Abstract
This article reports the results of necropsy, parasitologic, microbiologic, histopathologic, immunohistochemical, indirect immunofluorescence, biomolecular, and serologic investigations on 8 striped dolphins (Stenella coeruleoalba) found stranded from August to December 2007 on the Ligurian Sea coast of Italy. Severe, nonsuppurative meningoencephalitis was found in 4 animals, as characterized by prominent perivascular mononuclear cell cuffing and macrophage accumulations in neuropil. These lesions were associated with mild lymphocytic–plasmacytic infiltration of choroid plexuses in 1 dolphin. Toxoplasma gondii cysts and zoites, confirmed by immunohistochemical labeling, were scattered throughout the brain parenchyma of 2 of the 4 dolphins. No viral inclusions were seen in the brain of any animal. Other findings included severe bronchointerstitial pneumonia and pulmonary atelectasis, consolidation, and emphysema. Parasites were identified in a variety of organs, including lung (Halocerchus lagenorhynchi). Microbiologic and serologic examinations for Brucella spp were negative on all 8 dolphins. The 4 animals with meningoencephalitis had serum antibodies against T gondii (titers ranging from 1:80 to 1:320) but not against morbillivirus. In contrast, the other 4 dolphins were seropositive for morbillivirus (with titers ranging from 1:10 to 1:40) but seronegative for T gondii. No morbillivirus antigen or nucleic acid was detected in the tissues of any dolphin. It is concluded that the severe lung and brain lesions were the cause of death and that T gondii was the likely etiologic agent of the cerebral lesions. Morbillivirus infection was not considered to have contributed to death of these animals.
Toxoplasma gondii, a protozoan agent infecting a range of mammalian species worldwide, has been implicated as a cause of abortion and lethal systemic disease in several sea mammal species. 11 Although the potential of this protozoan to affect cetacean populations has not been thoroughly investigated thus far, a number of reports clearly suggest that T gondii infection is of potential concern to cetacean health and conservation. 9,11,32
T gondii is commonly believed to be an opportunistic pathogen for aquatic mammals, 24,26 as was the case in striped dolphins (Stenella coeruleoalba) during the dolphin morbillivirus (DMV) epidemic in the Mediterranean Sea from 1990 to 1992. 7,21
Between late 2006 and early 2007 another morbillivirus epidemic was reported in pilot whales (Globicephala melas) in the Mediterranean Sea near Gibraltar 12 and, in the following months, in pilot whales and striped dolphins along the Spanish coast. 28 Apart from its less dramatic scale, this more recent event shared many similarities with the Mediterranean striped dolphin die-off in this area from 1990 to 1992. 6,21 Nucleotide sequence analysis indicated that the DMV strain causing the recent mortality episodes in pilot whales and striped dolphins is closely related to that involved in the 1990–1992 die-off. 12,28
As in the earlier event, 4,5,21 unusually high striped dolphin mortality was recorded along the coast of Italy during this recent morbillivirus epidemic, including the Ligurian coast of northwestern Italy from August to December 2007 (F. Garibaldi and W. Mignone, personal communication). The aim of this study was to investigate possible causes of death of these animals, with emphasis on four cases of T gondii infection.
Materials and Methods
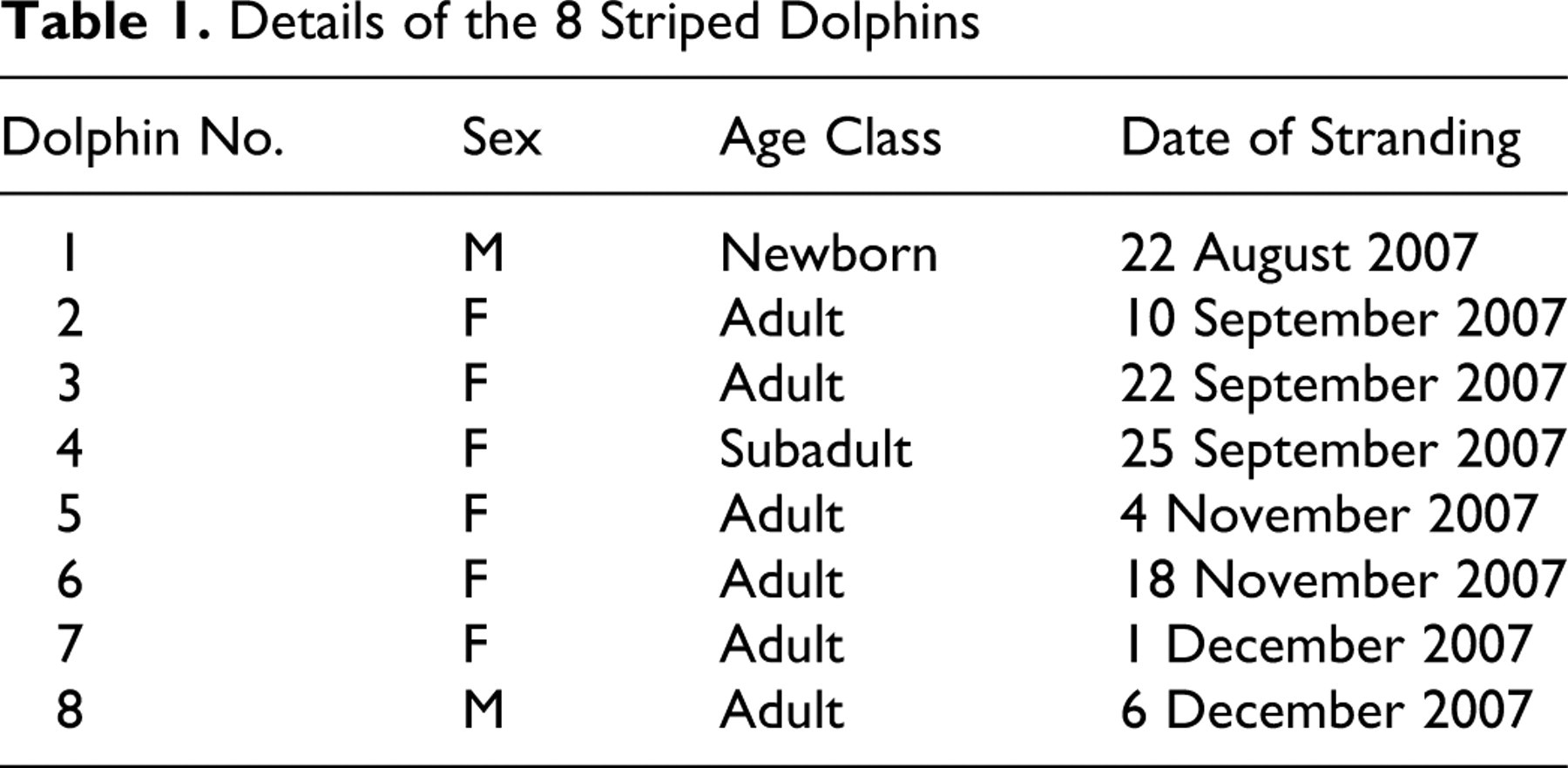
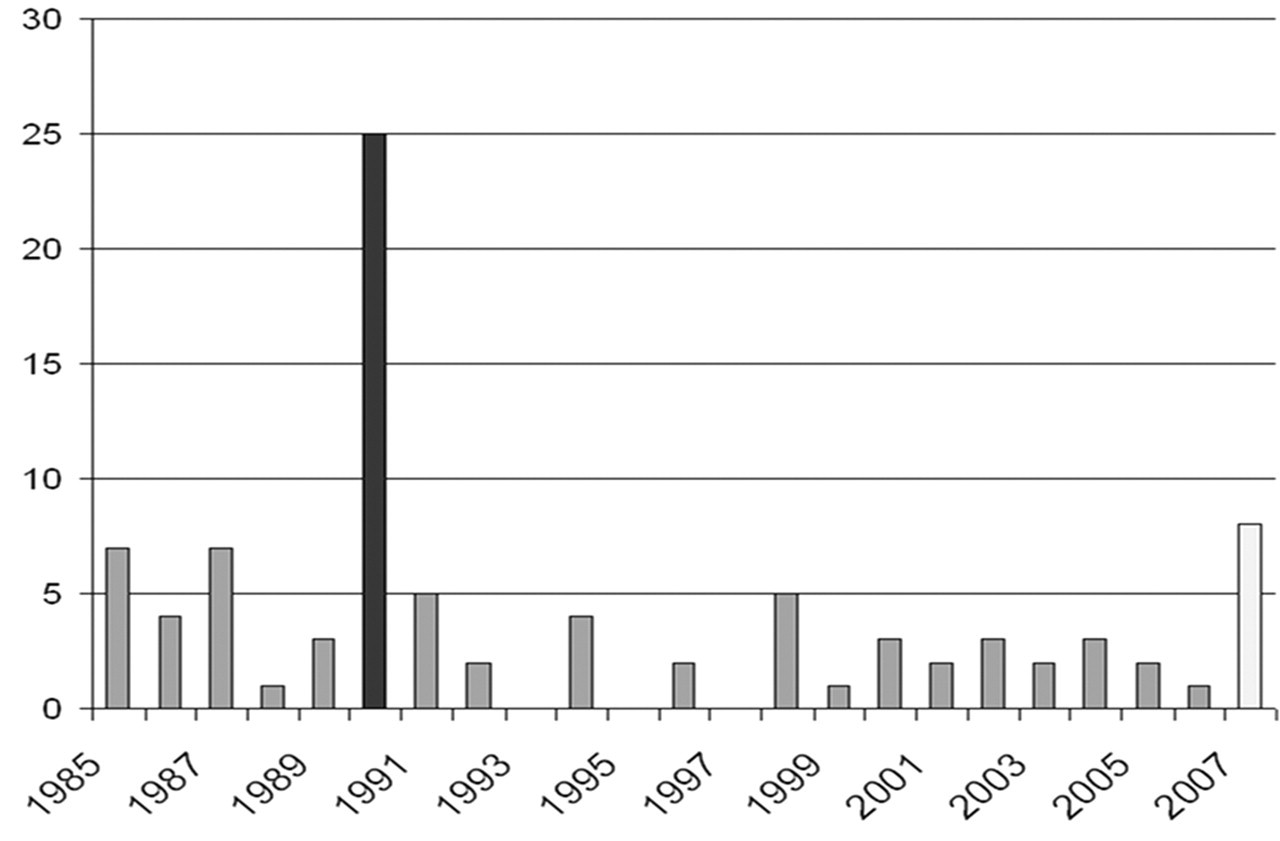
The study was carried out on 8 striped dolphins (Table 1 ) that had been found dead along the Ligurian Sea coast of Italy (Imperia province) from August to December 2007 (Fig. 1 ). The annual mean number of cetaceans found stranded during the previous 22 years (1985–2006) in this region is 4, with S coeruleoalba being the most commonly stranded species. The 2007 stranding figures show an increase over previous years (Fig. 2 ).
Details of the 8 Striped Dolphins
At necropsy, tissue samples were collected from a range of organs (lung, heart, stomach, intestine, liver, pancreas, kidney, adrenal gland, testis, ovary, uterus, spleen, mesenteric, tracheobronchial and pulmonary lymph nodes, skeletal muscle, and skin), along with the entire brain from each animal. Tissues were fixed in 10% neutral-buffered formalin or stored at 4°C and –20°C until used. At necropsy, a blood sample was collected from the heart chambers of each dolphin, and serum was removed by immediate centrifugation at 1,000 to 1,500 revolutions per minute for 15 minutes.
All tissues were embedded in paraffin wax, cut into 5-µm-thick sections, and stained with hematoxylin and eosin for light microscopic examination. Systematic tissue sampling was carried out on the brain of each animal with sections prepared from the telencephalon, diencephalon, mesencephalon, pons, cerebellum, and medulla oblongata. Periodic acid–Schiff (PAS) histochemical staining was performed on selected brain sections, whereas Gram staining was carried out on brain slices from 2 of the 8 dolphins under study.
Immunohistochemical and Indirect Immunofluorescence Labeling
Immunohistochemical (IHC) labeling of T gondii was carried out on brain and lung tissues from all dolphins. Antigen retrieval was performed by exposing poly-L-lysine-coated slides to a Tris buffer solution (pH 7.6) containing 0.1% trypsin and 0.1% calcium chloride. Endogenous peroxidases were inhibited by transfering sections to a solution of hydrogen peroxide in methanol, and nonspecific antigen binding sites were blocked by applying normal horse serum. Sections were then incubated with a commercially available goat polyclonal anti–T gondii antiserum solution (1:500; VMRD Inc, Pullman, WA). A secondary biotinylated horse anti-goat immunoglobulin G (IgG) solution (Sigma-Aldrich Inc, Saint Louis, MO) was applied to sections, followed by signal amplification with avidin-biotin-conjugated peroxidase. The reaction was visualized by means of 3,3′-diaminobenzidine (DAB) chromogen solution (Sigma-Aldrich Inc), followed by counterstaining with Mayer hematoxylin.
T gondii–infected ovine brain and canine lung were used as positive control tissues. For negative controls, the IHC technique was applied to these tissues but with omission of the primary antiserum or to sections of normal or pathologically unrelated ovine, canine, and dolphin tissues.
Indirect immunofluorescence (IIF) and IHC labeling for morbillivirus antigen was carried out on brain, lung, spleen, and lymph nodes from each animal. For IHC staining, antigen retrieval was performed by exposing poly-L-lysine-coated slides to a citrate buffer solution (pH 6). Endogenous peroxidases were inhibited by transferring sections to a solution of hydrogen peroxide in methanol, and nonspecific antigen binding sites were blocked by applying normal goat serum. Sections were subsequently incubated with a commercially available mouse monoclonal antibody (MoAb) solution (1:500) against canine distemper virus (CDV) nucleoprotein antigen (VMRD Inc) that recognizes the same epitope from different members of the Morbillivirus genus, including DMV. 28,34 A secondary biotinylated goat anti-mouse IgG solution (DAKO Corp, Carpinteria, CA) was applied to sections, followed by signal amplification with avidin-biotin-conjugated peroxidase. The reaction was visualized by means of DAB chromogen solution (Sigma-Aldrich Inc), and slides were counterstained with Mayer hematoxylin. 22,30 For IIF labeling of morbillivirus antigen, the same primary MoAb (1:100 dilution) was applied to tissue sections, followed by incubation for 3 hours with a goat anti-mouse IgG secondary antibody conjugated with fluorescein isothiocyanate (FITC) (KPL Inc, Gaithersburg, MD) and observation under a Zeiss Axiophot fluorescence microscope.
For both IHC and IIF staining, morbillivirus-positive brain, lung, spleen, and lymph node samples were used as control tissues. These included CDV-infected dog tissues, DMV-infected striped dolphin tissues, and phocine (phocid) distemper virus–infected common seal tissues. For negative controls, the IHC and IIF techniques were carried out on uninfected striped dolphin tissues and on tissues from known DMV-infected striped dolphins but with omission of the primary MoAb.
Microbiology
Detailed microbiologic investigations were carried out on brain and lung tissues of all 8 dolphins. Representative tissue samples were cultured onto 5% blood agar and subsequently incubated at 37°C for 48 hours under aerobic conditions (brain and lung) or microaerobic (5% CO2) conditions (brain only). Brain was also cultured on the following selective media under both aerobic and microaerobic conditions: Gassner agar, chocolate agar, Saboraud agar, Brucella agar, and Thayer-Martin-modified agar. All inoculated media were incubated at 37°C for 48 hours, except for Brucella-selective agar plates (which were incubated at 37°C for 10 days under microaerobic conditions) and Saboraud agar dishes (which were incubated for 10 days at room temperature). All cultures were checked daily.
Bacterial colonies were identified by means of commercial API galleries (bioMérieux Inc, Durham, NC). Suspect fungal colonies were subsequently transfered onto additional Saboraud agar plates and identified by means of the rRNA 16s Microseq 500 sequencing kit (Applied Biosystems, Foster City, CA). Identification was based on amplification of a 500-base-pair fragment from the 16s fungal ribosomal subunit 2,15,23 and the D2 fragment from the large subunit (LSU) of fungal rRNA. 16,17
Molecular Biology
A reverse transcription polymerase chain reaction (RT-PCR) technique with morbillivirus-specific genomic sequences was applied to tissue extracts from different regions of brain from all 8 dolphins. In this technique, 2 nucleoprotein gene fragments of 287 and 575 base pairs were selected as targets based on the universal morbillivirus primers P1 (5′-ACAGGATTGCTGAGGACCTAT-3′) and P2 (5′-GCACCGTACATGGTTATCTTG-3′) (GenBank accession No. EF451565), 13,34 as well as the DMV-specific primers DMV_REV (5′-AGATGGGCGAGACTGCACC-3′) and DMV_FW (5′-ATCAGGGCTCACTTTTGCATCCAGA-3′) (GenBank accession No. NC005283). Total RNA was extracted from 25 mg of each tissue sample by means of the RNeasy kit (Qiagen GmbH, Hilden, Germany) according to the manufacturer’s instructions. The assay was subsequently performed in 25 µl of reaction mixture containing 1X PCR Master Mix (Invitrogen Corporation, Carlsbad, CA), 10 U of RNase inhibitor, 50 U of MuLV RT (Applied Biosystems), 100 pmol/µl of each primer, and 2 µl of RNA. RT-PCR was carried out in a single-step reaction of 60 minutes at 48°C, followed by reverse transcriptase denaturation at 95°C for 2 minutes. Amplification was conducted by means of 35 cycles at 94°C for 45 seconds, at 48°C for 45 seconds, and at 72°C for 60 seconds followed by 7 minutes of final extension at 72°C.
Brain tissue extracts from a CDV-infected dog and a DMV-infected striped dolphin were used as positive control materials for the RT-PCR technique.
Serology
The sera from the 8 dolphins under study were evaluated by an immunofluorescence antibody test (IFAT) for the detection of anti–T gondii antibodies. A commercially available kit containing killed T gondii tachyzoites as substrate antigen and feline T gondii–positive and T gondii–negative control sera (Fuller Laboratories, Fullerton, CA) was used in this method. Briefly, 10 µl of serial twofold diluted sera, starting at 1:10, was added to 12-well slides. After 30 minutes of incubation at 37°C, a mixture of rabbit anti–bottlenose dolphin IgG (1:5000) was added for another 30 minutes at 37°C. The reaction was visualized by means of FITC fluorochrome-conjugated anti-rabbit IgG, and slides were observed under a Zeiss Axiophot fluorescence microscope. As in a study that compared various serologic tests for T gondii, 8 a cutoff titer of 1:40 was established, and endpoint dilution titers of positive sera were determined.
A virus neutralization assay to detect anti-morbillivirus antibodies was also performed, using the Onderstepoort strain of CDV. Briefly, 50 µl of each serial twofold diluted serum, starting at 1:5 dilution, and 50 µl of infectious culture medium containing 100 TCID50 of the virus were mixed and incubated in 96-well flat-bottomed plates for 1 hour at 37°C. One hundred microliters (1 × 105 cells/ml) of Vero cell suspension were then added to each well. Cultures were finally incubated for 3 days at 37°C with 5% CO2. Seropositivity was defined as a titer of 1 ≥ 20. 31,33
Finally, all sera were submitted to a rapid serum agglutination test for anti–Brucella spp antibodies based on Brucella abortus and B melitensis antigens. 19,29
Results
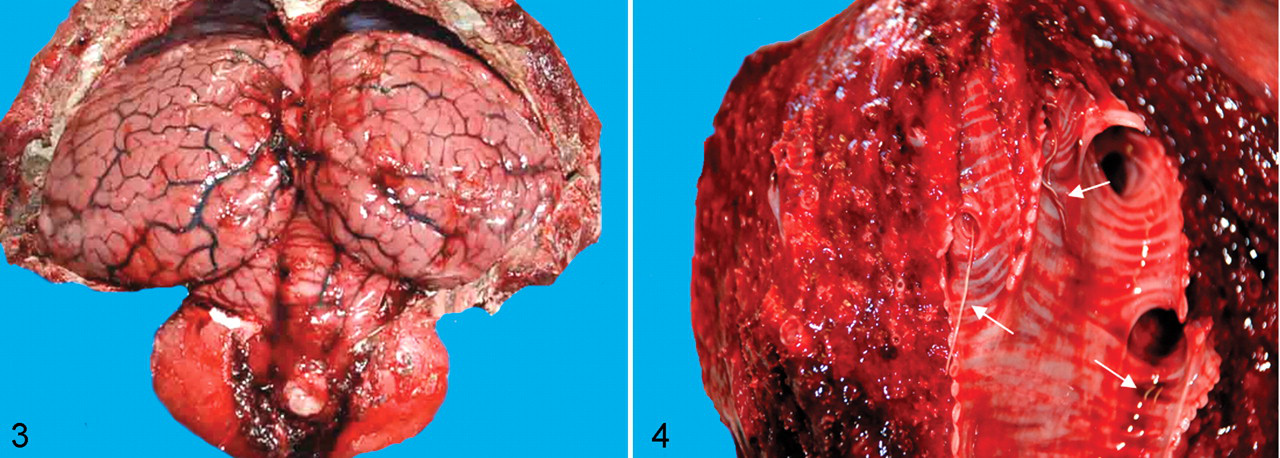
Table 2 summarizes the main pathologic findings in the 8 cetaceans under study. All carcases were well preserved, and some of the animals were in suboptimal nutritional status, as indicated by reduced blubber thickness. Gross evidence of meningeal hyperemia and brain edema was seen in 4 dolphins (3 females and 1 male) (Fig. 3). Two of these animals had large numbers of Phyllobothrium delphini and Monorygma grimaldii larval cysts in the subcutaneous tissue of perineal region and ventrolateral abdominal musculature, respectively (Table 3). Six animals (75%) had severe bilateral bronchointerstitial pneumonia, characterized by partial lung collapse, extensive areas of consolidation, and prominent subpleural emphysema (particularly in the apical regions of both lungs). Adult nematodes, morphologically consistent with Halocerchus lagenorhynchi (formerly H delphini), were present in the bronchial lumina of these dolphins (Fig. 4) and were associated with moderate catarrhal exudate and frothy fluid, indicative of pulmonary edema.
Nutritional Status, Gastric Content, and Main Lesions in the 8 Striped Dolphins a
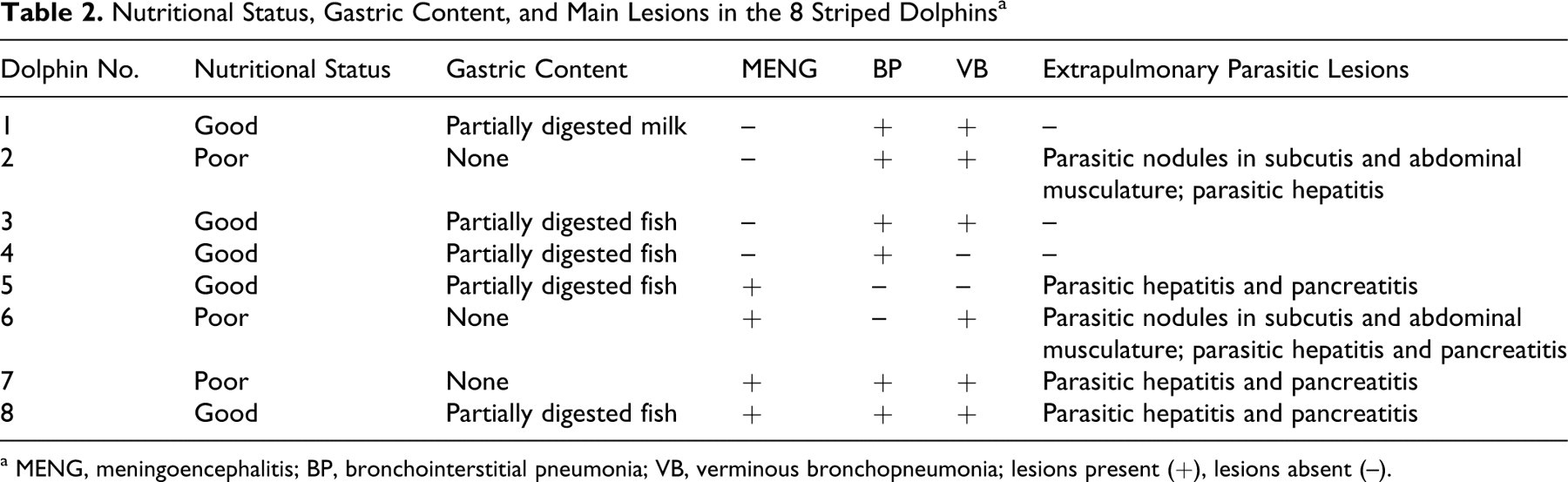
a MENG, meningoencephalitis; BP, bronchointerstitial pneumonia; VB, verminous bronchopneumonia; lesions present (+), lesions absent (–).
Microbiologic and Parasitologic Findings in the 8 Striped Dolphins a
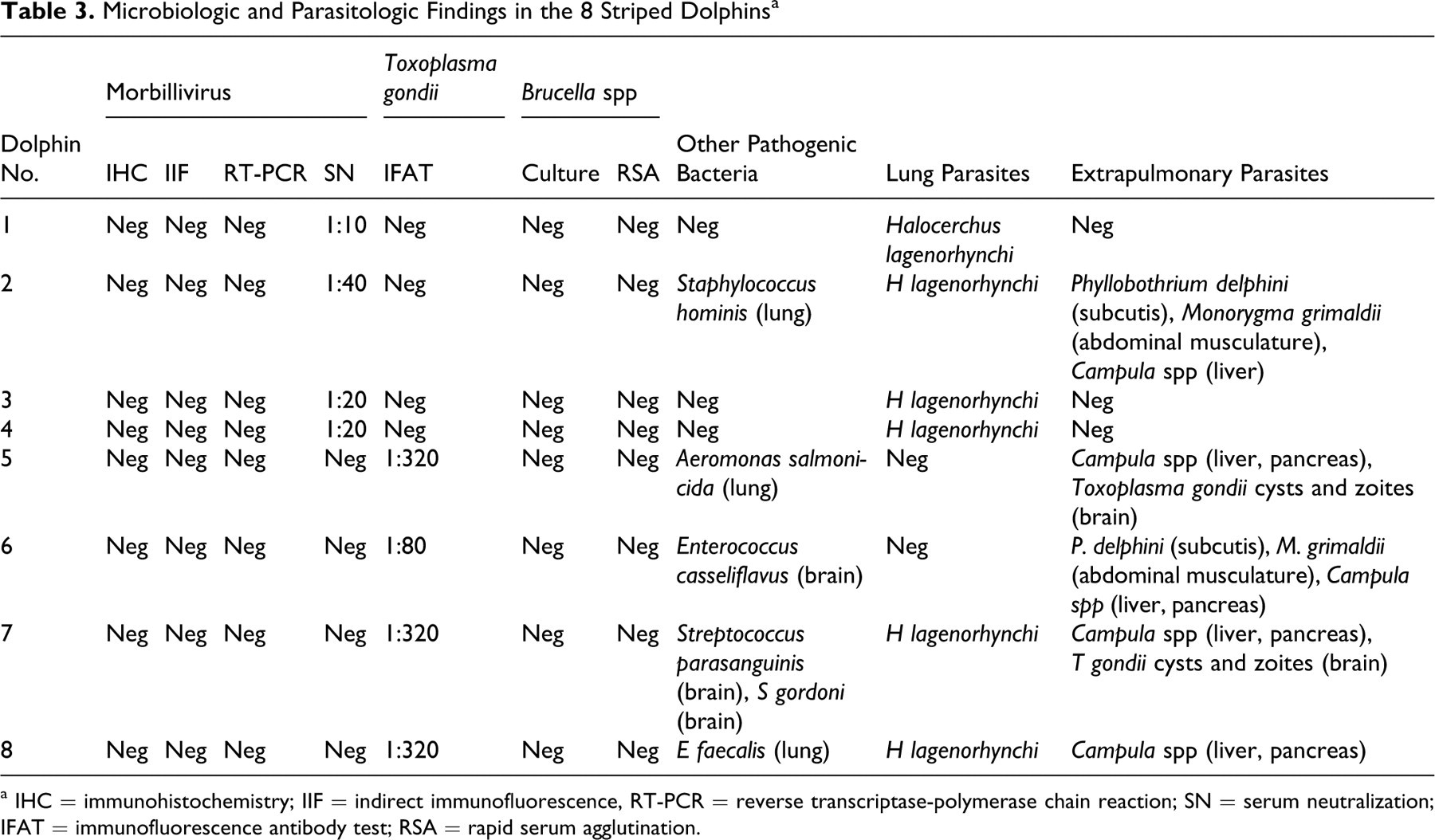
a IHC = immunohistochemistry; IIF = indirect immunofluorescence, RT-PCR = reverse transcriptase-polymerase chain reaction; SN = serum neutralization; IFAT = immunofluorescence antibody test; RSA = rapid serum agglutination.
Severe multifocal, subacute to chronic, nonsuppurative meningoencephalitis was present in 1 male and 3 female adult dolphins (50%). These lesions mainly involved the pons and mesencephalon and consisted of prominent perivascular mononuclear cell cuffing and focal microglial nodules scattered throughout the surrounding neuropil (Fig. 5). Macrophage accumulations in proximity to blood vessels and mild lymphoplasmacytic infiltration of choroid plexuses were also seen in the adult male dolphin. A microangiopathy in which the wall of affected vessels had an “onionlike” appearance was seen in all 4 of these animals. Astrogliosis and mild to moderate neuronal degeneration and necrosis characterized by vacuolation, chromatolysis, and shrinkage were also observed. Single, round, basophilic PAS-positive structures morphologically consistent with T gondii cysts (Fig. 6) were scattered throughout the brain parenchyma of 2 of these dolphins (Table 3). Viral inclusions were not seen in the brain of any animal, and no bacteria were apparent in Gram-stained brain tissue of 2 animals.
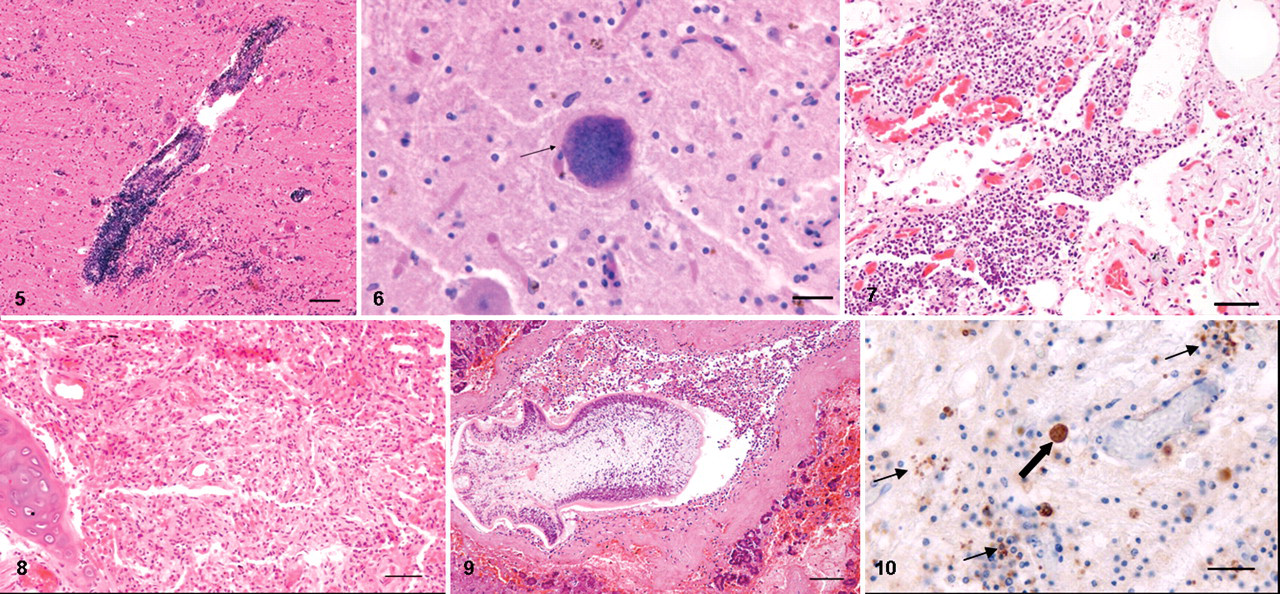
Microscopic lung lesions were characterized by seromucous exudation and macrophage infiltration of bronchial, bronchiolar, and alveolar lumina and by the presence of occasional double- to triple-nucleated alveolar syncytia. Lesions of severe chronic interstitial pneumonia, as characterized by lymphoid cell infiltration, extensive alveolar atelectasis, and marked septal fibrosis, were present in these animals (Figs. 7, 8). Parasitic granulomas were found scattered throughout both lungs.
Mild to moderate, multifocal, subacute to chronic lesions of nonsuppurative portal hepatitis were seen in 4 female and 1 male adult dolphins (62%). Four of these animals (50%) also had severe pancreatitis (Table 2), characterized by an infiltrate of lymphocytes, plasmacytes, and eosinophils and by the presence of extensive hemorrhagic and necrotic foci. Many metazoan parasites and ova, morphologically consistent with Campula spp, were observed in the hepatic and pancreatic (Fig. 9) parenchyma and ductal lumina of all 5 of these dolphins (Table 3).
No other significant microscopic findings were observed in any animal.
Immunohistochemistry and Indirect Immunofluorescence
Immunohistochemical labeling of T gondii cysts and zoites was seen scattered throughout the brain tissue of 2 dolphins that had meningoencephalitis (Fig. 10). T gondii antigen was not identified in any tissue from any of the other 6 dolphins. Immunohistochemical and IIF labeling for morbilliviral antigen was negative on all dolphins.
Microbiology
Several bacteria were isolated from the lung tissue of 3 dolphins and the brain tissue of 2 dolphins (Table 3). No Brucella spp or fungi were recovered from any animal.
Molecular Biology
Morbillivirus nucleic acid was not detected in the tissues of any dolphin.
Serology
The sera from the 4 dolphins that had severe lesions of meningoencephalitis were positive for anti–T gondii antibodies, at titers ranging from 1:80 to 1:320, but did not contain anti-CDV neutralizing antibodies. Conversely, anti-CDV neutralizing antibodies at titers ranging from 1:10 to 1:40 were detected in the sera of the 4 dolphins that did not have meningoencephalitis. No anti–T gondii antibodies were detected in sera of these animals. Antibodies to B abortus or B melitensis were not detected in any animal.
Discussion
All the dolphins investigated had parasitic infection and associated lesions in either lung or in liver, pancreas, subcutis, and abdominal skeletal musculature. Four (50%) had severe nonsuppurative meningoencephalitis and 6 (75%) had severe chronic bilateral pneumonia. These lesions almost certainly contributed to death, and their frequencies were similar to those previously reported in stranded cetaceans. 5 In this respect, the suboptimal nutritional status observed in some of the present dolphins is consistent with a subacute to chronic disease process.
The 4 dolphins that had lesions of meningoencephalitis had high serum titers of anti–T gondii antibodies, and histologic and immunohistochemical evidence of T gondii cysts and zoites was found in the brains of these animals. In a previous study in bottlenose dolphins, anti–T gondii antibody titers (detected via the modified agglutination test) were confirmed by IFAT and 1:40 identified as the prevalent positive titer. 8 Our serologic data were obtained by means of IFAT, which has been validated as a confirmatory assay for T gondii infection in sea otters (Enhydra lutris nereis), 26 a species in which T gondii meningoencephalitis is a common finding. 3 Toxoplasma infection, which appears to be common in some cetacean populations, including bottlenose dolphins from the coasts of Florida, South Carolina, and California, 9,11 has recently been linked to contamination of coastal waters with oocysts. 9,27 The epidemiology of T gondii infection of offshore species such as Risso’s dolphins (Grampus griseus) and striped dolphins is more difficult to explain but has been speculatively linked to the release of ship ballast waters contaminated by onboard rodents, cats, or contaminated soil. 32 Although T gondii is commonly regarded as an opportunistic pathogen in aquatic mammals, 24,26 as clearly documented in striped dolphins during the 1990–1992 Mediterranean Sea DMV epidemic, 7,21 the present findings suggest that T gondii had a primary etiologic role in the meningoencephalitis seen in the seropositive animals under study. A similar role has been suggested for captive and feral pinniped and cetacean species. 9,10,25 This hypothesis is supported by the negative results of all laboratory investigations for morbillivirus in these 4 animals, as well as by the negative results of the serologic and microbiologic investigations in all 8 dolphins for Brucella infection, which has been associated with meningoencephalitis in cetaceans, including striped dolphins. 14,19 The severe brain lesions in the present dolphins differed from those associated with Brucella infection in that meningitis was less prominent. In contrast, the severity of encephalitis was greater than that reported in Brucella-infected cetaceans. 14,19 Relatively high concentrations of immunotoxic heavy metals were measured in tissues from the liver (mercury, lead) and the kidney (cadmium) from the 8 dolphins under study. 35 However, it is not known if these metals had any etiologic role in the lesions found in these animals.
Enterococcus casseliflavus was recovered from the brain tissue of 1 dolphin that had meningoencephalitis, and Streptococcus parasanguinis and S gordoni were recovered from another. Although no information is available on the pathogenicity of these bacteria in sea mammals, E casseliflavus has been recently implicated as a cause of meningitis in human beings. 20 No microscopic alterations compatible with a microbial etiology were observed, and Gram staining did not reveal the presence of any bacteria in the cerebral tissue from these animals; as such, perimortem bacterial colonization seems likely.
The 8 striped dolphins under investigation were found stranded in a localized geographic area and over a limited period during which there was a considerable increase in reported strandings in that region (F. Garibaldi and W. Mignone, personal communication). These observations strongly support the hypothesis of a common source exposure as the cause of the increased striped dolphin mortality during 2007 in the Ligurian Sea. A similar pattern occurred in 2007 along the Spanish coastline in the western Mediterranean, where dozens of striped dolphins and several pilot whales died as a result of a DMV epidemic. 12,28 Morbillivirus infection was also recently reported in the brain tissue of a young striped dolphin found stranded in June 2007 on the northwestern Sardinian coast of Italy. 1 Again in 2007, a mass die-off of common seals (Phoca vitulina) and harbour porpoises (Phocoena phocoena) occurred along the regions of the Danish and Swedish coasts in which two major morbilliviral epidemics occurred in 1988 and 2002, respectively. The cause of that recent die-off is unknown. 18
Anti-morbillivirus antibodies were detected in the sera of 4 dolphins investigated in this study. Because the serologic test did not permit discrimination between immunoglobulin M and IgG antibodies, the timing of morbillivirus infection could not be determined. However, neither morbilliviral antigen nor nucleic acid was detected in the tissues of these dolphins, and the tissue lesions were not consistent with those of morbillivirus infection. 6,21
In conclusion, (1) the high antibody titers against T gondii in the 4 dolphins that had lesions of meningoencephalitis, (2) the presence of T gondii cysts and zoites in the brain of 2 of these animals, and (3) the absence of concurrent morbillivirus and Brucella spp infections support the hypothesis that this protozoan played a primary etiologic role in the development of the severe meningoencephalitis. The hypothesis that these lesions almost certainly caused the death and subsequent stranding of these animals is also supported by the lack of anti-Toxoplasma antibodies in the 4 dolphins that had no cerebral lesions.
Footnotes
The authors declared that they had no conflicts of interest with respect to their authorship or the publication of this article.
This work was carried out with a research grant (Progetto di Ricerca Cause di Mortalità e Studi Patogenetici in Cetacei Spiaggiati sulle Coste Italiane) from Ministero dell’Ambiente e della Tutela del Territorio e del Mare, Italy.
