Abstract
Multinucleated giant cells (MGCs) are a prominent histological feature of various mesenchymal neoplasms and are often considered a criterion of malignancy. Mesenchymal neoplasms with MGCs for which the cell lineage is unclear generally are referred to as giant cell sarcomas. Here we characterize the gross, histologic, and immunohistochemical features of 90 giant cell sarcomas in domestic pet rabbits. Based on the anatomic location and histologic and immunohistochemical findings, 18 cases were classified as histiocytic sarcomas (HS) and 72 cases as anaplastic sarcomas (AS). At postmortem examination, HS was either localized HS (n = 7) always affecting the lungs, or disseminated HS (n = 10) that affected the lungs (n = 10), liver (n = 6), kidneys (n = 4), pleura (n = 2), mediastinum (n = 2), heart (n = 4), skeletal muscle (n = 1), adipose tissue (n = 1), and lymph node (n = 1). Additionally, one cecal biopsy was consistent with HS. Microscopically, HS were characterized by sheets of neoplastic polygonal to round cells that contained single to several, often greatly enlarged nuclei as well as abundant cytoplasm. HS were always positive for CD204 and always negative for SMA and desmin. In contrast, AS arose most commonly from the skin or subcutis (n = 62) and rarely the skeletal muscle (n = 8) or abdominal organs (n = 2). In 29% of extra-abdominal AS, the tumor deeply invaded into surrounding connective tissue, skeletal muscle, tendons, and bone causing pathological fractures. Five of 9 postmortem cases metastasized to various organs often including the lungs. Microscopically, AS were characterized by sheets of spindle or pleomorphic cells admixed with variable numbers of MGCs. Immunohistochemically, AS were always negative for CD204 and often (71%) positive for SMA and/or desmin.
Keywords
Multinucleated giant cells (MGCs) are a prominent and common histological feature of many mesenchymal neoplasms of various domestic animals, including anaplastic sarcoma, 31 fibrosarcoma, 13 feline vaccine-associated sarcoma, 13,27 myxosaroma, 29 giant cell tumor of soft parts, 5 peripheral nerve sheath tumors, 7 perivascular wall tumors, 2 pleomorphic liposarcoma, 3 rhabdomyosarcoma, 25,31 skeletal and extraskeletal osteosarcoma, 17,31 giant cell glioblastoma, 12 osteoclastoma, 10 histiocytic sarcoma, 1,14,15,22 mast cell tumors, 28 extramedullary plasmacytoma, 21 and deciduosarcoma. 8 In contrast, MGCs have rarely been reported in epithelial neoplasms, such as anaplastic mammary carcinomas 9 and undifferentiated thyroid carcinomas. 26 The formation of MGCs in mesenchymal tumors is often a feature of malignancy and may be associated with metastatic spread and therapy resistance. 6 The generally accepted cause for multinucleation of neoplastic cells is endomitosis, which represents an abortive mitosis without cell division into 2 daughter cells (cytokinesis), due to physiological or pathological stress. 6
In many neoplasms with MGCs, a specific phenotype can be easily appreciated on standard histological examination based on characteristic features such as formation of osteoid in osteosarcomas. However, some mesenchymal neoplasms are classified as giant cell sarcomas, which is a morphological “umbrella” term applied to sarcomas that lack histological evidence of a specific cell lineage due to a high degree of anaplasia or nonspecific cell and tissue features. This group of neoplasms mainly, but not exclusively, includes anaplastic sarcoma (AS) with giant cells and histiocytic sarcoma (HS). While HS is usually identified by their expression of specific histiocytic lineage markers such as CD204, 14,15 AS (also referred to as pleomorphic sarcoma) represents a diverse group of neoplasms with various and sometimes controversial cell lineage. 23,24,32 In rabbits, one study identified 2 cases of AS within a total of 190 cases of cutaneous tumors and tumor-like lesions (1.1%). 31 Reports on HS in rabbits are sparse and include 3 case reports of cutaneous, pulmonary, and disseminated HS, respectively. 14,15,18 Giant cell sarcomas have not been fully characterized in rabbits and little is known about their microscopic characteristics and their biological behavior. In this study, we therefore describe the microscopic and immunohistochemical features of 90 giant cell sarcomas diagnosed at 3 different laboratories in the United States and Europe.
Materials and Methods
A total of 90 giant cell soft tissue sarcomas from pet rabbits had been collected from 3 veterinary pathology laboratories including 64 surgical biopsies and 26 postmortem examinations (13 performed by veterinary pathologists and 13 by referring veterinarians; Supplemental Table S1). At the Institute of Veterinary Pathology (IVP) of the Freie Universität Berlin, giant cell sarcoma in pet rabbits had been diagnosed during 16 postmortem examinations (13 performed by veterinary pathologists and 3 by the referring veterinarians) and examinations of 10 surgical biopsy samples between 1995 and 2018. At Northwest ZooPath (NWZP), giant cell tumors were diagnosed in 16 surgical biopsy samples and one postmortem examination performed by the referring veterinarian. The Zoo/Exotic Pathology Service (ZEPS) identified 38 giant cell tumors in surgical biopsies and another 9 cases in tissues collected during postmortem examination performed by the referring veterinarian. All cases were reviewed by pathologists at Michigan State University (MSU) and/or IVP and immunohistochemistry was performed at the MSU Veterinary Diagnostic Laboratory (cases from NWZP) or IVP (cases from IVP and ZEPS).
Immunohistochemistry with primary antibodies against vimentin, smooth muscle actin (SMA), desmin, S100, and CD204 (also known as macrophage scavenger receptor 1 [MSR1]) was performed according to methods stated in Supplemental Table S2. Additionally, all cases from IVP (n = 26) were stained by periodic acid-Schiff reaction (PAS) and Ziehl-Neelsen.
Results
All of the 90 included tumor cases comprised variable number of MGC cells and had a high degree of anaplasia. Other morphologic characteristics varied somewhat between cases. Based on the anatomic location and histological and immunohistochemical findings, cases were divided into 2 tumor entities, which are described separately in the following sections.
Tumor Type 1: Histiocytic Sarcoma (HS)
A total of 17 postmortem cases (7 were performed by veterinary pathologists and the remaining 10 by the referring veterinarian) and one cecal biopsy were included in this group. The median age was 8 years (mean age 7.8 years; range 3–11 years). Eleven rabbits were male (61%; 3 intact and 8 neutered) and 6 were female (33%; 4 intact and 2 neutered), and sex was not stated in one case. Breed included one case of each of the following: lion head, Teutoburger dwarf, and Dutch; breed was not reported in the remaining 15 cases.
In the 17 postmortem examinations, tumor was always present in the lungs and in 7 cases (41%) the lungs were the sole organ affected (localized HS; Fig. 1). The remaining 10 cases (59%) were disseminated HS affecting the lungs (n = 10) as well as the liver (n = 6), kidneys (n = 4), pleura (n = 2), mediastinum (n = 2), cardiac musculature (n = 2), skeletal musculature (n = 1), adipose tissue (n = 1), and mediastinal lymph nodes (n = 1; Fig. 2). In 4 cases (24%), a pneumothorax had been diagnosed by antemortem imaging (postmortem was performed by the referring veterinarian in all 4 cases), probably due to effacement of lung tissue by the tumor. Furthermore, a HS was found in the cecal wall of a surgical biopsy (information on neoplastic spread was not provided).
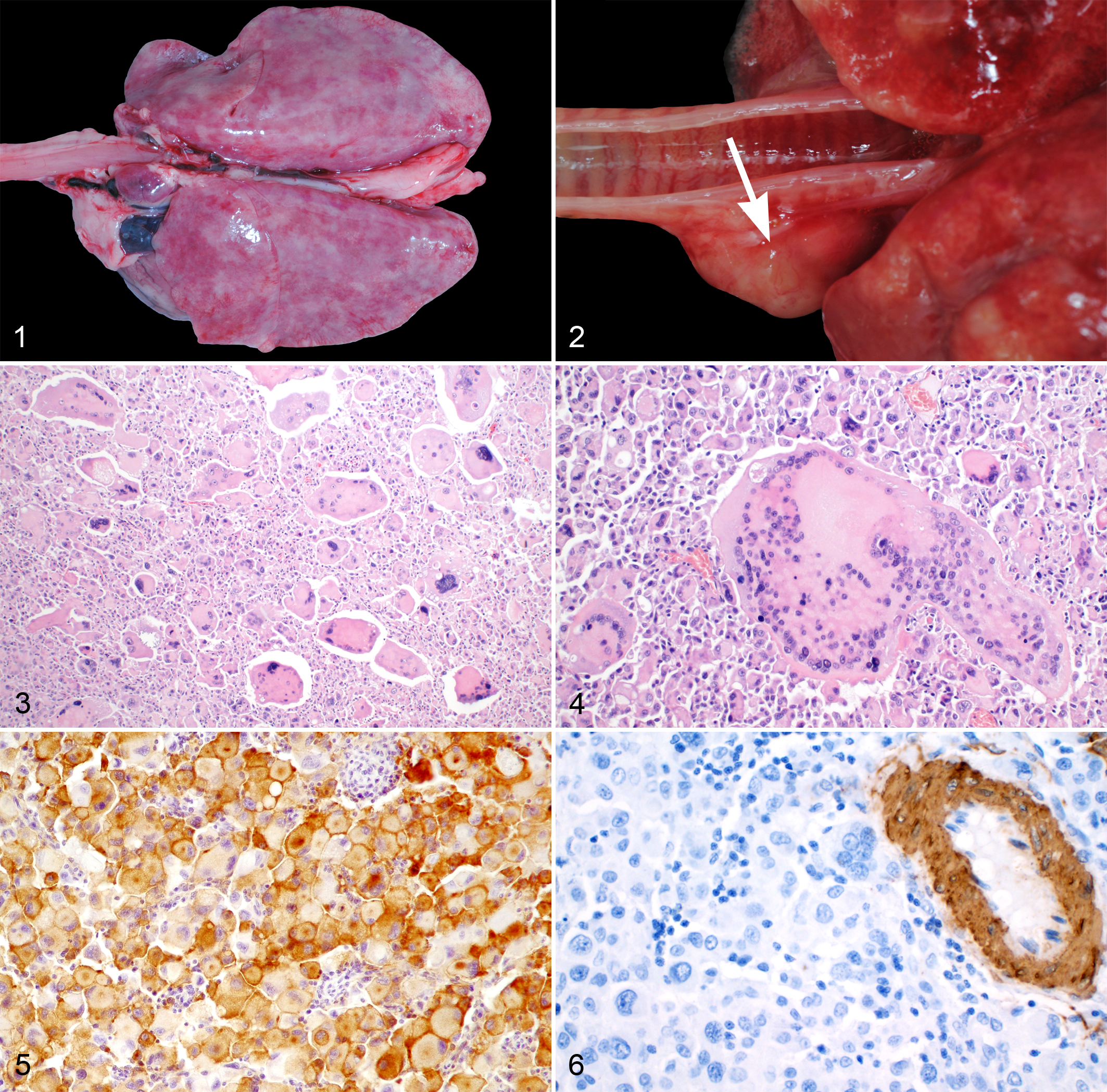
Grossly, all neoplastic masses were firm and tan-white, and those masses in the lungs had a multifocal-coalescing or diffuse pattern of infiltration resulting in a mottled appearance to the lungs. Histologically, the tumors were nonencapsulated, infiltrative, and highly cellular. In the lungs, the pulmonary architecture was nearly effaced by tumor in all cases. Neoplastic cells were mainly arranged in sheets and were highly pleomorphic (Fig. 3). Neoplastic cells were polygonal to round, contained abundant pale eosinophilic, often finely granular cytoplasm that stained positive for PAS in 10/10 cases (100%). Neoplastic cells contained round to oval, sometimes lobulated and often karyomegalic nuclei. Numerous cells contained several (up to 300) nuclei which often were located along the periphery of the cytoplasm similar to Langhans giant cells (Fig. 4). Nuclei mostly contained finely stippled chromatin with single or multiple, prominent nucleoli. The number of mitotic figures varied largely between cases ranging from 0 to 1 up to 8 mitoses in one high power field (hpf; 0.237 mm2). Atypical mitotic figures were encountered frequently. Large areas of necrosis, hemorrhage, and mixed inflammation were commonly observed within neoplastic masses. Hemophagocytosis and intracytoplasmic hemosiderin storage was observed in one disseminated HS. No bacteria, fungi, or foreign material were detected by routine microscopic examination or using specials stains in 10 cases (PAS, Ziehl-Neelson). In some cases, there was marked heterophilic inflammation.
Immunohistochemically, all cases tested had positive staining in almost all neoplastic cells for CD204 (18/18 cases; 100%; Fig. 5). Also, neoplastic cells were positive for vimentin in 18/18 cases (100%) and S100 in 6/18 cases (33%), and were negative for SMA and desmin in all cases (100%; Fig. 6 and Table 1). Based on histological and immunohistochemical examination, tumor cells were consistent with histiocytic lineage.
Immunohistochemical Findings in 90 Cases of Sarcoma With Giant Cells in Rabbits.
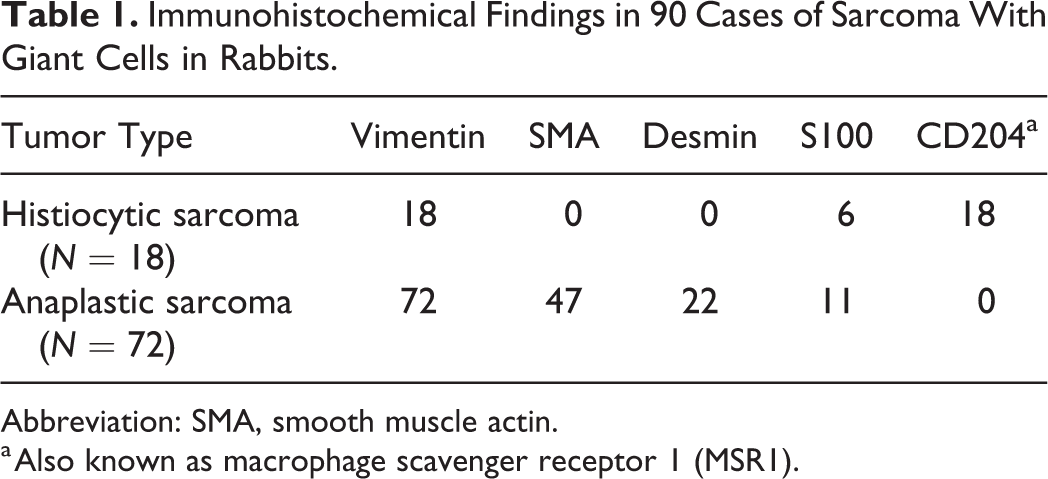
Abbreviation: SMA, smooth muscle actin.
a Also known as macrophage scavenger receptor 1 (MSR1).
Tumor Type 2: Anaplastic Sarcoma With Giant Cells (AS)
A total of 72 cases including 63 biopsies (61 surgical biopsies and 2 punch biopsies) and 9 postmortem examinations (6 were performed by a veterinary pathologist and 3 by the referring veterinarian) were included in this group. Median age was 7 years (mean age 6.7 years; range 2–15 years). Forty rabbits were male (56%; 23 intact, 17 neutered) and 24 were female (33%; 10 intact, 14 neutered), and sex was not given for 8 rabbits. Breeds included dwarf lop (4 cases), Dutch (4 cases), Flemish giant (2 cases), and one case each of English spot, rex, Netherlands, hermelin, and giant lop. Breed was not stated in the remaining 57 cases.
In 62 of 72 cases (86%; including 60 biopsy and 2 postmortem examinations), neoplastic masses were located in the dermis or subcutis. Single skin masses (cutaneous or subcutaneous) were observed in 55 rabbits (89%) while the number of skin masses was 2, 3, and multiple in 4, 1, and 2 rabbits, respectively. Of the skin masses, 67 different locations were recorded most commonly including the legs (25/62, 40%). Ten cases were in the foreleg, 13 in the hind leg (Fig. 7), and the specific leg was not stated for 2 cases. Remaining sites included thoracic wall (11/62, 18%), abdominal wall (10/62, 16%), head (8/62, 13%), inguinal region (5/62 cases, 8%), tail (2/62, 3%), neck (1/62, 2%), and unstated site (5/62, 8%). Four biopsies of cutaneous masses were submitted as recurrent growths. Furthermore, in 3 surgical biopsy cases and 5 postmortem cases (8/72, 11%), masses were primarily located in the skeletal musculature of the thoracic wall (n = 3), abdominal wall (n = 2), legs (n = 2), and pelvic musculature (n = 1). Additionally, 2 AS (3%) were detected in abdominal organs by postmortem examinations. The first was located in the liver, spleen, and mesenteric lymph node with unknown primary origin, and the second was in the liver.
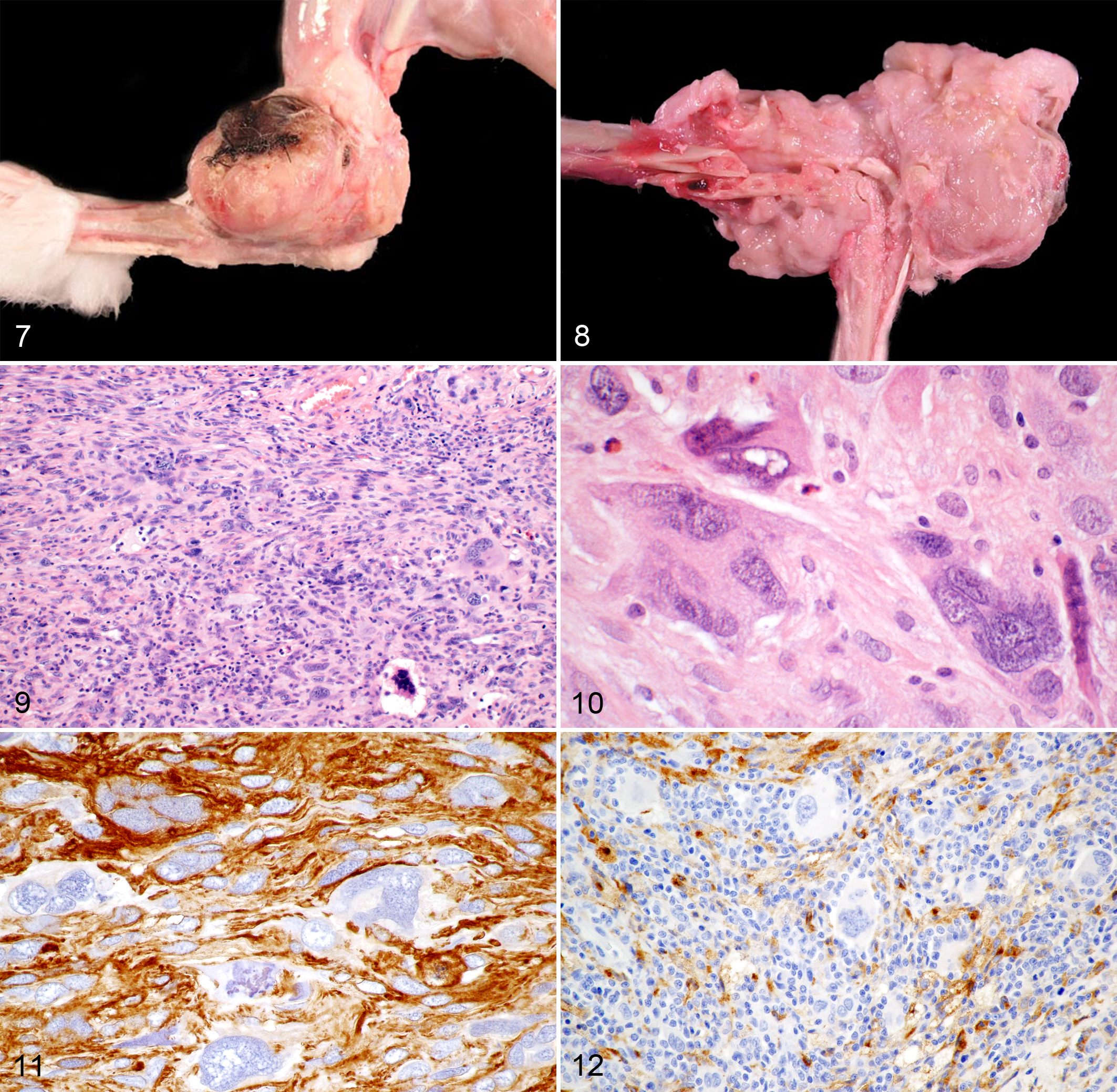
Grossly and histologically, neoplastic masses of the skin and musculature infiltrated into the surrounding connective tissue, deep muscle fascia, and skeletal musculature in 20/70 cases (29%, exclusively punch biopsies), into bone in 5/70 cases (7%; 2 cases with pathological fracture of femur or pelvis; Fig. 8), and into tendons in 2/70 cases (3%). Of the 7 postmortem cases with skin and muscle masses, 4/7 (57%) had distant metastasis to the lungs (n = 4), kidney (n = 2), liver (n = 1), and heart (n = 1).
Grossly, neoplastic masses were tan-white and firm. Histologically, the neoplastic masses were nonencapsulated, infiltrative, and highly cellular. Neoplastic cells were arranged in interlacing streams or in haphazardly arranged or storiform sheets. Whirling was observed in 3 cases. All cases were composed of 3 morphologically distinct cell populations (Fig. 9). The most frequently observed cells were spindle-shaped with mainly elongated, blunt-ending nuclei. There were few to numerous round to pleomorphic cells that greatly varied in size and had round to oval nuclei with a high degree of anisokaryosis. Characteristic for giant cell sarcoma, all tumors also contained variable numbers of MGCs with central, round to oval, sometimes indented nuclei (Fig. 10). MGCs had moderate to large amounts of pale eosinophilic, sometimes finely granular cytoplasm. PAS-positive granules were found in 4 of 16 cases tested (25%). Three cases contained few intracytoplasmic, empty vacuoles. Ziehl-Neelsen staining did not detect acid fast bacteria in the 16 cases tested. Nuclei had a stippled chromatin pattern and single or multiple nucleoli of variable size. The mitotic count within the neoplastic cell population ranged from 0–5 to 70 mitotic figures per 10 hpf. Atypical mitoses were common. Few neoplasms had small amounts of a myxomatous or mucinous matrix. Widespread areas of necrosis, hemorrhage, and mixed inflammation were common and skin neoplasms were often ulcerated.
Immunohistochemistry revealed that all cases were positive for vimentin (100%; Table 1) and 51/72 cases (71%) positive for (smooth) muscle markers (Fig. 11). Of those, SMA and desmin were both positive in 18/72 cases (25%), only SMA was positive in 29/72 cases (40%), and only desmin was positive in 4/72 cases (6%). S100 was positive in 11/72 cases (15%). Labeling of tumors cells against CD204 was negative in all examined cases (0/72; Fig. 12). However, there were few to abundant round to polygonal CD204-positive cells with membranous staining (consistent with reactive dendritic cell infiltration) intermixed with the spindle-shaped and multinucleated cell population.
Discussion
Giant cell sarcomas are a heterogeneous group of pleomorphic malignant neoplasms. Based on anatomic location and histological and immunohistochemical features, we were able to divide the examined cases into 2 tumor entities most consistent with HS and AS. The 2 entities were ultimately differentiated by detecting expression of CD204 in neoplastic cells. As has been previously reported, CD204 can be used to detect cells of histiocytic lineage in rabbits. 14 Besides other tumor types such as AS, granulomatous inflammation should be excluded. However, mycobacterial pneumonia is rarely described in rabbits, 20 and there was no evidence for acid-fast bacilli or other infectious agents in the present cases.
HS have only rarely been described in rabbits. 14,15,18 In dogs and cats, histiocytic neoplasms mainly arise from dendritic cells, including epithelial dendritic (Langerhans) cells and interstitial dendritic cells. 22,30 Canine HS are derived from interstitial dendritic cells, which can be found in almost all tissues. 22 Therefore, canine HS can originate in a wide range of tissues including spleen, lymph node, lung, skin, subcutis, and (peri)articular tissue. 1,22 Interestingly, in the present study of rabbits, HS mainly affected the lungs—and less commonly the liver or other organs—as has been reported in 1 of the 3 previous case reports. 18 It seems that rabbits have some predilection to develop HS in the lungs and—due to the effacement of the pulmonary architecture—may secondarily develop a pneumothorax. Only a single HS has been previously reported in the skin of a rabbit. 15 Rarely hemophagocytosis was observed similar to one case from the literature. 14
Dendritic cells of the Langerhans cell type, which colonize epithelia such as the pulmonary bronchial epithelium, are considered the origin for a feline histiocytic disease, called pulmonary Langerhans cell histiocytosis. 22 However, in contrast to feline pulmonary Langerhans cell histiocytosis, in rabbits the neoplastic histiocytic cells in the lungs were also positive for CD204, always effaced pulmonary tissue, had prominent bi- or multinucleated cell populations, and were accompanied by only minimal infiltration of lymphocytes and plasma cells. 4 Furthermore, co-expression of E-cadherin and CD204 has been reported for a condition in cats referred to as feline progressive histiocytosis that more closely resembles canine HS, and the HS in rabbits of this report. 30 Regardless, location, invasiveness, immunophenotype, and marked cellular pleomorphism ultimately support a diagnosis of a HS of the present cases.
In this study, AS were most frequently located in the skin/subcutis similar to the 2 previously described cases. 31 Only a few cases developed within the skeletal musculature or abdominal cavity. AS are often subdivided based on morphological pattern into storiform-pleomorphic, inflammatory, and giant cell variants. 23 The present study only included AS with MGCs. The histiogenesis for this diverse tumor entity is controversial. Historically, AS have been referred to as malignant fibrous histiocytomas; 16,23,24,32 however, this term should be avoided as a histiocytic cell origin is considered unlikely. Current evidence rather suggests that AS with MGCs most likely have a myofibroblastic lineage in dogs and cats. 11,13,23,24 Similarly, 71% of AS in rabbits in this study were positive for SMA and/or desmin, suggesting a myogenic differentiation, that is, smooth muscle, myofibroblastic, pericytic, or myoepithelial lineage. Whereas (cutaneous) rhabdomyosarcoma are also positive for desmin, they are negative (or only focally positive) for SMA. 25 Cutaneous smooth muscle tumors either arise from arrector pili or from smooth muscle of the dermal vasculature (angioleiomyosarcoma). 19 Cigar-shaped nuclei are usually a diagnostic criterion for smooth muscle cells, but may be absent in poorly differentiated leiomyosarcomas. Microscopic examination of hematoxylin-eosin–stained sections alone may be insufficient in differentiating poorly differentiated leiomyosarcomas from other soft tissue sarcomas. 19 Smooth muscle and desmin immunohistochemical labeling may be insufficient to reach a conclusive diagnosis since many perivascular wall tumors are also positive for SMA and/or desmin. 2 However, characteristic histological growth patterns of perivascular tumors such as whorling or staghorn formation were only rarely observed in this case series. Future studies need to show whether further smooth muscle markers such as smoothelin and caldesmon may prove helpful for differentiation. Some AS in this study did not label for smooth muscle markers, similar to the 2 previously described cases in rabbits. 31 It remains unknown whether these cases are less differentiated neoplasms of the same cell lineage or represent other types of soft tissue sarcomas such as pleomorphic fibrosarcomas.
In horses a spindle cell neoplasm with MGCs has been referred to as giant cell tumor of soft parts. 5 Similar to AS with MGCs in rabbits, equine giant cell tumors of soft parts are located in the dermis, subcutis, or less commonly in skeletal musculature predominately of the legs. 5 The histogenesis of this equine tumor also remains controversial. However, in contrast to AS with MGCs in dogs or rabbits, there is evidence that the giant cells in equine tumors may not represent neoplastic cells, but reactive histiocytic infiltrates. 5 Furthermore, equine giant cell tumors of soft parts are low-grade malignancies with minimal invasion into surrounding tissue and no distant metastasis, while AS with MGCs in rabbits are highly invasive and exhibit histologic features of malignancy, such as a high mitotic activity and considerable cellular anaplasia. Metastatic spread was also shown in some postmortem cases of the present case series. It is therefore unlikely that the neoplasms in horses and rabbits represent the same tumor type.
AS with MGCs in rabbits were highly invasive. Infiltration was commonly observed into surrounding connective tissue, deep fascia, skeletal musculature, tendons, and even bone, the latter with pathological fractures. Unfortunately, follow-up was not available for most cases, but in the few postmortem cases metastatic spread was frequent. Considering the high degree of invasiveness, frequent occurrence on the head or legs, and difficulty achieving complete excision, a high recurrence rate is expected and the overall prognosis should be considered guarded, similar to AS in dogs. 32
The current study indicates that giant cell sarcomas (ie, AS with MGCs, and HS) in rabbits can be differentiated by using immunohistochemistry for the histiocytic marker CD204, and this may be especially helpful in differentiating HS from AS with MGCs that have metastasized to viscera. 11,16 Of note, 2 of the described AS of abdominal organs had been initially diagnosed as HS and one disseminated HS had been initially diagnosed as a metastatic AS based on morphologic criteria alone. This supports the great benefit of immunohistochemistry for this tumor group. In contrast to dogs and cats, cutaneous and periarticular HS seem to be less common in rabbits and most HS occur in the lungs and liver causing rapidly progressive disease. 22 Further research is required to determine whether disseminated HS represents metastatic spread or spontaneous multicentric development.
In conclusion, giant cell sarcomas in rabbits may either represent AS or HS. While AS with MGCs occur most frequently in the skin or subcutis or, less commonly, in the skeletal muscle, HS occur primarily in the lungs. Immunohistochemical labeling with CD204 for HS and to a lesser extent SMA and desmin may be helpful in differentiating these 2 malignancies.
Supplemental Material
Supplemental Material, Combined_supplemental_materials-Bertram_et_al - Giant Cell Sarcomas in Domestic Rabbits (Oryctolagus cuniculus)
Supplemental Material, Combined_supplemental_materials-Bertram_et_al for Giant Cell Sarcomas in Domestic Rabbits (Oryctolagus cuniculus) by Christof A. Bertram, Michael M. Garner, Drury Reavill, Robert Klopfleisch and Matti Kiupel in Veterinary Pathology
Footnotes
Acknowledgements
We like to thank Dr. Ian Moore for his help compiling some of the data for a subset of the cutaneous rabbit sarcomas.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
