Abstract
Groups of gnotobiotic piglets were orally inoculated at 3 days of age with either Helicobacter heilmannii (Hh) or a newly described porcine-origin gastric Helicobacter pylori (Hp)-like bacterium. Three Hh-infected and 6 porcine Hp-like-infected swine were fed a milk replacement diet containing 5-10% (v/v) sterile corn syrup as a dietary source of fermentable carbohydrate. None of the piglets infected with Hh and supplemented with corn syrup developed gastric mucosal ulcers; 2 developed small erosive lesions in the pars esophagea. In contrast, all 6 dietary carbohydratesupplemented Hp-like-infected swine developed severe gastroesophageal ulcers; 1 of these exsanguinated into the stomach and died before the end of the experiment. Four of these 6 piglets had grossly evident partially digested blood in the intestinal lumens, indicative of bleeding into the gastrointestinal tract from the stomach. These data suggest that a high carbohydrate diet and gastric colonization by porcine Hp-like bacteria facilitate development of clinically significant gastroesophageal ulcers.
Introduction
Ulceration of the nonglandular esophageal portion of the stomach of feeder pigs, known as gastroesophageal ulceration (GEU), is a common and serious problem in swine production; economic losses are substantial. 26 The stratified squamous epithelium of the pars esophagea (pars) is devoid of mucous-producing glands and lacks the sodium bicarbonate buffering system characteristic of the gastric glandular mucosa. As a consequence, the pars is subject to damage by the acidic contents of the stomach. Others have described the pathogenesis of GEU lesions. 2, 9, 19, 26 As in humans with recurrent heart burn, reflux esophagitis, and Barrett's esophagus, it is believed that gastric-origin hydrogen ions and acidic metabolites of partial intragastric glycolysis enter and acidify the squamous epithelial cell cytoplasm. 15, 31 Cell membrane–bound Na-K-ATPase is disrupted, which results in accumulation of intracellular sodium ions and secondary accumulation of intracellular water, recognized histologically as acute cellular swelling, hydropic degeneration, and ultimately necrosis. 3, 9, 26 For erosive lesions, the underlying basement membrane remains intact and re-epithelialization of the damaged pars is rapid. Penetration of the basement membrane and continued acid-mediated damage to the underlying lamina propria is pivotal for the progression of erosions to ulcers. 23 Devitalized tissue may be secondarily colonized by commensal microbes, including fermentative anaerobes. 5, 12, 13, 16, 32
In humans as well as swine, there is a strong consensus that a relative or absolute increase in gastric hydrogen ions (acid) is the proximate cause of pars and esophageal damage. 26, 31 Thus, a therapeutic goal in humans is to elevate gastric pH toward neutrality through the use of bicarbonate buffering medications and/or to inhibit gastric hydrogen ion production by parietal cells with proton pump inhibitors. These medications provide symptomatic relief for patients affected with heart burn and reflux esophagitis and further implicate gastric hyperacidity in the pathogenesis of disease.
Feeder swine diets contain unsaturated fatty acids, short chain free fatty acids (acetate, propionate, butyrate, and lactate), or peroxidized fats, all of which elevate luminal acid concentration. 3 Finishing diets high in carbohydrate, such as corn and cornstarch, are also dietary sources of acidic metabolites. Incomplete glycolysis of cornstarch by hydrogen ions and/or the enzymatic actions of commensal fermentative microbes such as Lactobacillus and Bacillus species results in the generation of lactic, acetic, and propionic acids within the gastric compartment. 5, 12, 13, 16, 19, 32 In a previous study, we demonstrated that gastric colonization with these fermentative bacteria resulted in GEU if a dietary source of carbohydrate (corn syrup) was provided to colonized gnotobiotes. 19 Finally, in feeder swine, the physical form of the diet also influences the development of GEU. 26 In general, a finely ground (<3.5 mesh) diet is an important risk factor for ulcerogenesis, presumably because of the inability of these diets to “confine” released acids to the fermentation compartment of the glandular stomach. The loss of a pH gradient associated with finely ground diets permits cranial acid reflux into the pars esophagea.
In humans, gastric bacterial colonization by Helicobacter pylori (Hp), while often a subclinical infection, is now established as the cause of chronic superficial (active) gastritis and gastroduodenal ulcer disease. 11, 27, 33 The Helicobacter genus contains many different microbial pathogens and commensals. Helicobacter heilmannii (Hh) 10 is widely distributed in the stomachs of carnivores 7, 8, 14 and omnivores, including swine. 4, 6, 24, 25, 28– 30, 34 These microbes reside in extracellular mucus, deep in the gastric pits, and within the canaliculi of parietal cells in the fundus. 25 A bacterial component to the pathogenesis of porcine GEU was first suggested by reports that associated gastric colonization by Hh with an increased incidence of GEU in swine at the time of slaughter. 4, 28, 29 More recent studies have confirmed the high incidence of pigs colonized by this microbe. 6, 24, 30, 34 Yet, attempts to reproduce porcine GEU and mucosal ulcers with murine-passed Hh were unsuccessful. 19, 22 Moreover, the gastric niche for Hh appears to be predominately in the gastric pits of the fundus, an uncommon site of ulceration in swine and anatomically distant from the pars.
Recently, we reported the recovery of an Hp-like bacterium from young swine that is closely related to human Hp but distinct from Hh. 21 In a companion paper, 22 we demonstrated that one of these isolates, but not Hh, was pathogenic for young gnotobiotic swine and produced GEU as well as gastric glandular mucosal and duodenal mucosal ulcers in experimentally infected swine. These data suggest that the porcine gastric ulcer disease complex may involve gastric colonization with Hp-like bacteria. In this study, we report the reproduction of severe and fatal GEU in young gnotobiotic swine colonized by porcine Hp-like bacteria fed a liquid sow milk replacement diet supplemented with a source of fermentable liquid carbohydrate. 19
Materials and Methods
Piglets and liquid diets
A total of 22 gnotobiotic piglets from portions of 4 litters were used in these experiments. These were derived by Caesarian section and raised as described elsewhere. 17 The diet for gnotobiotic piglets consisted of a sterile liquid sow milk replacement formula (Similac) fed individually to each piglet three times daily, 200–300 ml/feeding. The volume of diet was adjusted over time to accommodate the increased nutritional requirements of the growing piglets. Dietary supplementation with liquid carbohydrate was accomplished by adding sterile corn syrup (Karo), 5% (v/v) to the milk diet at 6 days of age. 19 The supplement was increased to 10% (v/v) at 10–12 days of age and continued until termination at days 35 of age or when moribund (31 days of age in 1 pig).
Bacterial inocula
At 3 days of age, 9 piglets (Group A) were fasted for 12 hours and then orally inoculated with a murine gastric homogenate containing Hh. 8, 19, 22 No attempt was made to determine the number of infectious bacteria in this inocula since Hh is not culturable onto artificial media. Eleven piglets (Group B), separately housed from Group A pigs, were inoculated with porcine Hp-like bacteria (108 bacterial colony forming units [cfu] contained in 2.0 ml Brucella broth) at 3 days of age. The origins and porcine virulence of this porcine gastric bacterial pathogen have been recently reported. 21, 22 Two piglets (Group C) received Brucella broth alone as uninfected controls.
Experimental design and evaluation
The basic design used in this study was similar to those reported previously. 19 Dietary supplementation with corn syrup was introduced at 6 days of age (5% [v/v]) and increased to 10% (v/v) 5–6 days after the piglets had accommodated to the 5% (v/v) diet. Three Hh-infected piglets received the corn syrup supplement as did 6 of the porcine Hp-like–infected piglets. Before termination piglets were fasted overnight. On post-infection day (PID) 32, the fasted piglets were sedated and removed from the isolation units. After collection of a terminal clotted blood sample for serum, piglets were euthanatized with an intravenous overdose of sodium pentothal (Euthol). Stomachs were isolated, ligated at the distal esophagus and proximal duodenum, and removed. For bacterial culture and reisolation, the gastric mucosa from one-half of the stomach was detached from the underlying muscularis with a sterile scalpel blade, weighed, and then homogenized in 10% (w/v) Brucella broth. 17, 19, 21, 22 Ten-fold dilutions of gastric homogenate were plated in duplicate onto Skirrow's medium plates, and these were incubated for 4 days at 37°C, 5% (v/v) oxygen. Reisolates were confirmed to be Hp-like organisms by colony morphology, Gram's stain and morphology, and biochemical tests for urease and catalase activity as described. 17 In one instance, the GEU ulcer crater was dissected free of the surrounding gastric tissue and separately cultured for Hp-like organisms. As indicated, no attempt was made to recover Hh from Group A piglets; for these piglets, Warthin-Starry (WS)-stained gastric tissue section replicates were used to confirm Hh gastric infection. 19, 22
The gastric mucosal ulcers and GEU were photographed before immersion fixation of the opened stomachs in 10% (v/v) phosphate-buffered formalin solution for 24 hours. Tissue sections of the nonglandular (esophageal) cardia, the glandular cardia, the fundus, the antrum, pylorus, and proximal duodenum were collected and processed into paraffin blocks by routine methods. Replicate 5-micron tissue sections were deparaffinized through graded alcohols, rehydrated, and stained with hematoxylin and eosin and WS silver stains as described. 17, 19, 22
Results
Clinical signs
Piglets infected with either gastric bacterial species and maintained with a control milk replacement diet were clinically asymptomatic throughout the course of the experiment. Piglets that were fed a milk replacement diet and corn syrup were initially resistant to the diet but within several days consumed it completely between feedings as did their normal diet-fed counterparts. One of the Hp-like–infected piglets on the experimental diet died overnight on PID 30–31; it was necropsied 6–8 hours later.
Pathologic findings
At necropsy, the only extra-gastrointestinal lesion was seen in the livers of the carbohydrate-supplemented piglets. The livers were paler than normal and yellowish tan in color. Table 1 summarizes the gross findings in the stomachs from the piglets in this experiment. The gastric mucosa of all Helicobacter-infected pigs contained mucosal lymphoid follicles and associated mild submucosal edema, primarily in the lesser curvature and gastric antrum. These lesions were absent in the uninfected control piglets.
A summary of gross findings in the stomachs of gnotobiotic swine infected with gastricHelicobacters with or without dietary supplementation with liquid carbohydrate.
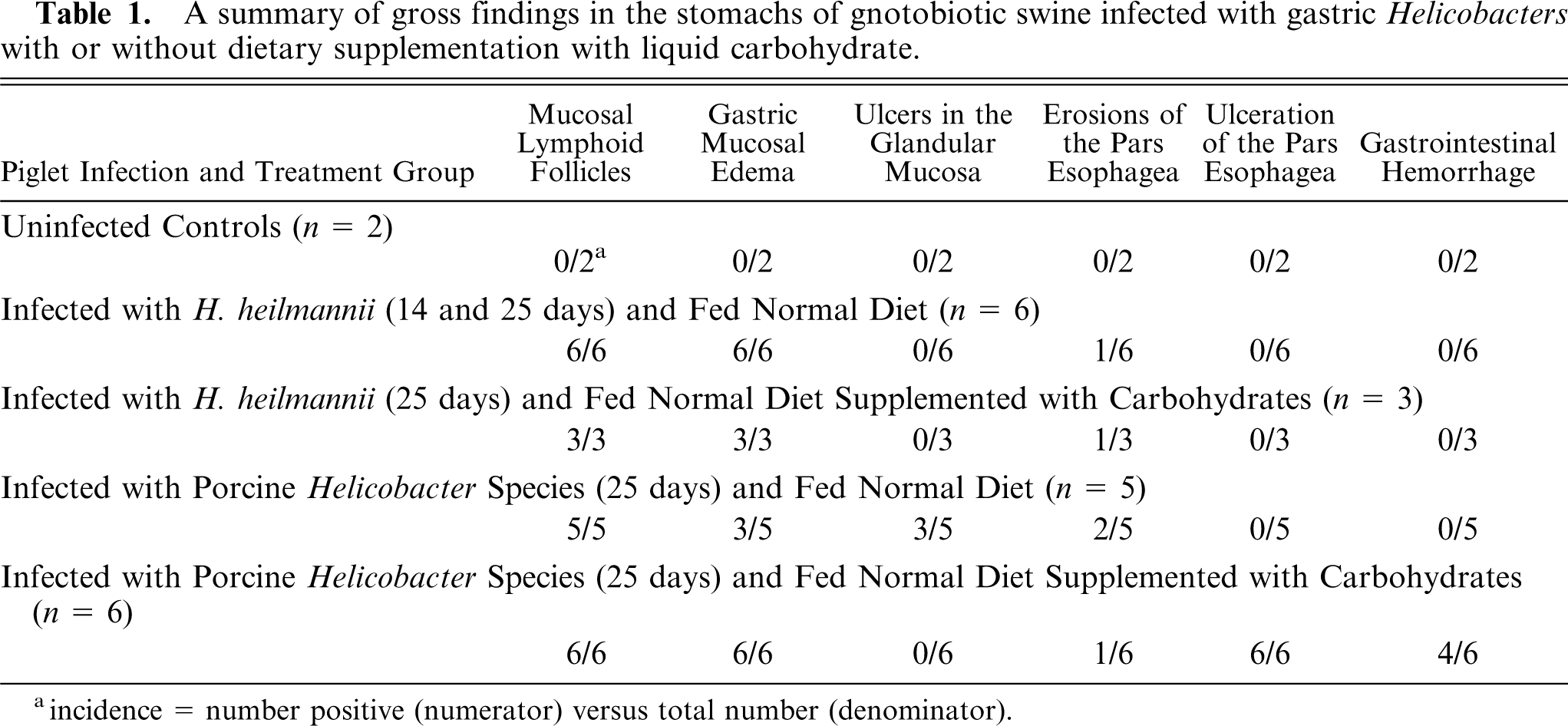
incidence = number positive (numerator) versus total number (denominator).
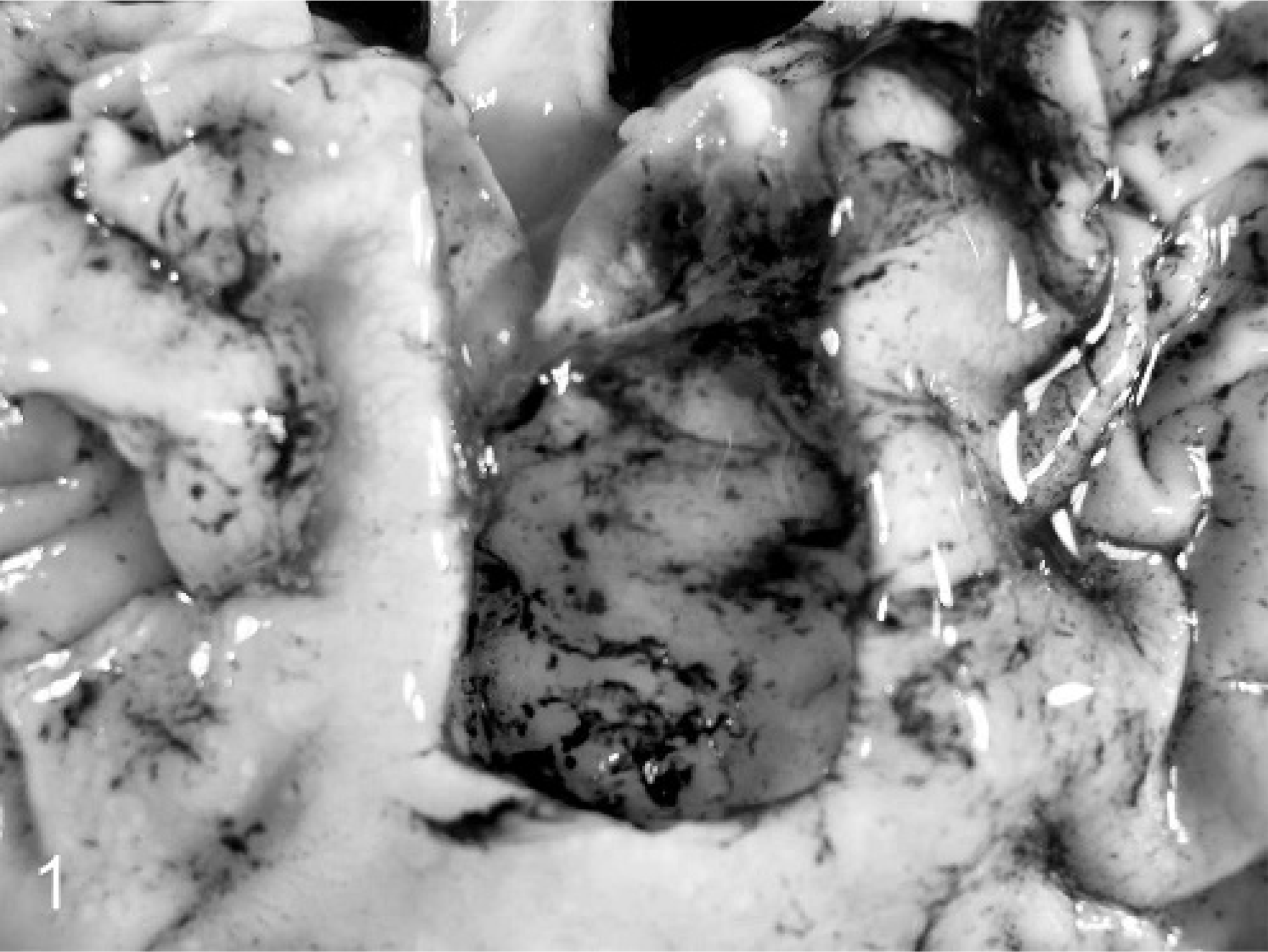
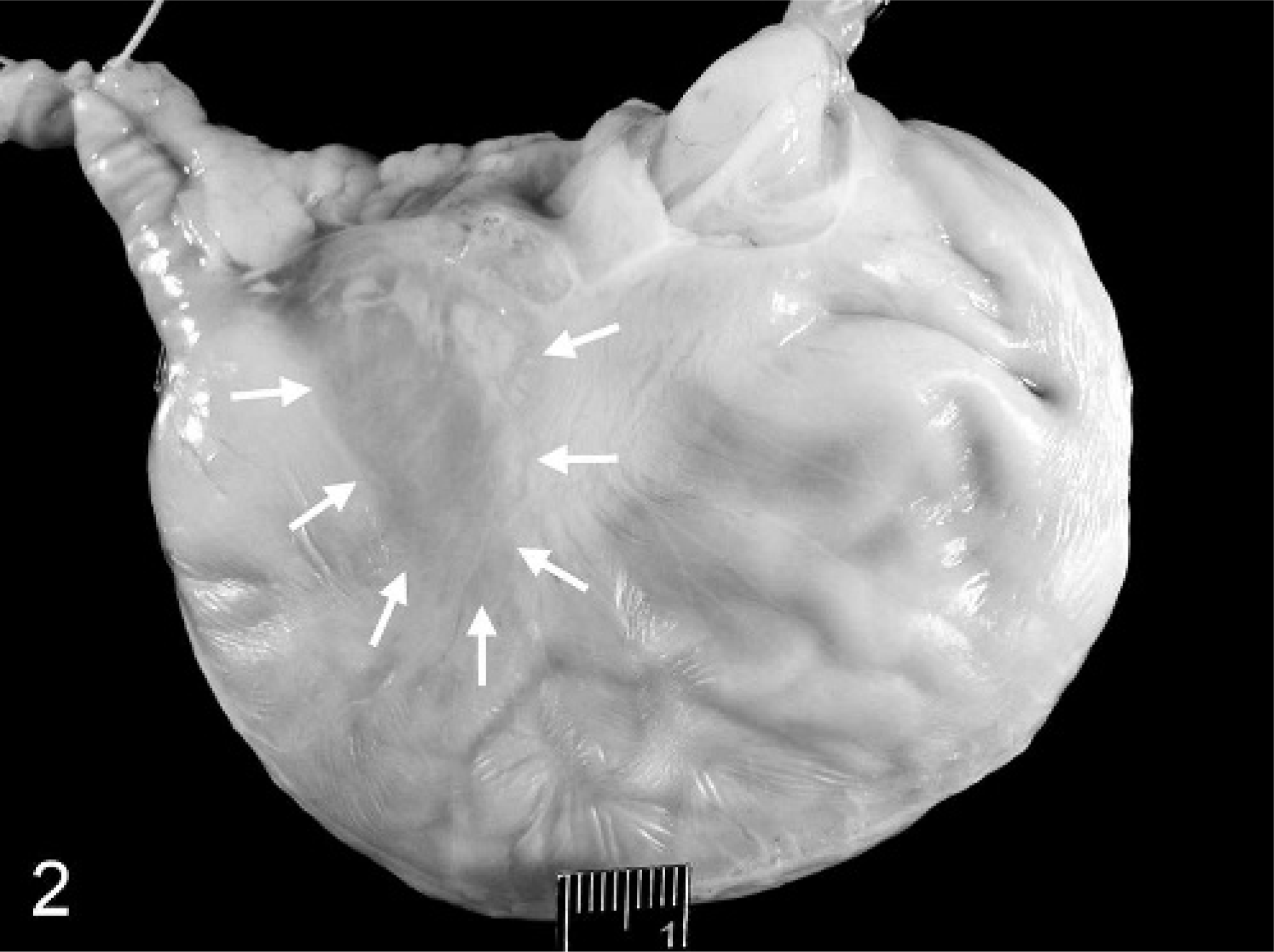
Small ulcers in the glandular gastric mucosa were detected in 3 of 11 piglets infected with porcine Hp-like bacteria and in none (0 of 9) of the piglets infected with Hh. A small erosive lesion of the pars was present in 1 of 6 Hh-infected piglets fed a normal milk diet and in 1 of 3 piglets fed the corn syrup–supplemented diet. In contrast, extensive and deep ulcerations of the pars were present in all 6 piglets infected with porcine Hp-like bacteria that were fed corn syrup. In these swine, the GEU were extensive (1.5–2.0 in. diameter) and deeply penetrating into the muscularis; the ulcer craters extended to cover the entire body of the pars (Fig. 1) and contained necrotic debris, gastric mucus, and partially digested blood. In 2 piglets, focal edema was present on the serosal surface of the greater curvature of the stomach opposite the GEU (Fig. 2). That this ulcerative process in the pars had been present for at least several days before termination was suggested by the presence of partially digested blood in the small and large intestine in 4 of the 6 piglets. The cause of death in the piglet found dead on PID 31 was hypovolemic shock secondary to ex-sanguination into the stomach. In that piglet, the stomach was threefold larger than expected and filled with clotted and partially digested blood and mucus (Fig. 3).
Gastric pars esophagea; a gnotobiotic piglet infected with porcine Hp-like bacteria fed a diet supplemented with 10% (v/v) corn syrup. An ulcer crater involving the entire pars esophagea contains partially digested blood and gastric mucus.
Stomach; a gnotobiotic piglet infected with porcine Hp-like bacteria fed a diet supplemented with 10% (v/v) corn syrup. Focal edema of the gastric serosa and muscularis (arrows) is present in the lesser curvature of the unopened stomach. The esophagus extends dorsally to the right, and the proximal duodenum and pancreas extend dorsally to the left. Bar = 1 cm.
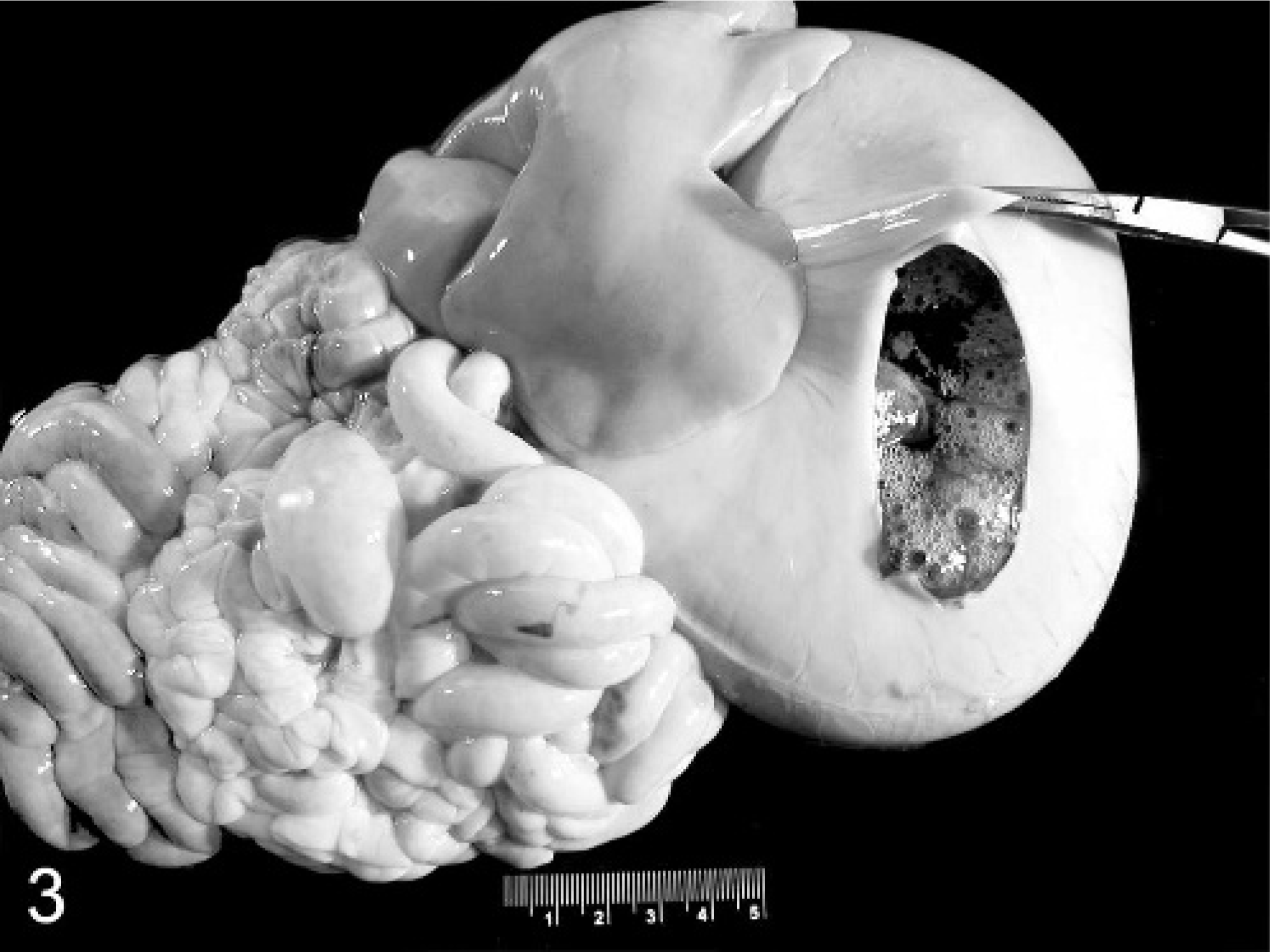
Stomach, liver, and intestinal tract; a gnotobiotic piglet infected with Hp-like bacteria and fed a liquid milk diet supplemented with 10% (v/v) sterile corn syrup. The stomach has been opened to illustrate gastric distention caused by accumulated intragastric blood and mucus. The liver is also pale. Bar = 5 cm.
Histologic examination of gastric mucosal tissue sections from infected but not uninfected control piglets confirmed the presence of lymphoid follicles embedded in the gastric mucosa. Lymphocytic inflammatory infiltrates in the lamina propria of the mucosa accompanied follicles. The ulcerative lesions in the pars were histologically comparable to those described in conventional swine (Fig. 4). The ulcer beds were devoid of epithelial cells and consisted of granulation tissue, hemorrhage, and necrotic debris. Inflammatory cells that accompanied these lesions were degenerate neutrophils. The edges of the ulcers contained hyperplastic remnants of nonglandular epithelium and inflammatory cell infiltrates and were distended by serous fluid accumulations.
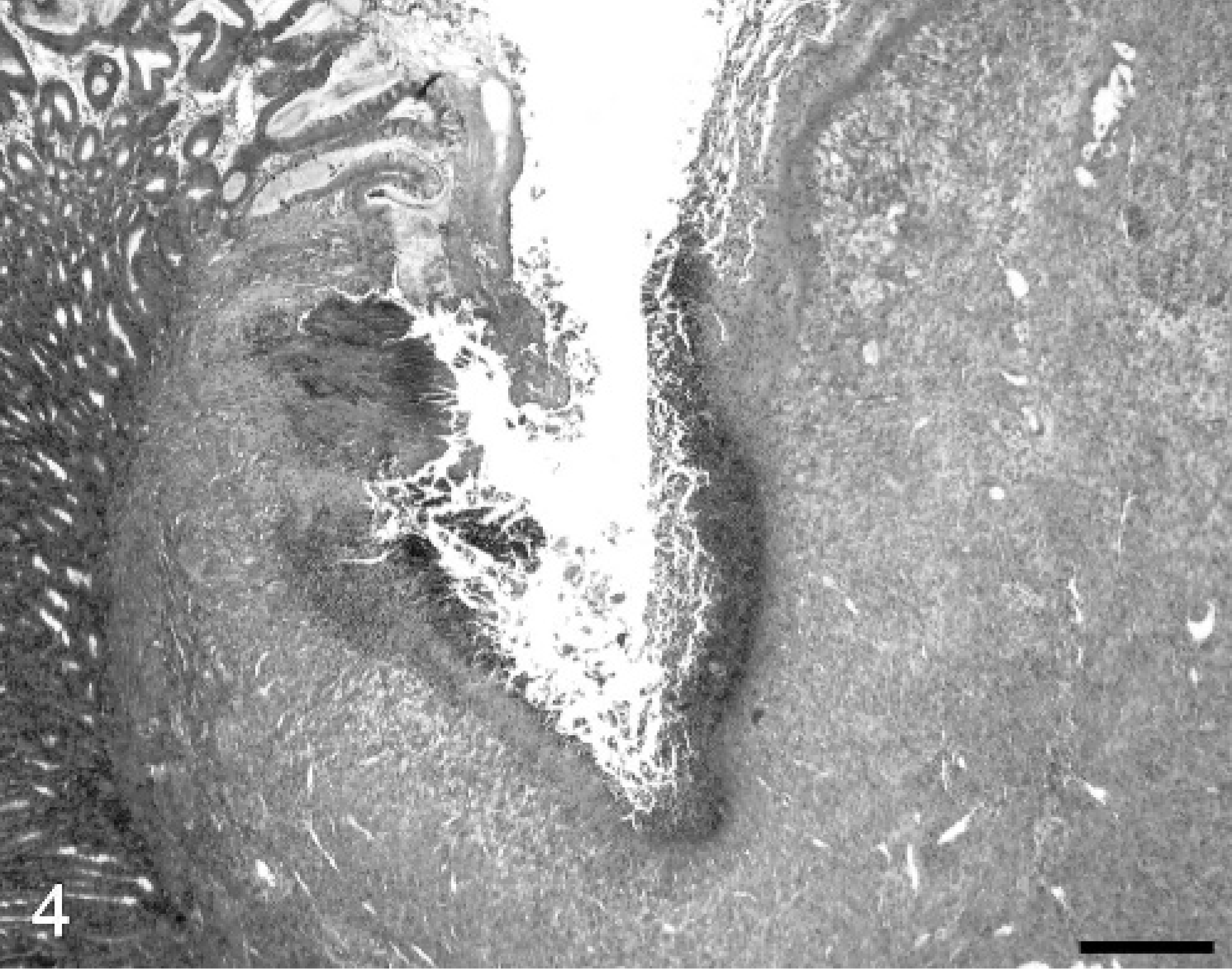
Pars esophagea and cardia; a gnotobiotic piglet infected with Hp-like bacteria and fed a liquid milk diet supplemented with 10% (v/v) sterile corn syrup. HE. Bar = 200 mm.
Microbiology
Four of 6 porcine Hp-like–infected piglets fed supplemental carbohydrate were cultured for quantitative recovery of microbes. Bacterial colony forming units contained in gastric mucosa ranged from 0.14 to 8.16 × 106 cfu/g. In 1 of these piglets, the ulcer crater was dissected free of the surrounding gastric mucosa and cultured separately. The ulcer contained 0.2 × 106 bacterial cfu, whereas the remaining glandular mucosa in that pig yielded 10-fold more organisms (2.94 × 106 cfu/g).
The WS silver stain was used to confirm gastric colonization in all pigs. All 9 Hh-infected swine contained readily detectible microbes of characteristic tightly spiraled morphology typical of Hh. Most of the Hh organisms were detected in the gastric fundus and antrum in extracellular locations associated with the lumenal surface epithelium or within the lumens of gastric pits. In gastric tissue section replicates from piglets infected with Hp-like organisms, the gastric mucosal cardia and antrum, particularly along the lesser curvature, was regularly colonized by short, curved, rod-shaped microbes of typical Helicobacter morphology. Organisms were abundant in the superficial mucus layer and adherent to the gastric glandular mucosa adjacent to the ulcerative lesions in the pars; WS-stained organisms were not detected within the ulcer craters in the pars.
Discussion
In this study, severe ulceration of the porcine pars esophagea was produced in gnotobiotic swine persistently colonized with porcine Hp-like bacteria when a readily fermentable source of dietary carbohydrate (corn syrup) was added to the sterile milk diet. Whereas these lesions were reproduced in young gnotobiotic swine, these experimentally produced GEU appear to be indistinguishable from naturally occurring GEU in older conventional swine. Although mild erosive lesions in the pars are occasionally detected in gnotobiotic piglets colonized with H. pylori, 18 Lactobacillus, or Bacillus and fed a sow milk replacement diet, 19 these lesions are mild, do not progress to ulceration with intragastric hemorrhage, and have never been associated with deaths in the isolation units. Importantly, colonization of corn syrup–supplemented gnotobiotes with Hh here and in a previous study 19 did not produce GEU. These data suggest that gastric colonization by Hh is not involved in the genesis of porcine GEU and that the association between gastric Hh colonization and GEU is coincidental, not causal. 4, 24, 28– 30
This appears not to be the case with a newly identified 19 porcine gastric Hp-like bacterium. Unlike monocolonization with Hh, 19, 22 or H. pylori, 17, 18 gastric colonization with porcine Hp-like organisms is associated with a high incidence of mild GEU (70%) and gastric and duodenal glandular mucosal ulceration (40%) when piglets are maintained on a control milk replacement diet. 22 In the present experiments, ulcers in the gastric glandular mucosa were identified in 2 of 5 porcine Hp-like–infected swine on a control diet. More importantly, however, was the combined influence of persistent gastric colonization with porcine Hp-like bacteria and dietary corn syrup as a source of fermentable carbohydrate upon the severity of GEU. All 6 Hp-like–infected piglets that received the corn syrup–supplemented diet developed severe GEU. One piglet died overnight on PID 30–31 without demonstrating definitive clinical signs of gastric ulceration before death; sudden death is a typical clinical finding in feeder swine GEU. 26 Three other piglets in this group had obvious evidence of intragastric bleeding (partially digested blood in the ulcer crater and adhered to gastric luminal mucus) that had been occurring for at least 1 day before termination as evidenced by the presence of partially digested blood in the small and large bowel.
As indicated elsewhere, 22 the preferred gastric niche for porcine Hp-like bacteria is the lesser curvature of the gastric cardia and antrum. This gastric colonization pattern in gnotobiotes is identical to that seen in H. pylori-infected gnotobiotic swine. 17, 20 The lesser curvature (cardia) is immediately adjacent to the pars esophagea, and it is tempting to attribute the propensity of porcine Hp-like–infected gnotobiotes to develop GEU to this anatomic proximity. Importantly, neither porcine Hp-like bacteria nor H. pylori adhere to stratified squamous epithelium of the pars nor does either organism colonize the intact pars esophagea. For H. pylori, colonization sites appear to be dictated by surface expression of glycoprotein residues, which interact with reciprocal carbohydrate residues (Lewis antigens) in gastric mucus and gastric epithelia. 1, 35 Lewis antigenic arrays in humans are also expressed in swine, and it is possible that the ease whereby gastric Helicobacter species initiate and perpetuate gastric colonization in swine is because of this molecular mimicry phenomenon.
Although this study provides additional experimental evidence for the role of gastric porcine Hp-like bacteria in the production of clinically significant porcine GEU, it does not provide a concise explanation for the actual mechanisms of ulcerogenesis nor do these studies exclude a role for other gastric commensals 19 in the genesis of GEU in older conventional swine. Neither excess dietary carbohydrate nor bacterial infection alone produced severe GEU. Rather, it appears that the combination of a carbohydrate-rich diet along with colonization by swine-origin Hp-like organisms acts synergistically to promote development of severe GEU. In commercial pork production units, diets high in fermentable carbohydrates facilitates development of severe GEU. 26 If the total amount of free acid (hydrogen ions) is the primary molecular determinant for development of GEU, the most likely source of excess hydrogen ions is the metabolic intermediates of complex carbohydrates that are generated by incomplete intragastric glycolysis. Acidic metabolites such as lactic and acetic acids are released during gastric fermentation. 2, 3 These metabolites may be generated by the fermentative capabilities of the Helicobacters or other gastric commensals, 19, 32 the hydrolytic effects of gastric acid, and perhaps even the action of swine-origin saccharidases upon the corn syrup substrate. The members of the Helicobacter genus are not regarded as strongly fermentative microbes, and the fermentative capabilities of the porcine Hp-like isolate are not yet known. It is possible that the actions of excess dietary carbohydrates are indirect and act to promote the overgrowth of Hp-like microbes within the gastric compartment. However, this possibility seems remote in that the number of bacterial cfu recovered from gastric mucosal homogenates was not greater than that recovered from swine fed a control milk diet. 17, 22
Demonstration that gastric microbes, including carbohydrate-fermenting commensals such as Lactobacillus and Bacillus 19 and now porcine-origin gastric Hp-like bacteria, may be involved in the development of GEU has several implications for the management of this disease and bacterial gastritis in conventional swine. Conventional wisdom suggests that the deleterious effects of GEU in feeder swine can be limited by restricting the amount of dietary carbohydrate in the diet and by providing diets to finishing swine in a physical form that maintains the gastric pH gradient within the stomach. 26 Indeed, both manipulations successfully reduce and may eliminate losses caused by GEU in commercial swine operations. If gastric microbes contribute to the pathogenesis of GEU, then perhaps targeted antimicrobial approaches to the control or prevention of gastric infection by gastric commensals and/or Hp-like bacteria may have a role in the control and management of porcine GEU.
Footnotes
Acknowledgements
We acknowledge the excellent technical assistance of Susan Ringler, Judith Dubena, and Amy Davis. Supported in part by USDA formula funds, Capricorn R and D, and grant number 1 R43 A1050028-01, NIH, PHS.
