Abstract
Tissue section replicates from lymphoid tissues and livers of gnotobiotic swine were examined by immunohistochemistry for the colocalization of porcine circovirus-2 (PCV-2) nucleocapsid and terminal deoxynucleotidyl transferase (TdT)-mediated incorporation of biotinylated nucleotides (UTP) onto the 3′-exposed hydroxyl groups (nick end labeling) nuclear deoxyribonucleic acid (TUNEL), a marker for apoptosis. Single- and dually stained replicates from uninfected controls, subclinically affected PCV-2-infected gnotobiotic pigs, PCV-2-infected piglets immunosuppressed with cyclosporine (Cys), and PCV-2-infected piglets with postweaning multisystemic wasting syndrome (PMWS) were evaluated. Thymuses were used as positive controls for apoptosis absent PCV-2, tissue sections from dogs given hyperthermic stress were examined as positive controls for induced TUNEL. Tissues from heat-stressed dogs contained TUNEL-positive cell nuclei in both lymphoid tissues and liver, TUNEL was greatest shortly after the delivery of the hyperthermic insult. In uninfected control and subclinically affected PCV-2-infected gnotobiotic pigs, rare hepatocytes and lymphoid cells were TUNEL positive, the frequency of these was similar to that seen in uninfected controls. In PMWS-affected and Cys-treated PCV-2 piglets, the only consistent strongly positive TUNEL signal was contained within the cytoplasm of virus-positive phagocytic mononuclear cells. In phagocytes, some PCV-2 inclusions were TUNEL positive. Collectively, these data indicate that apoptosis is not the primary mechanism of lymphoid depletion and hepatocyte loss in PMWS. Apoptosis associated with systemic viral diseases may be attributable to pyrexia rather than direct or indirect effects of viruses on target cells.
Postweaning multisystemic wasting syndrome (PMWS) of swine 12,21 is a viral infectious disease of increasing economic importance to the world pork industry. The primary cause of PMWS is infection with porcine circovirus–2 (PCV-2), a small, nonenveloped single-stranded circularized deoxyribonucleic acid (DNA) virus. 1,5,9,13,52 As a DNA virus, a nuclear phase of DNA replication is thought to be essential for the synthesis of viral DNA, and nuclear localization of either PCV-2 DNA or viral nucleocapsid protein 34,35 is used as the indices of the cell sources of new infectious viral progeny in vivo. 33,37 In epithelial cell monolayers, the PCV-2 replication cycle is accomplished without visible viral cytopathic effects. 1,19,48,51 It is likely that PCV-2 is minimally cytopathic in vivo as well because the amount of viral antigen regularly exceeds visual histologic evidence of tissue damage. Swine are the only farm animal species susceptible to PCV-2, and subclinical infection is common. 2,48,53 Reproduction of PMWS with PCV-2 has been accomplished in both conventional 2,4,6,8,22 and gnotobiotic swine. 14,15,30,31 Infection has been accomplished with cloned PCV-2 viral DNA. 17 In gnotobiotic piglets devoid of environmental stimulation and gut microbial flora, infection with PCV-2 results in subclinical infection. 29–31 In contrast, PMWS develops when PCV-2–infected gnotobiotes are coinfected with porcine parvovirus 31 or when infected piglets are parenterally immunostimulated. 30,31 In conventional swine, expression of PMWS is associated with the practice of vaccinations for Mycoplasma sp. pneumonia with adjuvanted biologicals designed to stimulate macrophages. 3,6,7,32
In gnotobiotic swine, liver failure is the proximate cause of death in experimental PMWS. 29–31 Depending on the potentiating cofactor, two forms of liver damage are seen in PCV-2–infected gnotobiotic piglets. In the viral coinfection 31 and immunostimulation 30 PMWS models, liver failure is characterized by hepatocyte depletion and multifocal to diffuse mixed (lymphocytic, plasmacytic and neutrophilic) granulomatous hepatitis. Even though hepatocyte loss is extensive, only rare hepatocytes contain viral nucleocapsid protein. 30 In contrast, gnotobiotic piglets infected with PCV-2 and immunosuppressed with cyclosporine (Cys) develop pantropic infection with prominent viral infection of hepatocytes. 29 Liver failure, although infrequent, as compared with respiratory and gastrointestinal disease, also occurs in conventional PCV-2–infected swine. 1,12,21,43,48 Gross and histologic hepatic lesions vary from focal mononuclear infiltrates in periportal triads and hepatic parenchyma to complete liver plate disorganization with hepatocyte loss, Councilman bodies, granulomatous inflammation, and extensive stromal fibrosis. 43 Unlike immunostimulation-induced PMWS in gnotobiotes, but similar to PMWS-like liver disease in immunosuppressed PCV-2–infected piglets, virus-positive hepatocytes are regular features of liver disease in field cases of PMWS. 1,43 Councilman bodies characteristic of apoptosis 26 are described in field cases of PMWS-associated hepatic disease 43 but have not been seen in experimental PMWS. 29–31
Generalized lymphadenopathy is also a prominent gross finding in swine affected with PMWS. 1,12,47,48 Affected lymph nodes are depleted of both paracortical regions and B-cell germinal centers and replaced by multifocal to diffuse granulomatous inflammation. 1,12,48 Syncytial giant cells are common, and intracytoplasmic viral inclusion bodies in phagocytic cells, when present, are diagnostic for PMWS. 12
Systemic lymphoid depletion has been attributed to apoptosis 49 or to virus infection of lymphocytes. 39 Changes in lymphoid tissues suggest that PMWS pigs are severely immunocompromised. 47,48 Apoptosis is implicated as a mechanism of virus-induced cell death in a number of viral diseases including human immunodeficiency virus, 10 morbilliviruses, including canine distemper virus, 16,20,38 feline immunodeficiency disease virus, 46 and PCV-2. 49 Apoptosis is an attractive explanation for lymphoid depletion because virus infections are known to subvert many cellular functions and it is likely that the apoptotic program is initiated by perturbed cellular functions in virus-infected cells. Apoptosis would facilitate release of new infectious viral progeny, particularly in those infections characterized by latency, persistence, or vertical integration into host cell DNA.
Other events that do not directly involve virus-cell interactions regularly accompany both local and systemic viral infections. These mechanisms, particularly secretion of proinflammatory cytokines, could initiate apoptosis independent of direct viral effects. In a conventional environment, systemic viral infections are also accompanied by pyrexia. 27 The febrile response is completely absent when similar infection experiments are performed within a gnotobiotic environment that lacks commensal microbes, endotoxin, or other bacterial products. 19 In distemper-infected gnotobiotic dogs, normothermia is a consistent feature of disease despite extensive viral infection of gastrointestinal and respiratory epithelia and lymphoid tissues. Similar findings are reported in gnotobiotic cats infected with feline panleukopenia virus. 42 For PCV-2–infected conventional swine, sustained fever of 104 F or greater is a characteristic of clinical disease 21 and rectal temperature elevations are used as a part of a scoring system to quantitate clinical severity of disease. 24 In several unpublished experiments, twice daily rectal temperatures were recorded in groups of PCV-2–infected gnotobiotes. All maintained normothermic values indistinguishable from those of uninfected controls regardless of the stage of the disease process (S. Krakowka, unpublished). Hyperthermic stress is known to initiate the induction of the apoptotic cell death program in vivo 45 and in vitro. 50 Thus, pyrexia, mediated by pyrogenic cytokines or bacterial endotoxins (or both), may also initiate apoptosis independent of cellular events associated with the viral replication cycle. The objective of this study was to determine whether morphologic and immunohistochemical evidence supports the proposition that virus-induced apoptosis is the mechanism responsible for lymphoid depletion and hepatocyte loss in two models of fatal PCV-2–mediated systemic disease in gnotobiotic swine. Our primary assay was the determination of the distribution of single-stranded DNA breaks by terminal deoxynucleotidyl transferase (TdT)–mediated dUTP nick end labeling (TUNEL) immunohistochemistry (IHC) and correlation of these sites to colocalized PCV-2 nucleocapsid protein.
Materials and Methods
Tissue samples
Archived paraffin-embedded blocks of formalin-fixed and cold ethanol–fixed liver, lymph nodes, spleen, and thymus were used as source materials. In brief, tissues from uninfected control gnotobiotic 28 piglets (n = 5); gnotobiotic piglets inoculated with PCV-2 alone (n = 6); PCV-2–infected gnotobiotic piglets immunostimulated 30 by subcutaneous immunizations with keyhole limpet hemocyanin emulsified in incomplete Freund's adjuvant and collected 13–14 days after immunization (n = 4) or at termination (n = 7); and gnotobiotic swine infected with PCV-2 and systemically immunosuppressed with Cys for 7 (n = 2), 14 (n = 2), 27 (n = 1), and 34 days (n = 3) after infection were studied. 29 To determine whether autolysis might induce single-stranded DNA breaks, which react in TUNEL IHC, tissue sections collected from six additional piglets infected with PCV-2 but not preserved by fixation in cold ethanol until 12 hours after death (ambient temperature, 22 C) were also stained with the TUNEL and PCV-2 reagents. Finally, as controls for the TUNEL IHC, absent PCV-2, archived replicate formalin-fixed, paraffin-embedded blocks of thymus, lymphoid tissues including spleen and liver from dogs (n = 8) subjected to controlled hyperthermia (core body temperature elevation to 39.5 C for 1 hour) were also studied. Clinical, immunologic, 25,41 gross, and histologic features of hyperthermia, including induction of heat stress protein 70 in the livers, lymph nodes, and thymi, have been reported. 40,41
Immunohistochemistry
Five-micron paraffin-embedded section replicates were cut and adhered onto glass microslides. After clearing with xylene, sections were rehydrated and then stained singly or in combination for: PCV-2 with a monoclonal antibody specific for the nucleocapsid, 35 a polyclonal rabbit anti-lysozyme (human) antibody, 49 and single-strand DNA breaks in cell nuclei (apoptosis) by TUNEL IHC. For the latter, a commercial TUNEL IHC kit (Trevigen Inc., Gaithersburg, MD 20877) was used according to manufacturer's instructions. As positive controls for apoptosisless PCV-2 infection, canine tissue sections with hyperthermia-induced apoptotic lesions were used. As controls for TUNEL staining in porcine tissueless PCV-2 infection, section replicates of thymus, an organ known for a high rate of apoptosis associated with T-cell maturation, 11 from uninfected gnotobiotic swine were used.
The general strategy for dual antigen staining was as follows. The predicted least prevalent antigen in the section was stained first and then developed with the biotin-conjugated appropriate secondary antiglobulin, streptavidin-horse-radish peroxidase (HRP-ABC) reagent, and chromogen. After quenching residual peroxidase with hydrogen peroxidase, the second antigen was labeled with the appropriate antibody followed by the biotinylated secondary antiglobulin, the ABC, and an HRP chromogen color contrasted to the first chromogen used. Dually stained sections were either lightly counterstained with hematoxylin or methyl green or were unstained, cover slipped, and examined. The chromogens used were: DAB (brown to black chromogen), Vector VIP (purple chromogen), Vector SG (blue chromogen), and Vector Nova Red (red chromogen). Section replicates stained for lysozyme and PCV-2, lysozyme was stained first and developed with Vector Nova Red substrate followed by staining for PCV-2 nucleocapsid developed with Vector VIP. For TUNEL and PCV-2, TUNEL was stained first and developed with excess DAB and then stained for PCV-2 developed with Vector SG. Controls consisted of singly stained and developed replicates with the second HRP substrate added after development with the first chromogen and singly stained replicates developed with each contrasting chromogen alone.
Results
PCV-2 nucleocapsid protein distribution in uninfected control and PCV-2–infected gnotobiotic piglets
Tissues from uninfected control gnotobiotic piglets were negative for PCV-2 nucleocapsid antigen, and infectious virus was not recovered from them. 30 The distribution of PCV-2 antigen in liver and lymphoid tissues in subclinically affected PCV-2–infected gnotobiotic swine has been reported previously. 30 In brief, PCV-2 nucleocapsid protein expression was scarce and restricted to the cytoplasm of mononuclear cells of histiocyte and macrophage morphologies in lymphoid tissues. In liver, occasional Kupffer/endothelial cells contained viral nucleocapsid as did single, widely scattered virus-positive inflammatory cells within mononuclear cell infiltrates in portal triads and parenchyma (Fig. 1).
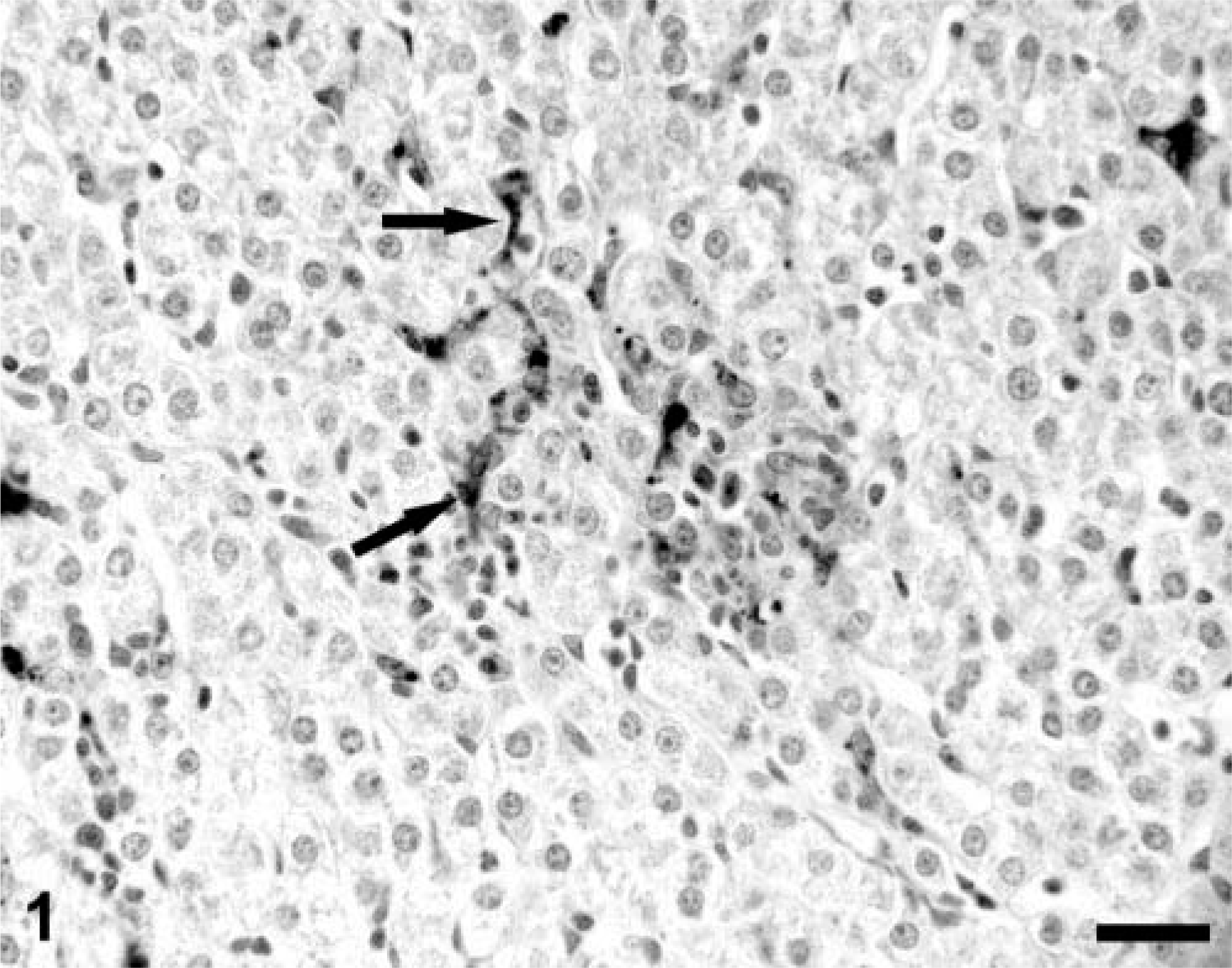
A section of liver from an asymptomatic PCV-2–infected gnotobiotic piglet illustrating focal lymphocytic and histiocytic hepatitis with several virus-positive inflammatory cell(s) in the focus. Kupffer cells (arrows) are also positive for viral nucleocapsid protein. Note that the hepatocytes, even within this infected focus, are PCV-2 nucleocapsid protein–negative. Anti–PCV-2 monoclonal antibody stained with biotinylated equine anti-mouse Ig, reacted with avidin-peroxidase, developed with DAB, and counterstained with hematoxylin. Bar = 115 µm.
PCV-2 nucleocapsid protein distribution in PMWS-affected gnotobiotic piglets
As reported, 30 extensive accumulation of PCV-2 nucleocapsid was seen in liver and lymph node sections from all PMWS piglets. Abundant amounts of viral nucleocapsid were widely distributed within the cytoplasm of cells of histiocytic, follicular dendritic, and phagocytic morphologies (Fig. 2A, B)). The latter often contained lightly basophilic cytoplasmic viral inclusion bodies characteristic of PMWS. In both lymphoid and liver sections, the predominant antigen-positive cell type(s) was macrophages and histiocytes identified by both morphologic appearance and by a positive immunohistochemical stain for cytoplasmic lysozyme. In lymph nodes, virus-positive macrophage-lineage cells were present both within the centers of developing or nascent lymphoid follicles (follicular dendritic cells) and lining medullary sinusoids. Most of the viral nucleocapsid in follicular dendritic cells was confined to the cytoplasm as finely dispersed granules of reaction product. Phagocytic cells lining sinusoids or in developing granulomas frequently contained large discrete nucleocapsid-positive cytoplasmic inclusion bodies. Rarely was nucleocapsid seen in nuclei of either histiocytes or dendritic cells. In PMWS-affected livers, sinusoidal Kupffer cells, possibly some endothelia, infiltrating cells of monocytic morphology, and resident histiocytic cells organized into discrete and coalescing granulomas were all strongly PCV-2 nucleocapsid positive. Viral antigen–positive hepatocytes, even in the most severe PMWS lesions, were rare and, when present, the label was confined to hepatocyte nuclei. Similar to lymph nodes, macrophages actively engaged in phagocytosis contained large cytoplasmic accumulations of virus-reactive product, whereas stellate cells of Kupffer and histiocytic morphologies contained diffusely distributed cytoplasmic PCV-2 nucleocapsid. In both, intranuclear PCV-2 nucleocapsid was uncommon when compared with the frequency of cytoplasmic nucleocapsid.
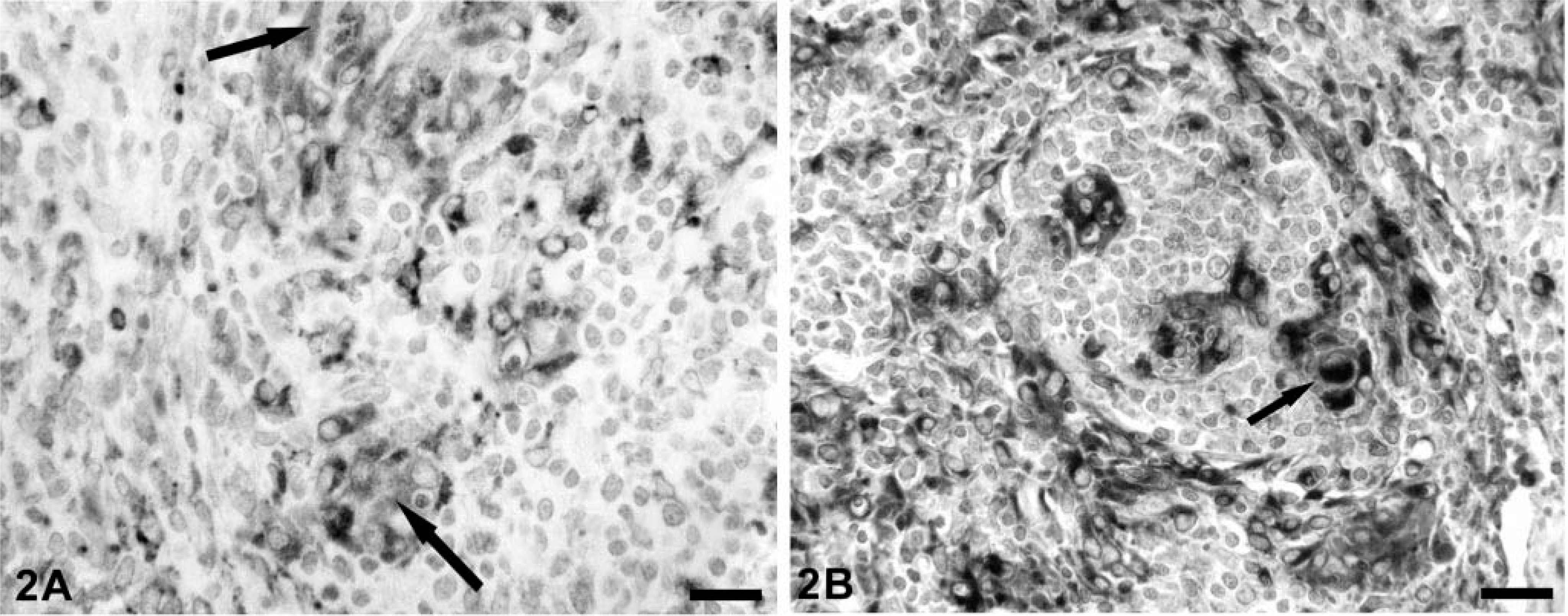
A section of lymph node from an immunostimulated
30
gnotobiotic piglet infected with PCV-2 demonstrating the distribution of PCV-2 nucleocapsid protein in follicular dendritic cells, histiocytes, and macrophages (
PCV-2 nucleocapsid protein distribution in Cys-treated PCV-2–infected gnotobiotic piglets
In lymphoid tissues, the general histologic features and distribution of PCV-2 nucleocapsid protein within the cytoplasm of monocytic cell–lineage cells in Cys-treated piglets were similar to those observed in PMWS pigs above. 30 However, as reported, 29 the distribution of PCV-2 nucleocapsid protein in liver section replicates from asymptomatic Cys-treated pigs was widespread not only in Kupffer and endothelial cells but also in hepatocytes (Fig. 3). Both nuclear and cytoplasmic staining was observed, and staining often occurred in the absence of other overt morphologic changes in virus-positive hepatocytes. In the single, fatally affected Cys-treated piglet (PID 27), hepatocytes were absent or showed severe degenerative-necrotic changes; many of these degenerate hepatocytes were strongly virus positive.
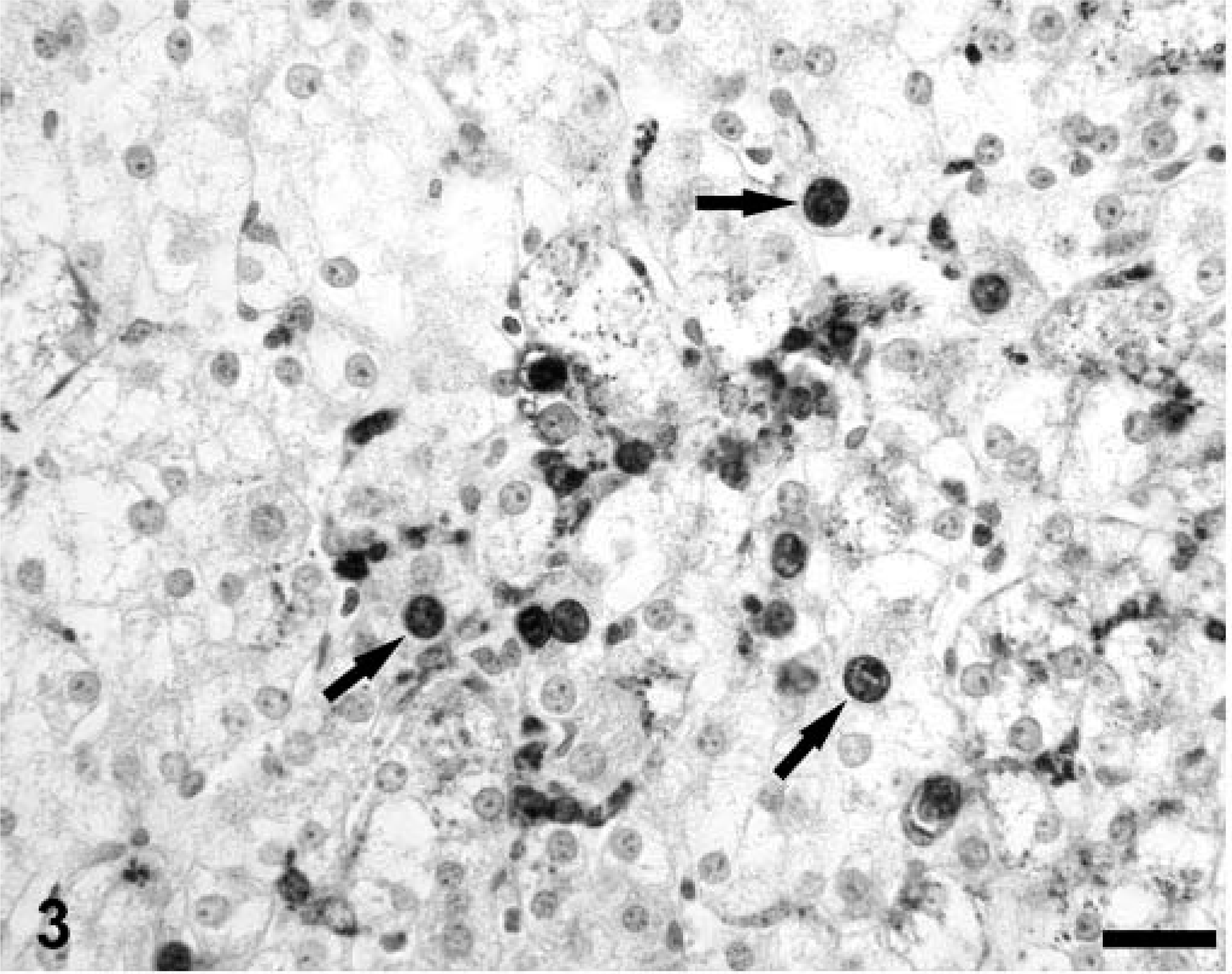
A section of liver from a PCV-2–infected gnotobiotic piglet immunosuppressed 29 with Cys illustrating the cellular distribution of PCV-2 nucleocapsid protein in hepatocyte nuclei (arrows) and cytoplasm. Kupffer cells and possibly endothelia also contain PCV-2 nucleocapsid protein. Anti–PCV-2 monoclonal antibody stained with biotinylated equine anti-mouse Ig, reacted with avidin peroxidase, developed with DAB, and counterstained with hematoxylin. Bar = 125 µm.
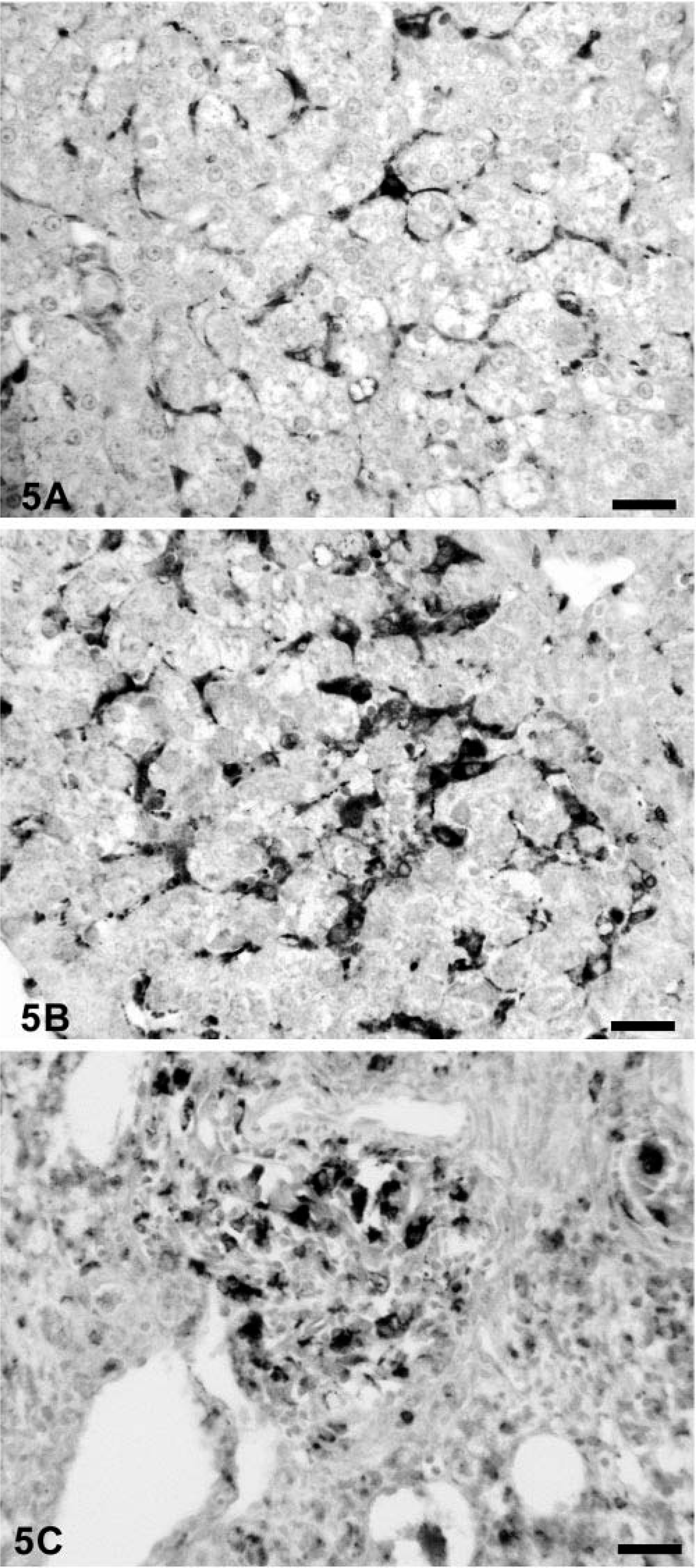
Distribution of cytoplasmic lysozyme in porcine tissues
Rabbit anti-human lysozyme identified histiocytes and macrophages in tissue section replicates of all pigs. In uninfected controls, Kupffer cells, follicular dendritic cells, and sinusoidal phagocytic cells were positive for lysozyme. Granulomas developing around lipid adjuvant droplets in draining regional lymph nodes of immunostimulated pigs 31 were strongly lysozyme positive (Fig. 4). When compared with the staining intensity in controls (Fig. 5A)), Kupffer cells (Fig. 5B)) and single cells within the mononuclear cell infiltrates in hepatic parenchyma of subclinically infected piglets, histiocytes/Kupffer cells were more strongly lysozyme positive. In PMWS-affected pigs, the majority of mononuclear cells within developing granulomas were lysozyme positive (Fig. 5C)). In areas of hepatocyte loss, macrophages assumed phagocytic rather than stellate morphology. In the former, the cytoplasmic lysozyme coalesced into cytoplasmic droplets, presumably phagolysosomes. This staining pattern was readily distinguished from the diffuse distribution of reaction product in inactive Kupffer cells of uninfected control gnotobiotes (Fig. 5A)) and PCV-2–alone infected piglets (Fig. 5B)).
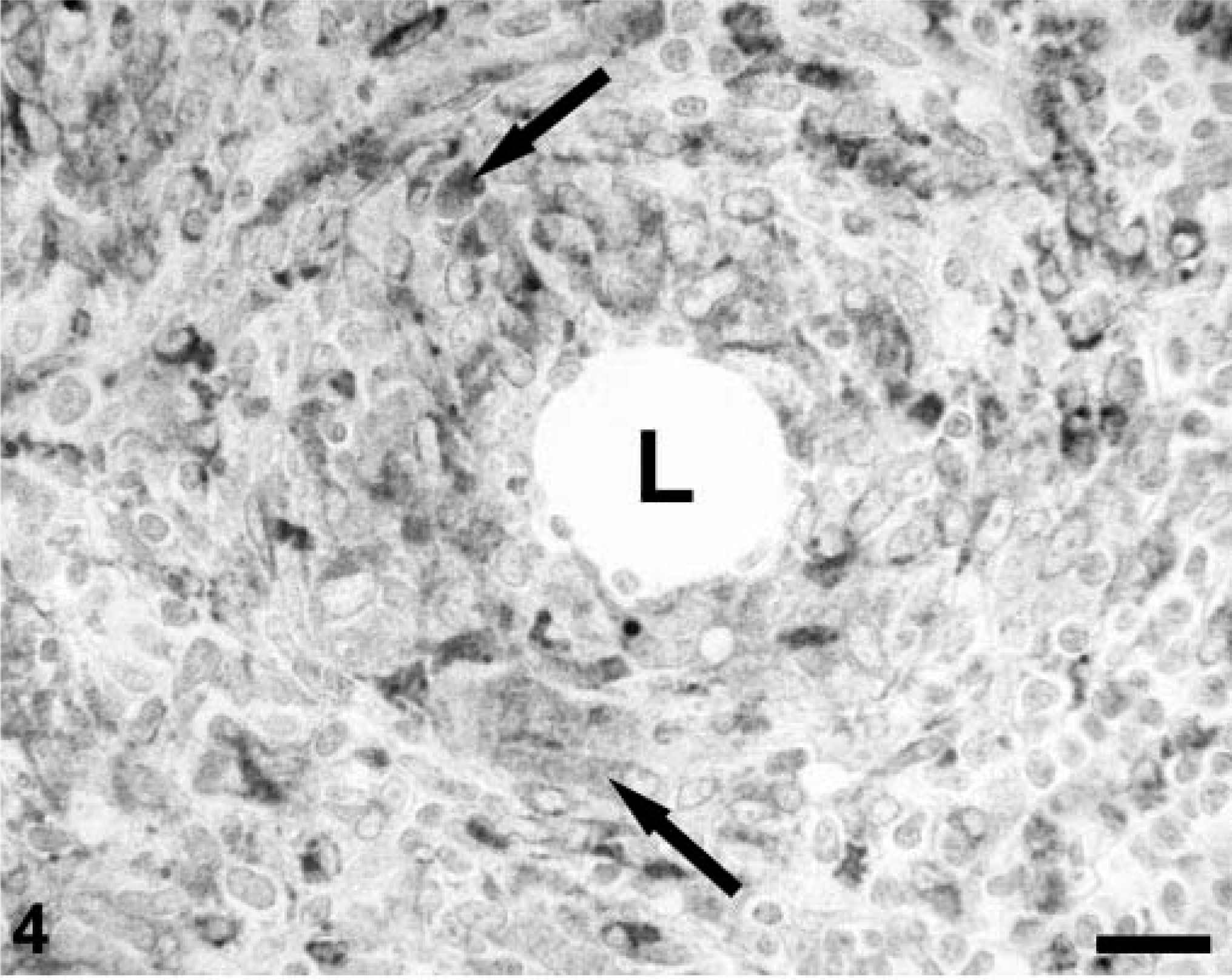
A regional lymph node from a gnotobiotic piglet infected with PCV-2 and immunostimulated as described 30 illustrating the distribution of lysozyme-positive macrophages (arrows) surrounding a lipid adjuvant droplet (L). Polyclonal rabbit anti-human lysozyme stained with biotinylated anti-rabbit Ig, reacted with avidin-peroxidase, developed with DAB, and counterstained with hematoxylin. Bar = 115 µm.
A section of liver from an uninfected gnotobiotic piglet (
TUNEL stains for apoptosis in tissues from heat-stressed dogs and thymuses of uninfected control gnotobiotic piglets
Cell nuclei in the liver and lymph node sections from eight dogs subjected to hyperthermic stress were TUNEL positive at 1 and 4 hours after the hyperthermic insult. TUNEL activity declined sharply as the tissues returned to normal morphologies such that the frequency of TUNEL-positive nuclei in convalescent tissues was no different than euthermic control tissues. Sections of thymus from uninfected control gnotobiotic swine (n = 5) and swine treated with Cys for 7–14 days (n = 4) were used as positive controls for apoptosis and TUNEL in porcine tissues. In both groups, cortical thymic nuclei were TUNEL positive, demonstrating that the TUNEL procedure was compatible with porcine tissues (not shown).
TUNEL stains for apoptosis in tissues from gnotobiotic swine infected with PCV-2
Lymph node sections from asymptomatic gnotobiotic piglets infected with PCV-2 were stained for single-stranded DNA breaks by TUNEL. Other than an occasional single cell of lymphoid morphology, interpreted as “background” apoptosis associated with normal lymphoid tissue turnover and renewal processes, both paracortical T-cell and primordial or atrophic germinal center-associated B-cell areas were TUNEL negative. In liver sections from asymptomatic PCV-2–infected piglets, TUNEL reactivity was rare and restricted to nuclei of hepatocytes and very occasional sinusoidal cells; the rare TUNEL-positive hepatocytes (Fig. 6) were not accompanied by inflammatory cell infiltrates.
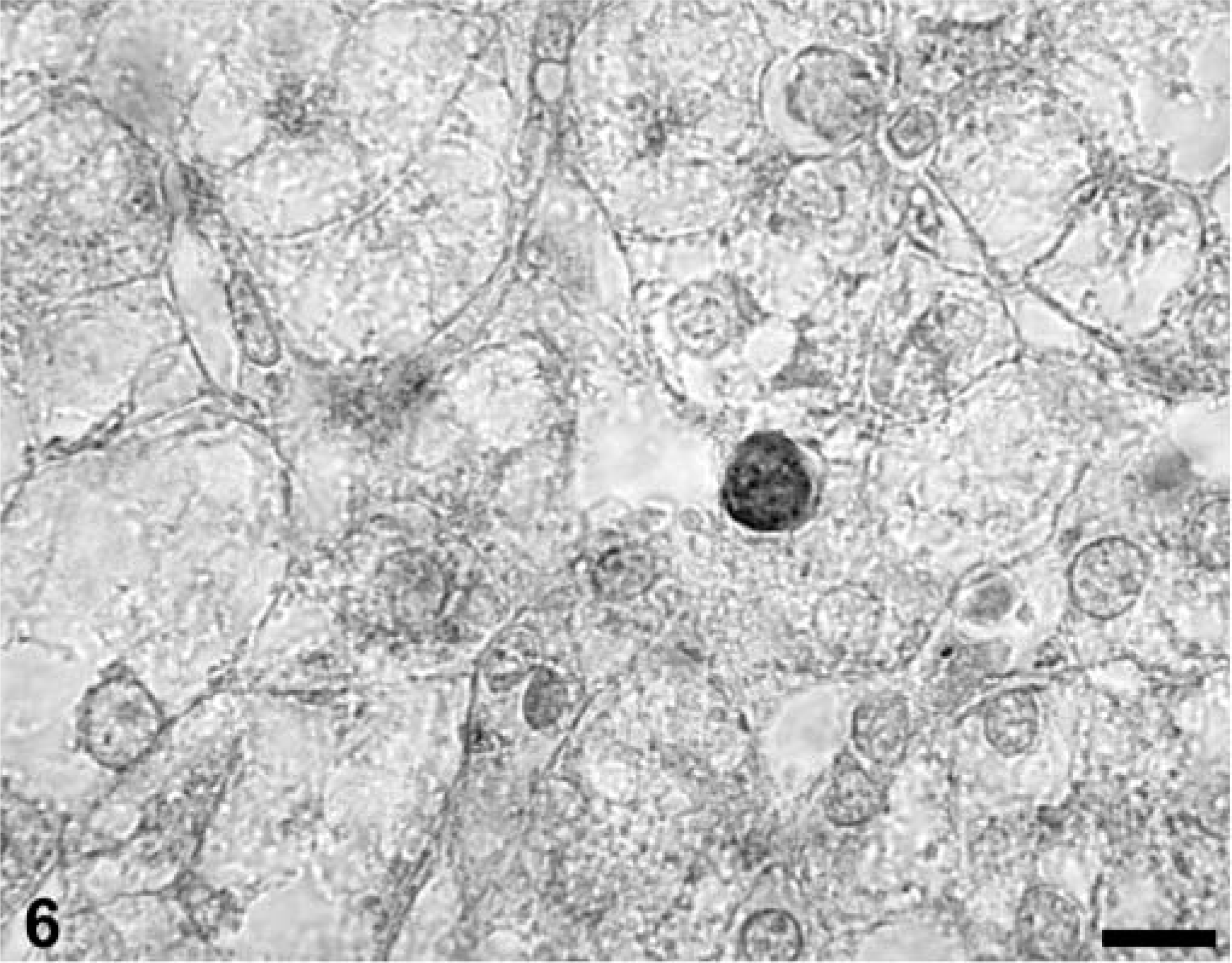
A section of liver from a piglet subclinically infected with PCV-2 illustrating a single TUNEL-positive hepatocyte nucleus. There is an absence of inflammatory cell infiltrates in the surrounding parenchyma. TACS 2 TdT-DAB In Situ Apoptosis Detection Kit (Trevigen Inc., Gaithersburg, MD), methyl green counterstain. Bar = 50 µm.
In lymphoid tissue sections from both PMWS-affected and immunosuppressed PCV-2–infected piglets, the vast majority of lymphocytes within both collapsed germinal centers and the depleted paracortex were TUNEL negative (Fig. 7). Nuclei of mononuclear phagocytes, even those containing cytoplasmic PCV-2 inclusion bodies, were routinely TUNEL negative although some TUNEL-positive cell nuclei were seen within developing granulomas. In contrast, TUNEL-positive cytoplasmic phagolysosomes were prominent in the cytoplasm of phagocytic cells lining the medullary spaces in lymph nodes and spleen. In these cells, viral inclusion bodies, when present, were often TUNEL positive (Fig. 8).
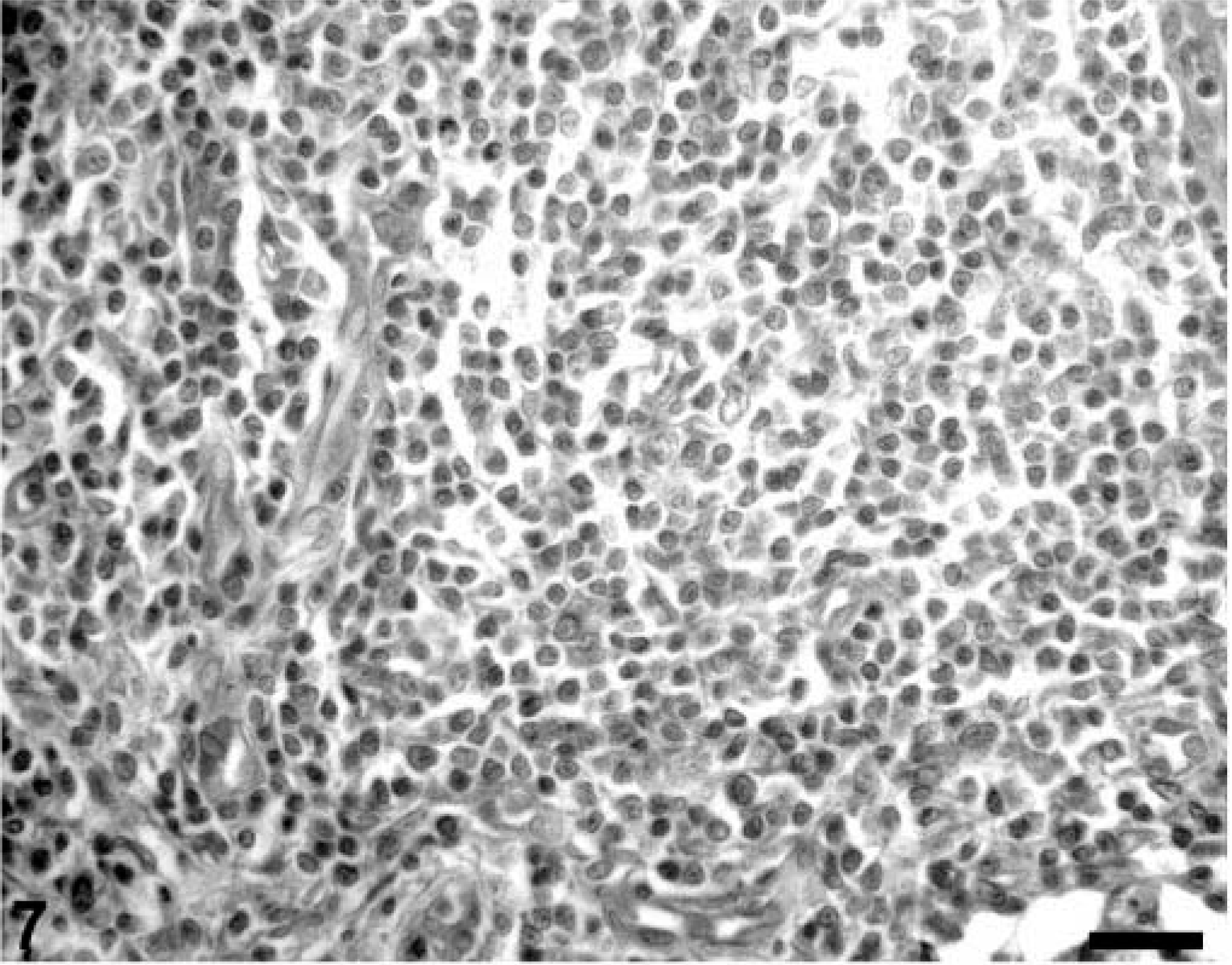
A section of lymph node from a PMWS-affected gnotobiotic piglet illustrating the absence of TUNEL-positive cell nuclei in a T cell–depleted region of that lymph node. TACS 2 TdT-DAB In Situ Apoptosis Detection Kit (Trevigen Inc., Gaithersburg, MD), methyl green counterstain. Bar = 125 µm.
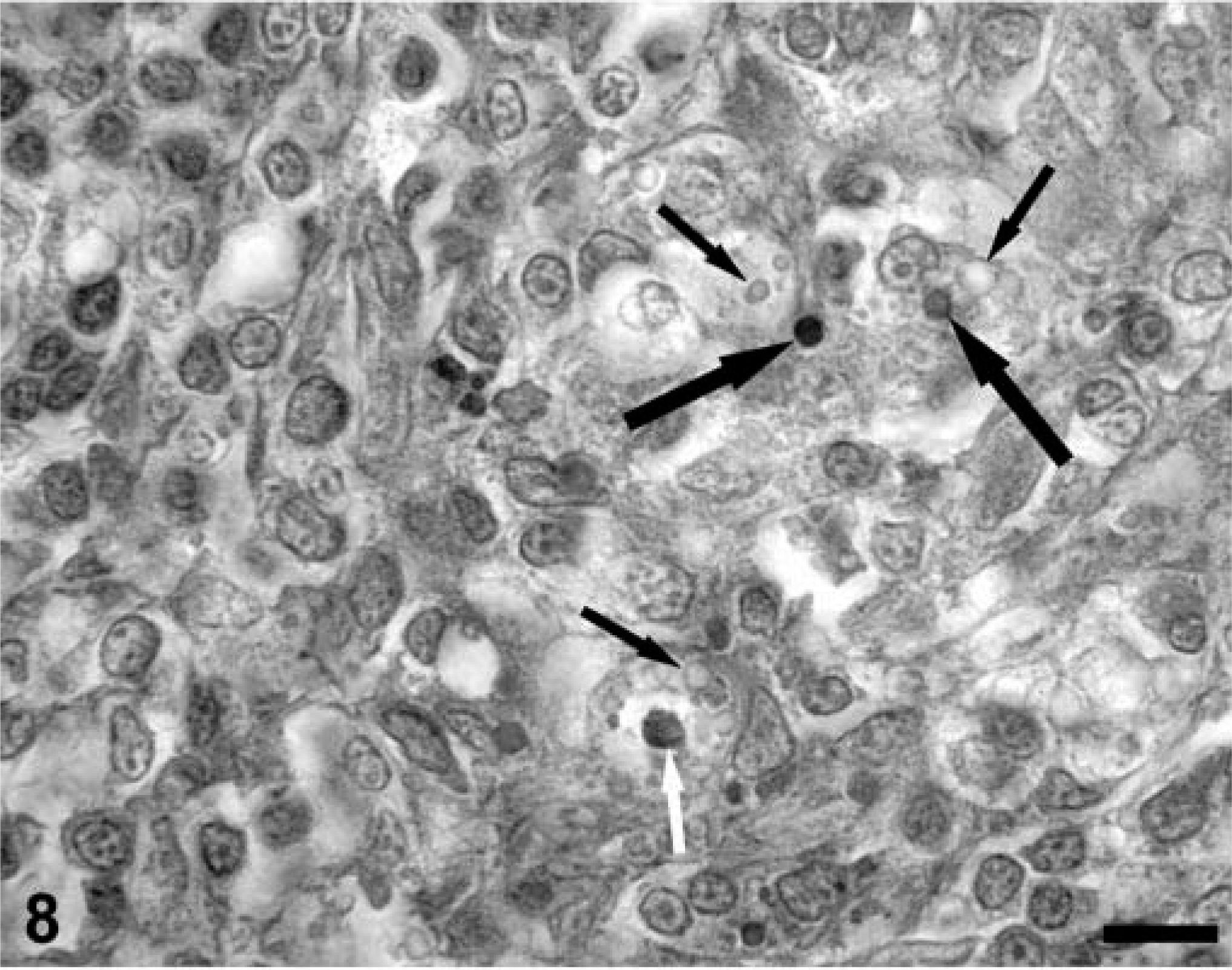
A section of a lymph node from a PMWS-affected gnotobiotic pig containing PCV-2 viral inclusion bodies and TUNEL-positive staining reaction within phagocytic cells. Some viral inclusion bodies stain for TUNEL (large black arrows), whereas other viral inclusions are TUNEL negative (small black arrows). Rare phagocytic cell nuclei are TUNEL positive (white arrow). TACS 2 TdT-DAB In Situ Apoptosis Detection Kit (Trevigen Inc., Gaithersburg, MD), methyl green counterstain. Bar = 125 µm.
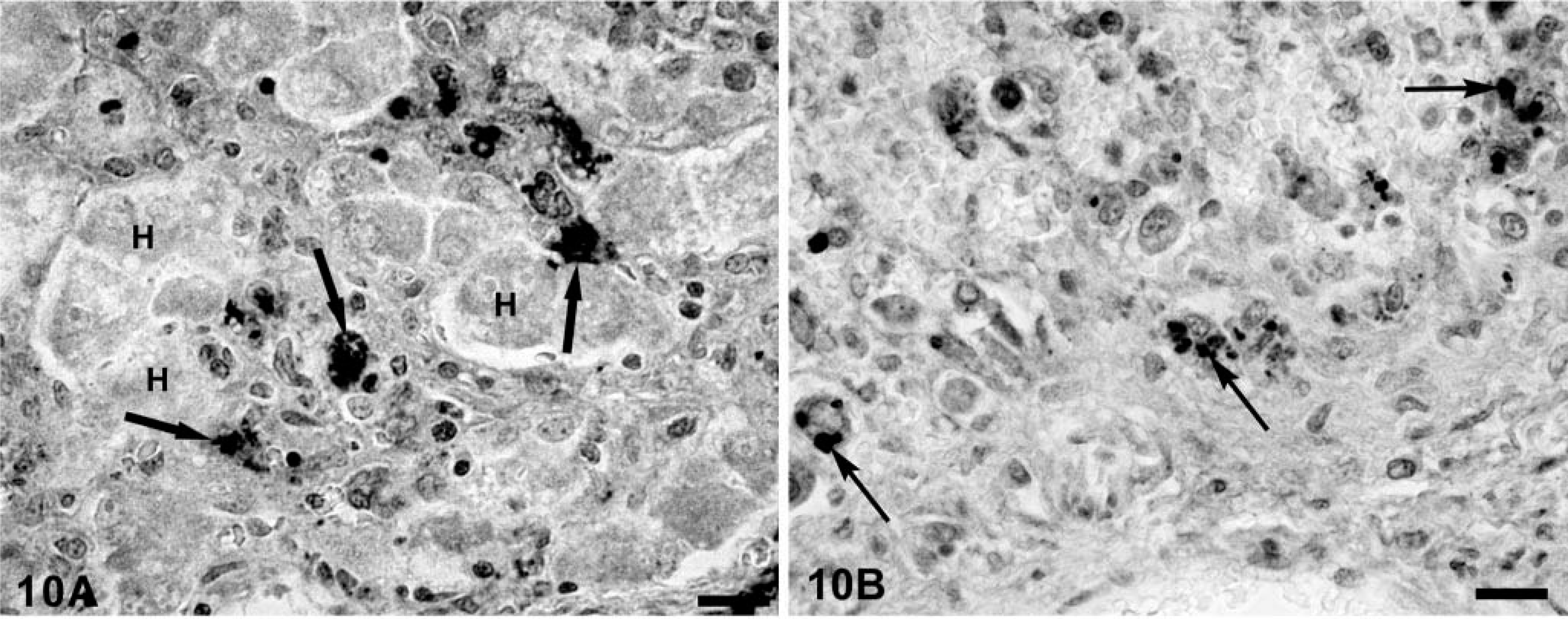
Multiple liver sections from both PMWS-affected and Cys-treated piglets were stained by TUNEL. In PMWS-affected liver sections, obviously damaged hepatocytes were TUNEL negative (Fig. 9), as were the few remaining intact hepatocytes. Notable was the absence of TUNEL-reactive DNA in hepatocyte nuclei from asymptomatic Cys-treated piglets despite the fact that many of these hepatocytes were strongly positive for nuclear and cytoplasmic PCV-2 nucleocapsid protein. In all fatally affected swine, a positive TUNEL signal was evident in the cytoplasm of infiltrating macrophages, closely associated with phagolysosomes (Fig. 10A, B)). Finally, tissue sections from six PCV-2–infected piglets in which preservation in cold ethanol fixative was delayed for 12 hours after death showed histologic evidence of autolytic change (data not shown). In these tissues, numerous cell nuclei of a variety of different cell types were weakly TUNEL positive (Fig. 11).
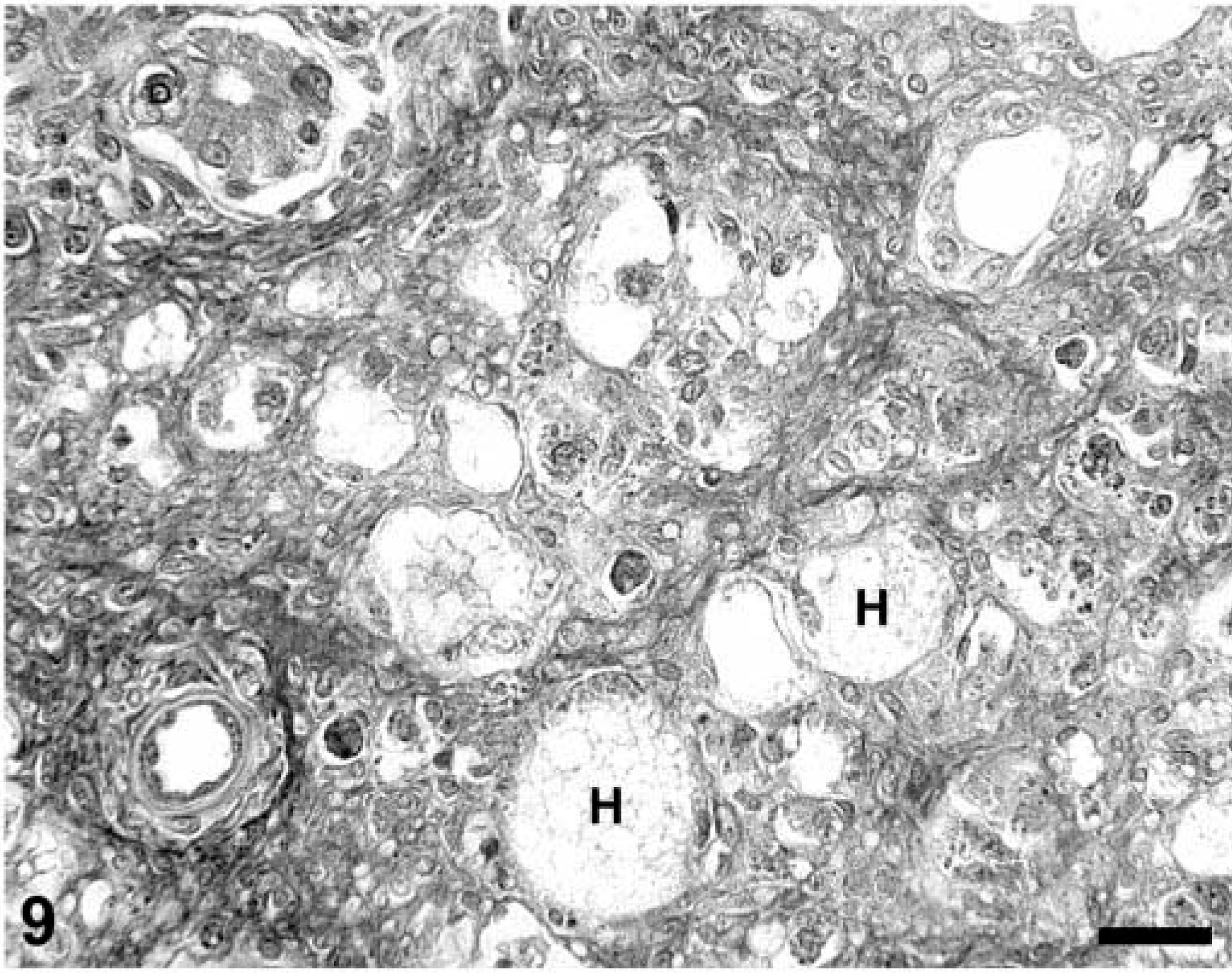
A section of liver from a PCV-2–infected gnotobiotic piglet with immunostimulation-induced 30 PMWS illustrating the absence of TUNEL-positive reaction in dead (H) and dying hepatocytes. Note the lack of intranuclear TUNEL reaction product in the nuclei of degenerate hepatocytes compared with Fig. 6. TACS 2 TdT-DAB In Situ Apoptosis Detection Kit (Trevigen Inc., Gaithersburg, MD), methyl green counterstain. Bar = 125 µm.
A section of liver from gnotobiotic piglets with developing PMWS illustrating TUNEL-positive cytoplasmic reaction product within macrophages, histiocytes, and Kupffer cells (arrows). Degenerating hepatocytes (H) are TUNEL negative (
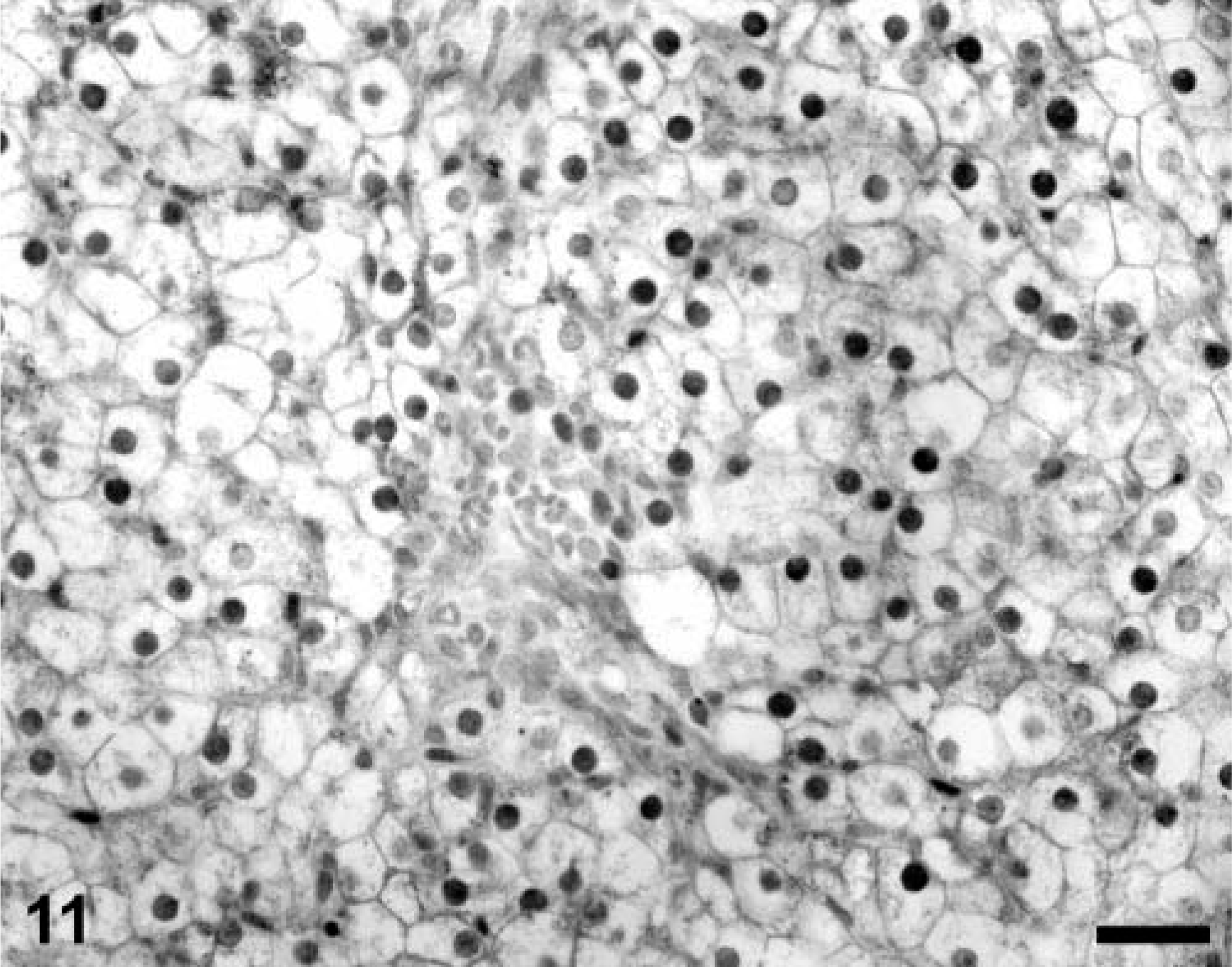
A section of autolytic liver (tissues preserved in fixative 12 hours after death) from a PCV-2–infected gnotobiotic piglet illustrating numerous weakly TUNEL-positive hepatocyte nuclei. Mild autolytic changes (cell swelling and rarefaction of cytoplasmic detail in hepatocytes) are consistent with the delay in tissue preservation by formalin fixation. TACS 2 TdT-DAB In Situ Apoptosis Detection Kit (Trevigen Inc., Gaithersburg, MD), methyl green counterstain. Bar = 125 µm.
Colocalization of PCV-2, lysozyme, and TUNEL in asymptomatic PCV-2–infected gnotobiotic piglets
In tissue sections from subclinically infected piglets, dual staining of liver and lymph node sections for lysozyme and PCV-2 confirmed that most of the cell-associated PCV-2 nucleocapsid protein was contained within lysozyme-positive cells. Section replicates stained for colocalization of PCV-2 with TUNEL demonstrated uncommon cytoplasmic accumulations of TUNEL in phagolysosomes of macrophages that also contained PCV-2 nucleocapsid. A few inflammatory cells infiltrating into hepatic parenchyma contained nuclear TUNEL reaction product, but these cells were PCV-2 negative. Hepatocytes in asymptomatic piglets treated with Cys were PCV-2 positive but TUNEL negative.
Colocalization of PCV-2, lysozyme, and TUNEL in PMWS-affected gnotobiotic piglets
In PMWS-affected lymphoid tissues, two different staining patterns for PCV-2 nucleocapsid were discerned. Follicular dendritic cells and histiocytes were lysozyme positive, TUNEL negative, and characteristically contained diffusely distributed cytoplasmic PCV-2 nucleocapsid protein. In sinusoidal phagocytic cells and granulomas, PCV-2 inclusion bodies stained strongly for viral nucleocapsid. Viral inclusions were also variably TUNEL positive (Fig. 8). In phagocytes, viral nucleocapsid colocalized with lysozyme-positive cytoplasmic organelles (phagolysosomes) indicating that these cells were of macrophage lineage. For both histiocytes/follicular dendritic cells and macrophages, significant TUNEL activity was restricted to the cytoplasm, which colocalized with both viral nucleocapsid and lysozyme as cytoplasmic inclusions. Although occasional phagocytic cell nuclei were TUNEL positive, this was never a dominant feature of the TUNEL distribution in PMWS-affected lymph nodes.
In liver tissue replicates from PMWS piglets, the amount and frequency of TUNEL reaction product that colocalized with viral nucleocapsid varied with the degree of hepatocyte loss in affected lobules. In severe lesions, the majority of hepatocytes were no longer structurally discernable. The few remaining hepatocytes were enlarged, contained centrally located nuclei, and were both PCV-2 nucleocapsid- and TUNEL negative. Ghost spaces formerly containing hepatocytes were frequently surrounded by phagocytic cells with cytoplasmic aggregates of viral nucleocapsid protein, TUNEL product, and lysozyme. In less severely affected livers, many swollen but histologically identifiable hepatocytes were still present. These hepatic plates were variably infiltrated with lysozyme- and viral nucleocapsid protein–positive granulomatous inflammatory cell infiltrates. Although occasional intra-nuclear TUNEL-positive inflammatory cells were identified in portal triads and inflammatory cell infiltrates of PMWS-affected animals, virtually all the TUNEL-positive product colocalized with PCV-2 nucleocapsid protein to the cytoplasm of macrophages and histiocytes within phagosomes (Fig. 10A, B)). In Cys-immunosuppressed piglets, virus-positive hepatocytes, even hepatocytes strongly positive for intranuclear PCV-2 nucleocapsid, were routinely TUNEL negative. As in PMWS livers, the liver section of the single, fatally affected Cys-treated piglet contained numerous TUNEL-positive foci within the cytoplasm of infiltrating macrophages, suggesting that phagosomes of these cells contained viral nucleocapsid and DNA fragments of both cellular and viral origin.
Discussion
This study was performed to discern the site(s) of viral nucleocapsid protein accumulation in PCV-2–infected gnotobiotic piglets and to test the hypothesis that apoptosis, induced by the direct cellular effects of virus infection, viral DNA, or viral proteins, accounts for systemic lymphoid depletion and hepatocyte loss characteristic of experimental PMWS 30,31 and hepatocyte death in immunosuppressed PCV-2–infected gnotobiotic piglets. 29 Section replicates from archived tissue blocks were used. At necropsy, these tissues were immediately preserved in either 10% phosphate-buffered formalin or cold (4 C) absolute ethanol to minimize effects of autolysis on the interpretation of morphologic changes. As documented elsewhere, 29–31 section replicates exhibited a spectrum of histologic lesions, from asymptomatic disease to those recognized as diagnostic for PMWS. 1,12,48 The gnotobiotic environment facilitated profound experimental immunosuppression with Cys, 29 a potent inhibitor of the inductive phases of the immune response. 23,54 The oral Cys doses used in these piglets (50.0 and 25.0 mg/kg) 29 were above those recommended for use in a conventional environment (10.0 to 15.0 mg/kg) where the risks of fatal opportunistic infections as a result of Cys-induced immunosuppression are high.
Both the immunostimulation 30,31 and immunosuppression 29 methods of reproducing clinically evident PCV-2–associated disease in gnotobiotic pigs are characterized by extensive hepatocellular damage evident as hepatic atrophy, icterus, hypoproteinemia, generalized edema, and terminal hemostatic defects. However, these two forms of liver failure differ from each other in that the immunostimulation model is characterized by progressive, mixed granulomatous hepatitis with PCV-2 restricted almost exclusively to infiltrating monocytic-lineage cells, Kupffer cells, and possibly sinusoidal endothelia, 30,31 whereas histologic changes in the liver sections from Cys-immunosuppressed pigs, although still characterized by infiltrating virus-positive histiocytes and phagocytes, lack associated infiltrating lymphocytes, plasma cells, neutrophils, and eosinophils. Moreover in Cys-treated swine, viral nucle-ocapsid protein is abundant in both hepatocyte nuclei and cytoplasm. 29 Accumulation of PCV-2 nucleocapsid in hepatocytes of immunosuppressed piglets is dramatic and closely resembles the distribution of viral DNA by in situ hybridization (ISH) in the more severe forms of PCV-associated hepatitis seen in conventional swine. 43
One explanation for the nucleocapsid protein–negative hepatocytes in either immunostimulated (PMWS affected) or asymptomatic PCV-2–infected gnotobiotes is that hepatocytes are virus positive during the pre-clinical stage of disease but immune and inflammatory responses clear released virus and kill virus-infected cells as the disease develops. This possibility was considered but discarded because we have examined many livers (and other tissues) from PCV-2–infected and immunostimulated gnotobiotic piglets before development of overt clinical signs of PMWS and could not document even a transient “hepatic phase” of infection (S. Krakowka, unpublished). It is also possible that hepatocyte infection may be accomplished directly by unencapsidated viral DNAs that are present in extremely high DNA copy numbers in PMWS liver homogenates. 36 This phenomenon may be a regular but undetected event during the evolution of PMWS, particularly if transcription and translation of virion proteins are truncated in hepatocytes such that they would be viral DNA–positive by ISH 43 yet PCV-2 nucleocapsid protein–negative by IHC. 30 This possibility seems unlikely in light of data that show that hepatocytes express both viral proteins if PCV-2–infected pigs are immunosuppressed with Cys. 29
If productive or even nonproductive viral infection of hepatocytes cannot be implicated as a direct mechanism for hepatocellular degeneration and death in the experimental immunostimulation model of PCV-2–associated PMWS, can other mechanisms be incriminated in the pathogenesis of liver injury? Although functional data are not yet available, the regular presence of inflammatory cell infiltrates in PMWS-affected livers suggests that proinflammatory mediators such as free oxygen and nitrogen radicals or cytokines like interleukin-1 and tumor necrosis factor produced and secreted by inflammatory cells could account for hepatocyte loss in PMWS. In fact, the absence of inflammatory cell infiltrates in the Cys-treated piglets and the lack of histologic indicators of hepatocyte injury even though viral protein is widely distributed within hepatocytes suggest that the inflammatory response characteristic of PMWS is central to hepatocellular injury. Another mechanism for hepatocyte destruction is that the apoptotic program is initiated and this phenomenon accounts for both hepatocyte loss and systemic lymphoid depletion characteristic of PMWS. The TUNEL IHC method detects single-strand DNA breaks in nuclear DNA (a hallmark of apoptosis) and is commonly used to distinguish between programmed cell death by apoptosis and cell death by misadventure (necrosis). As an index of apoptosis, TUNEL is relative insensitive as compared with biochemical measures of apoptosis such as DNA laddering or expression and upregulation of cellular capsases (S. Alldinger, personal communication). 11 Despite this, TUNEL IHC is useful in identifying apoptotic foci in histologic sections. Apoptosis is frequently implicated as a mechanism whereby viruses may cause cell death in the absence of direct virus-associated cytopathic effects. The latter is a characteristic feature of the PCV-2 viral replication cycle. 1,51 Indeed, apoptosis, as detected by TUNEL, has been demonstrated in lymphoid tissues from field cases of PMWS-affected swine collected at necropsy. 49 Induction of apoptosis directly by PCV-2 could readily account for systemic lymphoid depletion and hepatocyte loss characteristic of PMWS.
Contrary to expectations, 49 TUNEL IHC assays applied to PMWS lymph node section replicates did not identify increased intranuclear TUNEL-positive B or T lymphocytes in areas of lymphoid depletion above background levels observed in uninfected control or in subclinically infected piglets. Rather, TUNEL reaction product, if present, was found within and largely restricted to the cytoplasm of phagocytic mononuclear series cells in subcellular cytoplasmic structures compatible in size and location to phagolysosomes. In PMWS-affected livers, TUNEL-specific reaction product was rarely identified in hepatocytes, even in those hepatocytes severely damaged by the disease process. The histologic features of hepatocyte damage in PMWS (cellular swelling, decreased cytoplasmic acidiphillia, and enlarged pleomorphic nuclei) are also not compatible with the morphology of apoptosis as is commonly recognized in tissue sections. 11 Were apoptotic mechanisms independent of direct viral infection involved in the genesis of the hepatocyte loss, TUNEL-positive hepatocytes should have been identified in less severely affected liver section replicates and also in virus-positive liver sections from Cys-treated piglets; they were not. Rather, TUNEL was almost exclusively detected within the cytoplasm of macrophages and histiocytes of obvious active phagocytic morphology. Thus, TUNEL-positive phagolysosomes in macrophages within and adjacent to areas of extensive hepatocyte destruction suggest that phagocytic cells are actively involved in the removal of dead and dying hepatic parenchyma and that the source of the free DNAs for end-labeling with the TUNEL procedure was ingested and fragmented nuclear or viral DNAs (or both).
If induction of apoptosis by a direct viral effect(s) cannot be implicated in either experimental model of fatal PCV-2–associated disease, are there other explanations for the presence of TUNEL-positive cell nuclei indicative of apoptosis 49 in field cases of PMWS? A trivial technical explanation for false-positive single-stranded DNA breaks in tissues is autolysis. Although not strongly positive, weakly staining intranuclear TUNEL reaction product was observed in both lymphocytes and hepatocytes in sections prepared from mildly autolytic porcine tissues. These findings indicate that autolytic processes may result in occasional false-positive reactions with TUNEL IHC and emphasize the need for selection of proper controls for TUNEL immonohistochemical stains. Thus, the degree of autolysis may, in part, explain false-positive TUNEL reactions generally, and such an effect may be a caveat to the interpretation of TUNEL activity reported in diagnostic specimens 49 in which mild or moderate autolysis often coexists with PMWS lesions. More important, however, is the effect of generalized pyrexia, an indirect consequence of many systemic viral infections including PCV-2, on apoptotic processes in vivo. In conventional swine with overt or developing PMWS, persistently elevated body core temperature is a consistent clinical feature of disease. In fact, persistent fever is one element used in clinical scoring of the severity of disease in experimentally induced PMWS in conventional swine. 24 Although the physiologic basis for fever has not been formally determined in PMWS-affected swine, there is no reason to suspect that the febrile response in PMWS-affected swine differs from that observed in other viral infectious diseases. Virtually all systemic virus infections are associated with pyrexia of variable duration and severity. Pyrogenic signals responsible for fever are well known and consist of the proinflammatory cytokines and numerous bacterial products including bacterial endotoxins absorbed from the gut. It has long been known that experimental viral infections, when performed within a gnotobiotic environment, are accompanied by a truncated febrile response. 18 There is a general consensus that the pyrexia of viral infections is mediated by products of bacterial commensal organisms that reside in the gut and elsewhere.
Thermal stress or heat shock is an excellent model for the controlled induction of the pyrexia of infection, and this laboratory has developed techniques to deliver controlled sublethal hyperthermic insult to core body canine tissues using an extracorporeal perfusion apparatus. 41 The immunologic, hematologic, and physiologic consequences of hyperthermia in a canine model are described in detail elsewhere. 25,40,41 In this study, archived canine tissue blocks, collected at various time intervals after 1 hour thermal stress insult, were used as positive controls for heat stress–induced apoptosis and TUNEL IHC, absent PCV-2 infection. Previously published data unequivocally demonstrate that even this brief elevation in core body temperature induces heat shock protein 70 synthesis in target tissues 41 and is sufficient to initiate the apoptotic program in heat-stressed liver and lymph nodes. Thus, the positive TUNEL reaction in heat-stressed tissues and the TUNEL-negative data generated in PMWS-affected gnotobiotic swine strongly suggest that apoptotic phenomena documented in conventional PMWS-affected pigs are more likely mediated by elevated core body temperatures (pyrexia) in PMWS-affected swine and are not the direct effects of the viral infection itself. In light of these findings, perhaps the contributions of apoptosis to the pathogenesis of other viral diseases is mediated by the indirect effects of systemic infection (fever) and not by direct effects of the virus on infected cells themselves.
Footnotes
Acknowledgements
This research was supported by research grants from The National Pork Producers Council, Ames, IA, and The Ohio Animal Health Foundation. G. Allan, B. Meehan, and F. McNeilly were supported by a European Union Project QLK2-CT-1999-00445. In Canada, J. Ellis was supported by a grant from the Canadian Natural Sciences and Engineering Research Council Collaborative Research Opportunities 234281-00. M. Oglesbee was supported by grant RO1 NS31693, NIH, PHS. The excellent technical assistance of Susan S. Ringler, Judith Younger, and Michelle Newkirk is appreciated.
