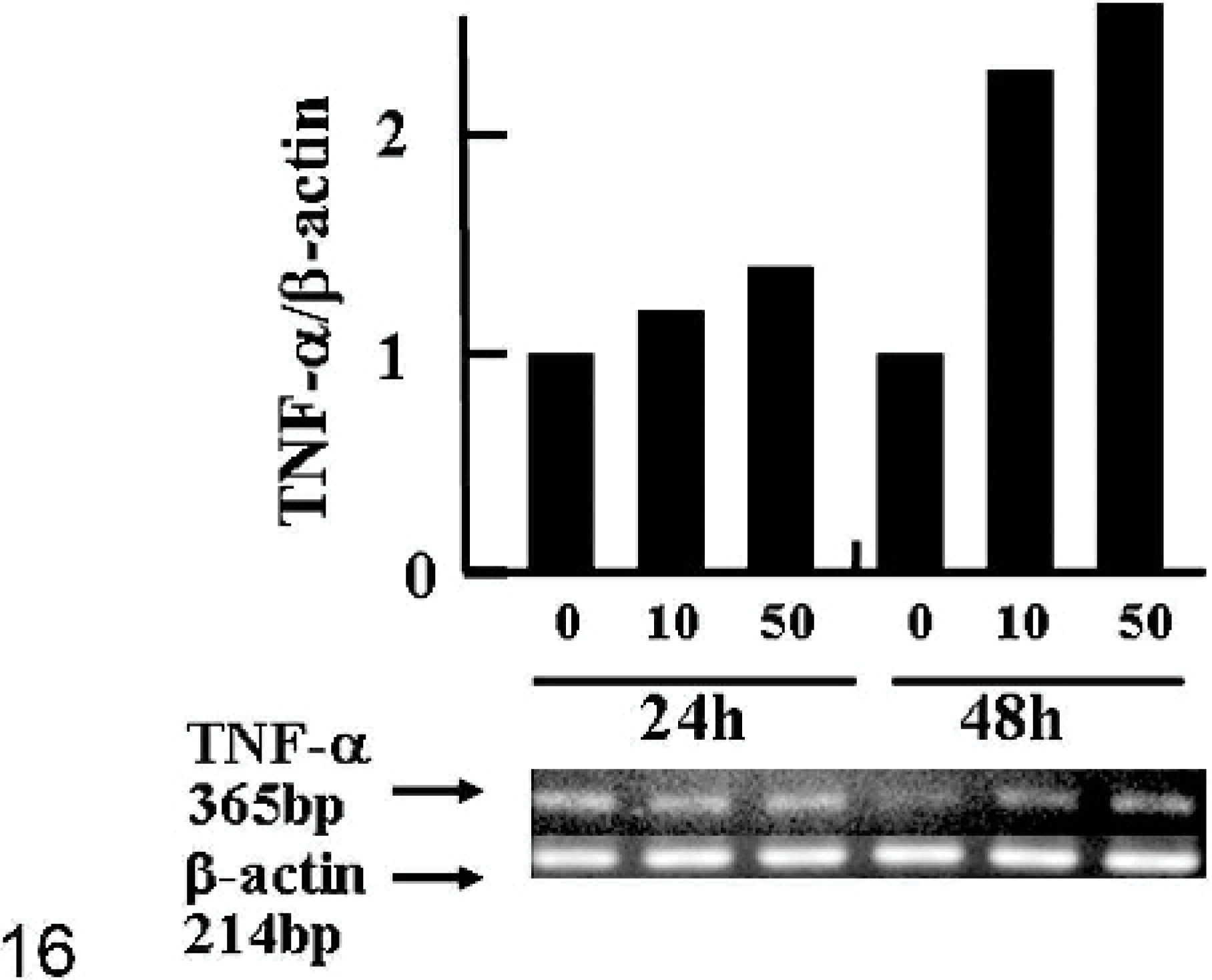
Fig. 16. The expression of mRNA of TNF-α in KJ-A cells incubated with LPS (0, 10, or 50 μg/ml) for 24 and 48 hours by RT-PCR. The bands of TNF-α at each concentration are normalized semiquantitatively by those for β-actin, and expressed relative to the intensity of control (0 μg/ml; level = 1) at each incubation time.
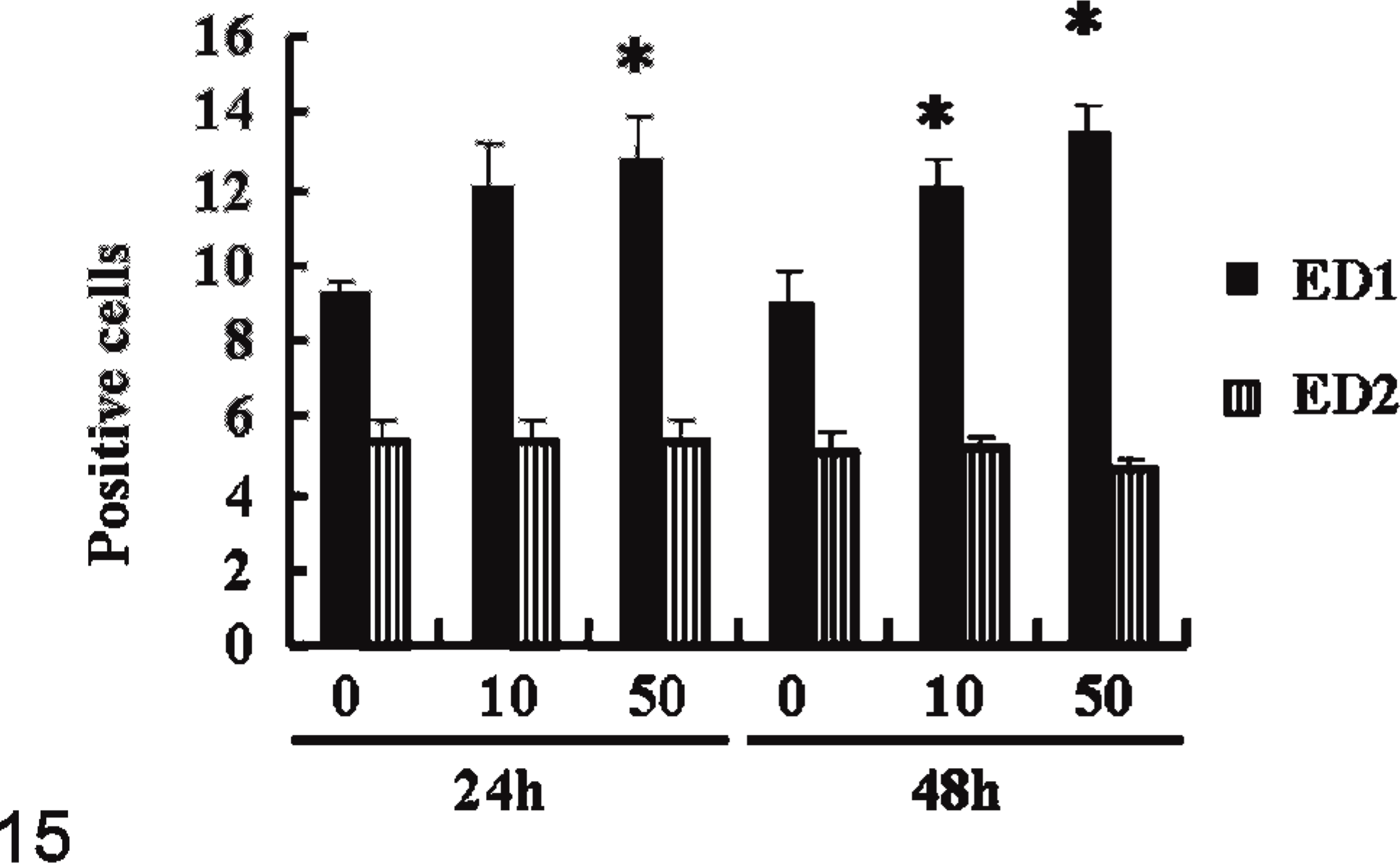
Fig. 15. Phenotypic changes (%) of KJ-A cells by immunohistochemistry with ED1 and ED2 at 24 and 48 hours after LPS (0, 10, or 50 μg/ml) addition. ∗ = significantly different from 0 μg/ml at each incubation time at P < .5. Values represent means ± SD.
Discussion
MFH is a pleomorphic soft tissue sarcoma consisting mainly of histiocytic cells and spindle-shaped fibroblastic cells in varying proportions.7,28,35 In addition, undifferentiated mesenchymal cells and giant cells may be present in tumor tissues.7,35 Thus, MFH shows a broad spectrum of histologic patterns. Depending on the predominant cell types and amount of intercellular material, human MFHs are classically divided into storiform-pleomorphic (common), myxoid, xanthogranulomatous (or inflammatory), and giant cell types.7,35 Although the pathogenesis of MFH has long been a matter of controversy, the presence of cells with histiocytic nature supported by collagenous stroma seems to be a hallmark for the diagnosis of MFH.2,6 The present primary tumor exhibited histology of interlacing bundles consisting of oval and fusiform cells with fibrous matrix. Although the primary tumor gave an appearance of fibrosarcoma, the immunohistochemical analysis revealed that a number of ED1-positive cells were present in tumor tissues. Furthermore, cells with cytoplasmic lysosomal granules were detected by electron microcopy. These observations indicated that the primary tumor consisted of neoplastic cells with a histiocytic phenotype. In human MFHs, many neoplastic cells stain positive for CD68, a monoclonal antibody specific for human histiocytes and macropghages.2 In rats, MFH has been reported to develop in the subcutis, and histologically rat MFHs show interlacing bundles or whorling patterns supported by fibrous components.6 Histiocytic sarcomas, whose origin has been considered histiocytes belonging to the mononuclear phagocyte system (MPS) lineage, also develop in the subcutis of aged rats.6,21 Besides the subcutis, histiocytic sarcomas occur in multiple organs, including the liver, uterus, and bone marrow. However, histiocytic sarcomas differ morphologically from MFHs in that the former consist of sheets of round to fusiform cells with granulomatous areas.6,21,39 Taken together, the present primary tumor was regarded as an MFH that had developed in the subcutis.
To further investigate the cellular nature of the present tumor, we established a homotransplantable tumor line (KJ), and cultured cell lines (KJ-P and KJ-A). Histologic and electron microscopic findings for KJ tumors and tumors induced by KJ-P and KJ-A corresponded to those for the original tumor. Furthermore, neoplastic cells in these derivatives reacted to lysosomal enzyme markers such as ACP and Non-SE. ACP and Non-SE have been used as markers for tumors originating from myelomonocytes and histiocytes.4,14 Immunohistochemically, many neoplastic cells in the KJ tumors demonstrated positive reactions to ED1. ED1 is used to identify infiltrating macrophages in lesions of rats.12,36 The antigens recognized by ED1 are mainly on membranes of cytoplasmic granules, especially phagolysosomes, of macrophages, and thus, the degree of ED1 expression may depend on phagocytic activity.3 Although the number of ED2-positive cells was smaller than that of ED1-positive cells, we confirmed the presence of ED2-reacting neoplastic cells in KJ tumors. ED2 reacts with cell membrane antigens of rat resident macrophages (e.g., Kupffer cells in the liver).5,12,39 These enzyme/immunohistochemical analyses indicated the presence of neoplastic cells with histiocytic/macrophage-like phenotypes in the current tumor. OX6 labels MHC class II antigens expressing in activated dendritic cells/macrophages in rats.36,39 There were no cells reacting to OX6 in the present rat MFH, although neoplastic cells in canine MFHs have been reported to express MHC class II antigens demonstrated by immunohistochemistry.18
Some investigators have considered that histiocytic cells in human MFHs may be nonneoplastic, infiltrative macrophages induced by monocyte chemoattractants produced by neoplastic cells.10,29,36 However, cultured KJ-P and KJ-A cells showed positive reactions to ED1, ED2, ACP, and Non-SE, suggesting that cells with histiocytic nature in the present rat MFH are neoplastic.
LPS is well known to modulate macrophage functions. In particular, LPS stimulates production of proinflammatory cytokines such as TNF-α in peritoneal and alveolar macrophages.1,31,34 Besides increased numbers of ED1-positive cells, indicative of an enhanced macrophage function, the present study showed a dose-dependent increase in TNF-α mRNA in LPS-treated KJ-A cells. Increased TNF-α mRNA expression by LPS stimulation suggests a potential for macrophage functions of KJ-A cells. Cultured cell lines established from human MFHs produced TNF-α, interleukin (IL)-6, IL-8, and macrophage-colony stimulating factor (M-CSF).19,42 Although possible productions of various factors in histiocytic KJ-A cells or in fibroblastic cells to compare with the histiocytic cell line should be further investigated, inflammatory cytokines released by neooplastic cells might be related to the complicated morphogenesis of MFH.36
In conclusion, the present study demonstrated that in vivo and in vitro tumor lines (KJ, KJ-P, and KJ-A) established from a rat subcutaneous MFH possessed histiocytic and macrophage-like characteristics. It has been considered that undifferentiated stem cells seen occasionally in human MFHs may have a potential for multidirectional differentiation towards osteoblasts, myoblasts, and lipoblasts.9,17,23,27,41,42 Thus, the latest WHO (2002) classification for human soft tissue tumors considers MFH a synonym for undifferentiated pleomorphic sarcomas.8 The present study revealed no evidence that neoplastic cells differentiating into myogenic cells (no expression of desmin and α-SMA, and no reaction with PTAH stain), neurogenic cells (no expression of S-100 protein), osteogenic cells (no reactivity for ALP), or lipoblastic cells (no reaction with oil red O stain) were present. It has been also postulated that both histiocytic cells and fibroblastic cells in MFHs may be derived from a common undifferentiated stem cell;13,23,27,28 alternatively, the histiocytic cells derived from the stem cell may assume the appearance and function of fibroblasts (facultative fibroblasts).7,15 Histiocytes and macrophages are in the MPS lineage.10 A cell line (HS-P) that had been isolated from a spontaneous histiocytic sarcoma arising in the liver of an aged F344 rat39 gave positive reactions to ED1, ED2, ACE, and Non-SE, in agreement with enzyme/immunohistochemical features of KJ-P and KJ-A. Based on these findings, neoplastic histiocytic cells in the present rat MFH may be involved in the MPS lineage. The tumor lines (KJ, KJ-P, and KJ-A) would prove useful for investigating the morphogenesis of MFH, in terms of the mesenchymal differentiation and relationship between bone-marrow and mesenchymal stem cells.
Footnotes
Acknowledgement
This study was supported in part by a Grant-in-Aid (No. 18658110) for Exploratory Research, the Ministry of Education, Culture, Sports, Science and Technology, Japan.
