Abstract
During an extensive study regarding the epidemiology of larval Eustrongylides infestation in a free-ranging endangered population of dice snakes (Natrix tessellata) from Histria, Romania, an adult female was euthanized to evaluate pathologic changes. Parasites appeared as nodules at various locations: in subcutaneous connective tissues, on the serosae of the intestines and liver. Histologic sections revealed nematode larvae surrounded by a capsule, forming a parasitic granuloma with 3 layers: macrophage layer, lymphocyte layer, and fibrous capsule. Differences between newly formed and mature granulomas consisted mainly in the eosinophilic infiltration. Other types of parasitic granulomas of reptiles are discussed in comparison with our findings.
Keywords
Similar to other wild vertebrates, reptiles are commonly infected by a wide range of nematodes, serving either as their definitive or intermediate hosts. Despite this, reports describing the apparent disease caused by larval nematodes in reptilian hosts are not frequent. In this paper, we report on pathologic changes induced by larval stages of dioctophymatid nematodes in the European dice snake, Natrix tessellata.
Nematodes of the genus Eustrongylides parasitize the proventriculus of piscivorous birds. The life cycle is heteroxenous, larval stages develop in fish; however, infections of other poikilothermic hosts were described. 8 There are several reports of infection with Eustrongylides larvae in snakes, but only a few of these reports focused on pathology of infection. 1, 6 Moreover, none of these studies focused on the pathology of Eustrogylides from the Palaearctic region.
During a field study performed in Histria, Constanţa County, Romania (44.54°N, 28.93°E) in 2005, we found a population of dice snakes (Natrix tessellata) heavily infected with larvae of Eustrongylides. Live snakes were carefully inspected for the occurrence of subcutaneous nodules caused by Eustrongylides larvae; epidemiologic data will be presented elsewhere. To describe pathologic changes, a single heavily infected adult female snake was selected and euthanized by using 200 mg thiopental sodium (thiopental ICN) i.p. injection. A necropsy was performed; organs with macroscopic parasitic nodules, namely subcutaneous tissue, esophagus, stomach, intestine, and liver were preserved in 10% formalin for histologic examination. Six living larvae were manually extracted from subcutaneous nodules and were preserved in 4% hot formalin for identification. Identification was made based on morphologic features by F. Moravec. Histologic sections were processed and stained routinely with hematoxylin and eosin (HE).
The examined snake was in moderate nutritional state, showing no apparent behavioral changes before euthanasia. Numerous irregular swellings in the dorsal and lateral aspects of the mid body (in the area of the stomach) were the only recorded lesions before the necropsy. These nodules caused an overall swelling of body thickness in that area (Fig. 1). The consistency of the nodules was firm, and they had little mobility under the skin. Superficial skin lesions were not observed adjacent to the nodules.
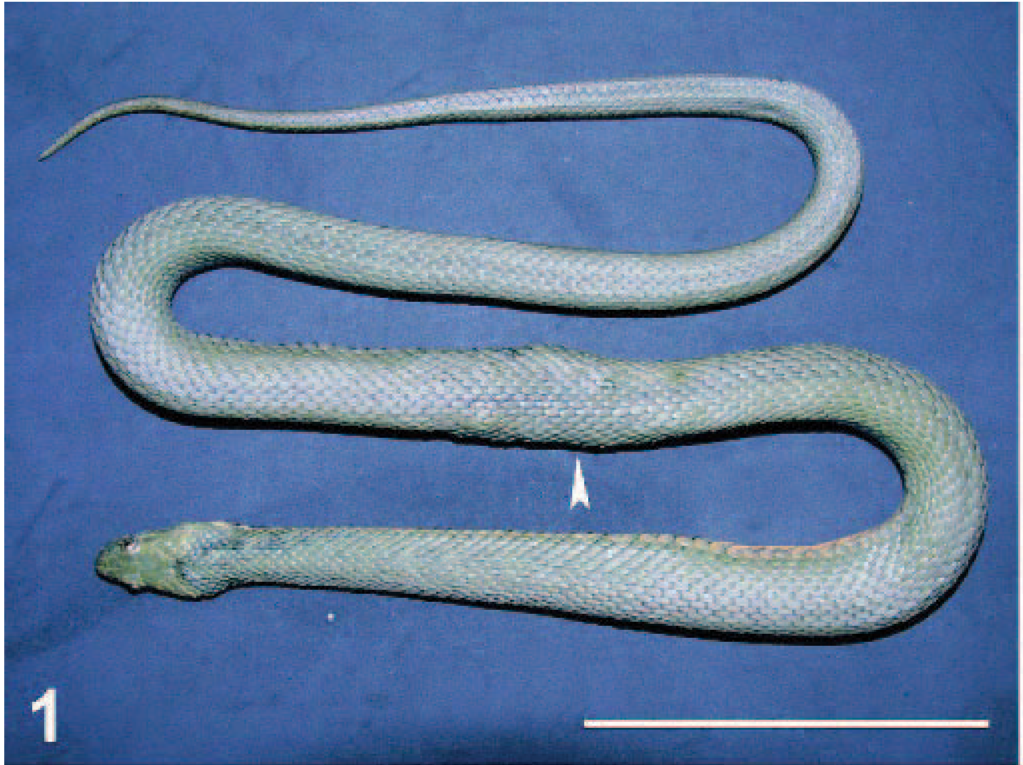
Body; dice snake. Swellings are visible in the mid body of the snake (arrowhead).
During necropsy, a large number of nodules were noticed in the subcutaneous connective tissue, corresponding to swellings. These were not only in the dorsal area but also on the lateral and the ventral aspects (Fig. 2). After opening the body cavity, nodules were also observed on pleuroperitoneum, esophagus, intestine, stomach, and liver. The nodules measured 2 to 9 mm, with rounded, oval, or irregular shapes, and were yellow to brown-red. Some nodules contained coiled nematodes, whereas others contained firm yellow to brownish material. In both cases, the contents of nodules were surrounded by a dense fibrocollagenous capsule, adherent to adjacent connective tissues.
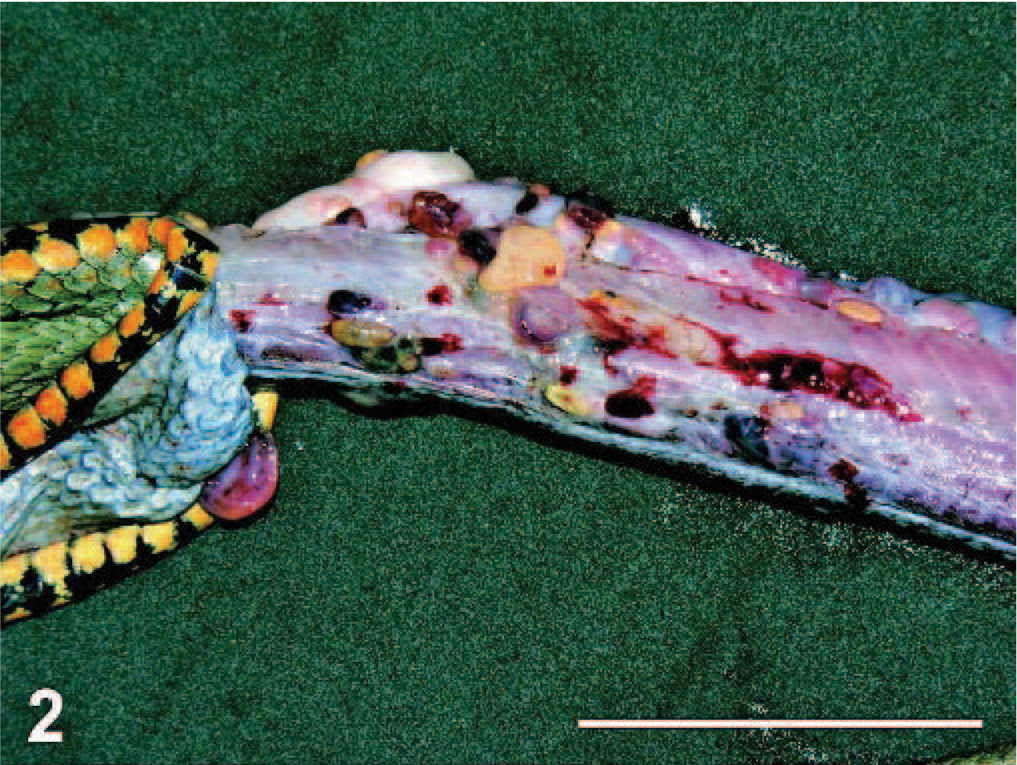
Body; dice snake. Nodules are attached to the surrounding connective tissue.
In most nodules, a single nematode was present; however, nodules with 2 larvae were also observed. Nematodes were reddish, coiled inside the nodule, and measured between 40 and 80 mm. Subsequently, nematodes were identified as fourth-stage larvae of Eustrongylides sp. Based on the larval morphology, specific identification is impossible. 8
Microscopically, the nodules were found to be granulomas in different stages of maturation, localized in subcutaneous fatty tissue, in the subserosa of the small and the large intestines and also in the subserosa of liver (Fig. 3). In the central part of some granulomas, structures of nematodes were still distinguishable, whereas in others, the larvae were in various stages of degeneration and/or were replaced by a central area of necrosis. Mineralization was observed in the central part of necrosis of some of the granulomas from the stomach (Fig. 4). In the liver, the capsules of granulomas containing nondegenerated parasites (newly formed granulomas) were thinner than in ones with degenerated larvae (mature granulomas). The granulomatous capsules consisted of 3 layers (Fig. 5): i) the inner layer formed mainly by macrophages, ii) the lymphocyte layer, and iii) the outer fibrous capsule. Eosinophilic infiltration varied. In newly formed granulomas, eosinophiles were low in number in the subcutaneous localization but were numerous in subcapsular hepatic granulomas (Fig. 6). In mature granulomas, the intensity of eosinophilic infiltration was variable in all parasitized tissues. In 1 mature granuloma, nuclei of some macrophages were pyknotic. Heterophilic cells were also present.
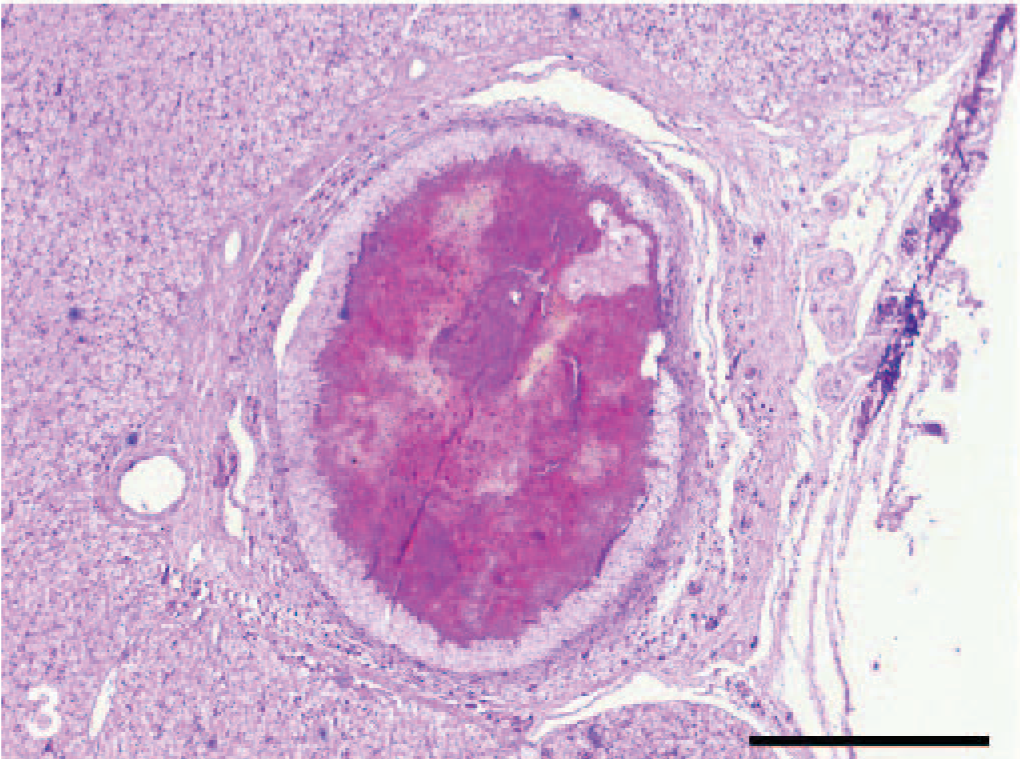
Liver; dice snake. Central necrosis is present within subcapsular granuloma. HE.
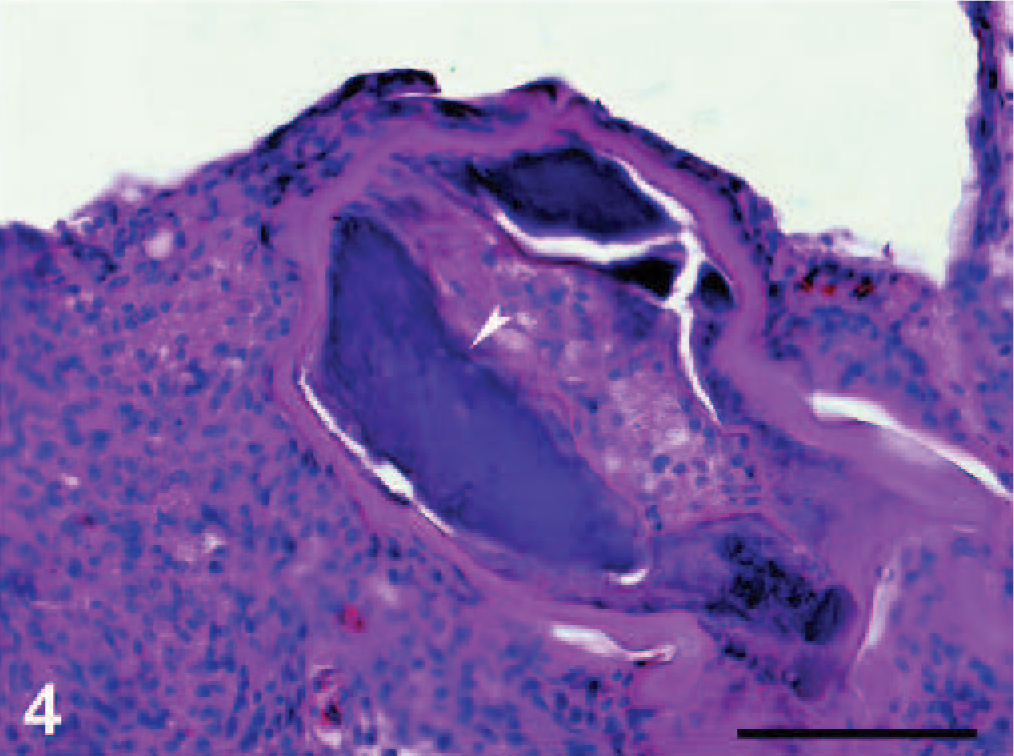
Stomach; dice snake. Central necrosis and mineralization (arrowhead) is present in subserosal granuloma. HE.
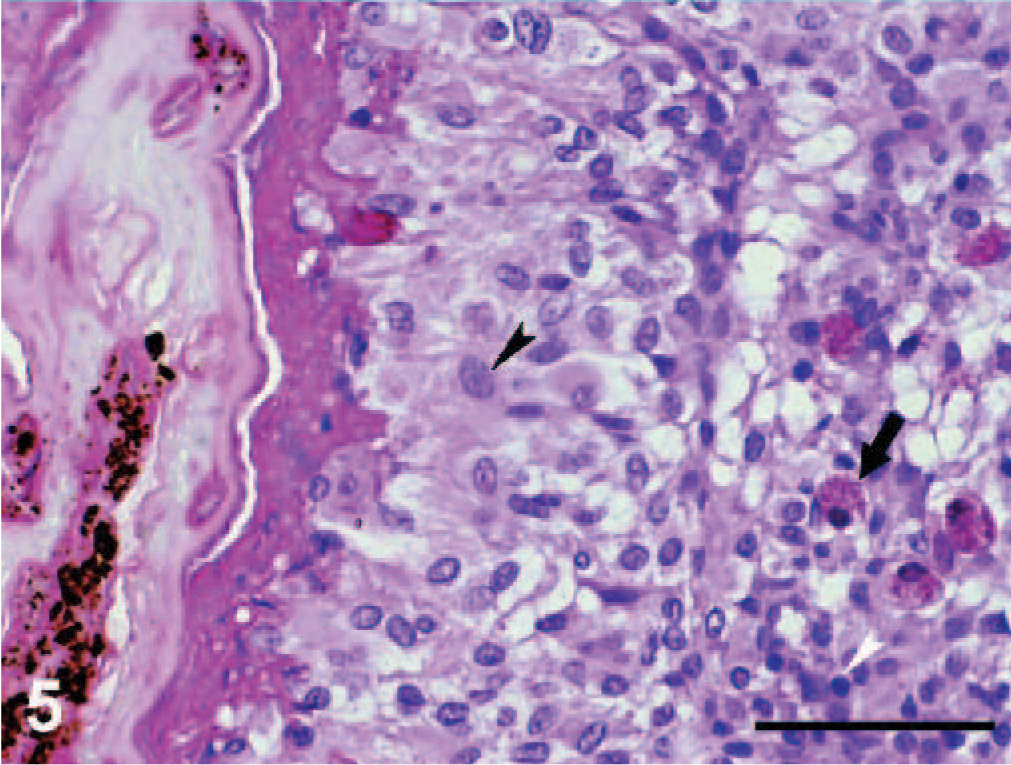
Liver; dice snake. Central necrosis, macrophage layer (black arrowhead) and lymphocyte layer (white arrowhead) with eosinophilic infiltrate (arrow) are forming the 3 layers of parasitic granuloma. HE.
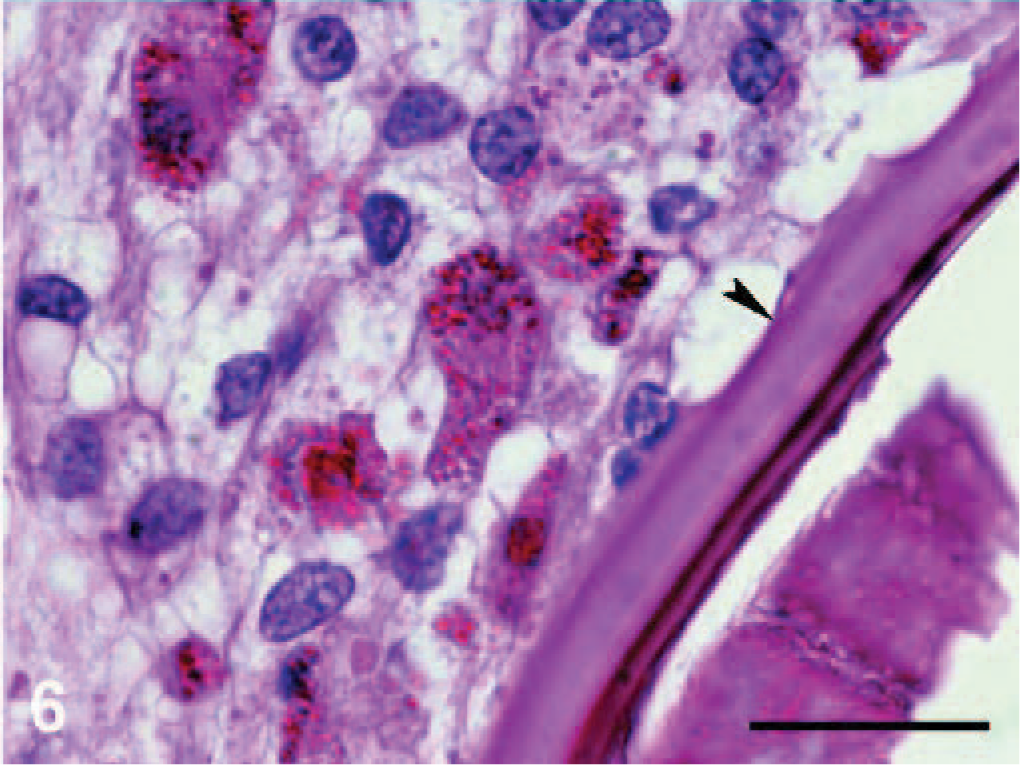
Liver; dice snake. Eosinophilic infiltrate and macrophages are present near cuticle of nematode (arrowhead) in early stage subcapsular granuloma. HE.
Different organs and tissues of free-ranging reptiles are commonly found to be infected by larval nematodes. There are reports describing various histologic lesions induced by these parasites in lizards, snakes, and chelonians, with different histologic features. It seems that encapsulation in the connective tissue is characteristic to larvae of Eustrongylides, as shown in the present paper and in previous studies, 1 whereas other encapsulated nematode larvae are most commonly found in the submucosal or muscular layer of digestive tube. 3, 5, 7 Dracunculus species are able to induce nongranular but pustular lesions in snakes. 4
As in our report, in most cases, 2 types of granulomas are found: newly formed, containing viable undamaged larvae, and mature, with degenerating or degenerated larvae. 1, 5, 7
A common characteristic of nematode-induced granulomas in reptile is the presence of an outer layer. Granulomas induced by physalopterid larvae in snakes and lizards are collagenous, whereas larvae of Ascarops in lizards, 2, 3 Spiroxys in turtles, 7 and Eustrongylides in snakes 1 are contained by granulomas surrounded by a fibrous layer. In recently formed granulomas produced by larvae of Spiroxys in turtles, granulomas were not surrounded by a fibrous capsule. 7
Histiocytes and lymphocytes are commonly found in granulomas induced by various nematode larvae in different tissues and hosts: physalopterid larvae in gastric submucosa and subserosa of lizards and snakes, 5 Ascarops larvae in gastric submucosa, 3 and liver parenchyma of lizards, 2 but are absent in granulomas produced by Spiroxys in turtles 7 and Eustrongylides in snakes. 1 Our report sustains this observation.
The eosinophilic infiltrate during the early stage of infection was found to be massive in infections caused by Spiroxys in turtles 7 and Eustrongylides in snakes 1 and, in both cases, decreased in intensity in mature granulomas. Our results show the same stage dependent eosinophilic infiltrate only in subcapsular liver granulomas.
Apparently, snakes can be considered dead-end hosts in the life cycle of Eustrongylides nematodes that probably leads to continuous death of larvae and consequent pathology. Although lesions induced by larval Eustrongylides in snakes are severe, little is known about how they can influence the health of snakes. Mortality caused by Eustrongylides larvae was reported in captive red-sided garter snakes (Thamnophis sirtalis parietalis) 6 but without histopathologic evaluation.
Footnotes
Acknowledgements
We thank Frantisek Moravec for the parasite identification. This study was funded by the CNCSIS grant TD196 (to AM), GAČR 206/03/1544 (to MS), GAČR 524/03/D104 (to DM) and by a stipend from the Swedish Institute (SK). We thank Prof. Fredric Frye for kindly reviewing the manuscript and Lucia Bel and Bogdan Fersedi for field help. We are indebted to ARBDD for issuing research permits.
