Abstract
Human granulocytic anaplasmosis (HGA), an emerging disease of public health concern in many areas of the world, is caused by Anaplasma phagocytophilum. Small animal models of A phagocytophilum in laboratory mice have been developed and used to study the pathogenesis of HGA. In this study, we characterized the pathologic changes in acute infection of C3H/HeJ mice experimentally infected with the NY18 isolate of A phagocytophilum. Although no clinical signs were noted, acute infection was associated with gross splenomegaly, microscopic inflammatory lesions in the lung and liver, hyperplastic lesions on the spleen, and clinical pathology abnormalities including neutropenia and monocytosis. This study emphasizes the use of well-defined animal models as a valuable tool for the study of A phagocytophilum infections.
Keywords
Human granulocytic anaplasmosis (HGA), previously known as human granulocytic ehrlichiosis, is an emerging disease of public health concern in many areas of the world. HGA is caused by the intracytoplasmic, gram-negative rickettsia, Anaplasma phagocytophilum. 22 Clinical manifestations of HGA include fever, headache, myalgia, anemia, leukopenia, thrombocytopenia, and elevated liver enzymes 3, 4, 6, 16, 21, 25, 40 Prior to the emergence of HGA in 1994, the pathogen was described in Europe in sheep, goats, and cattle as the cause tick-borne fever and in the United States and Europe as the cause of equine granulocytic ehrlichiosis. 5, 11, 28, 29, 34– 36, 42, 48 Tick vectors of A phagocytophilum, Ixodes spp., are commonly found in regions of the United States, Europe, and Asia. 14, 20, 33, 34, 37, 41, 43 The white-footed mouse, Peromyscus leucopus, is considered to be the main reservoir host in North America, but raccoons, gray squirrels, and cottontail rabbits become naturally infected with A phagocytophilum and may also serve as reservoir hosts, thus contributing to the maintenance of the natural cycle of infection. 26, 33
Laboratory mice have been developed as small animal models of A phagocytophilum and used to study the pathogenesis of HGA. While mice do not consistently develop signs of clinical disease as described in humans, horses, or nonhuman primates, they do develop an acute infection and associated pathologic changes. 1, 7– 10, 12, 13, 15, 23, 24, 31, 38, 39, 42, 46 In this study we report the results of a pathologic study of acute A phagocytophilum infection in C3H/HeJ mice and discuss the relative importance and advantages of this mouse model in contrast to other A phagocytophilum models and hosts.
Materials and Methods
Experimental design
Seven groups of 3 mice each were inoculated with A phagocytophilum–infected HL-60 cultured cells (NY18 isolate). 2 One group of 14 mice was inoculated with uninfected HL-60 cells to serve as controls. Two control and 3 infected mice were exsanguinated and necropsied at 4 hours postinoculation (P.I.) (day 0) and on days 2, 4, 7, 10, 14, and 20, P.I. The response of mice to A phagocytophilum infections was followed by clinical observation and by necropsy, histopathology, and hematology studies. Infection of mice was confirmed by polymerase chain reaction (PCR), isolation, and propagation in HL-60 cell cultures and by light microscopy (LM) demonstration of morulae in neutrophils from stained blood smears.
Mice
Six-week-old, female C3H/HeJ–specific pathogen-free mice, susceptible to A phagocytophilum infection, were used for this study (Jackson Laboratories, Bar Harbor, Maine). This mouse strain is unresponsive to bacterial lipopolysaccharide (LPS), in part because of a defect in macrophage activation. 30, 44, 47 Mice were cared for by the Oklahoma State University Laboratory Animal Research Unit in accordance with specifications detailed in Institutional Guide for the Care and Use of Laboratory Animals.
A phagocytophilum isolate
The NY18 isolate of A phagocytophilum was propagated in cultures of human undifferentiated HL-60 promyelocytic cell line. 2 Infected and uninfected cell cultures were maintained at 37°C in RPMI medium as previously reported. 27 The A phagocytophilum–infected HL-60 cells were used for inoculations of mice when 90%–100% of the cell cultures were infected as determined by detection of intracellular morulae in stained cytospin cell smears (Hema-3 Stain, Fisher Scientific, Middletown, VA). Cultures collected on the day of inoculation were centrifuged and resuspended in serum-free RPMI 1640 medium to the concentration of 2 × 106 cells/ml. Mice were infected by intraperitoneal inoculation with 0.5 ml (1 × 106 cells) of the infected or uninfected cell culture samples.
Clinical observations
All mice were examined daily for clinical symptoms of A phagocytophilum infection, including hair ruffling, postural changes, lethargy, anorexia, and respiratory distress.
Collection of blood and analysis
Mice were sedated with carbon dioxide gas, followed by euthanasia by exsanguination via cardiocentesis, using a 25-gauge needle attached to a 1-ml syringe. Blood samples were transferred to 500 μl of ethylenediaminetetraacetic acid (EDTA)-coated pediatric collection blood tubes and stored at 4°C. Blood smears were made and stained with Diff Quick (Fisher Scientific) and examined with LM for the presence of A phagocytophilum morulae. Complete blood counts and differential white blood cell analysis were performed using an automated hematology analyzer (Cell Dyn 3500, Abbot; Hollinston, MA). All data analyses were performed using SAS Version 9.1 (Cary, NC). Two-way analysis of variance was used to test blood parameters for differences due to day or treatment (control or infected). If a significant interaction was found between day and treatment (P < .10), mean differences between control and infected means were compared on each day, correcting for multiple comparisons using the Bonferroni procedure. For neutrophils, lymphocytes, and monocytes, percentage of white blood cell values were used as they were less variable between days.
PCR for detection of A phagocytophilum in blood samples
Total DNA was extracted from blood samples collected on each sampling day using TriReagent (Sigma, St. Louis, MO) according to manufacturer's instructions. The DNA was resuspended in sterile distilled water and stored at -20°C until used in PCRs. The Anaplasma 16S rDNA PCR was used for determining infected mice. A fragment of 468 nucleotides comprising 16S rDNA positions -1 - 467 of the Anaplasma marginale Florida strain reference sequence (Genbank accession number AF309867) was amplified by PCR using oligonucleotide primers 16SANA-F (5′-CAG AGT TTG ATC CTG GCT CAG AAC G-3′) and 16SANA-R (5′-GAG TTT GCC GGG ACT TCT TCT GTA-3′) as described previously. 18, 45 The A phagocytophilum species-specific major surface protein (msp)4 PCR was used to corroborate pathogen identification as reported previously. 18 The msp4 and 16S rDNA gene sequences were amplified from 1 μl (0.1–10 ng) of DNA by PCR using 10 pmol of each primer in a 50-μl volume (1.5 mM MgSO4, 0.2 mM dNTP, 1X AMV/Tfl 5X reaction buffer, 5 u Tfl DNA polymerase) using the Access RT-PCR system (Promega, Madison, WI). Reactions were performed in an automated DNA thermal cycler (Eppendorf Mastercycler personal, Westbury, NY) for 35 cycles. PCR products were electrophoresed on 1% agarose gels to check the size of amplified fragments by comparison to a DNA molecular weight marker (1 Kb DNA Ladder, Promega).
Culture
Blood samples were collected from infected and control mice necropsied on days 7 and 20 P.I. and placed in EDTA-treated tubes. Blood (100 μl) from each mouse was added to a flask containing 1 × 106 HL-60 cells /ml in 5 ml of medium and maintained as described previously. 19 Samples of the cell cultures were collected daily, and Cytospin smears were prepared, stained with Hema-3 (Fisher Scientific), and examined by LM for the presence of A phagocytophilum morulae.
Collection of tissues and histopathology
The organs and tissues were examined in situ, dissected, collected, and fixed in neutral buffered, 10% formalin solution. Histopathologic examination was performed on adrenal glands, bone marrow (humerus, sternum, and vertebra), bone marrow (smears), brain, eyes, harderian gland, heart, joints (humerus, sternum, and vertebra—articular surfaces), kidneys, large intestine, liver, lung, mandibular lymph nodes, mesenteric lymph nodes, ovaries, pancreas, peripheral blood (smear), pituitary gland, salivary gland, small intestine, spinal cord (cervical, thoracic, and lumbar), spleen, stomach, thymus, urinary bladder, uterus, and vagina. Formalin-fixed tissues were processed routinely, embedded in paraffin, sectioned at approximately 5 μm, stained with HE, and evaluated for microscopic lesions by 2 pathologists independently. Blood and bone marrow smears were prepared, fixed, stained using the Diff-Quick stain (Fisher Scientific), and evaluated microscopically for the presence of A phagocytophilum morulae. The distribution and severity of the lesions were recorded.
Results
Clinical observations
All experimental animals survived to their scheduled euthanasia times. Through the duration of the study, overt signs of illness were not observed in any of the experimental animals.
Infection of mice with A phagocytophilum
PCR analysis of blood samples identified A phagocytophilum DNA in 0/3, 2/3, 2/3, 3/3, 0/3, and 1/3 of the mice on days 0, 2, 4, 10, 14, and 20 P.I., respectively. Blood samples from all uninfected control mice were PCR negative. The sequence of the msp4 DNA amplified from infected mice was the same as the PCR sequence derived from the NY18 isolate infection in cultured HL-60 cells (Genbank accession number AY530194). 17 All HL-60 cell cultures inoculated with blood from infected mice became infected with A phagocytophilum. Cultures inoculated with blood from day 7 were positive for morulae after 4 days in culture and reached 100% infection by day 8. Cultures inoculated with blood from day 20 were positive after 2 days and reached 100% infection by day 7. Occasionally, morulae were recognized in the cytoplasm of neutrophils in peripheral blood of infected mice at day 7, although the infection in these cells was too low to perform a cell count. Morulae were not seen in the bone marrow cells on infected mice.
Gross and microscopic findings
Significant gross findings were limited to the spleen of infected mice. Grossly, the splenic capsule was taut, the borders were rounded, and the capsular and cut surfaces had scattered <1-mm-diameter white foci, interpreted as hypertrophic lymphoid follicles (Fig. 1).
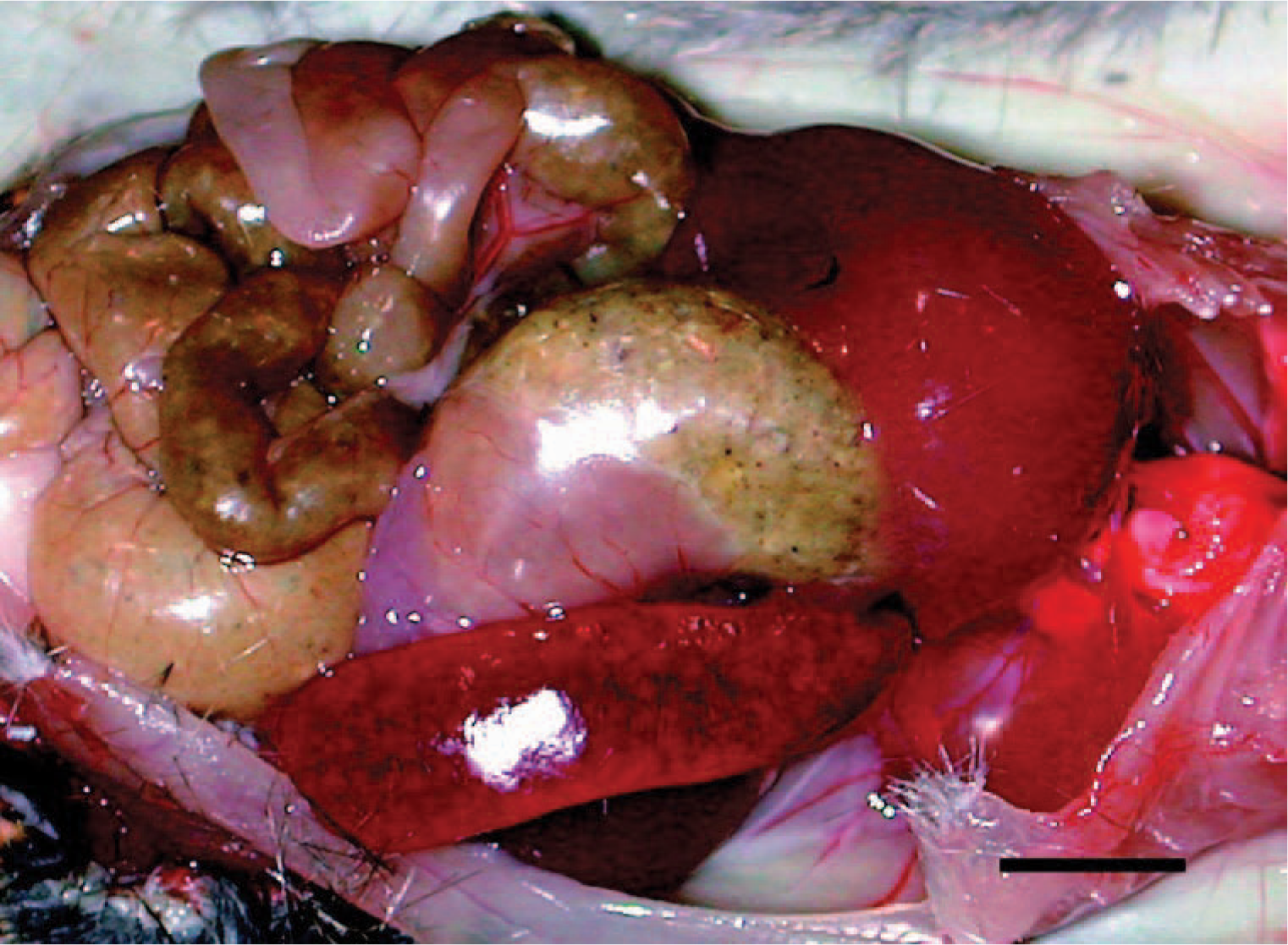
Spleen; A phagocytophilum–infected C3H/HeJ mouse No. 27; day 14; splenomegaly. The spleen is enlarged about twice normal; the splenic capsule is taut, the borders are rounded, and the capsular and cut surfaces had scattered <1-mm-diameter white foci, interpreted as hypertrophic lymphoid follicles. Bar = 1 cm.
Lesions were also observed microscopically in the lungs, spleens, and livers of infected mice. The lesions were first observed in the lung of 1/3, 1/3, 3/3, 3/3, 3/3, and 2/3 infected mice inoculated at days 0 (4 hours P.I.), 2, 4, 7, 10, and 14, respectively. The lesions in the lung were inflammatory and were characterized initially as a slight hypercellularity of the alveolar walls, with mild hyperemia, and increased numbers of neutrophils within the capillaries; there was mild accumulation of foamy macrophages, some erythrocytes, and slight deposition of proteinaceous material and fibrin within the alveoli (Fig. 2). This was followed by multifocal, perivascular collections of a few lymphocytes, plasma cells, macrophages, and occasional neutrophils that extended into the adjacent interstitium and alveolar septa (Fig. 3). Occasionally, the walls of a few pulmonary vessels had mildly thickened walls, with hypercellular tunicas and lined by hypertrophic endothelium (Fig. 4) as compared with the controls (Fig. 5). Pulmonary arteriolitis was observed in the infected mice (Fig. 6), which was not apparent in the controls (Fig. 7).
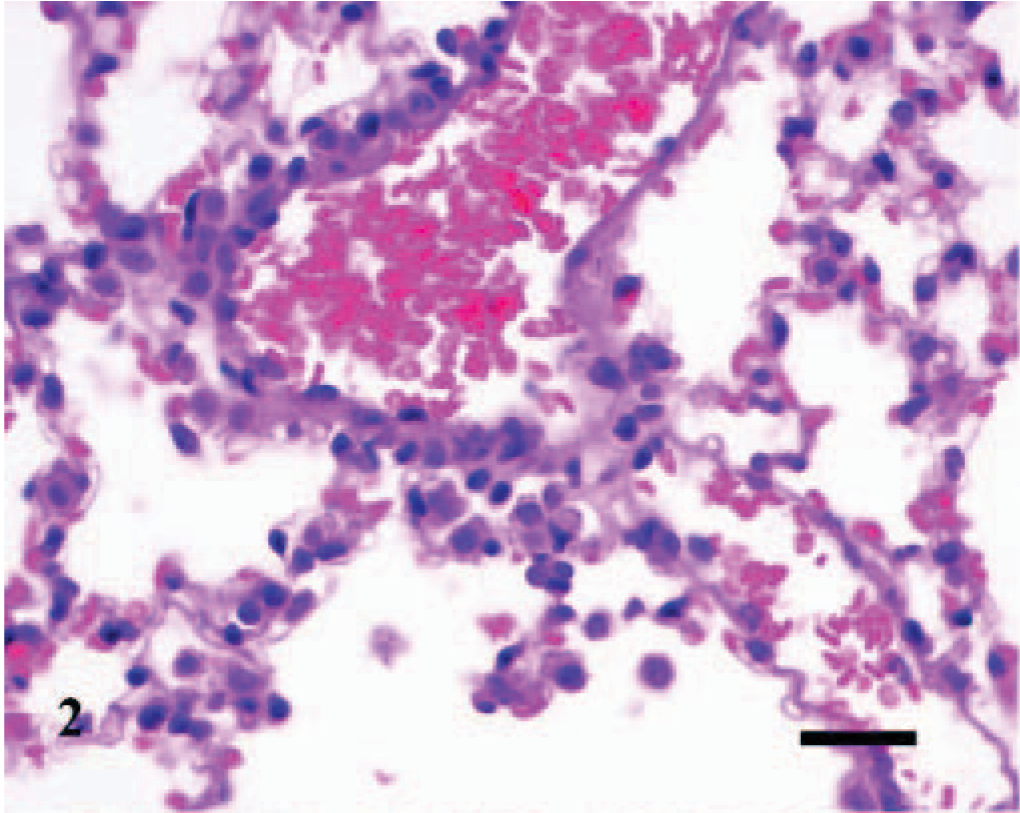
Lung; A phagocytophilum–infected C3H/HeJ mouse No. 3; day 0. The alveolar walls are slightly thickened with mild hyperemia and increased numbers of leukocytes; the alveoli contain mild accumulation of foamy macrophages, some erythrocytes, and slight deposition of proteinaceous material. HE; Bar = 25 μm.
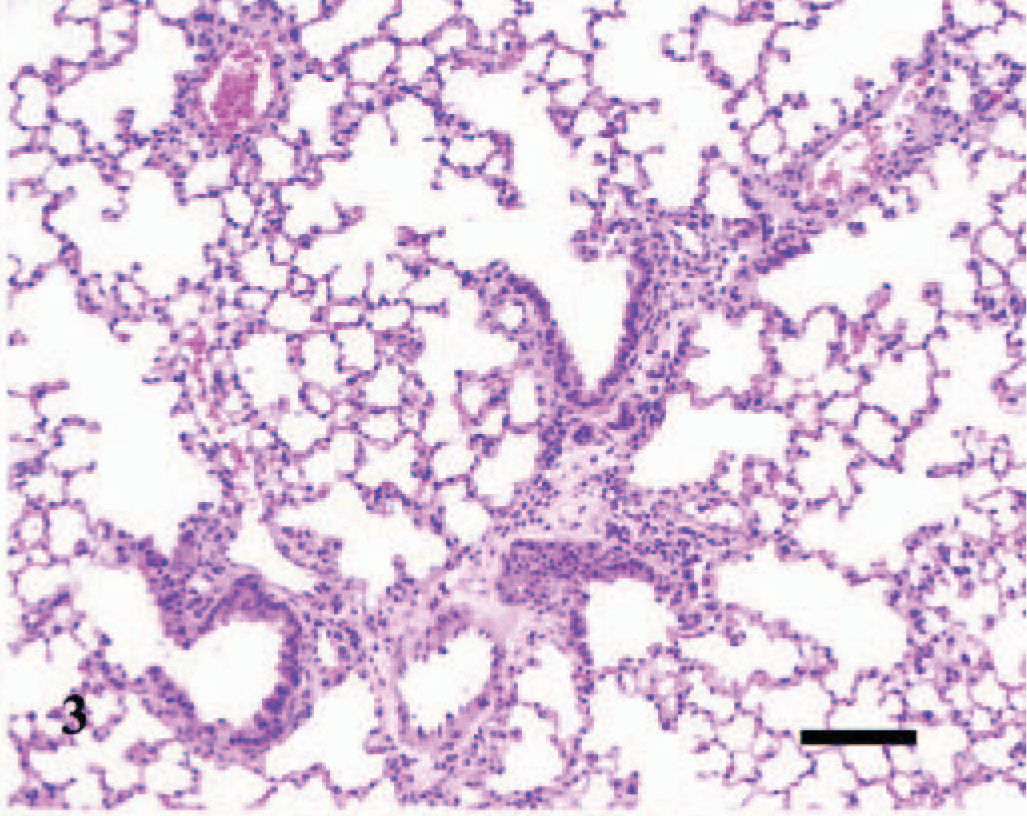
Lung; A phagocytophilum–infected C3H/HeJ mouse No. 22; day 10; interstitial pneumonia. There is diffuse hypercellularity of the lung and mild, multifocal, collections of leukocytes about blood vessels and bronchioles. HE; Bar = 100 μm.
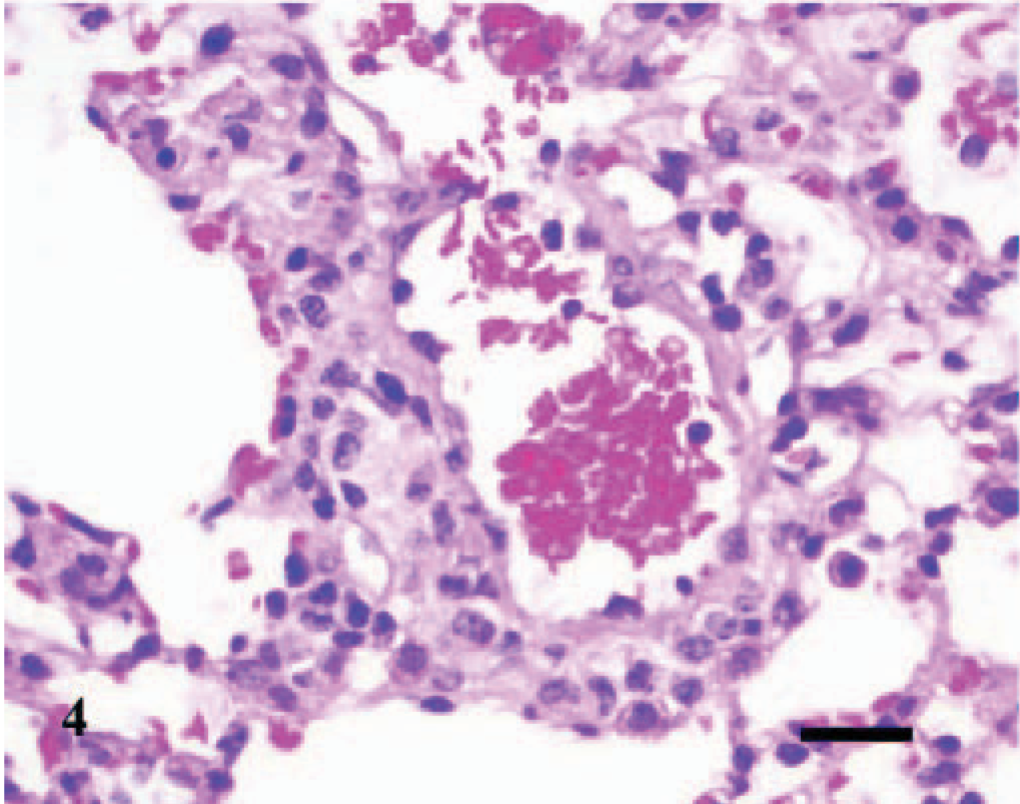
Lung; A phagocytophilum–infected C3H/HeJ mouse No. 12; day 4. The wall of a few pulmonary vessels is mildly thickened, with a hypercellular tunica media and lined by hypertrophic endothelium. HE; Bar = 25 μm.
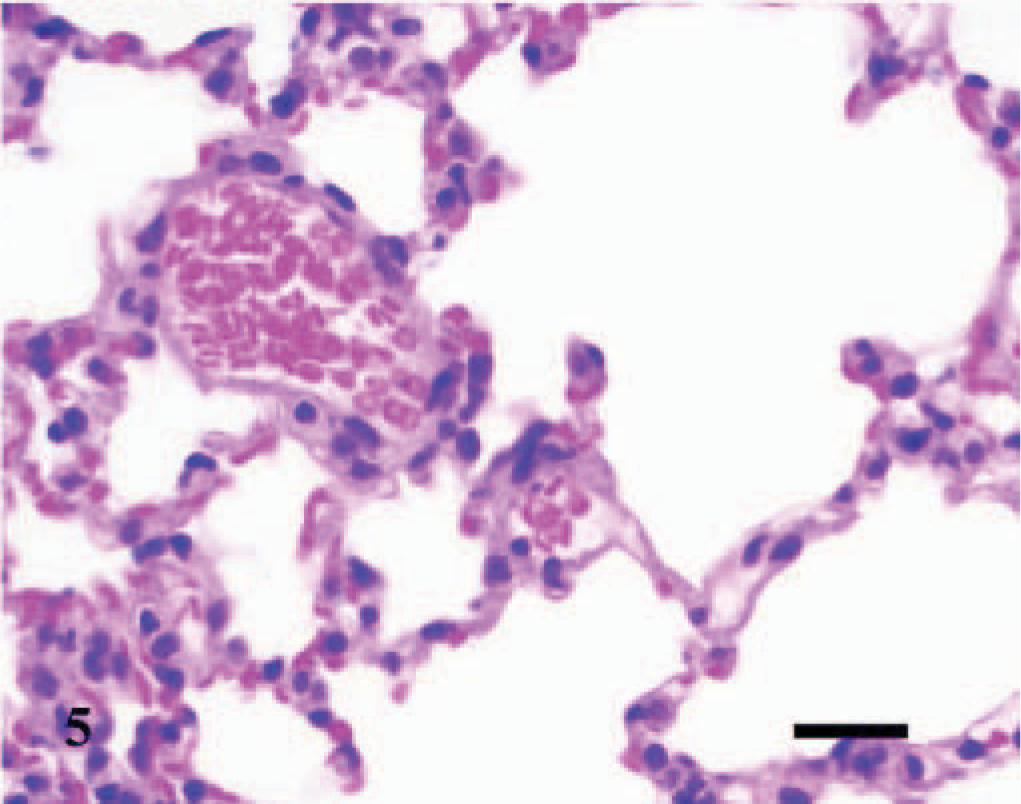
Lung; control C3H/HeJ mouse No. 19; day 7. Cross section through normal pulmonary tissue. HE; Bar = 100 μm.
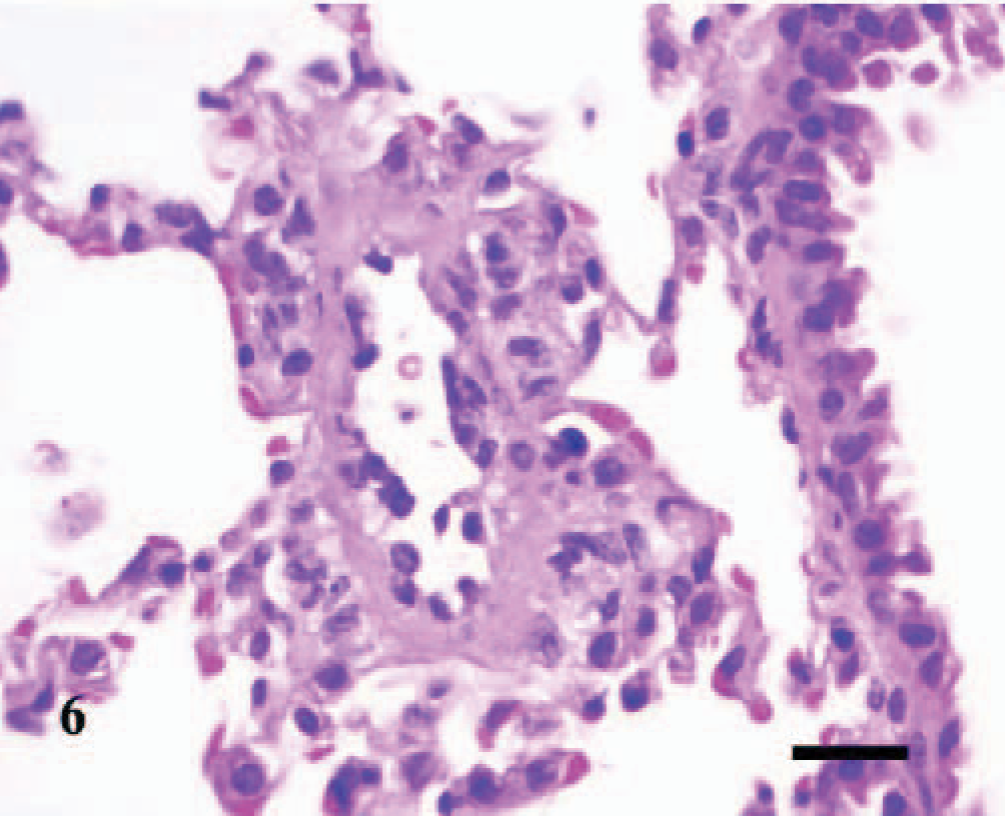
Lung; A phagocytophilum–infected C3H/HeJ mouse No. 12; day 4; pulmonary arteriolitis. The walls of the pulmonary arteriole are thickened and hypercellular. Collections of few lymphocytes and macrophages/monocytes and plasma cells exist beneath the hypertrophic endothelium, within the tunica media and tunica adventitia, and extend into the adjacent perivascular tissues and alveolar septa. HE; Bar = 25 μm.
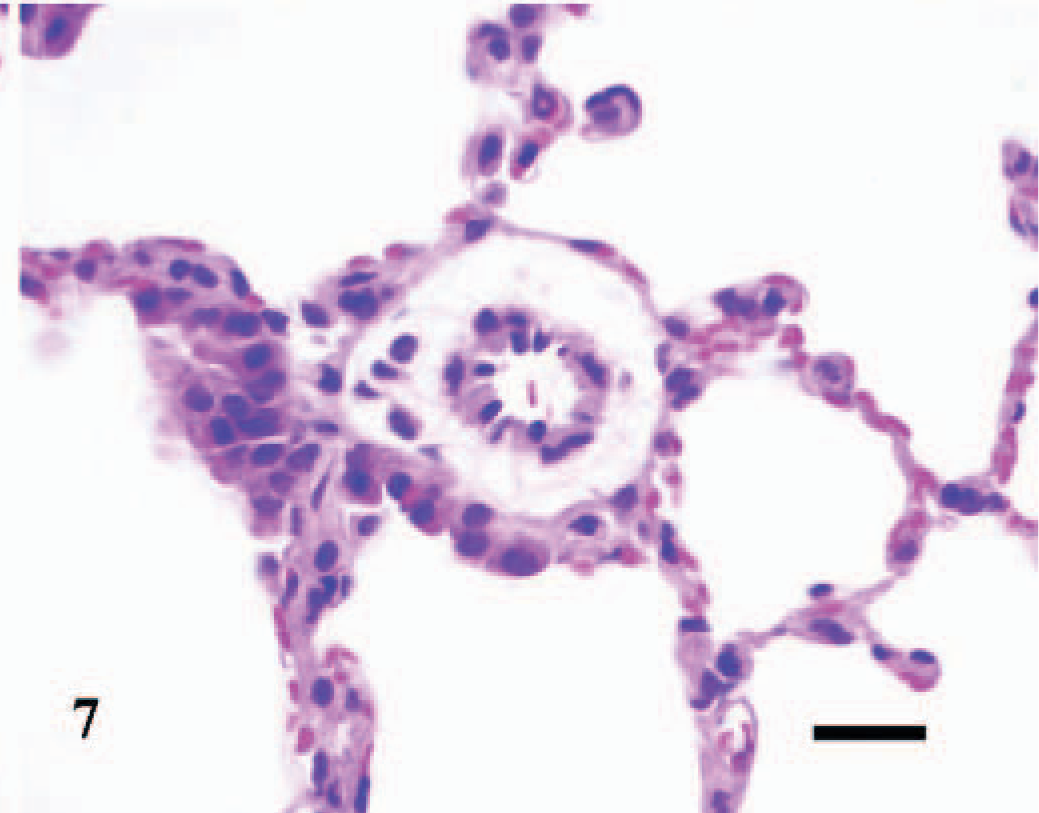
Lung; control C3H/HeJ mouse No. 19; day 7. Cross section through a normal pulmonary arteriole. HE; Bar = 25 μm.
The splenomegaly recognized grossly was characterized microscopically by expansion of both the red and white pulp (Fig. 8) as compared with sections of spleens from the uninfected control mice (Fig. 9). In the splenic red pulp, there was marked hematopoietic hyperplasia (including erythroid, myeloid, and megakaryocytic elements), which was first observed in the subcapsular areas and along muscle trabecula and later extended into the adjacent red pulp areas between the lymphoid follicles (Fig. 10). The splenic white pulp was also prominent and consisted of increased numbers and size of lymphoid follicles and periarteriolar sheaths (Fig. 11). Evidence of splenic inflammation, lymphoid depletion, or lymphocyte necrosis was not detected microscopically in any of the infected or control animals.
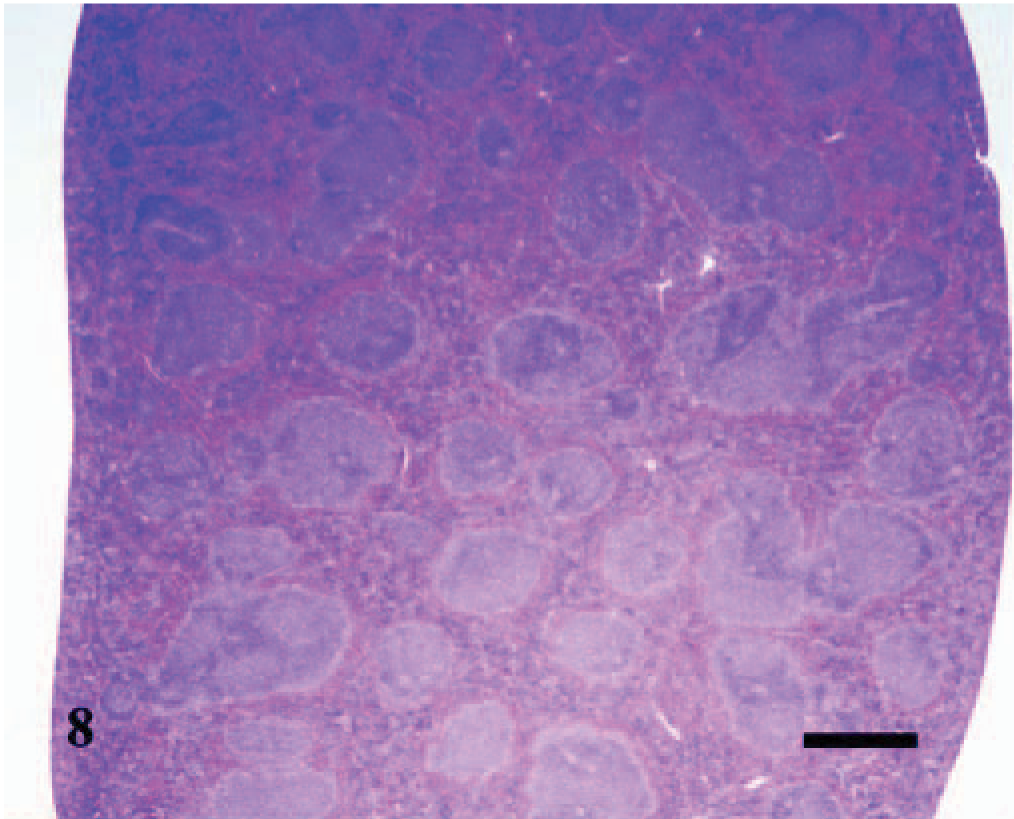
Spleen; A phagocytophilum–infected C3H/HeJ mouse No. 27; day 14; splenomegaly. The red and white pulp are expanded and hypercellular. HE; Bar = 800 μm.
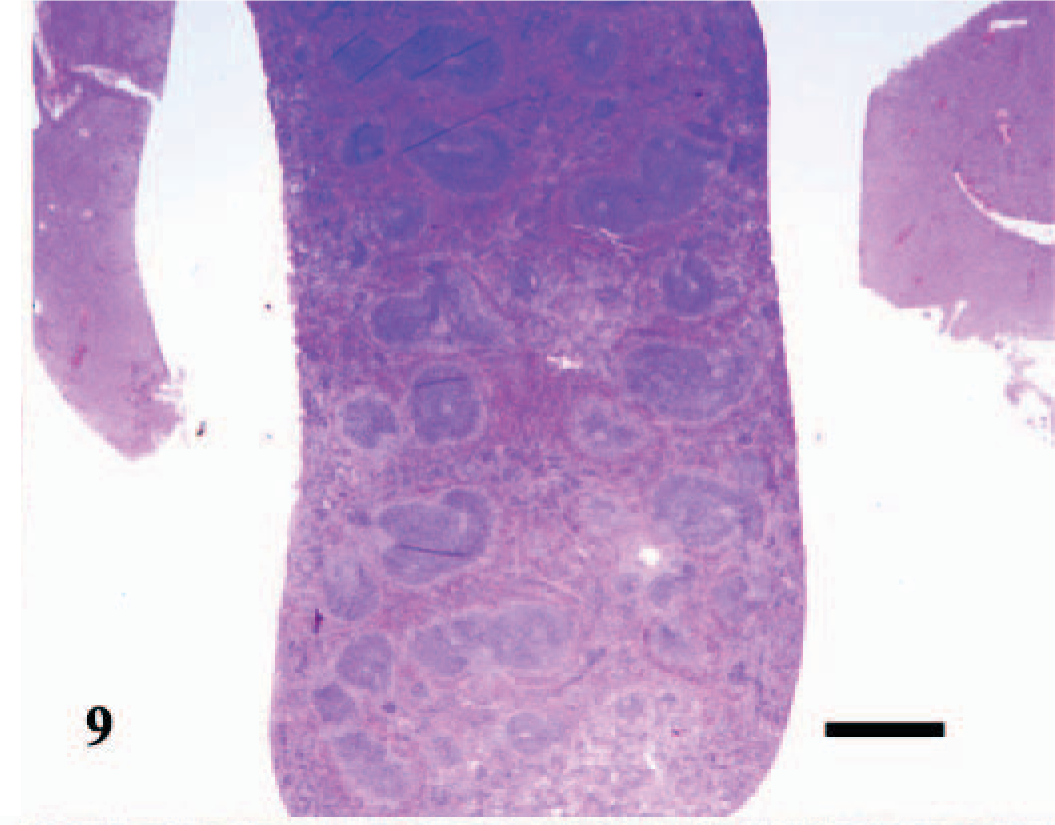
Spleen; control C3H/HeJ mouse No. 34; day 20. Cross section through normal splenic tissue. HE; Bar = 800 μm.
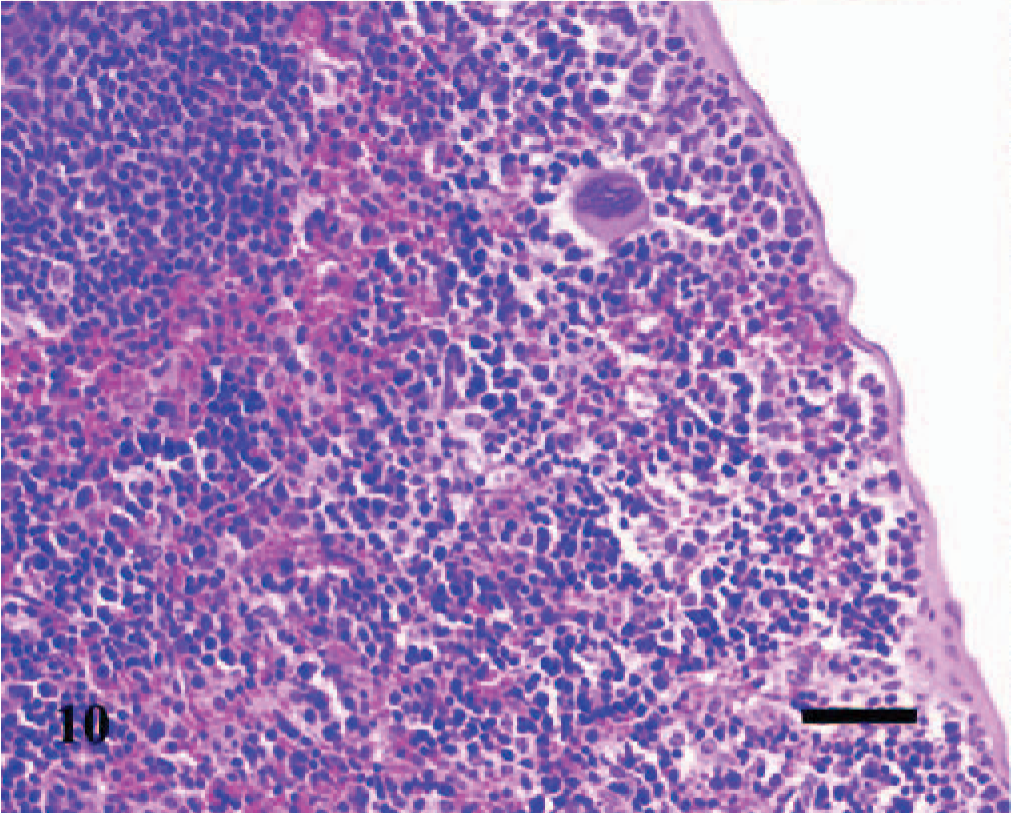
Spleen; A phagocytophilum–infected C3H/HeJ mouse No. 27; day 14. In the red pulp, a heterogeneous collection of hematopoietic cells expand the subcapsular areas and extend into the adjacent red pulp areas between the lymphoid follicles. HE; Bar = 40 μm.
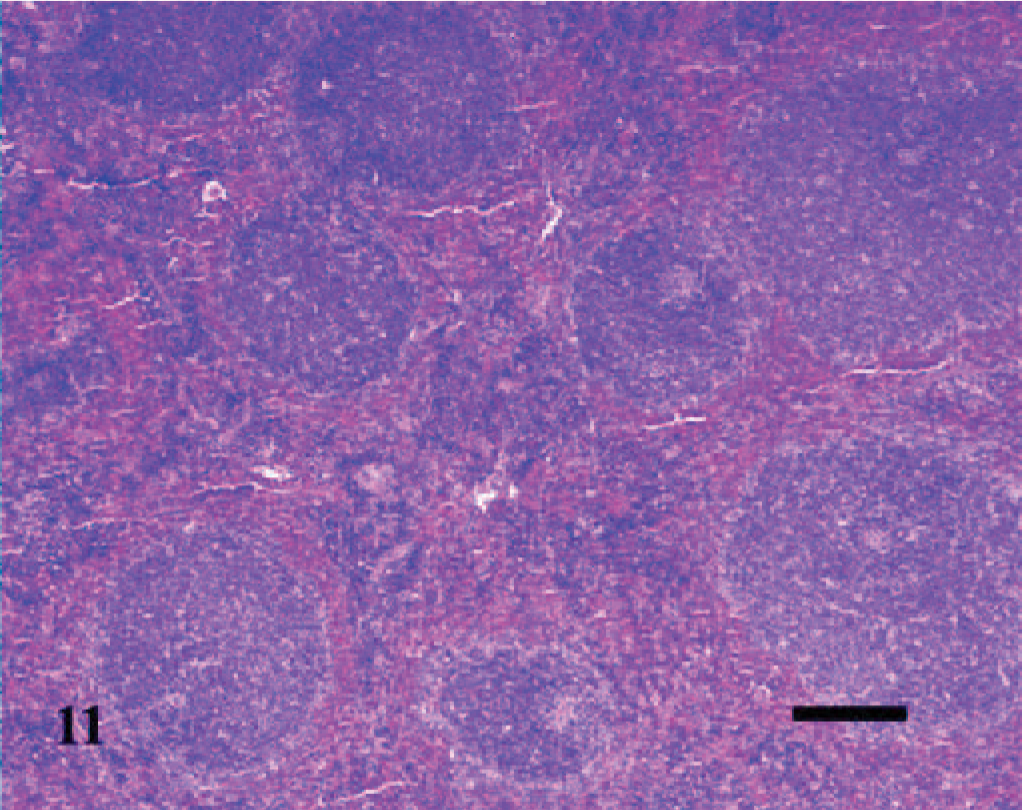
Spleen; A phagocytophilum–infected C3H/HeJ mouse No. 27; day 14. Increased numbers of irregular lymphoid follicles and periarteriolar sheaths exist among the hypercellular red pulp. HE; Bar = 100 μm.
As in the lung, hepatic lesions were inflammatory and were observed in 1/3, 3/3, 2/3, and 1/3 infected mice inoculated at days 2, 4, 7, and 10, respectively. Early lesions were characterized by a few, discrete sinusoidal collections of macrophages, neutrophils, and some necrotic cellular debris; occasionally, there were discrete foci of hepatocellular apoptotic necrosis in adjacent areas (Fig. 12). Subsequent hepatic lesions were followed by periportal collections of some lymphocytes, plasma cells, macrophages, and few neutrophils in perivascular spaces; and in some mice, the portal infiltrates extended or breached into adjacent hepatic lobules (Fig. 13).
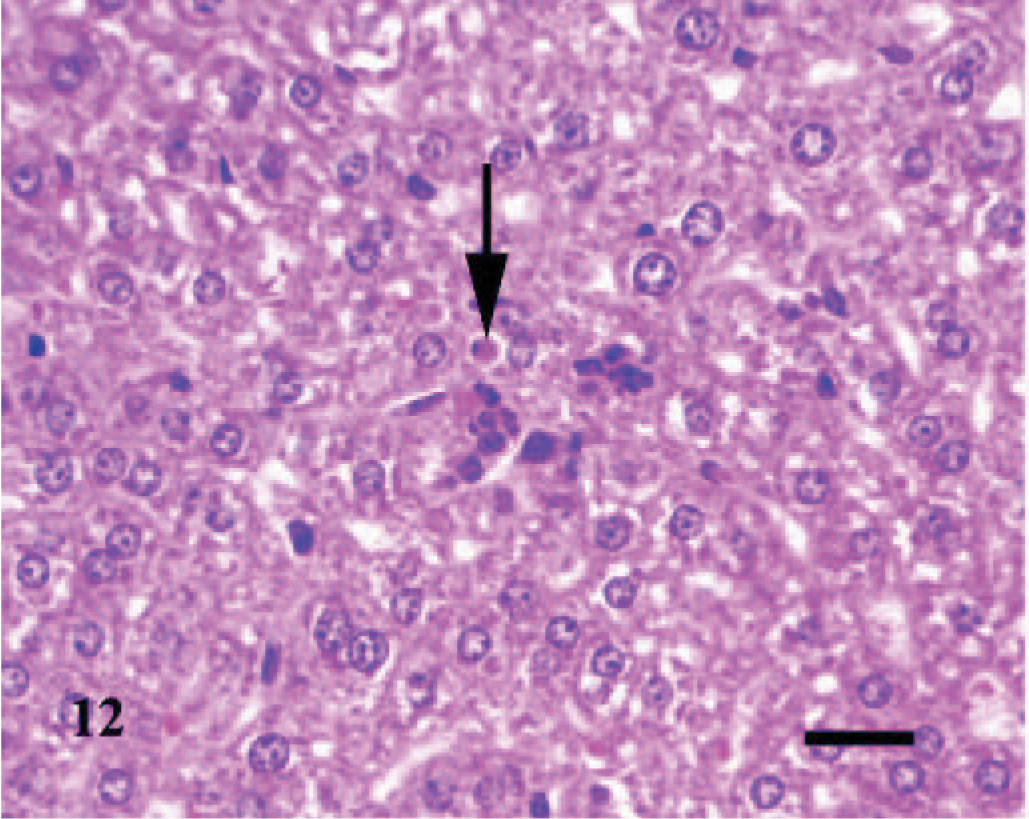
Liver; A phagocytophilum–infected C3H/HeJ mouse No. 8; day 4. There are discrete sinusoidal collections of macrophages, neutrophils, and some necrotic cellular debris; the arrow depicts apoptotic necrosis of a hepatocyte. HE; Bar = 25 μm.
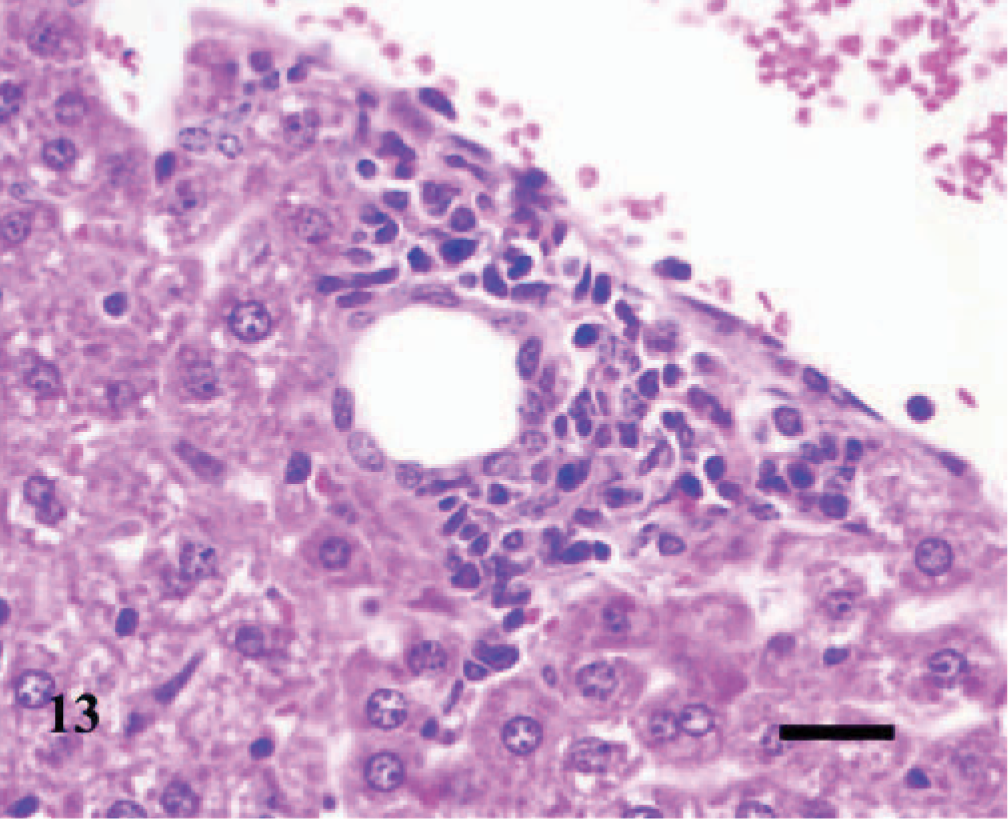
Liver; A phagocytophilum–infected C3H/HeJ mouse No. 13; day 7; portal hepatitis. A mild collection of lymphocytes, plasma cells, macrophages, and few neutrophils expands the periportal area and extends into adjacent hepatic lobule. HE; Bar = 25 μm.
Microscopic lesions were not observed in the adrenal gland, bone marrow (humerus, sternum, and vertebra), brain, eyes, harderian gland, heart, joints (humerus, sternum, and vertebra—articular surfaces), kidneys, large intestine, mandibular lymph nodes, mesenteric lymph nodes, ovaries, pancreas, pituitary gland, salivary gland, small intestine, spinal cord (cervical, thoracic, and lumbar), stomach, thymus, urinary bladder, uterus, or vagina.
Clinical pathology of blood samples
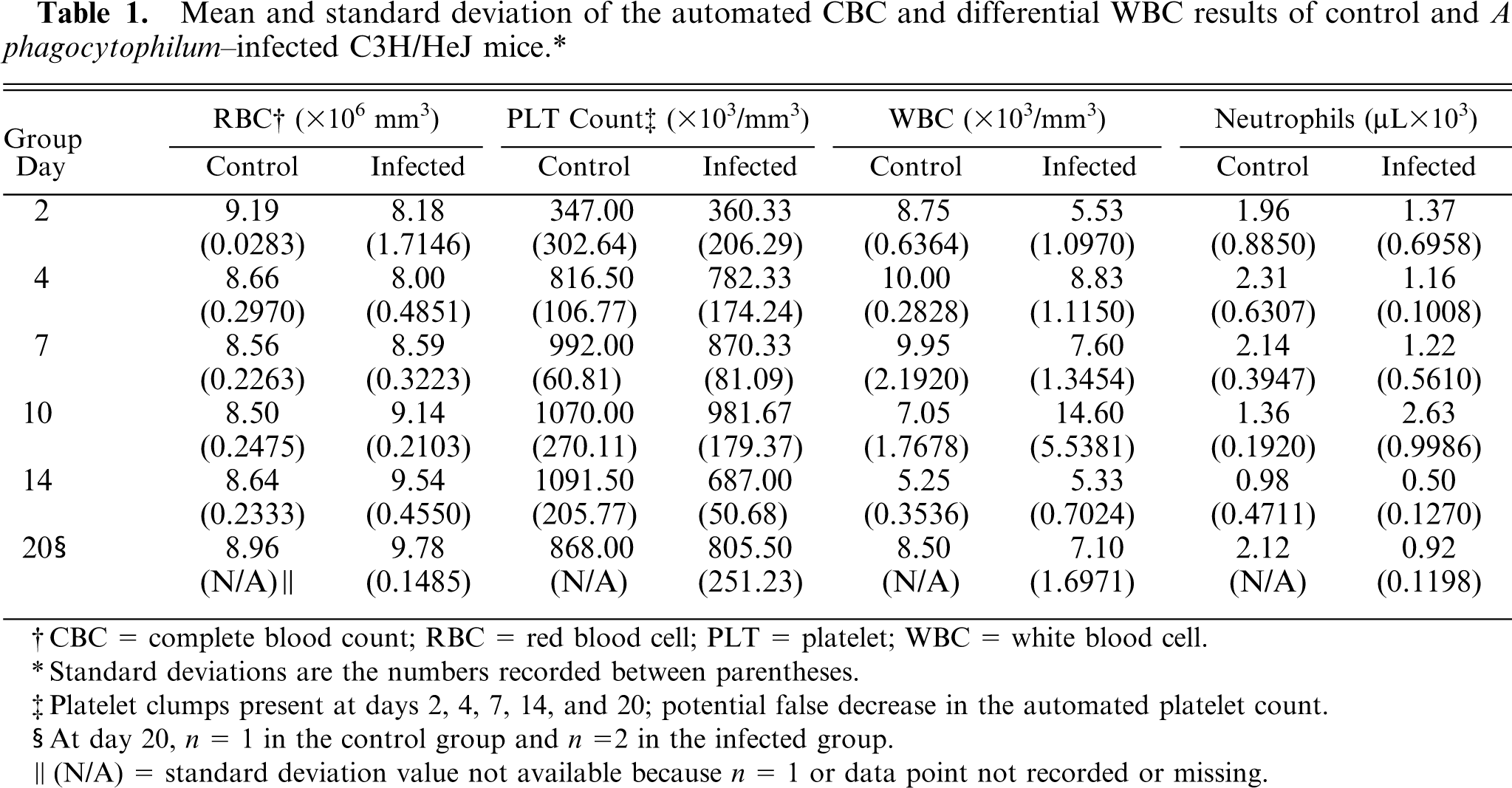
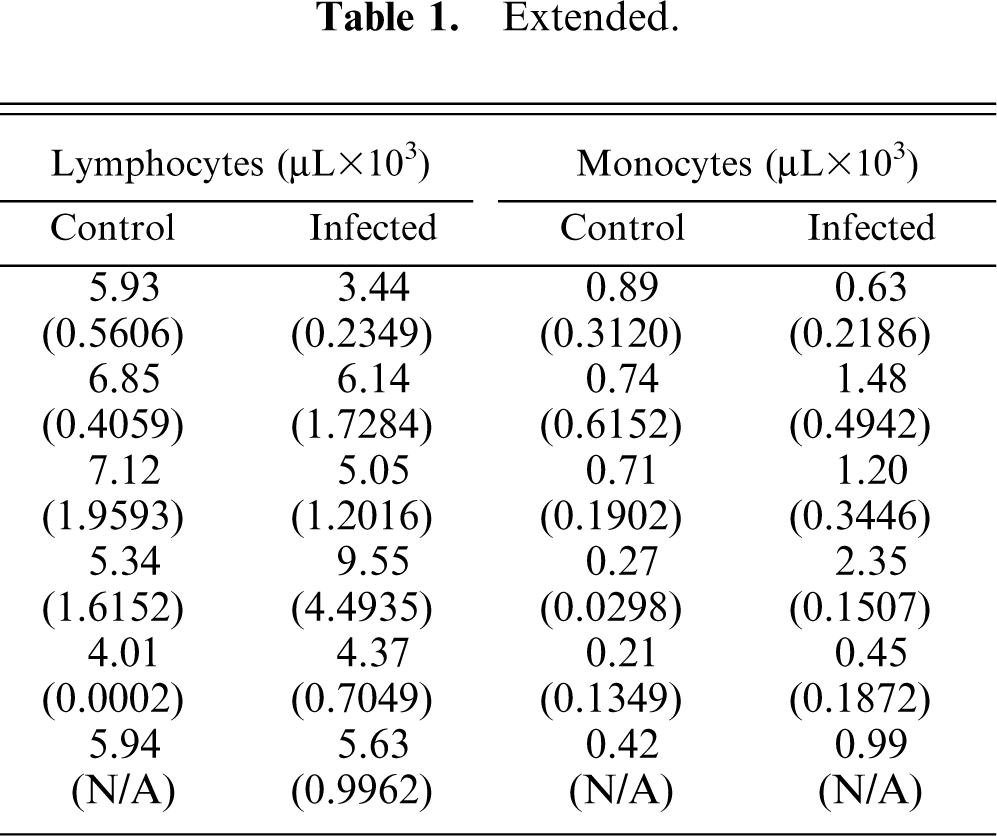
Table 1 summarizes the clinical pathology data generated on mouse blood. Because of low blood volume obtained at day 0, the tabulated data only reflect values of blood samples collected from day 2 through day 20 P.I. A significant difference between control and infected mice existed for both neutrophils and monocytes. Infected mice had significantly decreased numbers of neutrophils (P = .0085) and elevated numbers of monocytes (P = .0038). There were significant treatment-day differences between platelet count and white blood cell counts (P = .0258); however, evidence of platelet clumps at various days potentially caused a false decrease in the automated platelet counts. A significant interaction effect between treatment and day existed for white blood cell counts (P = 0.0266). Thus, controls-versus-infected differences were examined for each day separately. On day 10, infected mice had significantly higher white blood cell counts than control mice (P = .0023).
Discussion
As reported previously, C3H/HeJ mice experimentally inoculated with A phagocytophilum did not present clinical signs of disease; however, this pathologic study demonstrated that the C3H/HeJ mouse model system was useful for defining the parameters of acute infection. 13, 30, 38, 46
Microscopically, lesions were present in the spleen, lung, and liver. An apparent neutropenia, monocytosis, and intracytoplasmic morulae in neutrophils, characteristic of A phagocytophilum infection, were also seen. The pulmonary and hepatic lesions observed in the current mouse study are consistent with the lesions described in a recent study in which the lesions associated with infection with A phagocytophilum were compared between human, equine, and ruminant species. 32 As in our study, these species developed mild interstitial pneumonia (with perivascular collections of lymphocytes, plasma cells, and macrophages) as well as mild, periportal hepatitis, with hepatocellular apoptotic necrosis. In contrast, the splenic pathology described for these species consisted of lymphoid depletion and lymphocyte necrosis, which differ from the observations made on our C3H/HeJ mouse model. 32 Comparable lesions have been described in another study, in which nonhuman primates were used as animal models of granulocytic anaplasmosis (ehrlichiosis). 23 While the pulmonary lesions observed, including the interstitial pneumonia and vasculitis, had been described before in immunodeficient mice (C3H-SCID), very little has been documented about their occurrence in immunocompetent C3H/HeJ mice (albeit with defective activation of macrophages and unresponsive to bacterial lipopolysaccharide). 13, 30, 44, 47, 48 In one study, a mild interstitial pneumonia, but with no associated vascular lesions, was described in the C3H/HeJ mouse. 38
In contrast to the human disease and to other studies using the immunocompetent, LPS-responsive, C3H/HeNCr mice, the finding of significant monocytosis by day 10 P.I. appears to be unique in the C3H/HeJ mice. 46 While anemia is a feature of granulocytic anaplasmosis in humans and in at least 1 study using the C3H/HeJ mice strain, in this study infected C3H/HeJ mice had no evidence of anemia. 30 Whereas in some studies using C3H/HeN and C57BL/6J mice, thrombocytopenia is reported as a feature of mice infected with A phagocytophilum, in this study, the C3H/HeJ mice did not develop thrombocytopenia. 10 In support of our observations, another study also found no evidence of thrombocytopenia in A phagocytophilum–infected C3H/HeJ mice. 38
Although little is known about the pathogenesis of granulocytic anaplasmosis, evidence suggests that the immune system of the host plays a significant role in the development of the lesions observed in our study. A role as likely contributors in the pathogenesis of this disease has been proposed for both gamma interferon (IFN-γ) and IL-10. 12, 30, 32, 38, 39
While relative differences between and among the various mouse models of granulocytic anaplasmosis may exist, the C3H/HeJ mouse model system provides a useful model for defining the role of the response of the host to infection with this pathogen in the control of acute anaplasmosis infections and contributes to our overall understanding of the pathogenesis of A phagocytophilum.
Mean and standard deviation of the automated CBC and differential WBC results of control and A phagocytophilum–infected C3H/HeJ mice.∗
CBC = complete blood count; RBC = red blood cell; PLT = platelet; WBC = white blood cell.
Standard deviations are the numbers recorded between parentheses.
Platelet clumps present at days 2, 4, 7, 14, and 20; potential false decrease in the automated platelet count.
At day 20, n = 1 in the control group and n = 2 in the infected group.
(N/A) = standard deviation value not available because n = 1 or data point not recorded or missing.
Footnotes
Acknowledgements
This research was supported by the project No. 1669 of the Oklahoma Agricultural Experiment Station, the Endowed Chair for Food Animal Research (K. M. Kocan, College of Veterinary Medicine, Oklahoma State University), NIH Centers for Biomedical Research Excellence through a subcontract to J. de la Fuente from the Oklahoma Medical Research Foundation, and by the Georgia Department of Agriculture through a contract to the Athens Veterinary Diagnostic Laboratory, University of Georgia. C. Almazán is supported by grants-in-aid from the CONACYT and Promep (University of Tamaulipas), Mexico. Dr. R. Alleman (University of Florida) is acknowledged for supplying HL-60 cells and the NY18 isolate of A phagocytophilum. Janet J. Rogers (Core Sequencing Facility, Department of Biochemistry and Molecular Biology, Noble Research Center, Oklahoma State University) is acknowledged for DNA sequencing. Dr. Deborah Keys is recognized for her professional statistical analysis and consultations. The technical assistance of Victoria Naranjo, Jodene Bane, Jackie Forest, Ryan Riddle, and James Pellerin was essential for the completion of this study.
