Abstract
A 5-year-old Shetland Sheepdog was presented with a history of weakness, ataxia, anemia, thrombocytopenia, and occasional seizures. The dog had been treated for 6 months with prednisone for inflammatory bowel disease. A positive titer for Ehrlichia canis was detected 6 months before referral. The initial physical examination revealed a weak, laterally recumbent dog with pale mucous membranes. Neurologic examination revealed multiple neurologic deficits. A complete blood cell count (CBC) revealed normochromic, normocytic, nonregenerative anemia; lymphopenia; thrombocytopenia; and neutrophilic and monocytic leukocytosis. urinalysis revealed proteinuria, with a specific gravity of 1.045. The dog was unresponsive to treatment and died. At necropsy, there was severe serofibrinous peritonitis and pleuritis, with randomly scattered dark brown necrotic foci present in multiple organs, including liver, spleen, kidney, and pancreatic lymph node. Histologically, there were extensive regions of parenchymal necrosis surrounded by neutrophils admixed with epithelioid macrophages, lymphocytes, and pigmented fungal organisms. Numerous brown, 2 to 6 μm in diameter, septate, branching hyphae, subsequently identified as Ochroconis gallopavum (formerly Dactylaria constricta var. gallopava), were observed.
Phaeohyphomycosis (Greek phaeo, gray or black) is a collective term for cutaneous and systemic diseases caused by several genera of black molds that develop in tissue in the form of dark-walled septate mycelia. The term dematiaceous is used for pigmented fungi and requires demonstration of melanin pigment, which imparts brown-black discoloration to the fungus. These fungi are ubiquitous, opportunistic, markedly pathogenic, and generally produce disease in immunocompromised or chronically ill debilitated animals, as well as those exposed to long-term corticosteroid or broad-spectrum antibiotic therapy.
Numerous members of the phaeohyphomycosis group have been reported to cause superficial, cutaneous, subcutaneous, and systemic disease in animal and humans. In particular, cats are more susceptible than other animals. 6 Some of the genera that are reportedly pathogenic in dogs and cats include Curvularia and Fonsecaea pedrosoi, which cause cutaneous disease. 10, 13 Cladophialophora bantiana (synonyms Torula bantiana, Cladosporium bantianum, Cladosporium trichoides, Xylohypha emmonsii, and Xylohypha bantiana) is part of the normal fungal flora on the hair of dogs and causes cutaneous, systemic, or localized cerebral disease in dogs and cats. 1, 5, 19 Systemic disease in dogs has been reported due to Bipolaris spicifera and Candida glabrata infection. 24 Moniliella suaveolens, Exophiala spinifera, Phialophora verrucosa, and Alternaria alternata cause cutaneous and subcutaneous disease in cats. 2, 6, 18
The nomenclature of Ochroconis gallopavum has been controversial. 15 The fungus O. gallopavum was initially termed Diplorhinotrichum gallopavum and was later reclassified as Dactylaria gallopava in 1968. 3 In 1983, the organism was further reclassified and assigned to the genus Ochroconis. 7 However, this classification has not entirely been accepted. Salkin and Dixon 22 used morphologic features and proposed D. gallopava be regarded as D. constricta var. gallopava. Therefore, some investigators still prefer to use the genus Dactylaria. Some microscopically similar species include Ochroconis humicola, O. constricta, O. tshawytschae, and Scolecobasidium terreum. Ochroconis gallopavum is known for its neurotropic potential and predilection to cause severe necrotizing encephalitis in humans 15 ; cats; 20 and birds, including poultry, 4, 11 grey-winged trumpeters (Psophia creptans), 14 and Japanese quail (Coturnix coturnix japonica). 23 Infections in fish, amphibians, and reptiles have also been reported. 12
Ochroconis gallopavum is a thermophilic dematiaceous fungus and favors soil and decaying vegetation that is undergoing a composting phenomenon associated with the generation of heat and acidic environment. It has been isolated from broiler house litter where similar environmental conditions prevail. It is also a contaminant of effluents of hot springs and nuclear reactors, thermal soils, and self-heated coal waste piles. This report describes an unusual case of systemic phaeohyphomycosis caused by Ochroconis gallopavum, without involvement of the central nervous system.
A 5-year old, intact female Shetland Sheepdog was presented with a 2-month history of decreased appetite, weakness, ataxia, nystagmus, anemia, and possible occasional seizure activity, and a 1-month history of thrombocytopenia. The dog had been treated for 6 months with prednisone (0.5 mg/kg PO EOD [every other day]) and metronidazole (13 mg/kg PO q 12 hours) for inflammatory bowel disease, and 2 injections of imidocarb, 1 week apart, for treatment of Ehrlichia canis because of a positive titer on a Triple Snap Test (IDEXX; Westbrook, ME). The dog had also received a whole-blood transfusion and erythropoietin injections (500 units SC EOD) 3 weeks before referral.
Initial physical examination revealed an extremely weak, laterally recumbent, moderately underweight dog, with pale mucous membranes and bruising associated with recent venipunctures. Neurologic examination revealed mild depression; mild head tilt to the right; decreased menace response; intermittent horizontal, vertical, or rotary positional nystagmus; wide excursions of head; absent conscious proprioception (CP) of the pelvic limbs, decreased CP of thoracic limbs (right > left); increased extensor tone; and hypertonic pelvic limbs, with bilateral crossed extensor reflexes. Neurologic examination suggested multifocal disease of forebrain (seizures and decreased menace), brainstem (central vestibular disease), and spinal cord lesions between T2 and L3.
A CBC revealed a normochromic, normocytic, nonregenerative anemia with a packed cell volume (PCV) of 16% (reference interval [RI]: 33–55%), red blood cell count of 2.33 × 106/μl (RI: 5.5–8.5 × 106/μl), and a reticulocyte count of 0. A thrombocytopenia with a manual platelet count of 15 × 103/μl (RI: 200–500 × 103/μl), and monocytosis of 3.6 × 103/μl (RI: 0.15–1.3 × 103/μl) was noted. Left-shift neutrophilic leukocytosis of 23,352 × 103/μl (RI: 3–12 × 103/μl), lymphopenia of 0.556 × 103/μl (RI: 1–5 × 103/μl), and eosinopenia of 0.0 × 103/μl (RI: 0.1–1.0 × 103/μl) were also observed in the later stage. Serum chemistry profile revealed an increased total protein of 8.4 g/dl (RI: 4.8–7.3 g/dl) because of hyperglobulinemia of 6.0 g/dl (RI: 2.5–4.5 g/dl). Urinalysis revealed a urine specific gravity of 1.045, with a 3+ protein on dipstick (>300 mg/dl), a 3+ sulfosalicylic acid (approximately 200–500 mg/dl), and an inactive sediment. Inadequate regenerative response to anemia (PCV 16%, mean corpuscular volume [MCV] 78.8 fl [RI: 60–75 fl]), mean corpuscular hemoglobin concentration [MCHC] 35.0 g/dl (RI: 23–36 g/dl) and reticulocytes at 4% (RI: 0–1.5%) was observed in later stage during treatment for ehrlichiosis.
Primary differentials for the nonregenerative anemia and thrombocytopenia included chronic infectious and inflammatory diseases, such as ehrlichiosis or other infectious agents (histoplasmosis or toxoplasmosis), development of erythropoietin autoantibodies, or malignant neoplasia; multifocal neurologic disease differentials included metronidazole toxicity, neoplasia, or inflammatory/infectious diseases, such as chronic ehrlichiosis, canine distemper, toxoplasmosis, or granulomatous meningoencephalomyelitis.
The dog was admitted into the intensive care unit and was administered Normosol-R (4.0 ml/kg/hour), ranitidine (2 mg/kg intravenous[IV] Q 8 hours), doxycycline (5 mg/kg PO q 12 hours), and 4 injections of dexamethasone sodium phosphate (0.3 mg/kg IV Q 8 hours), followed by prednisone (2 mg/kg PO Q 12 hours). Ehrlichia canis titers were positive at 1 ° 40,960 (1 ° 80 or greater is considered positive). The dog was unresponsive to treatments, and its condition continued to deteriorate over the next 7 days with culmination in death.
At necropsy, the dog was in poor nutritional condition characterized by bony prominences and marked paucity of visceral and subcutaneous adipose tissue. The mucous membranes and subcutis were mildly icteric. The subcutaneous tissue in the ventral cervical and thoracic region was expanded by edema. There was severe peritonitis and pleuritis, with large quantities of serofibrinous effusion present in the abdominal and pleural cavities, and multiple, ecchymotic hemorrhages throughout the omentum. Multiple, randomly scattered foci, with slightly raised, pale yellow margins and indented soft, friable necrotic centers were present on the diaphragmatic and visceral hepatic capsular surfaces and in the parenchyma. A focally extensive pale region of acute infarction rimmed by a red zone of hemorrhage was located near the edge of the left lateral lobe. There were numerous fibrinous interlobular adhesions. The serosal blood vessels of the gall bladder were distended, and the gall bladder contained thick, viscous bile. The spleen was moderately enlarged. Multiple, discrete, variably sized, slightly raised, dark-brown necrotic foci were present on the capsular surfaces and in the parenchyma of spleen, renal cortices, and pancreatic lymph node. There was mild cerebellar coning and the leptomeninges over the frontal lobes were multifocally cloudy.
Representative tissues, collected during necropsy, were fixed in 10% neutral buffered formalin, routinely processed, and embedded in paraffin. Four- to 5-μm tissue sections were stained with hematoxylin and eosin (HE), Fontana-Masson, periodic acid–Schiff, and Grocott methenamine silver (GMS).
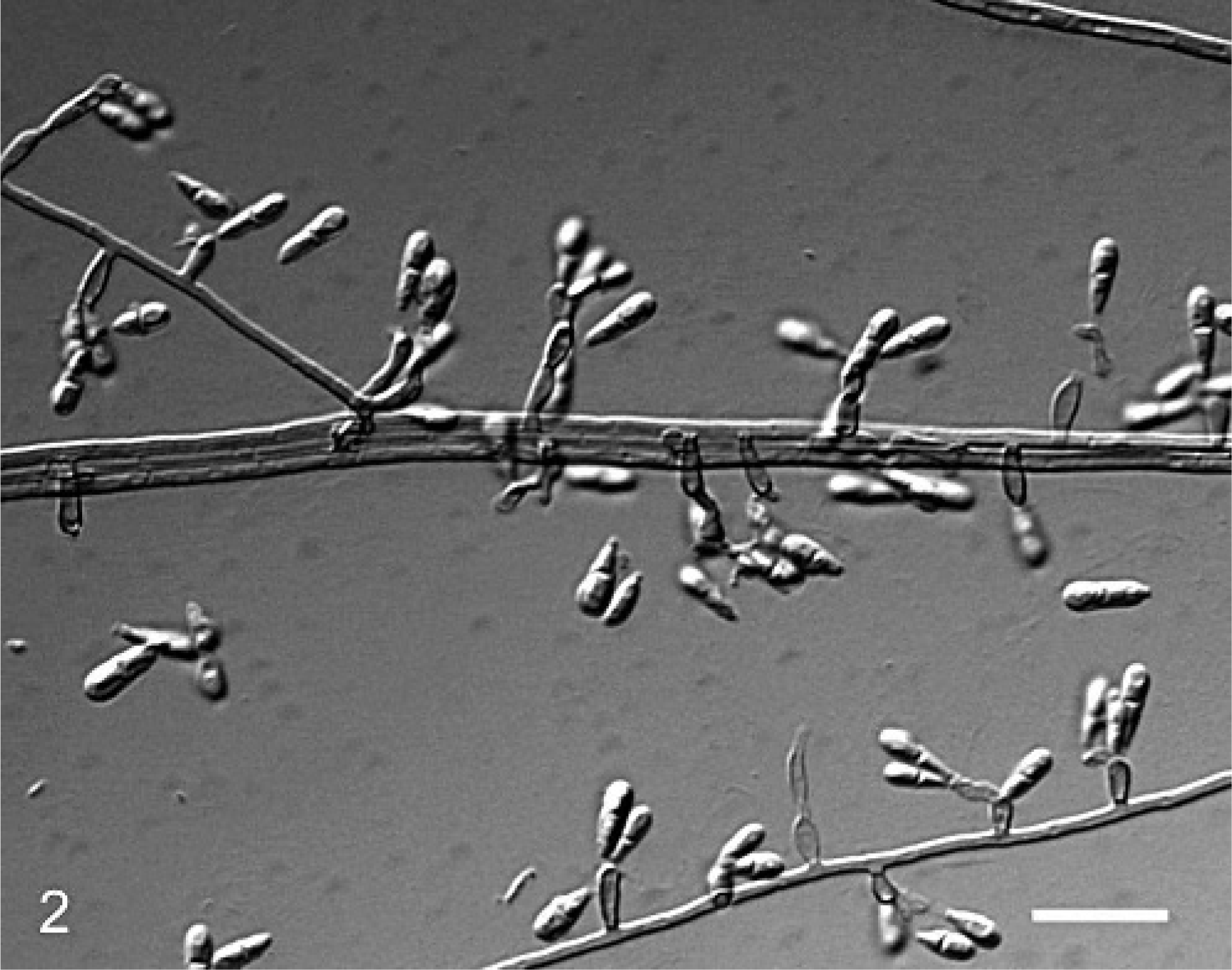
Microscopically, inflammatory and confluent necrotic foci were similar in all affected organs (liver, kidneys, spleen, and pancreatic lymph node). There were extensive regions of parenchymal necrosis, with no architectural details that merely contained abundant cellular debris. These regions were rimmed by a zone of intact and degenerate neutrophils admixed with rare epithelioid macrophages and lymphocytes, as well as numerous pigmented fungal organisms (Fig. 1). Often these regions were associated with blood vessels in which tunics were effaced by fibrin, inflammatory cells, and fungal elements. Numerous, poorly stained bundles and streams of brown and rarely translucent dematiaceous hyphae were embedded in or surrounding the necrotic regions. Fungal hyphae were 2 to 4 μm in diameter, septate, occasionally wavy with relatively parallel contoured walls (Fig. 2). There was irregular branching of hyphae frequently at right angles. Hyphae rarely contained spherical, intercalary or terminal focal bulbous dilatations up to 7 μm in diameter. The brown-black pigment of the hyphal wall was seen readily in unstained tissue sections and with Fontana-Masson stain.
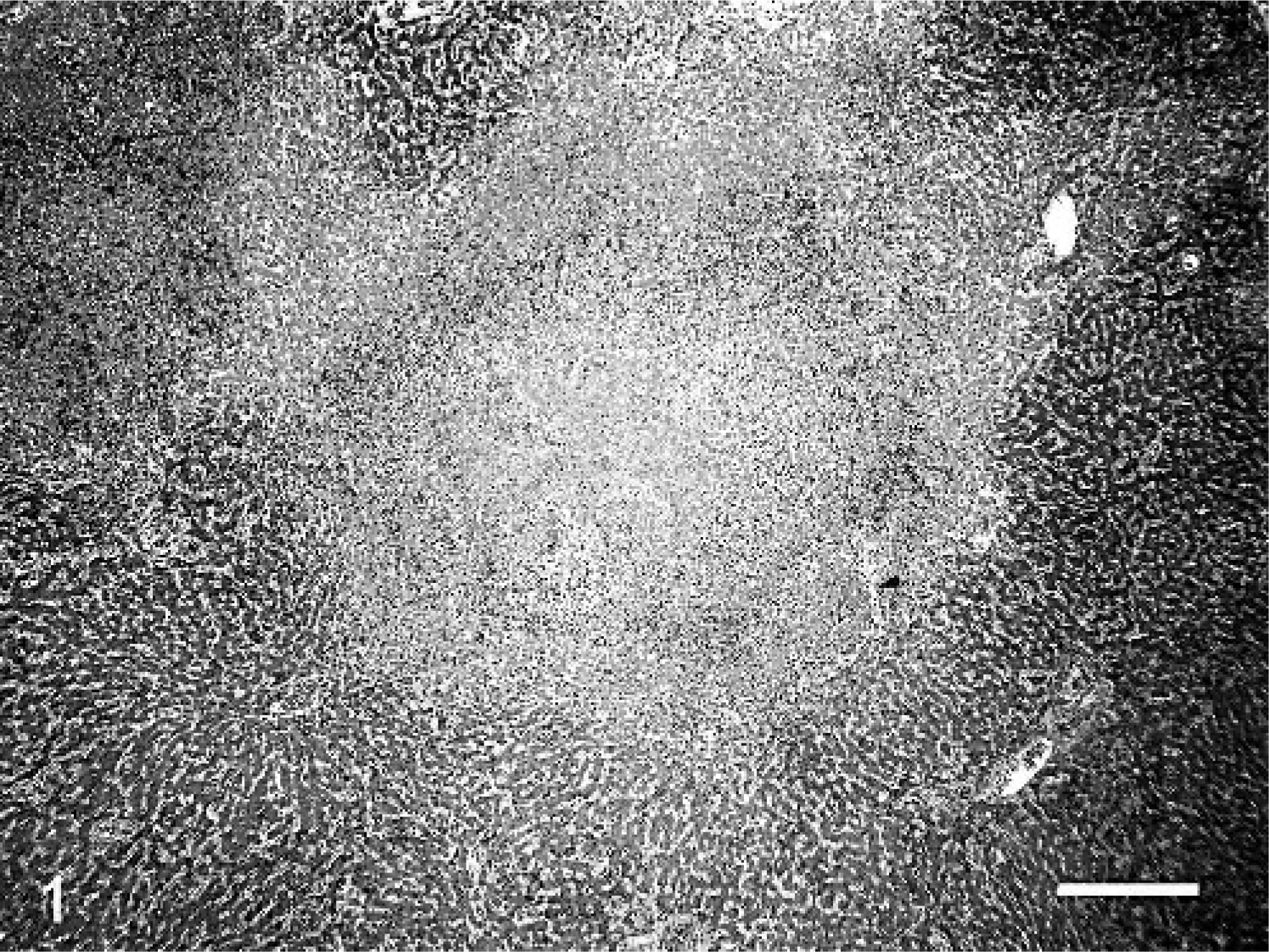
Liver; dog. Photomicrograph of the liver parenchyma effaced by severe necrosis and surrounded by zone of neutrophils and macrophages. Note the thrombosed vessel (arrowhead). HE. Bar = 80 μm.
Liver; dog. Note the septate, branching (arrow head), hyphae with relatively parallel contoured walls. GMS stain. Bar = 12 μm.
In addition, Kupffer cells were diffusely hypertrophied. Bile canalicular cholestasis was occasionally observed. The serosal surfaces of stomach, small intestine, and urinary and gall bladder were regionally thickened by abundant granulation tissue admixed with few scattered plasma cells and macrophages, and were lined by severely hyperplastic mesothelial cells. Numerous fungal hyphae were associated with the gall-bladder serosa. Mild interstitial pneumonia was present and was characterized by diffuse thickening of alveolar septa by mononuclear cells, megakaryocytes, focal depositions of collagen, and marked endothelial swelling. In several regions, alveolar lumens contained fibrin, plasma cells, and few foamy macrophages. The capillary lumens were often occluded by fibrin thrombi. A mild membranous glomerulopathy characterized by segmental thickening of mesangium by a homogenous eosinophilic material was present. Multiple collecting tubules, ducts, and predominantly loops of Henle in the medulla contained small quantities of proteinaceous material. The bone marrow was hyperplastic and contained 1 to 6 megakaryocytes per high-power field with abundant metarubricytes and numerous monocytic precursors admixed with increased numbers of plasma cells. Granulocytic precursors were rarely observed.
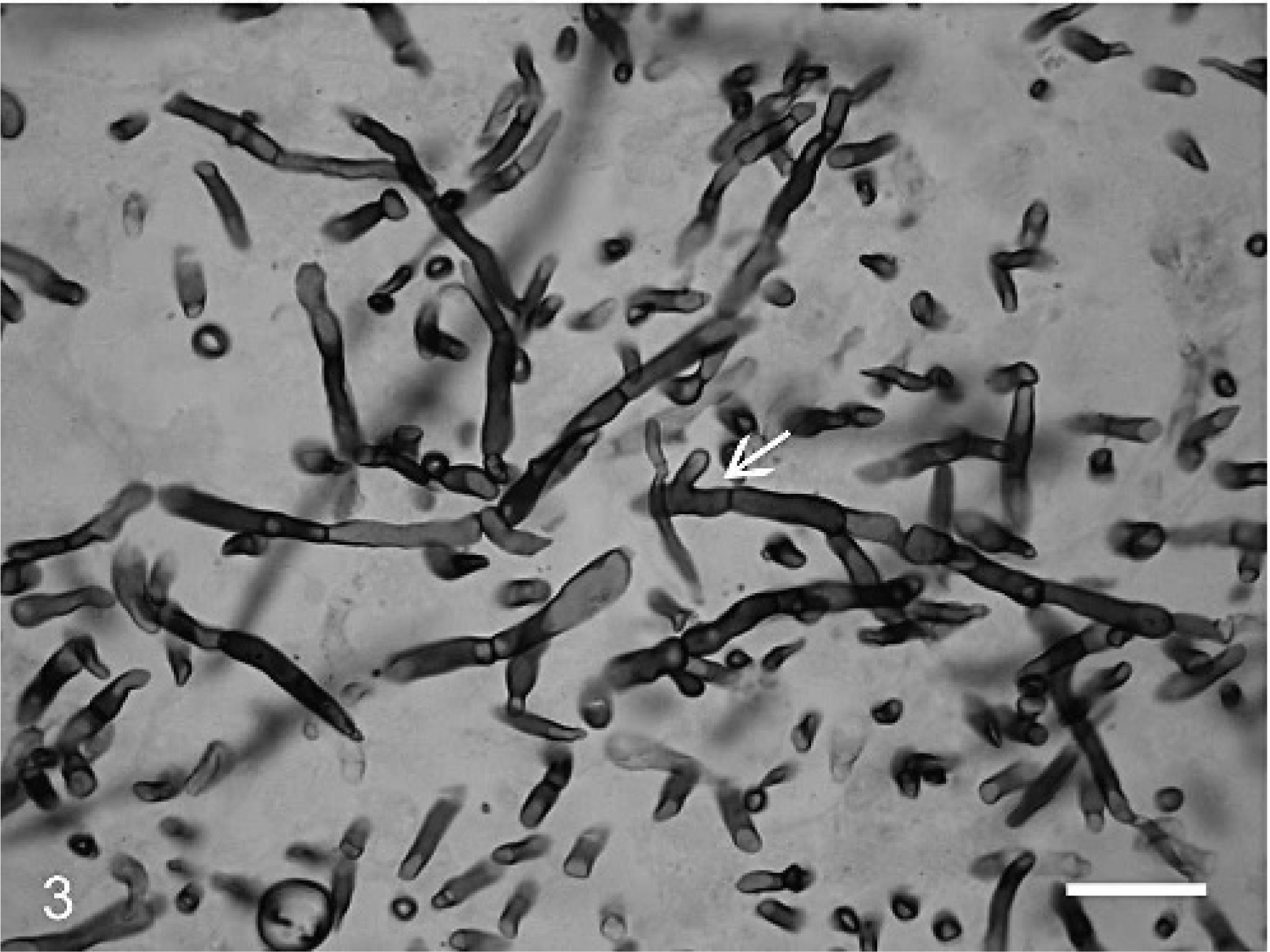
Based upon the morphologic features of darkly-pigmented hyphae in tissue, a provisional diagnosis of systemic phaeohyphomycosis was made. The liver sections were submitted for microbial culture. The morphologic and physiologic characteristics of Ochroconis gallopavum have been described. 9 Colonies on potato dextrose agar were brown, flat, and velvety, with a mahogany diffusing pigment. The isolate exhibited growth at 42°C and failed to grow on media containing cycloheximide. The microscopic morphology of the isolate consisted of brown, septate hyphae with 2-celled, smooth-walled to verruculose clavate conidia. Conidia were borne on fragile denticles, were somewhat constricted at the septum, and measured 11–18 × 2.5–4.5 μm (Fig. 3). Based upon these characteristics, the isolate was identified as Ochroconis gallopavum by the Fungus Testing Laboratory at the University of Texas Health Science Center at San Antonio, Texas.
O. gallopavum slide culture reveals septate, branching, brownish, hyphae with short, hyaline conidiophores and two-celled conidia borne on denticles. Unstained colonies on agar. Bar = 15 μm.
Lesions from chronic ehrlichiosis were also observed and included perivascular plasmacytic lymphocytic infiltrate in multiple organs (brain, spinal cord, liver, gall bladder, pancreas, and kidneys), interstitial pneumonia, and glomerulopathy with proteinuria. Marked splenic and hepatic hemosiderosis was also present.
The concept of phaeohyphomycosis has been reviewed and the terms superficial, cutaneous, subcutaneous, and systemic phaeohyphomycosis are proposed for the major categories of human phaeohyphomycosis. 17 Superficial infections are limited to the epidermis and hair follicles, whereas cutaneous lesions involve epidermis and dermal tissues. Both superficial and cutaneous forms are believed to result from the colonization of epidermis and hair follicles. The cerebral, cutaneous, and subcutaneous forms are most common in cats, whereas systemic and cerebral forms are common in dogs. For better understanding of this heterogeneous group of diseases, we propose analogous terminology in veterinary medicine. This classification gives direct information about the broad anatomical location and extent of host involvement.
The portal of entry and exact mechanism by which members of phaeohyphomycosis cause systemic disease or localized primary cerebral infection are unknown. The cutaneous and subcutaneous phaeohyphomycosis are presumed to occur through traumatic inoculation or secondary contamination of preexisting cutaneous lesions, and cerebral and systemic disease is proposed to occur via respiratory exposure to conidia or via hematogenous spread from the localized cutaneous form. 19 In this dog, the presence of lesions in multiple organs and hyphae within the vascular tunics are consistent with a hematogenous infection. The source of infection in this dog is most likely environmental, because Ochroconis is usually soil borne. The initial route of entry in our case remains undetermined. In the absence of cutaneous or subcutaneous lesions, we suspect inhalation of the Ochroconis conidia or, alternatively, contamination of the bruising associated with venipunctures. Respiratory exposure to conidia, with subsequent germination and dissemination, has been shown to produce the disease experimentally in poultry. 4 The absence of neurotropism in this disseminated infection is unusual; the cause(s) for this are not elucidated in this case. Multiple histologic sections of the brain representing frontal, parietal, temporal, and occipital lobes; cerebellum; and brain stem; and sections representing cervical, thoracic, lumbar, and sacrum regions of the spinal cord were examined. However, fungus or similar lesions induced by O. gallopavum in other organs were not observed in the sections. Neurologic symptoms in this case are believed to be caused by Ehrlichia sp. induced plasmacytic perivasculitis involving the brain and spinal cord. Alternatively, this perivascular infiltrates of plasma cells could be from undetected fungus.
Phaeohyphomycosis in humans and animals is commonly associated with immunosuppression. Cladophialophora bantiana has been isolated from a dog with canine distemper. 8 We speculate immune suppression induced by long-term corticosteroid therapy and concomitant Ehrlichia canis infection as predisposing factors in our dog. Lymphopenia was observed in this dog. However, we did not characterize T-or B-cell lymphopenia in this case, but we suspect disrupted cell-mediated immune responses, particularly a reduction in macrophage activation. Murine experiments conducted by Whitcomb suggested that cellular immunity plays an important role in the prevention of phaeohyphomycosis. 25
These fungi are saprophytes and may be part of normal cutaneous flora; therefore, the disease generally occurs in immunocompromised animals. However, in cats, the majority of cases occur in healthy animals. 5, 21 Lobetti 16 evaluated white cell and differential counts, serum protein electrophoresis, and serum immunoglobulin fractions in 2 dogs with systemic phaeohyphomycosis in the absence of primary immunodeficiency. The results included left-shift neutrophilic leukocytosis, lymphopenia, and hyperglobulinemia. The investigator concluded that the humoral response and neutrophil functions were normal. 16 However, lymphopenia may have accounted for the systemic disease. Cell-mediated immunity, in particular, activation of macrophages by helper T cells plays a crucial role in controlling fungal infections. Persistent leukopenia and lymphopenia have also been described in multiple dogs with systemic or cerebral disease. 1, 19 It appears that lymphopenia and secondary disruption of cell-mediated immunity may act as a predisposing factor for systemic phaeohyphomycosis.
Ochroconis and other etiologic agents of phaeohyphomycosis are increasingly recognized as emerging pathogens in immunocompromised animals and humans. Therefore, phaeohyphomycosis infection in domestic animals is a public health and an occupational health hazard because of risk of exposure during biopsy, necropsy, and handling.
Footnotes
Acknowledgements
We thank Dr. A. W. Confer for reviewing the manuscript and for his valuable comments. Professional contributions of Elizabeth Thompson (Fungus Testing Laboratory, University of Texas Health Science Center, San Antonio, Texas) in identifying the fungus is highly appreciated. The assistance of Ms Betty Handlin in preparing the manuscript is appreciated.
