Abstract
Flat-Coated Retrievers seem to be at increased risk of developing soft-tissue sarcomas, and undifferentiated round cell or spindle cell sarcomas account for approximately 59% of sarcomas in the breed. In an attempt to classify these tumors further, formalin-fixed, paraffin-embedded sections from 14 undifferentiated sarcomas from Flat-Coated Retrievers were reviewed and examined with a panel of histologic and immunohistochemical stains. The panel included vimentin, desmin, Myo D1, smooth muscle actin, cytokeratin, S100, von Willebrand factor (factor VIII), Mac 387, CD3, major histocompatibility complex (MHC) class II, and CD79a. The majority of the sarcomas showed greater than 70% staining for MHC class II. We conclude that these undifferentiated sarcomas in Flat-Coated Retrievers belong to a spectrum of tumors with varying proportions of characteristic cell types and morphologic features, some of which fit the diagnostic criteria for malignant fibrous histiocytoma. Many of these sarcomas seem to have a significant myofibroblast component and a mild or moderate T cell infiltrate but the precise cell lineage is still uncertain.
In a recent survey of the type of tumors occurring in the Flat-Coated Retriever, we found that soft-tissue sarcomas accounted for 26% of all tumors and that undifferentiated round cell and spindle cell tumors made up approximately 59% of these sarcomas.15 Many of the undifferentiated sarcomas we examined in this study were located in skeletal muscle or around joints. Possible differential diagnoses included fibrosarcoma, rhabdomyosarcoma, and synovial sarcoma, although malignant fibrous histiocytoma (MFH), leiomyosarcoma, hemangiopericytoma, and neurofibrosarcoma also were considered. Because the histogenesis of this large group of sarcomas was not obvious from conventional light microscopic examination of sections stained with hematoxylin and eosin (HE), we selected a small group of undifferentiated sarcomas for which sufficient fixed tissue was available for histopathologic review and further examination with a panel of histologic and immunohistochemical stains.
Materials and Methods
Buffered formalin-fixed tumor specimens (variable fixation times) were submitted to the University of Cambridge for histopathologic examination as part of a survey on the occurrence of tumor types in Flat-Coated Retrievers. Sections were cut from paraffin-embedded specimens and stained with HE for histopathologic examination. Fourteen undifferentiated sarcomas were selected for review and further analysis with a panel of histologic and immunohistochemical stains. The conventional stains used were van Gieson, Masson trichrome, reticulin, and phosphotungstic acid hematoxylin (PTAH). The immunohistochemical antibodies used were vimentin (1:200, 3B4, mouse anti-bovine monoclonal, DAKO), desmin (1:50, DE-R-11, mouse anti-porcine monoclonal, DAKO), Myo D1 (1:50, mouse anti-human monoclonal, DAKO), smooth muscle actin (1:8,000, 1A4, mouse anti-human monoclonal, Sigma), cytokeratin (1:200, MNF116, mouse anti-human monoclonal, DAKO), S100 (1:50, rabbit anti-ovine polyclonal, DAKO), von Willebrand factor (1:400, rabbit anti-human polyclonal, DAKO), Mac 387 (1:100, mouse anti-human monoclonal, DAKO), CD3 (1:300, rabbit anti-human polyclonal, DAKO), major histocompatibility complex (MHC) class II (1:100, TAL.1B5, mouse anti-human monoclonal, DAKO), and CD79a (1:25, HM57, mouse anti-human monoclonal, DAKO). For immunohistochemistry, 3-μm-thick serial sections were cut from the appropriate blocks onto Vectabond-coated (Vector Laboratories) slides, deparaffinized in xylene, and rinsed in absolute alcohol; endogenous peroxidase activity was inhibited with 0.5% hydrogen peroxide in methanol for 10 minutes. Tissue sections used for desmin, Myo D1, and CD3 were heat treated for 2 minutes in 0.01 M citrate buffer, pH 6, in a pressure cooker (CD79a and MHC class II heated in a microwave for 10 minutes) for antigen exposure before application of the primary antibody. Tissue sections for vimentin, cytokeratin, S100, myoglobin, von Willebrand factor (factor VIII), and Mac 387 were treated with 0.1% trypsin at 37 C for 20 minutes. To reduce nonspecific binding, slides were incubated in 20% rabbit serum (for subsequent monoclonal) or 20% porcine serum (for subsequent polyclonal) in Tris-buffered saline for 10 minutes before application of the primary antibody for 60 minutes at room temperature. After rinsing, slides were incubated for 30 minutes with biotinylated rabbit anti-mouse (mouse monoclonal, DAKO) or biotinylated pig anti-rabbit (rabbit polyclonal, DAKO) antibodies at 1:400 in 3% normal human serum, followed by 30 minutes incubation with avidin–biotinylated complex (ABC, DAKO). Peroxidase activity was demonstrated with diaminobenzidine solution for 10 minutes and slides were counterstained lightly with Gill's hematoxylin for 2 minutes, before rinsing, dehydrating, clearing, and mounting with coverslips.
Positive control tissues included dog small intestine, muscle, spinal cord, and reactive lymph node, although other normal tissues on many of the tumor sections also acted as internal positive and negative controls for many of the antibodies. In addition, two fibrosarcomas and one lymphoma were included for comparison with the group of undifferentiated sarcomas. Negative controls included normal rabbit and mouse sera in place of the primary antibody.
Results
After reviewing the HE-stained sections, the undifferentiated sarcomas were classified histologically as malignant fibrous histiocytomas and grouped subjectively according to the classification system detailed in the Wold Health Organization International Classification of Tumors of Domestic Animals, vol. II.10 Seven tumors were classified as inflammatory (Fig. 1). These tumors displayed occasional bizarre histiocytoid cells, partially concealed by an inflammatory cell infiltrate of lymphocytes, plasma cells, eosinophils, and occasional neutrophils. Two tumors were classified as giant cell. These tumors had numerous multinucleated giant cells mixed with spindle cells and mononuclear histiocytic cells (Fig. 2). Five tumors were classified as storiform–pleomorphic (Fig. 3). These tumors consisted of fibroblast-like cells arranged in cartwheel (storiform) patterns and histiocytoid cells, typically with an infiltrate of lymphocytes, plasma cells, and occasional neutrophil. Histiocytoid cells were frequently karyomegalic or multinucleate, with nuclear atypia. In addition, the cohort also included two fibrosarcomas and a lymphoma. The fibrosarcomas displayed small elongated spindle cells arranged in bundles and whorls with the production of an obvious collagen (Masson trichome positive) matrix. The lymphoma displayed 90% positive staining for CD79a (B cell marker).
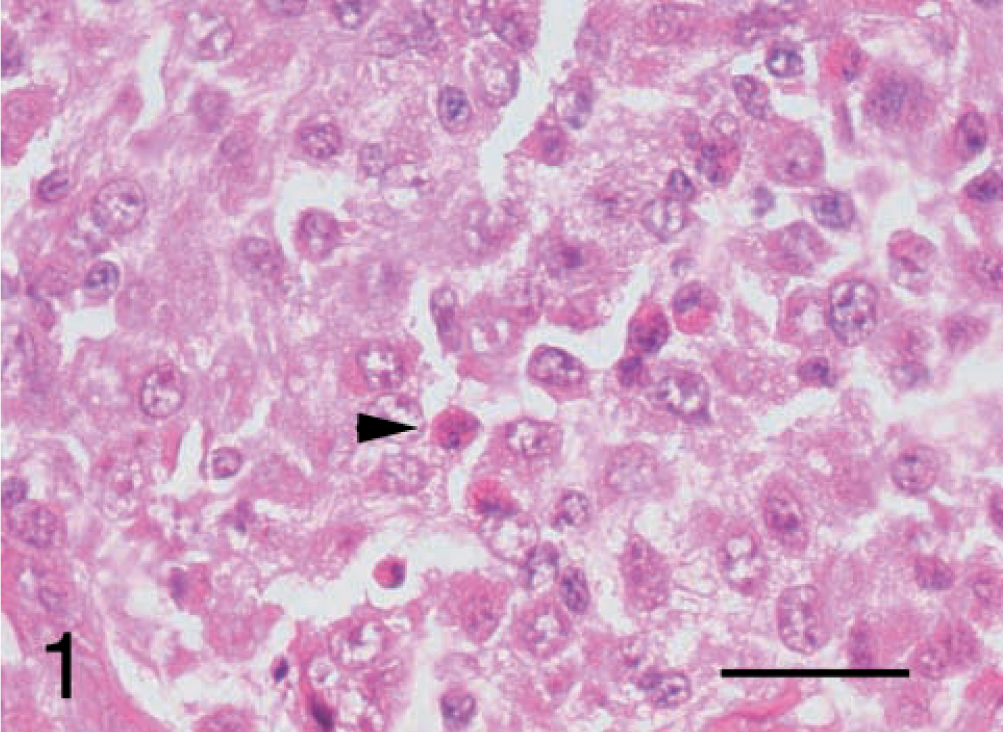
Malignant fibrous histiocytoma, subtype inflammatory; dog; tumor No. 6. The large influx of inflammatory cells is evident. The arrowhead indicates an eosinophil. HE. Bar = 25 μm.
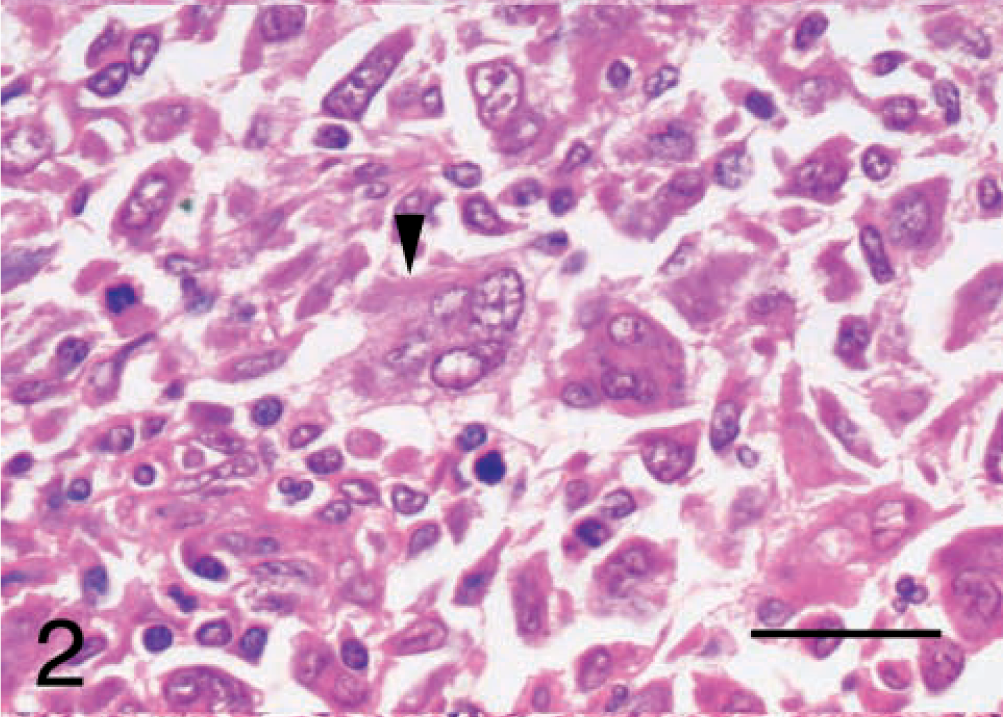
Malignant fibrous histiocytoma, subtype giant cell; dog; tumor No 8. The arrowhead indicates a multinucleate giant cell. HE. Bar = 25 μm.
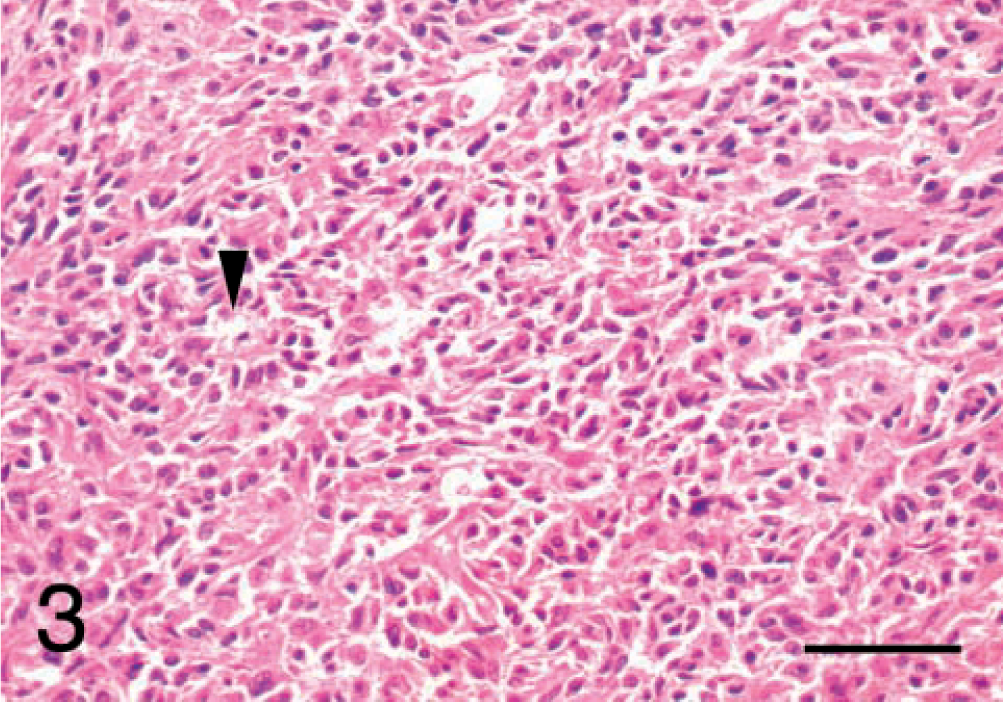
Malignant fibrous histiocytoma, subtype storiform–pleomorphic; dog No. 10. The arrowhead indicates the typical cartwheel arrangment of the malignant cells. HE. Bar = 50 μm.
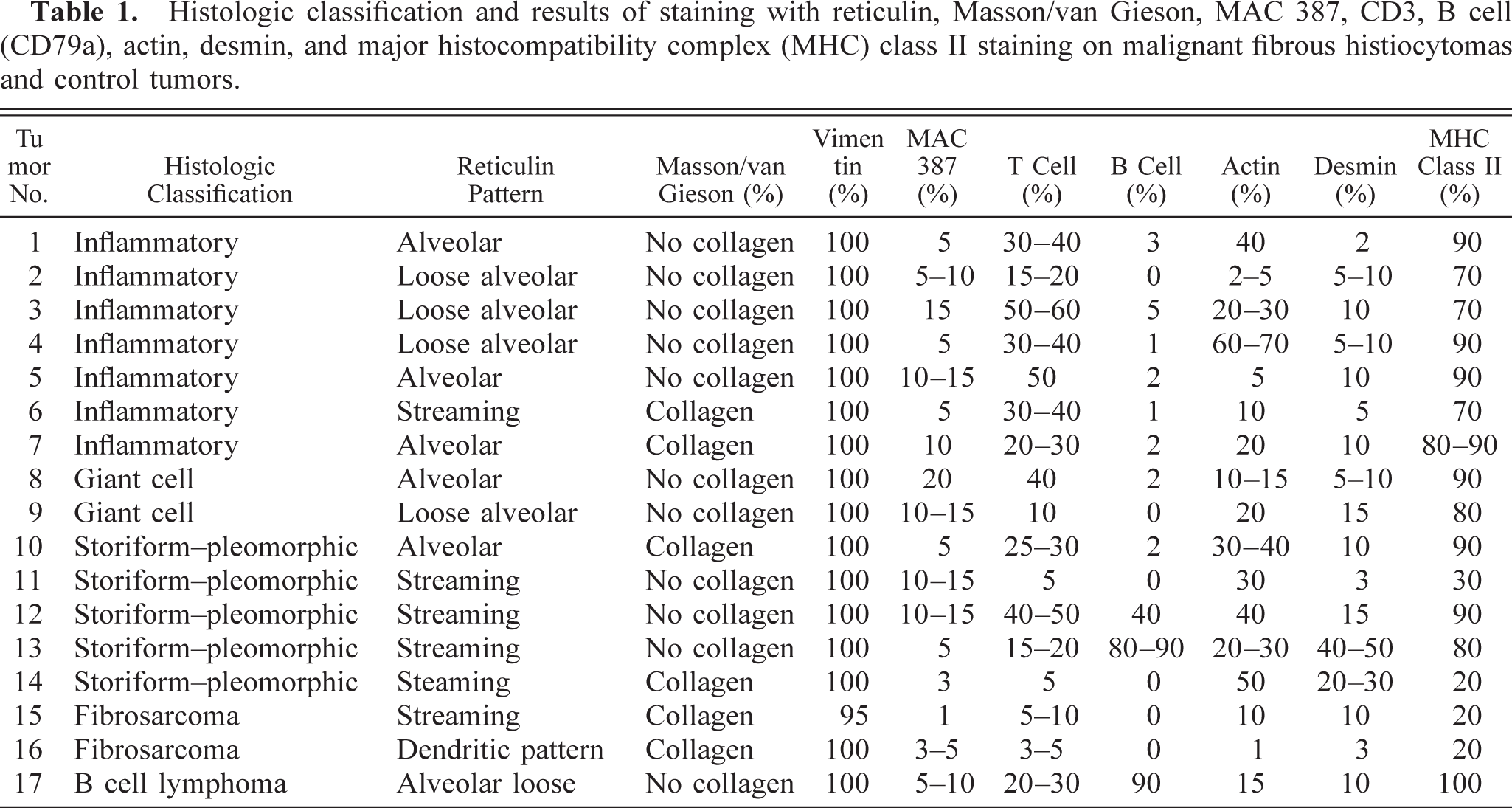
The results for the conventional histological stains are shown in Table 1. The Masson trichrome and van Gieson stains helped to identify intercellular collagen matrix. Both fibrosarcomas, two inflammatory tumors (Nos. 6 and 7), and two storiform–pleomorphic tumors (Nos. 10 and 14) showed evidence of collagen production. However, none of the tumors in the giant cell subtype groups showed evidence of collagen production. Muscle striations were not visible in any of the tumors stained with PTAH (results not shown). Several patterns were obvious with reticulin staining. The main ones were either dense streaming bundles of fibers or fibers arranged in an alveolar pattern. The alveoli were either large and loose or smaller, surrounding each cell. In general, the inflammatory and giant cell tumors were associated with a loose or small alveolar reticulin pattern and the storiform–pleomorphic tumors were associated with a streaming reticulin pattern.
Histologic classification and results of staining with reticulin, Masson/van Gieson, MAC 387, CD3, B cell (CD79a), actin, desmin, and major histocompatibility complex (MHC) class II staining on malignant fibrous histiocytomas and control tumors.
The results of the immunohistochemical stains that showed a positive result are summarized in Table 1 and illustrated in Figs. 4 and 5. For all antibodies, immunoreactivity was obvious in the positive control sections but was not detected in the negative controls. Vimentin immunoreactivity was detected in the majority of tumor cells in all 17 tumor sections, confirming their mesenchymal origin (Fig. 5). In contrast, none of the 17 tumors showed immunoreactivity for cytokeratin, Myo D1, S100, or von Willebrand factor (results not shown), although certain normal cells within some sections stained positively, that is, epidermis, bile ducts, and lung alveoli with cytokeratin; dermal melanocytes and follicular dendritic cells in lymph node with S100; skeletal muscle weakly with Myo D1; and blood vessel endothelium with von Willebrand factor.
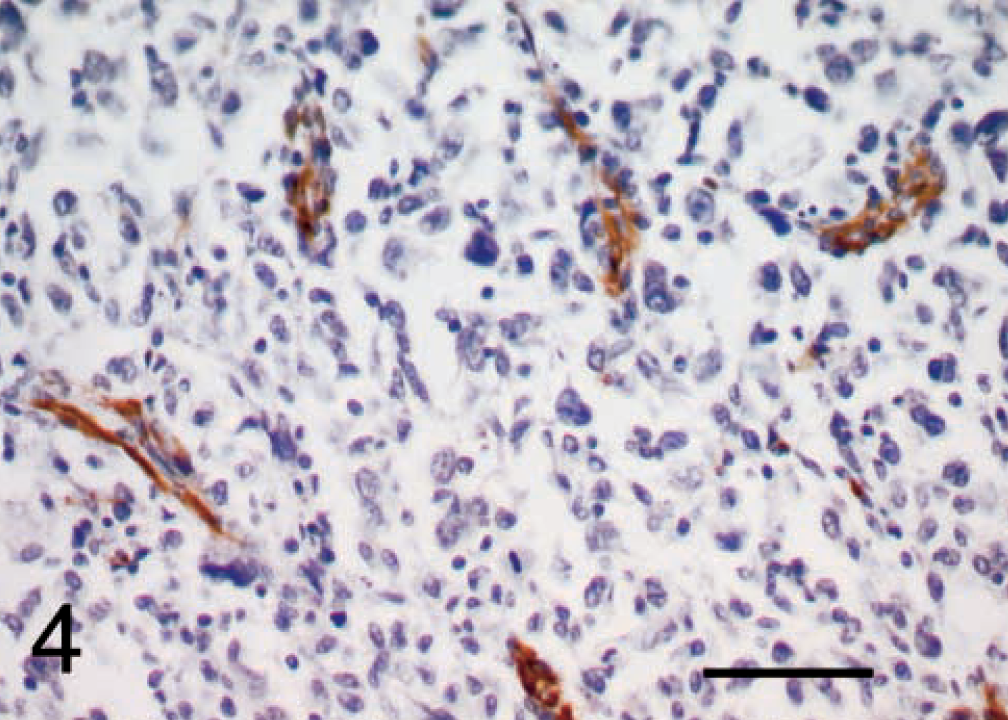
Malignant fibrous histiocytoma, subtype giant cell; dog No. 8. Actin immunoreactivity. The immunoreactive cells were not tumor cells, but rather were smooth muscle cells of blood vessels, stromal connective tissue cells, and other cells between the tumor cells arranged in a dendritic pattern. ABC, Gill's hematoxylin counterstain. Bar = 50 μm.
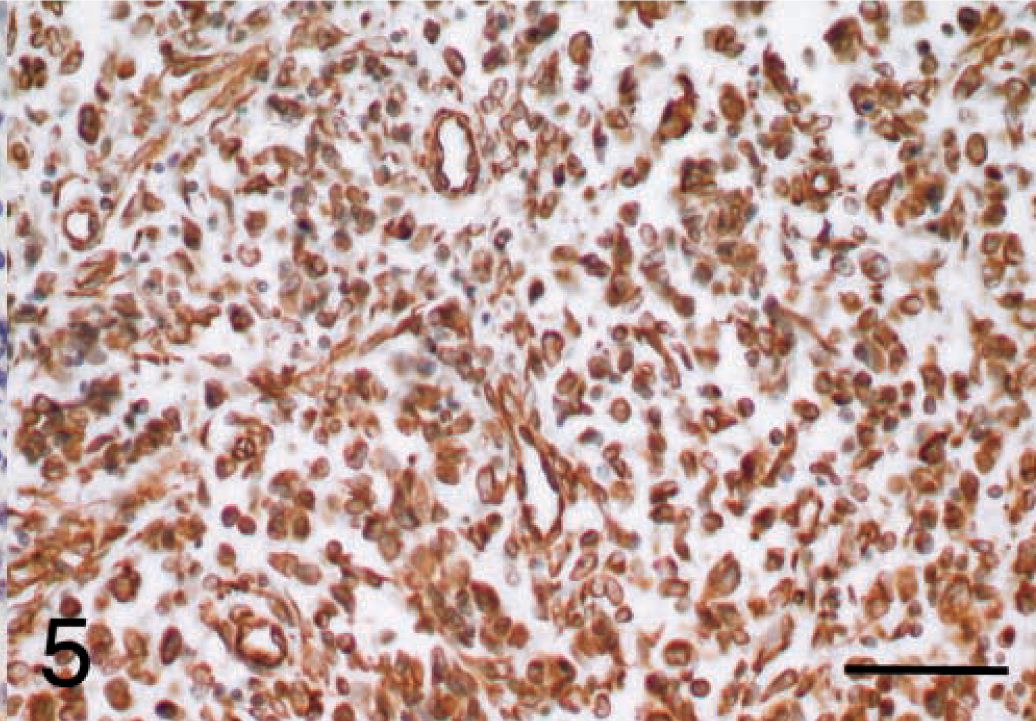
Malignant fibrous histiocytoma, subtype giant cell; dog; tumor No. 8. Vimentin immunoreactivity was detected in the majority of tumor cells in all 17 tumor sections, confirming their mesenchymal origin. ABC, Gill's hematoxylin counterstain. Bar = 50 μm.
Desmin and actin staining was variable. Usually, less than 15% of cells in each section were immunoreactive for desmin, although a higher percentage of cells stained positively in two tumors in the storiform–pleomorphic subgroup (No. 14, 20–30% and No. 13, 40–50% of cells). The staining was cellular although occasionally, background stroma without any obvious cellular component appeared to stain. A higher proportion of cells in each tumor were immunoreactive for actin, particularly in No. 14 and No. 4, where more than 50% of cells stained. However, for both actin and desmin, the immunoreactive cells were not tumor cells, but rather smooth muscle cells of blood vessels, stromal connective tissue cells, and other cells between the tumor cells arranged in a dendritic pattern (Fig. 4).
Mac 387 immunoreactivity was detected in only a small subpopulation of monocytes in both tumor and control sections.
In all except three tumors (No. 13, 80–90%; No. 12, 40%; No. 17, 90%), CD79a immunoreactivity was detected in only a very low percentage of cells, indicating minimal infiltration of B cells. Tumor No. 13 displayed 80–90% positivity with CD79a. Most of the staining was of the cytoplasmic granules present in the surrounding hepatocytes and this result was attributed to artifact. Tumor No. 17 was a confirmed B cell lymphoma and No. 12 was a MFH within a lymph node, hence the high B cell staining in these tumors. In immunoreactive cells, the stain was associated with the cytoplasmic membrane and produced a dark halo around the nucleus, although some nuclear staining was also apparent. In positive control sections of a reactive lymph node, staining was restricted to B cell areas, as expected.
A higher proportion of cells in each section were immunoreactive for CD3 (approximately 50% of cells in tumor Nos. 5, 12, and 3), confirming that most tumors had a mild or moderate lymphoid infiltrate consisting mainly of T cells (Fig. 6).
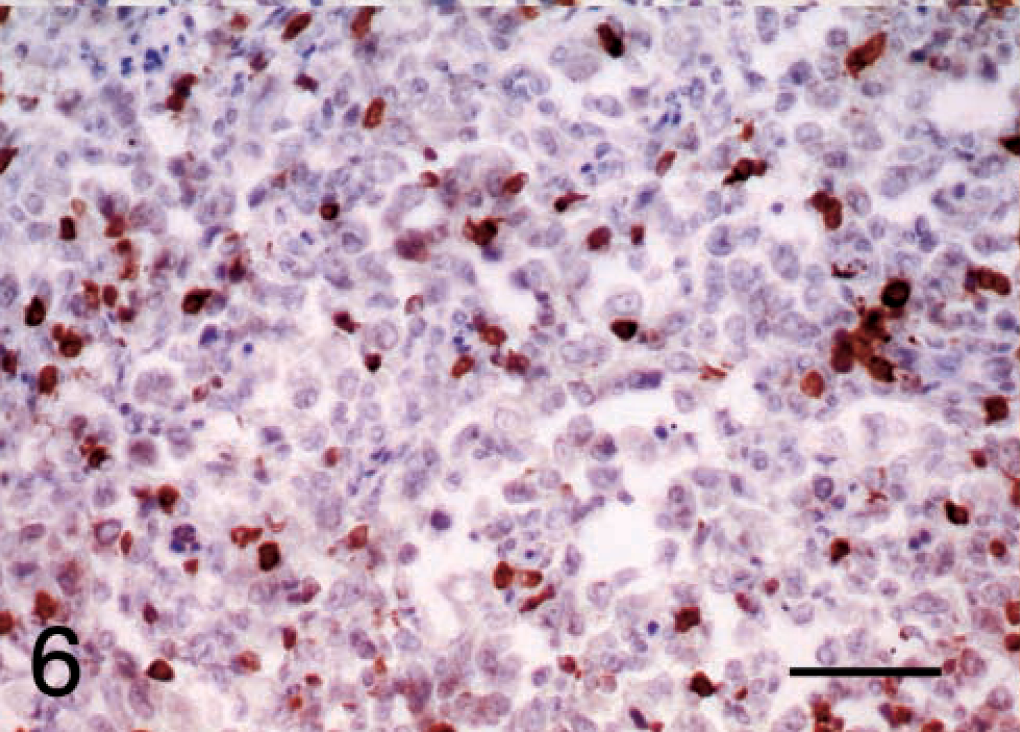
Malignant fibrous histiocytoma, subtype giant cell; dog; tumor No. 8. CD3 immunoreactivity confirms that most tumors had a mild or moderate lymphoid infiltrate consisting mainly of T cells. ABC, Gill's hematoxylin counter stain. Bar = 50 μm.
Twelve out of 14 of the MFH displayed levels of MHC class II staining that were greater than 70% (Fig. 7). All three subtypes of MFH were included in this group. Two storiform–pleomorphic MFHs showed reduced MHC class II immunostaining (tumor No. 11, which was 30%, and tumor No. 14, which was 20%). Both of the fibrosarcomas (tumor Nos. 15 and 16) displayed 20% staining with MHC class II. The B cell lymphoma (tumor No. 17) displayed 100% staining with MHC class II, but this is not surprising because B cells express MHC class II on their surface.11
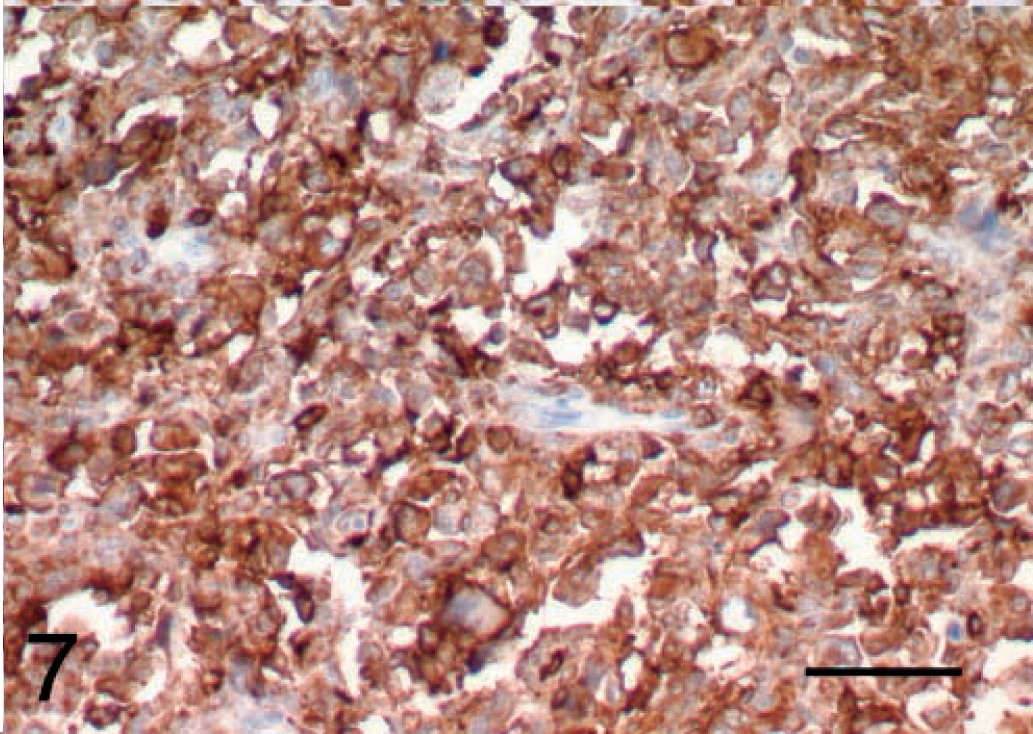
Malignant fibrous histiocytoma, subtype giant cell; dog; tumor No. 8. MHC class II immunoreactivity can be observed on most cells of this tumor. ABC, Gill's hematoxylin counter stain. Bar = 50 μm.
Discussion
A histologic review of 14 undifferentiated sarcomas taken from Flat-Coated Retrievers has indicated that these tumors could be reclassified as MFH, a malignant pleocellular neoplasm thought to arise from primitive mesenchymal cells.10 In the Flat-Coated Retriever, MFH seems to be part of a spectrum of tumors with varying proportions of characteristic cell types and morphologic features. The veterinary literature contains several reports of MFH occurring in purebred and mixed-breed dogs,2,5,9,12,16,18,19,22–27 but only one report included a Flat-Coated Retriever.12 In contrast, another retriever breed, the Golden Retriever, is one of the breeds most commonly affected with MFH.12,16,23,26 Interestingly, anaplastic sarcomas, which affect Flat-Coated Retrievers in the UK,15 also are reported to affect both Flat-Coated Retrievers and Golden Retrievers in the USA (Kisseberth, personal communication), suggesting that these two retriever breeds may be affected by the same sarcomas.
MFH is generally found in older dogs, but the tumor has been reported in a puppy.18 In this study, the mean age of affected dogs was 8.5 years (range 5–11 years). MFH is described as an extraskeletal, soft-tissue tumor and usually is considered to be locally aggressive but not highly metastatic in terms of clinical behavior.26 In the present study, nine of the dogs had presented with a primary limb mass, six sited deep in the musculature of the upper forelimb and three around the stifle. Two of these animals were euthanatized at the time of diagnosis and three others were known to have developed metastases after amputation of the limb. Four dogs had presented with massive and or widespread tumors involving one or more of the liver, spleen, mediastinum, and pleura. All of these cases were euthanatized and diagnosed at postmortem examination. Thus, in this relatively small number of dogs, MFH in the Flat-Coated Retriever seems to be more aggressive and to be associated with a higher rate of metastasis than is usual for this type of tumor. The giant cell variant of MFH in dogs previously has been reported as an aggressive tumor associated with poor prognosis.26
Microscopically, MFH consists of three components: fibroblastic cells, histiocytic round or polygonal cells, and multinucleated giant cells.6 Human MFH has been divided into five subtypes based on the pattern and predominance of the cell types: storiform–pleomorphic, inflammatory, giant cell, myxoid, and angiomatoid.10 Only the first three subtypes have been found with any consistency in the dog. By using histochemical and immunohistochemical staining, we were able to confirm these three MFH subtypes within the tumors in this series. Few other studies of canine MFH have presented immunohistochemical details and those that have were on small case series including a variety of breeds.9,16,25 In one large study of 32 immunostained fibrohistiocytic nodules of the spleen, it was unclear how many of the cases were MFH.23 Thus, the present study appears to be the first to report detailed imunohistochemical staining results for a moderately large group of confirmed cases of MFH.
Masson trichrome and van Gieson stains helped to demonstrate intercellular collagen matrix production in both fibrosarcomas. In addition, collagen production was also noted in two inflammatory MFHs (tumor Nos. 6 and 7) and two storiform–pleomorphic MFHs (tumor Nos. 10 and 14); however, collagen production is not unusual in MFH.20 PTAH staining failed to demonstrate any cross striations in the tumor cells, although this may not be surprising because morphologically, none of cells appeared straplike or showed obvious myoid differentiation. Reticulin staining helped to group the tumors broadly according to a pattern of streaming bundles of fibers or to an alveolar pattern. In general, the inflammatory and giant cell tumors were associated with a loose or small alveolar reticulin pattern and the storiform–pleomorphic tumors were associated with a streaming reticulin pattern.
All 17 tumors showed immunoreactivity for vimentin. Vimentin expression has been widely used in both humans and dogs to confirm the mesenchymal origin of various tumors.3,8,14,17,22 Thus, we concluded that all 17 tumors were sarcomas, as diagnosed on initial HE-stained sections. In contrast, none of the tumors showed immunoreactivity with the cytokeratin antibody used in this study.
Desmin and actin staining of the tumors helped to visualize a background network of supporting stromal cells that otherwise was undetected on HE-stained sections. Desmin has been used as an immunohistochemical marker for both canine rhabdomyosarcomas and leiomyosarcomas, whereas smooth muscle actin is restricted to leiomyosarcomas.1,22 Desmin immunoreactivity was low (<15% of cells stained) in all except two storiform–pleomorphic tumors. One other report has demonstrated desmin immunoreactivity in four storiform–pleomorphic cases of MFH,9 but others have shown variable immunoreactivity.16,25 Actin immunoreactivity was seen at levels of greater than 15% of tumor cells, in 11 of the 17 tumors in our study, and was present in all of the subgroups. However, the cells that stained were either vascular smooth muscle or stromal cells. The stromal cells were most likely to be myofibroblasts because these cells stain with actin and to a more variable extent, desmin.3,13
Myofibroblasts have been seen in human fibroblastic tumors and in tumors containing fibroblasts and histiocytes.4 In a study of mesenchymal tumors of the canine spleen, Spangler and coworkers22 attributed a high proportion of actin and desmin immunoreactivity in fibrosarcomas, leiomyosarcomas, and undifferentiated sarcomas to myofibroblasts and even postulated a common tumor histogenesis from a population of splenic myofibroblasts. In our study, the myofibroblast content of the tumors was not considered sufficient for a diagnosis of myofibrosarcoma, except in tumor No. 14 (storiform–pleomorphic) and No. 4 (inflammatory), which had very high actin immunoreactivity. It is unclear why these two tumors should have such a high stromal, possibly myofibroblast component, unless they represent a stromal reaction to the tumor cells. Interestingly, Hendrick and coworkers9 also demonstrated high actin immunoreactivity (>50% tumor cells staining) in three splenic tumors of storiform–pleomorphic MFH with inflammation, but lower immunoreactivity in three other cases.
S100 immunoreactivity was not demonstrated in any of the MFH tumors in our study. Although other studies agree with this finding,16,25 weak S100 immunoreactivity has been seen in a few other examples.9,23
B and T cell staining confirmed that a lymphoid response to the tumors was present. The majority of tumors had a mild or moderate T cell infiltrate, as would be expected as part of a cell-mediated immune response. Only three tumors had a significant B cell content. One of these (tumor No. 2) was situated within a lymph node and so the B lymphocyte content was not unexpected. The second tumor was in liver (tumor No. 13) and staining here was considered to be an artifact induced by liver enzymes. The lymphoma (tumor No. 17), included as a control, displayed 90% staining with CD79b antibody.
A human histiocyte–monocyte marker, Mac 387, was used to see if it would detect canine histiocytes in the tumor sections, but even on control tissue (reactive lymph node), Mac 387 only reacted with a small proportion of monocytes. When used on sections of canine malignant histiocytosis, no immunoreactivity of histiocytes was detected and this was attributed to a lack of complementary antigenic epitopes of the human antigen.7 Unfortunately, no other histiocyte marker is commercially available for fixed canine tissue at the present time, although an anti-canine mouse monoclonal for CD18 has been made and used on frozen tissue by one group.21 α1-Anti-chymotrypsin and lysozyme immunoreactivity have been used by some researchers to try and demonstrate the presence of histiocytes in MFH.16,25 However, we have had very inconsistent results with these antibodies, and did not include them in our panel.
Twelve out of 14 MFHs demonstrated greater than 70% MHC class II positivity. All three subtypes of MFH were included in this group. Canine MHC class II antigens are constitutively expressed on T lymphocytes, antigen-presenting cells such as macrophages, and B lymphocytes.11 Although the high class II expression seen in 12 out of 14 of the MFHs may represent macrophages, the possibility also exists that the class II expression may have been induced on the MFH cells by cytokine stimuli. Because no MFH tumor displayed 100% MHC class II staining, it is likely that MFH is not an exclusively histiocytic tumor, but rather is made up of cells with a differentiation toward either fibroblasts or macrophages.18
Although the histiocytes in MFH are thought by some to be neoplastic, the origin of both histiocyte-like cells and multinucleated giant cells in MFH has been the subject of much debate.6,12 A recent report of a histiocytic infiltrate within a human MFH transplanted into SCID mice suggests that MFHs induce the infiltration of reactive monocytes and macrophages into the tumor,6 leading the authors to infer that histiocyte-like cells in MFH should be considered as a reactive monocyte–macrophage lineage rather than as an element of the neoplasm. This finding also was suggested by Schneider and coworkers.20 Thus, the histiocytic-looking cells in the Flat-Coated Retriever sarcomas also may not be a neoplastic component of the tumor.
In conclusion, we believe that a proportion of the undifferentiated sarcomas previously reported to affect the Flat-Coated Retriever can be reclassified as MFHs. Special staining techniques assisted this classification. Immunostaining was able to demonstrate the mild to moderate lymphoid infiltrate present in the MFH, particularly in the inflammatory subtype, and both histochemical staining and actin and desmin immunostaining were able to help in the classification of MFH into the three different subgroups. The tissue of origin of MFH remains uncertain.
Footnotes
Acknowledgements
We would like to acknowledge Lisa Happerfield, Alison Warn, Madeline Fordham, and Andrea Kemp for technical assistance in conventional and immunohistochemical staining of tissue sections. We would like to thank Dr. Nick Coleman of the Department of Histopathology, Addenbrookes Hospital, Cambridge, and Mark Stidworthy of the Department of Clinical Veterinary Medicine, Cambridge, for help in the histopathologic classification of the MFH; and Nick Woodger and Dr. Ross Harley of the Department of Clinical Veterinary Medicine for their help with production of the photomicrographs. JSM is in receipt of an Alison Alston Canine Award from the Royal College of Veterinary Surgeons.
