Abstract
Stillbirth and neonatal mortality are substantial problems in captive bottlenose dolphins (Tursiops truncatus). The cause of these problems often is unknown. We report a case of Escherichia coli septicemia in a male 3-day-old bottlenose dolphin calf. Lesions included omphalitis, synovitis, and hepatic necrosis associated with the presence of Gram-negative bacilli. E. coli was isolated in pure culture from multiple organs. A serum gammaglobulin level of 1.5 g/L indicated a lack of maternally acquired immunity. The observed failure to nurse may have resulted from brain injury due to perinatal asphyxia. Evidence for perinatal asphyxia was the diffuse presence of a moderate amount of meconium in the lungs.
Stillbirth and mortality in the first 3 months after birth are substantial problems in captive breeding programs of bottlenose dolphins (Tursiops truncatus). 4 Because the causes of these problems often are not known, it is difficult to take appropriate remedial actions. Performing necropsies and reporting on such cases could improve our knowledge in this area. Here we report a case of Escherichia coli septicemia possibly resulting from lack of maternally acquired immunity in a bottlenose dolphin calf.
A male bottlenose dolphin calf was born on the 17th of May 2000 at the Dolfinarium Harderwijk, The Netherlands. The cow was 8 years old, born in captivity, and had not given birth before. The date of conception was unknown. The cow was kept in a 10,000-m3 pool, together with other bottlenose dolphins, throughout pregnancy and parturition. The pool contained natural seawater, which was filtered by dual layer sand carbon filters. Filter speed was 1,000 m3 per hour. From March 2003 to January 2005, the water of the basin was tested 13 times at approximately monthly intervals for the presence of coliform bacteria including E. coli. Water samples were diluted 10 times with 0.1% pepton, applied to 3M EC Petrifilm (3 M Microbiology, St. Paul, MN), and incubated for 24 hours at 41°C. No coliforms were found in any of the samples.
The calf was delivered 135 minutes after the tail flukes became visible in the vulva. No complications were noted during labor. No nursing was observed from birth to death, despite the observation of milk leaking from the cow's teats during the first few days after parturition. At 75 hours after birth the calf's breathing frequency rose from 4 to 8 breaths per minute. The calf died 82 hours after birth (Fig. 1).

Bottlenose dolphin calf, lateral view of carcass. Ruler = 33 cm.
Gross necropsy was performed according to a standard protocol. 8 The nutritional condition was poor, based on the deep indentation dorsally in between the head and the body, and the lack of fat in the interventricular grooves of the heart. No ingesta were present in the gastric compartments or intestinal tract. Multiple hemorrhages, ranging from 1 mm to 5 cm in diameter, were observed in the subcutis of the left and right dorsolateral sides of the head, the subcutis of the left and right sides of the mandible, and in the muscle tissue dorsal and lateral to the atlanto-occipital joint. Petechial hemorrhages were found 5 cm caudal to the pylorus in a 2-cm-long segment in the mucosa of the duodenum and in the hepatic capsule in a 1-cm-wide band on either side of the falciform ligament. There was pulmonary edema and congestion, characterized by abundant white foamy fluid in the bronchi and diffuse reddening of the lung parenchyma, respectively. The atlanto-occipital joint contained 1 to 2 mL of slightly viscous, red-yellow fluid and had a dark-red synovial membrane, indicating synovitis. The surrounding muscle was edematous. The blubber layer was diffusely yellow, indicating neonatal jaundice.
The following tissues were sampled for histology: adrenal gland, atlanto-occipital joint, aorta, brain, bronchus, cerebellum, cerebrum, cervical spinal cord, colon, duodenum, esophagus, fore stomach, fundic stomach, heart, pituitary gland, jejunum, kidney, liver, lung, mesenteric lymph node, muscle, pancreas, pulmonary lymph node, pyloric stomach, skin erosion on mandible, spleen, thymus, thyroid, trachea, tracheo-bronchial lymph node, umbilicus, urachus, and urinary bladder. Tissue samples were fixed in 10% neutral-buffered formalin, routinely processed, and embedded in paraffin. Five-micrometer-thick sections were mounted on glass slides and stained with hematoxylin and eosin (HE). Selected lung sections were stained with monoclonal antibody AE1/AE3 (Neomarkers, Fremont, CA) to identify keratin-containing cells. Sections of adrenal gland and navel were stained with Gram stain to characterize bacteria. For quantification of the proportion of lung parenchyma containing keratin squames or meconium, 50 arbitrarily chosen fields (40× objective) of HE-stained lung sections from each lung were examined for the presence of keratin squames, meconium, or both.
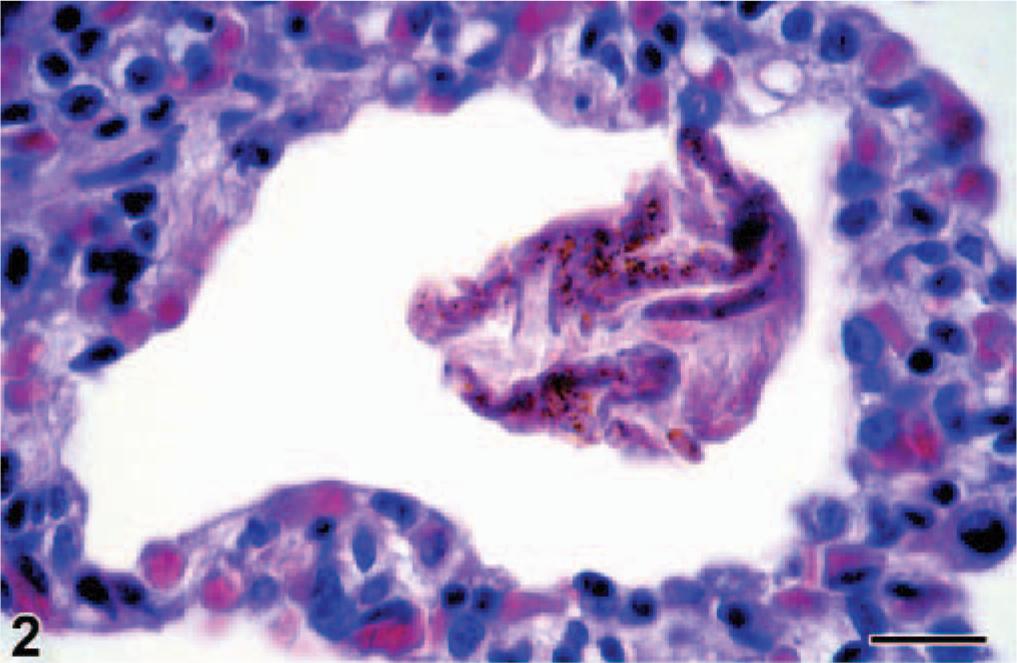
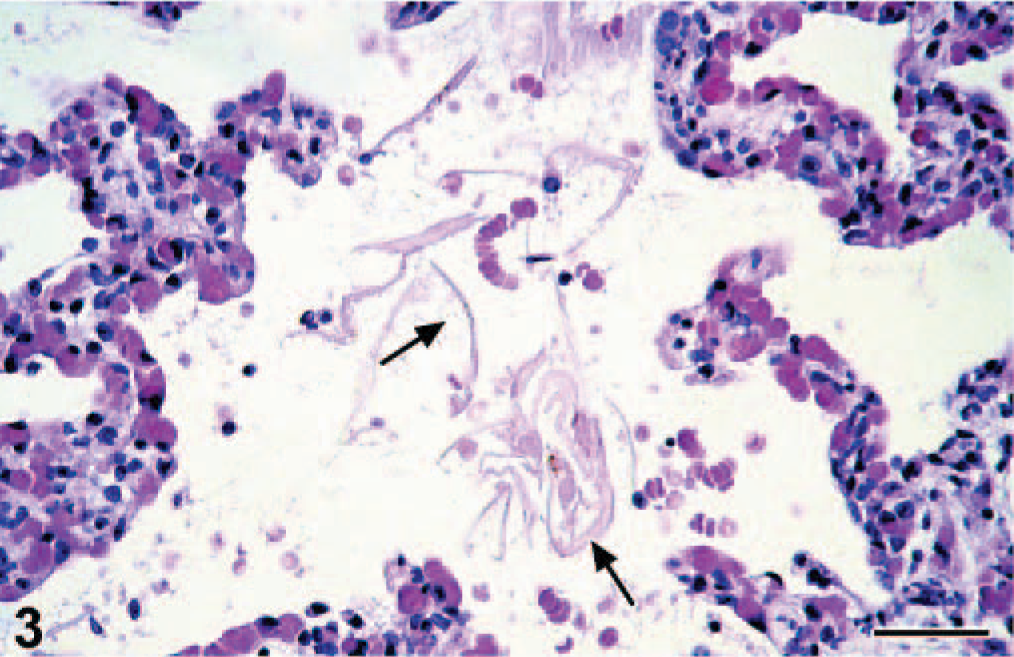
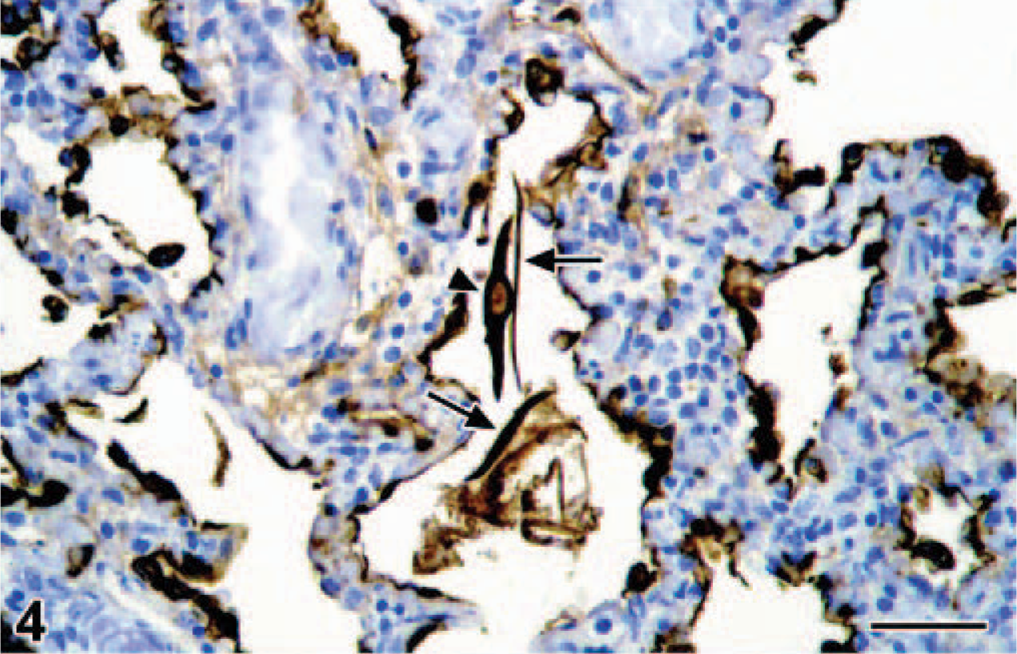
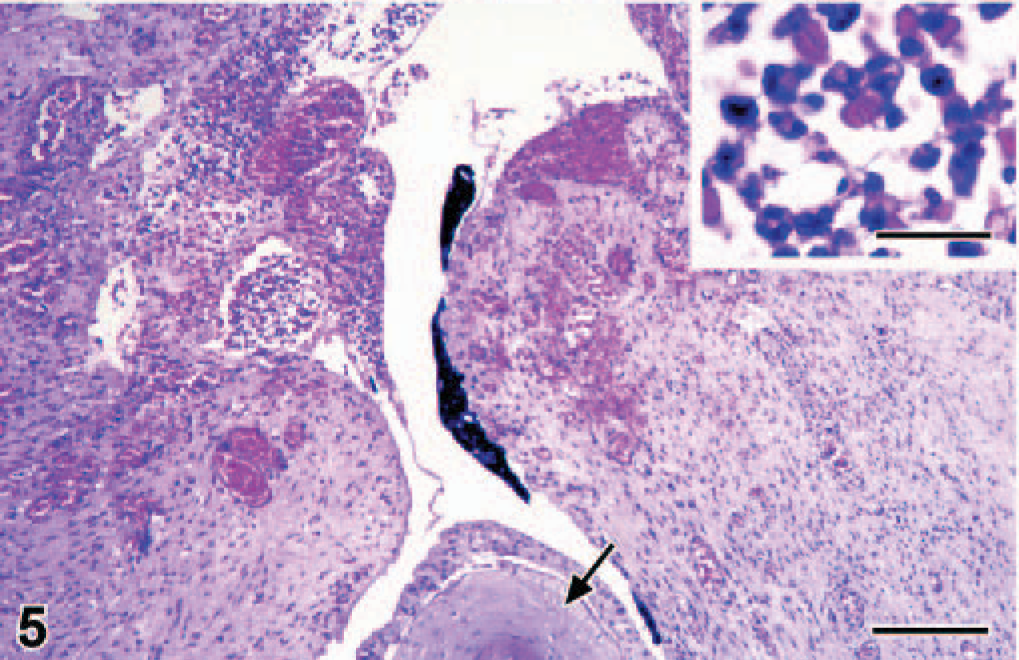
By histology, the lungs had diffuse congestion, edema, and aspiration of meconium. The meconium was present in moderate amounts in the lumina of alveoli and bronchioles and was visible as amorphous material containing yellow to brown granules (Fig. 2). Meconium was detected in 33% of the 40× objective fields of lung sections examined. Also present were fibrin and large numbers of filamentous pink structures, some with a central nucleus (Fig. 3). By immunohistochemistry, these structures were positive for keratin (Fig. 4) and were identified as keratin squames, which are sloughed epithelial cells from the oronasal region and amniotic fluid. 5 Keratin squames were detected in 58% of the 40× objective fields of lung sections examined. There was a severe purulent and hemorrhagic omphalitis, characterized by many neutrophils and erythrocytes mixed with aggregates of Gram-negative bacilli in the umbilical vessels and surrounding connective tissue (Fig. 5). A similar purulent and hemorrhagic infiltrate with bacilli was observed in the subintima of the synovial membrane of the atlanto-occipital joint and in the adjacent edematous muscle. Some of the bacilli were found in the cytoplasm of the neutrophils, confirming bacterial infection during life. The liver had multiple random foci of hemorrhage and necrosis. In these areas, the hepatocytes were dissociated and had irregular vacuolation and loss of definition of the cytoplasm. In multiple tissues, there were aggregates of Gram-negative bacilli in the blood vessels. Tissues affected were trachea, bronchus, tracheo-bronchial lymph node, synovial membrane of the atlanto-occipital joint, cerebellum, cervical spinal cord, urinary bladder, adrenal cortex (Fig. 6), myocardium, thymus, pancreas, hypothalamus, esophagus, pylorus, small and large intestine, liver, eye, and aorta.
Lung; bottlenose dolphin calf. The lumen of the pulmonary alveolus contains keratin squames mixed with meconium, which is characterized by brown granules. HE. Bar = 10 μm.
Lung; bottlenose dolphin calf. The lumina of the pulmonary alveoli contain multiple keratin squames (arrows), visible as filamentous pink structures. HE. Bar = 25 μm.
Lung; bottlenose dolphin calf. The keratin squames (arrows) in the lumina of the pulmonary alveoli express keratin. A nucleus (arrowhead) is visible in one keratin squame. The epithelial cells lining the alveolar walls also express keratin. Avidin-biotin-peroxidase complex method for keratin counterstained with Mayer' hematoxylin Counterstain. Bar = 25 μm.
Navel; bottlenose dolphin calf. There is hemorrhage and infiltration by neutrophils in the connective tissue subjacent to the skin of the navel (arrow). The amorphous black material is the inspissated remnant of an umbilical vessel. HE. Bar = 150 μm. Inset: Higher magnification of the neutrophils and erythrocytes in the connective tissue of the navel. HE. Bar = 15 μm.
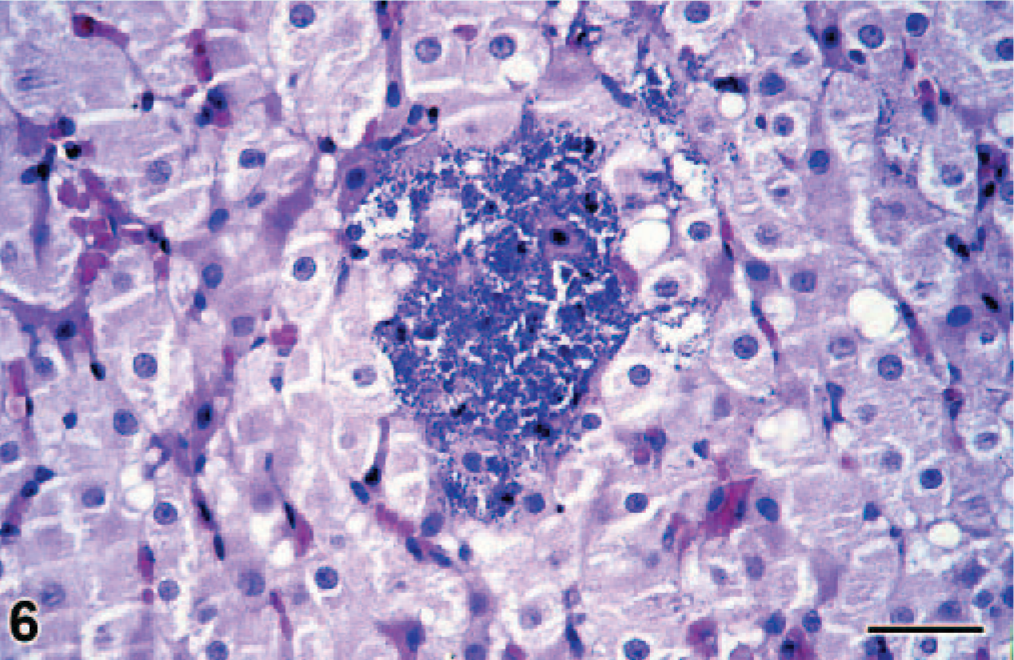
Adrenal gland; bottlenose dolphin calf. There is a large aggregate of bacilli in the adrenal cortex. HE. Bar = 25 μm.
Bacteriologic examination was performed on swabs from the cerebellar meninges, spinal cord, atlanto-occipital joint, bronchi and umbilical vein, and on tissue samples from brain, liver, lung, spleen, kidney, and intestine. Tissue samples were superficially heat sterilized by quickly passing the specimen through a flame. All samples were inoculated on 2 blood agar plates and a MacConkey agar plate apart from intestine, which was inoculated in selenite enrichment broth for Salmonella sp. retrieval and subsequently inoculated on a brilliant green agar plate. All plates were incubated at 37°C. For each sample, a blood agar plate was inoculated under anaerobic conditions, a chocolate agar plate under micro-aerophilic conditions, and the remaining plates under aerobic conditions. After 24 hours, suspect colonies were inoculated in tubes containing triple sugar iron, urea, indole, and ornithine decarboxylase for bacterial identification. All cultures were kept at 37°C for 48 hours before being judged negative.
E. coli was isolated in abundance in pure culture from all tissue specimens except for the spleen. Spleen may have tested negative because the sample was small and therefore completely sterilized by superficial heat sterilization. Salmonella sp. was not isolated from the intestine.
The serum gammaglobulin level was measured by agarose gel electrophoresis (Paragon system, Beckman Coulter Inc, Fullerton, CA). This neonate had a gammaglobulin level of 1.5 g/L in the postmortem serum.
To our knowledge, this is the first reported case of E. coli septicemia in a bottlenose dolphin calf. The diagnosis was based on the detection in multiple organs of bacterial aggregates—some associated with hemorrhage and neutrophil infiltration—and the isolation of E. coli in pure culture. E. coli is an important cause of neonatal septicemia in domestic animals and is associated with lesions similar to those found in this case. 2 Infection of the bottlenose dolphin calf likely occurred during birth, as happens in other species, including horses. 3 In the bottlenose dolphin, the anus is located at the caudal tip of the genital slit. Defecation during birth will be done directly onto the calf. During nursing attempts, the calf is positioned thus that defecation of the cow will lead to the calf passing through a fairly concentrated volume of feces diluted with salt water. There was no evidence that the water of the basin was contaminated with E. coli, and because E. coli survives poorly in salt water, 10 contamination of the water was unlikely. Repeated testing from 2003 to 2005 confirmed this supposition. The portal of entry was most likely the umbilicus because it had the most marked infiltration of neutrophils and was therefore probably the oldest site of inflammation. It is noteworthy that no inflammatory changes were visible in the umbilicus on gross necropsy.
The E. coli septicemia probably resulted from lack of maternally acquired immunity. The bottlenose dolphin has an epitheliochorial placenta, 12 which prevents in utero transfer of maternal antibodies to the fetus. The bottlenose dolphin calf is therefore dependent upon colostrum to acquire maternal antibodies, including those against ubiquitous pathogens such as E. coli. Diagnosis of lack of maternally acquired immunity was based on the concentration of gammaglobulin, 1.5 g/L, in postmortem serum. This is low compared with serum gammaglobulin levels in adult bottlenose dolphins (5–20 g/L, 7 adults, C. E. van Elk unpublished data) and in neonatal bottlenose dolphins (3–7 g/L, 6 neonates that died within 13 days after birth). 1
The lack of maternally acquired immunity in this dolphin probably resulted from failure to nurse. This finding was based on the lack of observed nursing attempts, despite the fact that milk was seen leaking from the cow's teats during the first few days after birth. Furthermore, it was based on the absence of colostrum in the digestive tract and the poor nutritional condition of the dead calf.
Failure to nurse may have resulted from brain injury resulting from perinatal asphyxia. Perinatal asphyxia might have been caused by either compression of the umbilical cord during birth or premature separation of the placenta. The duration of the birth, defined by the time needed from the expulsion of the tail flukes to the expulsion of the entire calf, was within normal ranges. In this dolphin, birth took 135 minutes versus an average of 94 minutes (range 45–240 minutes) for live births in other marine mammal facilities and an average of 188 minutes (range 20–600 minutes) for stillborn calves. 7 The duration of the birth does have a relation with the outcome, but it is not completely clear-cut. This was a primiparous cow and it might be speculated that because of the tighter constriction of the birth canal in a primiparous mother, umbilical compression is more likely to occur. In dolphins, the birth canal has no bony elements, and birth is realized by forcing the neonate through the cervix and vulva. Perinatal asphyxia in foals may cause brain damage associated with behavioral abnormalities such as loss of coordinated swallowing and suckling reflexes, and inability to locate the udder. 6 The same is true for infants, where mild hypoxic brain damage results in a decreased level of alertness and delayed onset of normal suckling behavior. 11 Although we did not find any brain lesions by routine histology, this does not preclude mild brain damage in this dolphin.
Evidence for perinatal asphyxia in this calf was the diffuse presence of a moderate amount of meconium and large numbers of keratin squames in both lungs. Because of perinatal asphyxia, a distressed fetus will often pass meconium into the amniotic fluid. Gasping during the later phase of asphyxia may then cause in utero aspiration of the meconium. 5 However, it should be noted that a small amount of meconium can be found in the majority of neonates in cattle. 9 In future necropsies on neonatal bottlenose dolphins, in which perinatal asphyxia can be excluded, it should be determined whether or not meconium is present in the pulmonary alveoli. If so, quantification of the amount of meconium and keratin squames in the pulmonary alveoli may be needed to determine whether it is an incidental finding or a sign of perinatal asphyxia.
In conclusion, here we report a case of E. coli septicemia in a bottlenose dolphin calf, associated with meconium aspiration, lack of maternally acquired immunity, and omphalitis. We speculate that the sequence of events was perinatal asphyxia, brain damage, failure to nurse, lack of maternally acquired immunity, omphalitis caused by E. coli infection, and E. coli septicemia. Pathologists examining stillborn calves and neonatal mortality of bottlenose dolphins should take into account perinatal asphyxia as a possible underlying cause, and specifically look for associated lesions. The knowledge gained from performing and reporting on necropsies of stillborn calves and neonates will be instrumental in improving the reproductive success of bottlenose dolphins in captivity.
Footnotes
Acknowledgements
We are grateful to Dr. Jaap Wagenaar and Ms. Marian Brockhuizen-Stins of the Veterinary Medical Diagnostic Center of the University of Utrecht, the Clinical Chemistry department of the St. Jansdal Hospital in Harderwijk, and to Ms. Fiona Read and Ms. Vivian Emmer for technical assistance. We thank the Dolfinarium in Harderwijk for their commitment to and support of this research.
