Abstract
Mycotic meningoencephalitis in dogs may manifest as a primary disease of the central nervous system or as a part of disseminated infection. Fungi belonging to the genus Bipolaris are saprophytic plant pathogens and can cause disease in humans. The authors report a case of Bipolaris infection in a dog with granulomatous meningoencephalitis, nephritis, and vasculitis. The clinical and histological features resembled those of the more common aspergillosis, thus warranting confirmation by molecular methods. Polymerase chain reaction and sequence analysis identified Bipolaris from the brain lesion, indicating its involvement in the disease. To the authors' knowledge, this is the first reported case of meningoencephalitis caused by this fungus in a domestic animal.
In dogs, a number of fungal agents cause central nervous system (CNS) infection either as primary intracranial disease or as a component of disseminated infection. Fungi such as Cryptococcus neoformans, Histoplasma capsulatum, Blastomyces dermatitidis, Coccidioides immitis, Aspergillus sp, Fusarium, and Cladophialophora bantiana are known to cause CNS infection. 1,3,6,9,12 In this report, we describe a disseminated mycosis with CNS involvement in a dog as caused by Bipolaris sp (previously known as Drechslera spicifera 10 ).
Clinical History
An 8-year-old spayed female crossbred Labrador Retriever was presented as an emergency to the Veterinary Medical Teaching Hospital at Texas A&M University. The dog had a history of progressive weight loss, inappetence, polyuria, and polydipsia for 2 months, as well as a recent onset of vomiting, right-sided head tilt, nystagmus, and loss of sensation on the right side of the face. Abnormal findings upon neurologic examination included postural deficits, falling to both sides, and altered mentation. A diagnosis of central vestibular disease was based on clinical signs and neuroanatomic localization. The animal was euthanized owing to the poor prognosis.
Necropsy and Histologic Findings
At necropsy, the brain was grossly normal without apparent foci of discoloration or malacia. The right renal pelvis was moderately dilated and contained scanty soft tan-to-yellow material. Both kidneys had a few small spherical or irregular cavitations (up to 8 mm in diameter) at the corticomedullary junction. Representative tissues were fixed in 10% neutral buffered formalin, routinely processed, embedded in paraffin wax, and sectioned at 4 to 6 μm for histologic and histochemical evaluation.
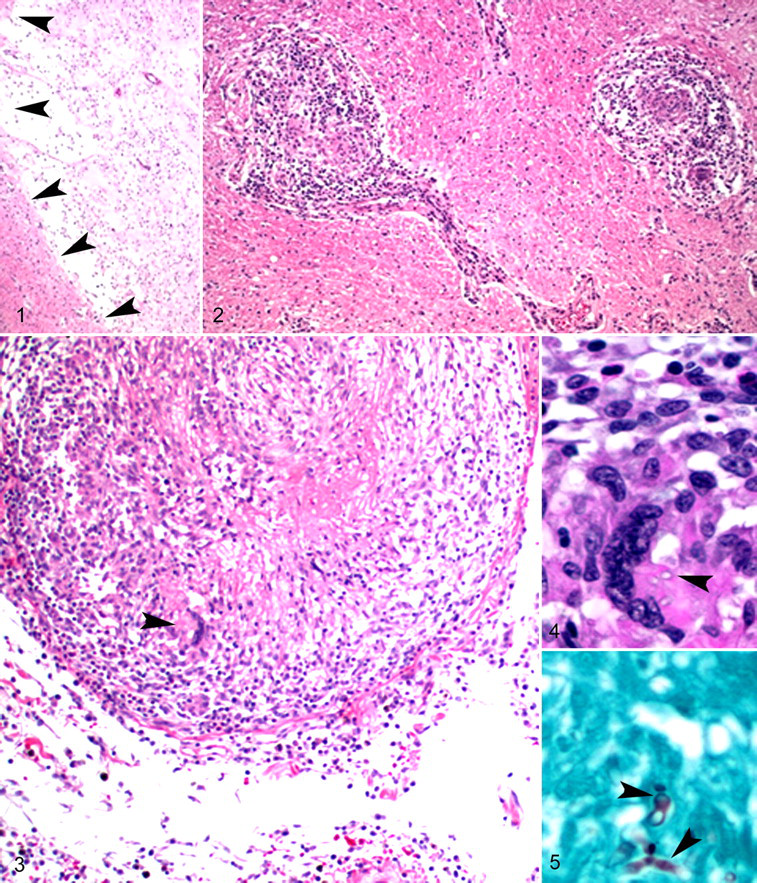
Histologic changes were distributed through the cerebrum, pons, medulla oblongata, and surrounding leptomeninges. Multifocal pallor and cavitation were in the cerebral cortex (Fig. 1 ). Multifocally, the parenchyma of the cerebral cortex and medulla oblongata was obscured by discrete, noncaseating, unencapsulated, focally coalescing granulomas ranging from as small as 50 μm to several millimeters in diameter and composed of macrophages, lymphocytes, fewer plasma cells, and rare multinucleated giant cells (Fig. 2 ). Spaces separated these granulomas from the contiguous neuropil. The leptomeninges were expanded up to 10-fold by similar inflammatory cells and edema. A few meningeal blood vessels were enlarged, occluded by the inflammatory cells, fibrin, cellular debris and edema, and they had disrupted walls (Fig. 3 ). The perivascular space was expanded by fibrosis. A few multinucleated cells had intracytoplasmic fungal organisms (Fig. 4 ). Grocott’s methenamine silver accentuated numerous irregularly septate and nondichotomously branching fungal hyphae with 3- to 6-μm-wide nonparallel walls in the cerebral lesions (Fig. 5 ). Renal sections had similar granulomas, with intralesional fungal hyphae in the cortex, medulla, and pelvis.
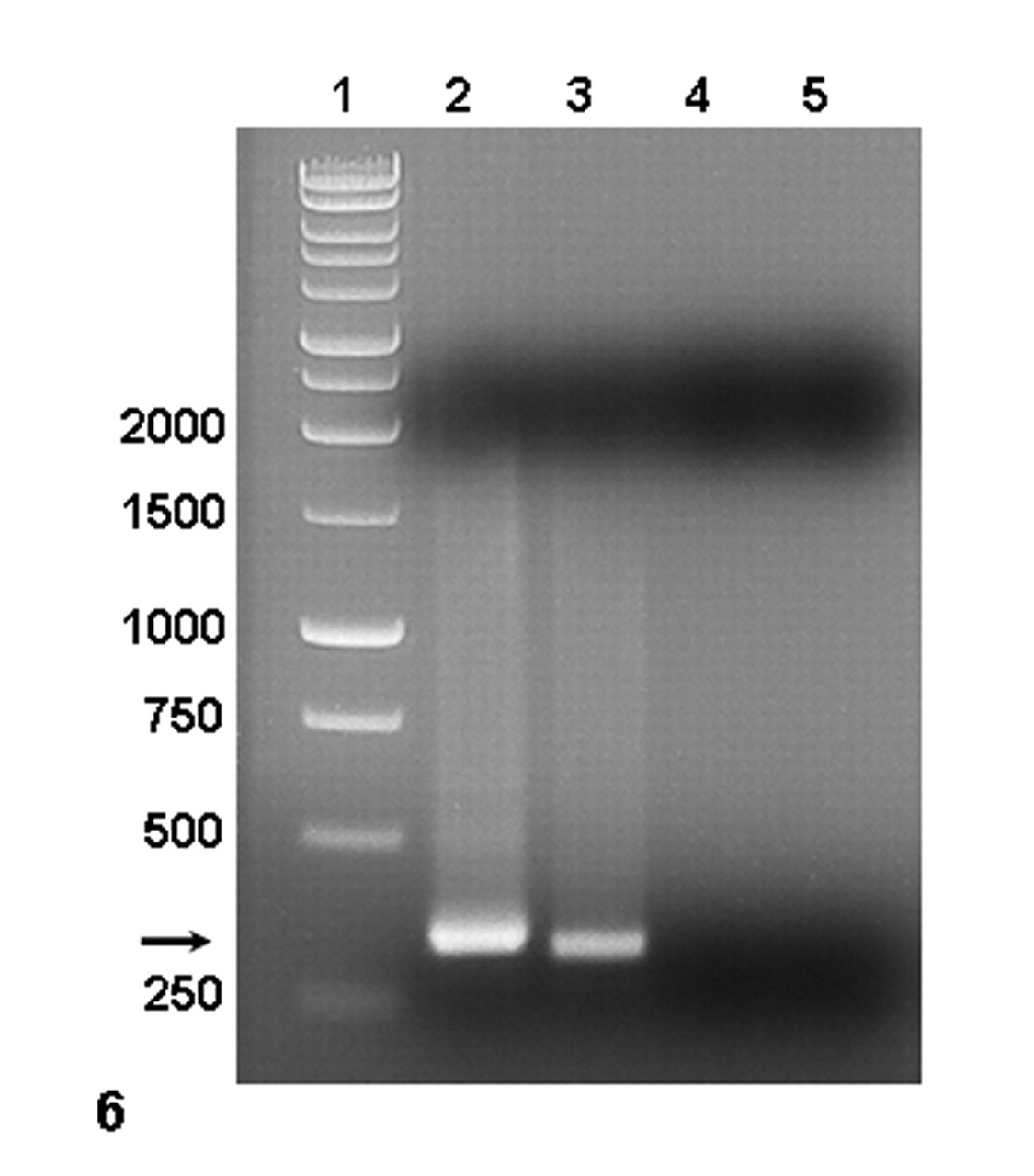
Agarose gel electrophoresis of polymerase chain reaction–amplified fungal rDNA: ITS3 and ITS4. Lane 1, 1 Kb DNA ladder (numbers on the left are in kilobases); lane 2, test sample; lane 3, positive control (Candida albicans); lane 4, kidney from an age-matched dog with no history of fungal infection; lane 5, no template control.
Molecular Diagnostics
To identify the organism, DNA was extracted from formalin-fixed, paraffin-embedded brain and kidney (QIAamp DNA Mini Kit, Qiagen, Valencia, CA). Extracted DNA was quantified with the NanoDrop 1000 spectrophotometer (Thermo Fisher Scientific, Waltham, MA). The universal fungal oligonucleotide primers were used for polymerase chain reaction (PCR) detection: ITS3 (5′-GCATCGATGAA GAACGCAGC-3′) and ITS4 (5′-TCCTCCGCTTATTGATATGC-3′), which amplify portions of the 5.8S and 28S rDNA subunits and the ITS2 region. 16 The PCR conditions were as follows: incubation at 95°C for 5 minutes; 35 cycles of 95°C for 30 seconds, 55°C for 30 seconds, and 72°C for 30 seconds; and a final extension step at 72°C for 5 minutes. A thermocycler (Mastercycler, Eppendorf, Westbury, NY) was used for amplification. The PCR products were visualized with electrophoresis on 1% agarose gel containing ethidium bromide. The PCR assay contained one positive control (Candida albicans in-house control) and two negative controls (DNA extracted from brain of an age-matched dog with no history of fungal infection and no template control). The PCR-amplified product was sequenced (GENEWIZ, South Plainfield, NJ), and data were used to search the GenBank database (US National Library of Medicine, Bethesda, MD) to identify the fungus with the help of the BLASTn program (http://www.ncbi.nlm.nih.gov/BLAST). Amplification of the DNA sample with ITS3 and ITS4 primers resulted in a fragment of approximately 350 base pairs (Fig. 6 ). A search with the BLASTn algorithm to compare the sequence of the amplified fungal rDNA with existing sequences in the GenBank database revealed 100% homology with Bipolaris sp (GenBank accession Nos. AF455407 and AY004776).
Discussion
Central vestibular disease results from dysfunction of the vestibular nuclei in the brain stem or from disease affecting the caudal cerebellar peduncles or the flocculonodular lobe of the cerebellum. 13 Common diseases associated with central vestibular disease in dogs include canine distemper, toxoplasmosis, neosporidiosis, cryptococcosis, aspergillosis, granulomatous meningoencephalitis, Lyme disease, Rocky Mountain spotted fever, and ehrlichiosis, as well as neoplasms. Organophosphorus toxicity, hepatic encephalopathy, and thiamine deficiency are less frequent causes. Antemortem diagnosis is typically based on analysis of cerebrospinal fluid, antibody titer, culture, and diagnostic imaging, such as magnetic resonance imaging. Diagnostic investigations including imaging, cerebrospinal fluid analysis, and fungal culture were not performed in this case.
The pathogenesis of mycotic encephalitis is often unclear. In humans, systemic fungal disease is more common in immunocompromised individuals. Fungal pathogens can enter the body through respiratory, gastrointestinal, and cutaneous routes. The presence of hyphae in vessels suggests hematogenous dissemination in this dog. Numerous histiocytic intracytoplasmic and extracellular fungi were observed in the blood vessels of the leptomeninges and the kidneys. Given the month-long clinical signs of weight loss and polyuria/polydipsia and the sudden onset of CNS disease, we presume that the encephalitis was secondary to primary renal disease. There was no history of steroid administration in this patient or other evidence of immunosuppression.
The genus Bipolaris contains approximately 47 species of ubiquitous, mostly plant pathogens, and it is most frequently associated with grasses, plant material, decaying food, and soil. Three species—B spicifera, B australiensis, and B hawaiiensis are dematiaceous fungi and may cause phaeohyphomycosis. Recently, Bipolaris sp has been reported to cause human disease, including meningitis, particularly in immunocompromised patients. 2,4,7,8,14 Reports of Bipolaris infection in animals are sparse. 5,10 A concurrent systemic mycosis with B spicifera and Torulopsis glabrata involving skin, kidney, pericardium, and heart in a dog was reported without CNS involvement. 15
Reports of cerebral phaeohyphomycosis caused by dematiaceous fungi in the dog are uncommon. Cladosporium cladosporioides was isolated from a case of granulomatous encephalitis and nephritis in a German Shepherd. 11 The case reported here describes involvement of Bipolaris in a dog with CNS disease. Bipolaris is a dematiaceous fungus; however, in tissue sections, the fungal hyphae were poorly pigmented in contrast to the intensely pigmented fungi commonly observed in phaeohyphomycosis. The level of pigmentation in phaeohyphomyces depends on variables such as the rate of growth, viability, age, and availability of the nutrients. Based on the pigment content, the fungus in this case resembled nondematiaceous Hyphomyces, such as Aspergillus. However, immunohistochemistry with a genus-specific antibody failed to demonstrate Aspergillus in the brain or kidney (data not shown), thus decreasing the index of suspicion of its involvement in this case.
The most reliable, rapid, and sensitive method of identifying a fungal agent is PCR. It eliminates the shortcomings of culture (i.e., longer time to grow and additional laboratory tests for identification) and the challenges of morphologic identification. The isolated DNA from the brain and kidney had 100% homology to Bipolaris, thus corroborating its involvement in the disease.
Histologically, mycotic infection in the CNS is typically associated with granulomatous inflammation with involvement of meninges and vessels, 3,9,11 and the histologic distribution and nature of cerebral lesions in the current case are compatible with this. Scattered cavitations with loss of neuropil suggest ischemia from occlusive vasculitis. Given the low specificity of the histologic features, a definitive identification of the fungal pathogen is often difficult and warrants molecular strategies for confirmation.
In summary, a case of fungal meningoencephalitis in a dog with disseminated mycosis was associated with infection with Bipolaris sp. Clinical signs suggested central vestibular disease. To the best of our knowledge, this is the first report of clinical disease caused by Bipolaris sp with CNS involvement in a domestic animal. This organism should be included in the differential diagnosis for mycotic meningoencephalitis.
Footnotes
Acknowledgements
The financial support to Dr Sims by GlaxoSmithKline for residency training through the ACVP/STP Coalition is thankfully acknowledged. We thank Dr Salvatore Frasca Jr for advice on experimental design and Ms Sarah Jones and her group for preparation and staining of tissue sections.
The authors declared that they had no conflicts of interest with respect to their authorship or the publication of this article.
The authors declared that they received no financial support for their research and/or authorship of this article.
