Abstract
Rare cases of thymic granulomatous lesions were found in pigs. The lesions consisted of epithelioid cells, multinucleated giant cells, and lymphocytes. Such lesions also were observed in the mesenteric lymph nodes, spleen, kidney, and stomach. The cytoplasm of the majority of giant cells and some epithlioid cells was periodic acid-Schiff (PAS) positive. All cells were positive for vimentin, lysozyme, and desmin. Ultrastructurally, the giant cells were rich in organella and attached to adjacent epithelioid cells by membrane interdigitation. The cells included numerous coated vesicles and granules. No etiologic pathogen, including porcine circovirus type 2, was detected in the lesions. This is the rare case of idiopathic thymic granulomatous lesion in pigs.
Systemic granulomatous lesions in pigs have been reported in cases of mycobacterial infection, 7, 9 brucellosis, 3 mucormycosis, 10 and postweaning multisystemic wasting syndrome (PMWS). 2, 4, 6, 8 The lesions were observed mainly in the lymph nodes, 2– 4, 6– 9 tonsils, 2, 6, 7 liver, 2, 3 spleen, 2, 4, 6 Peyer patches, 2, 6 kidneys, 4 reproductive organs, 3 bone, 3 stomach, 10 and heart. 4 In the present report we describe systemic granulomatous lesions in pigs that were much more severe in the thymus. Although there are a few articles describing thymic granuloma in PMWS pigs, 2, 6 spontaneous thymic granulomatous lesions of other etiologies have never been reported in pigs.
The lesions were found by chance in pigs when a toxicologic experiment was carried out using T-2 toxin. Three 42-day-old female pigs from a litter (father: Large Yorkshire × Dutch Landrace; mother: Landrace × Large Yorkshire) were subcutaneously injected with T-2 toxin (0.3 mg/kg) dissolved in dimethyl sulfoxide (DMSO). Two pigs from the same litter were injected with DMSO as a control.
The pigs were euthanatized at 24 hours after injection. At autopsy no gross lesions were observed in either T-2–treated or control animals. Tissues were fixed in 10% neutral buffered formalin, embedded in paraffin, sectioned at 4 μm, and stained with hematoxylin and eosin (HE). Some sections were stained with periodic acid–Schiff (PAS), Gram's stain, Gomori methenamine silver, and Ziehl-Neelsen.
Paraffin sections also were subjected to immunohistochemistry using the labeled streptavidin biotin (LSAB) method. Primary antibodies used were anti-cow cytokeratin, anti-chicken desmin, anti-cow vimentin, anti-human smooth muscle actin, anti-human lysozyme, and anti-rabbit alpha-sarcomeric actin (DAKO, Kyoto, Japan). Biotinylated goat anti-rabbit immunoglobulin G (IgG) antibody or anti-mouse IgG antibody (Kirkegaard Perry Laboratories, Gaithersburg, Maryland, USA) was used as a secondary antibody. To detect porcine circovirus type 2 (PCV-2), biotinylated PCV-2 antibody (a gift from Dr. Kawashima, National Institute of Animal Health, Japan) was used. Positive signals were visualized by treating the sections with 3-3′-diaminobenzidine tetrahydrochloride.
For electron microscopic analysis, small pieces of formalin-fixed tissues of the thymus and mesenteric lymph nodes were refixed in 1% osmium tetroxide and embedded in epoxy resin. Ultrathin sections were double-stained with uranyl acetate and lead citrate and were examined using a transmission electron microscope (JEM-1200EX; JEOL, Tokyo, Japan).
In addition, polymerase chain reaction (PCR) for detecting PCV-2 was carried out. DNAs were extracted from the paraffin sections of thymus and lymph nodes according to the procedure by Kim and Chae. 5 The protocol for PCR, including primer setting and cycles, was set according to the report by Kawashima et al. 4 The positive template used in PCR was prepared from PCV-2–infected cells (a gift from Dr. Kawashima).
Histologically, granulomatous lesions were observed in various organs of both T-2–treated and control animals. Lesion distribution is shown in Table 1. The lesions consisted of epithelioid cells, multinucleated giant cells, and lymphocytes. The most severe lesions were observed in the thymus. The thymic cortex was markedly atrophic, and the granulomatous lesions were spread in the medulla (Fig. 1). The same lesions were found in the mesenteric lymph nodes. On the other hand, lesions were localized in parts of the spleen, kidneys, and stomach. The giant cells possessed from several to more than 10 nuclei and abundant cytoplasm (Fig. 2). The epithelioid cells were round or oval in shape and had 1 or 2 nucleoli. The cytoplasm of all multinucleated giant cells and some epithelioid cells was PAS positive. No causative pathogens were detected in the lesions by various stainings such as Gram's, Gomori methenamine silver, and Ziehl-Neelsen.
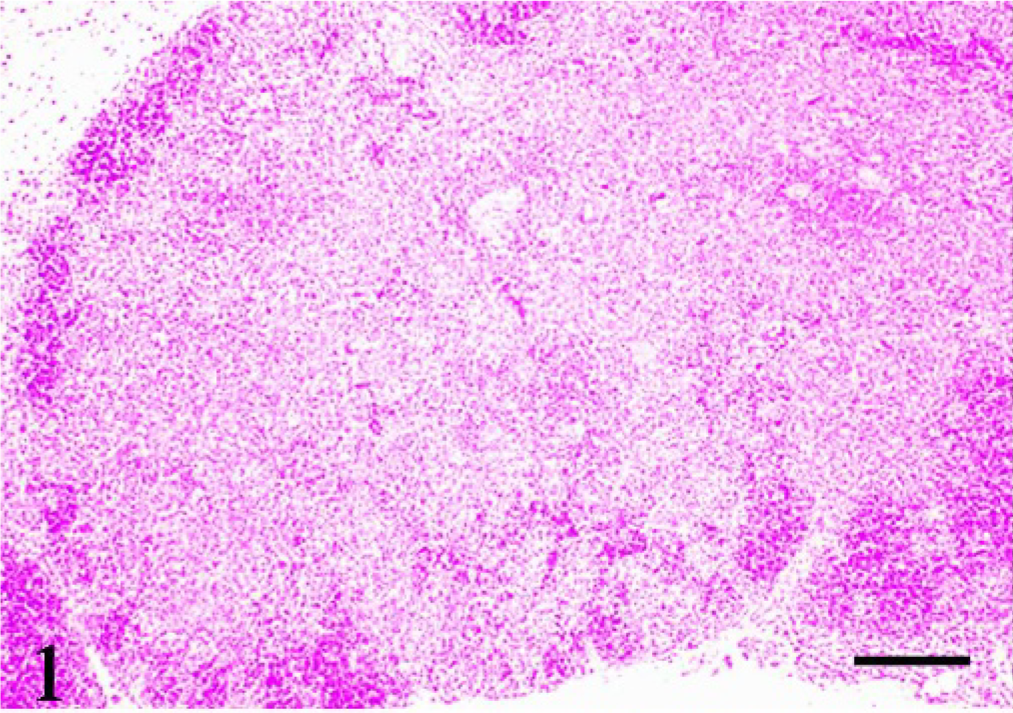
Thymus; pig No. 2. Marked atrophy of the cortex and expanded granulomatous lesion in the medulla. HE. Bar = 100 μm.
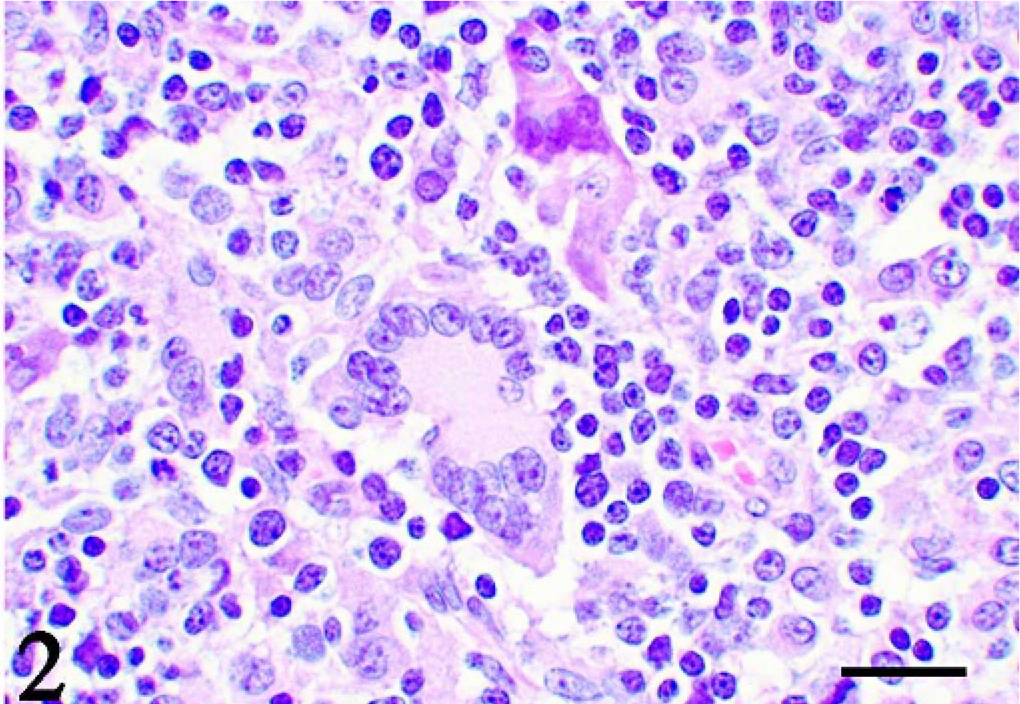
Thymus; pig No. 2. Granulomatous lesion consists of multinucleated giant cells, epithelioid cells, and lymphocytes. HE. Bar = 10 μm.
The distribution of granulomatous lesions in pigs. ∗
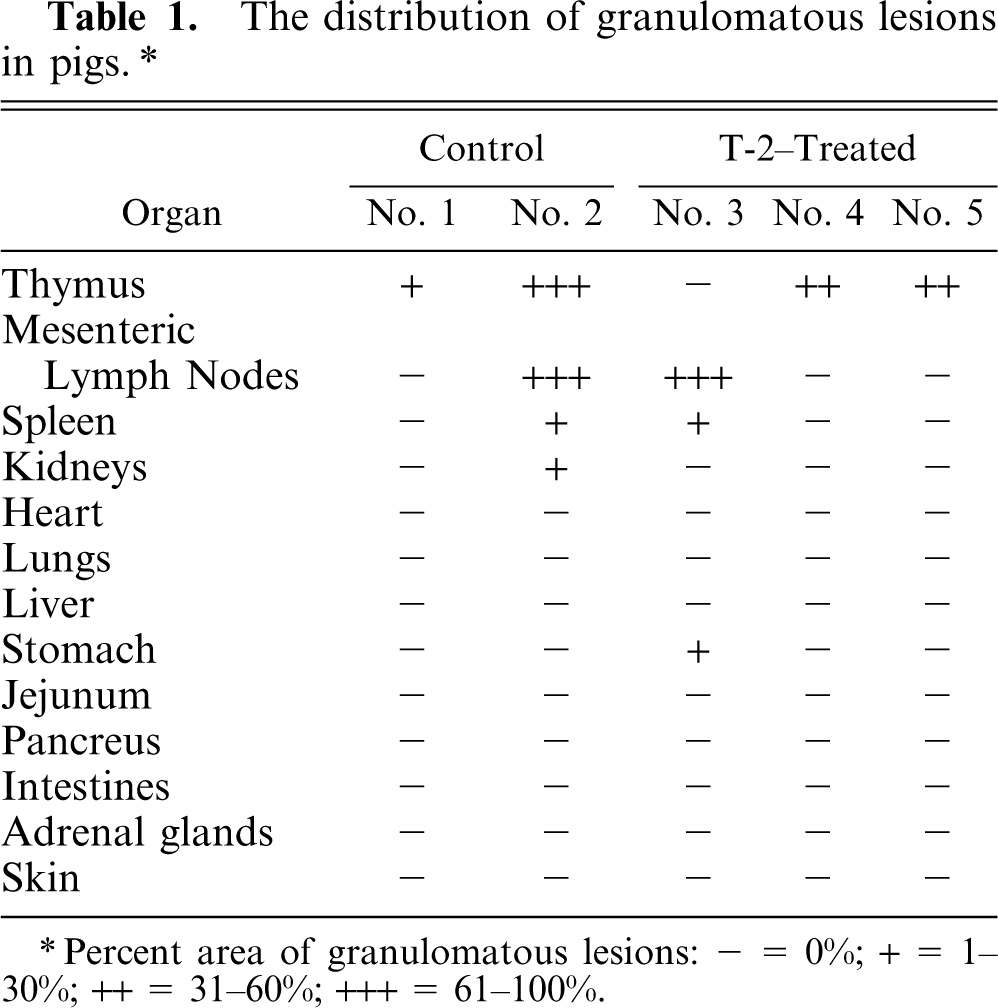
Percent area of granulomatous lesions: − = 0%; + = 1–30%; ++ = 31–60%; +++ = 61–100%.
Immunohistochemistry revealed that both epithelioid and giant cells were strongly positive for vimentin (Fig. 3) and desmin (Fig. 4) and were weakly positive for lysozyme (Fig. 5), whereas they were negative for cytokeratin and both smooth muscle and sarcomeric actins. No signals of PCV-2 infection were observed in any of the tissues examined.
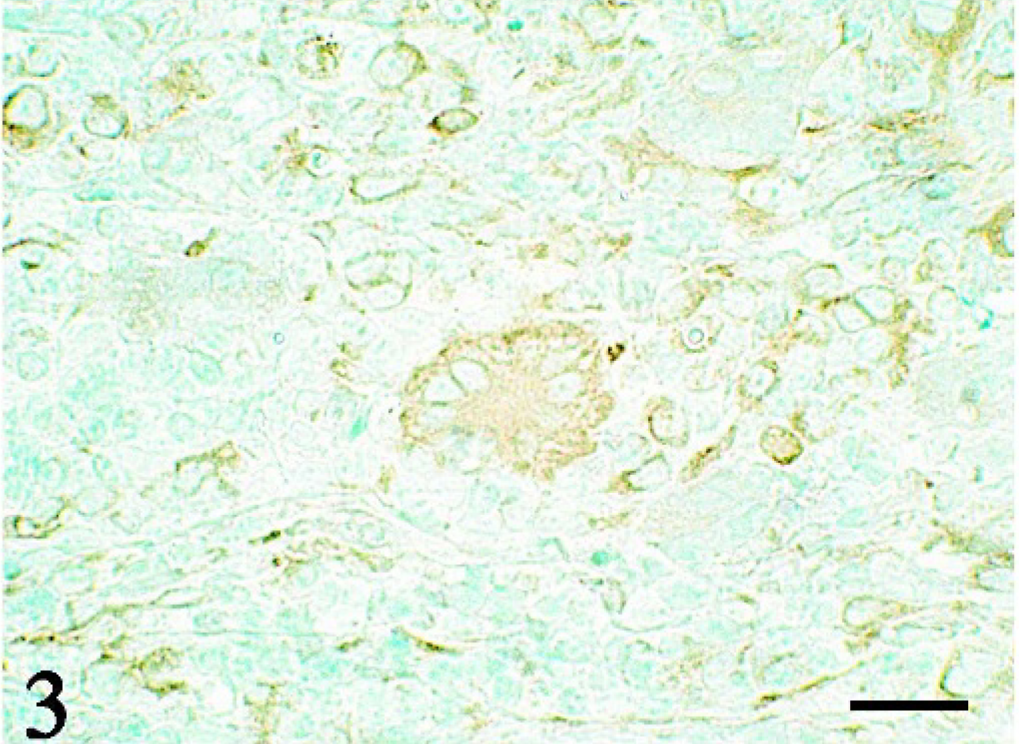
Thymus; pig No. 2. Giant cells and epithelioid cells are strongly positive for vimentin. LSAB immunostaining with methylgreen counterstaining. Bar = 10 μm.
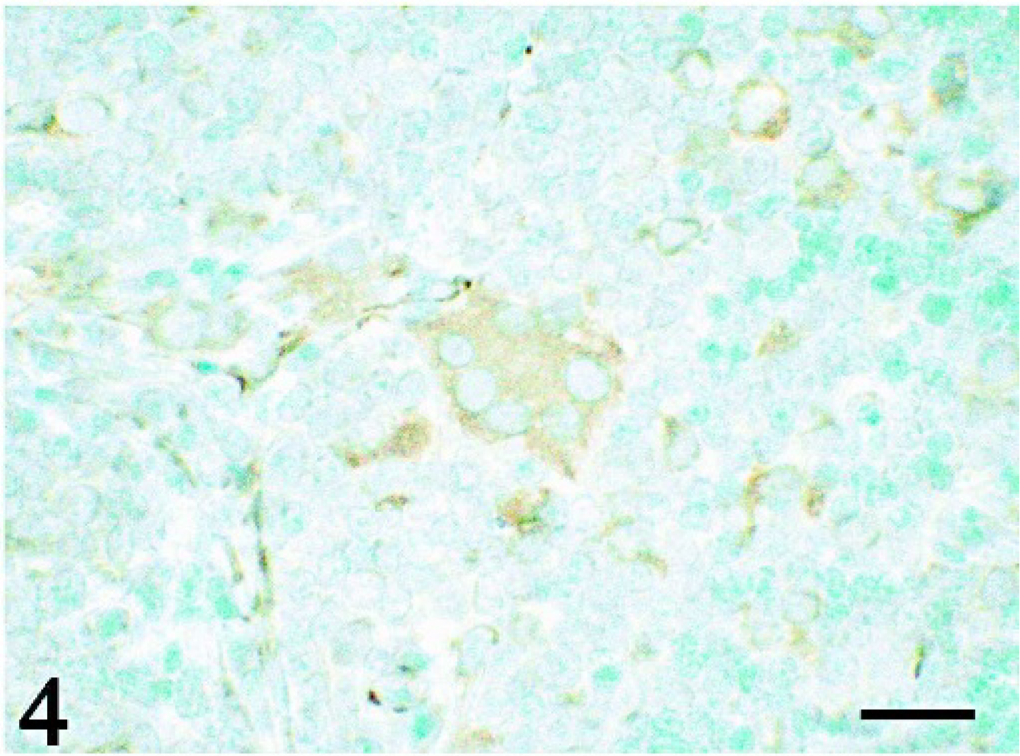
Thymus; pig No. 2. Giant cells and epithelioid cells are positive for desmin. LSAB immunostaining with methylgreen counterstaining. Bar = 10 μm.
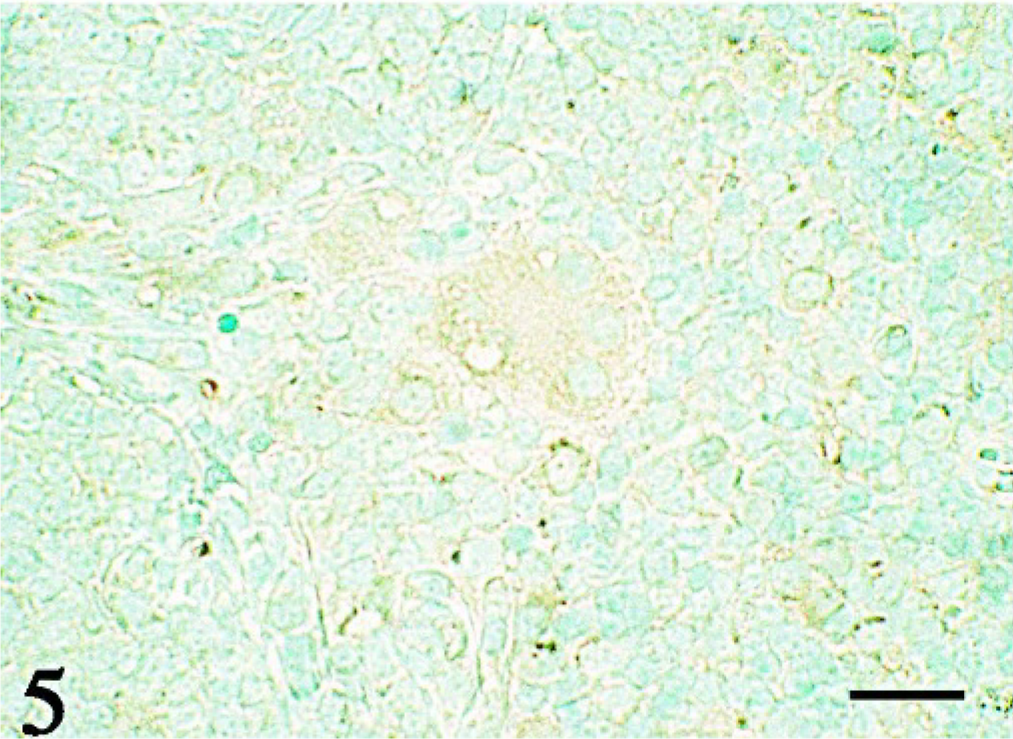
Thymus; pig No. 2. Giant cells and epithelioid cells are weakly positive for lysozyme. LSAB immunostaining with methylgreen counterstaining. Bar = 10 μm.
Ultrastructually, the giant cells were rich in organella and attached to the adjacent epithelioid cells by membrane interdigitation (Fig. 6). Rough endoplasmic reticulum was slightly dilated and branched to form an intracytoplasmic labyrinth. There were numerous coated vesicles and fine granules as well (Fig. 6). The diameter of the coated vesicle was 0.08 μm, and the diameter of the granule was between 0.1 and 0.4 μm. The surface of the epithelioid cells was smooth, and the cells included fewer organella and granules. No intermediate filament bundles were observed in giant cells or epithelioid cells.
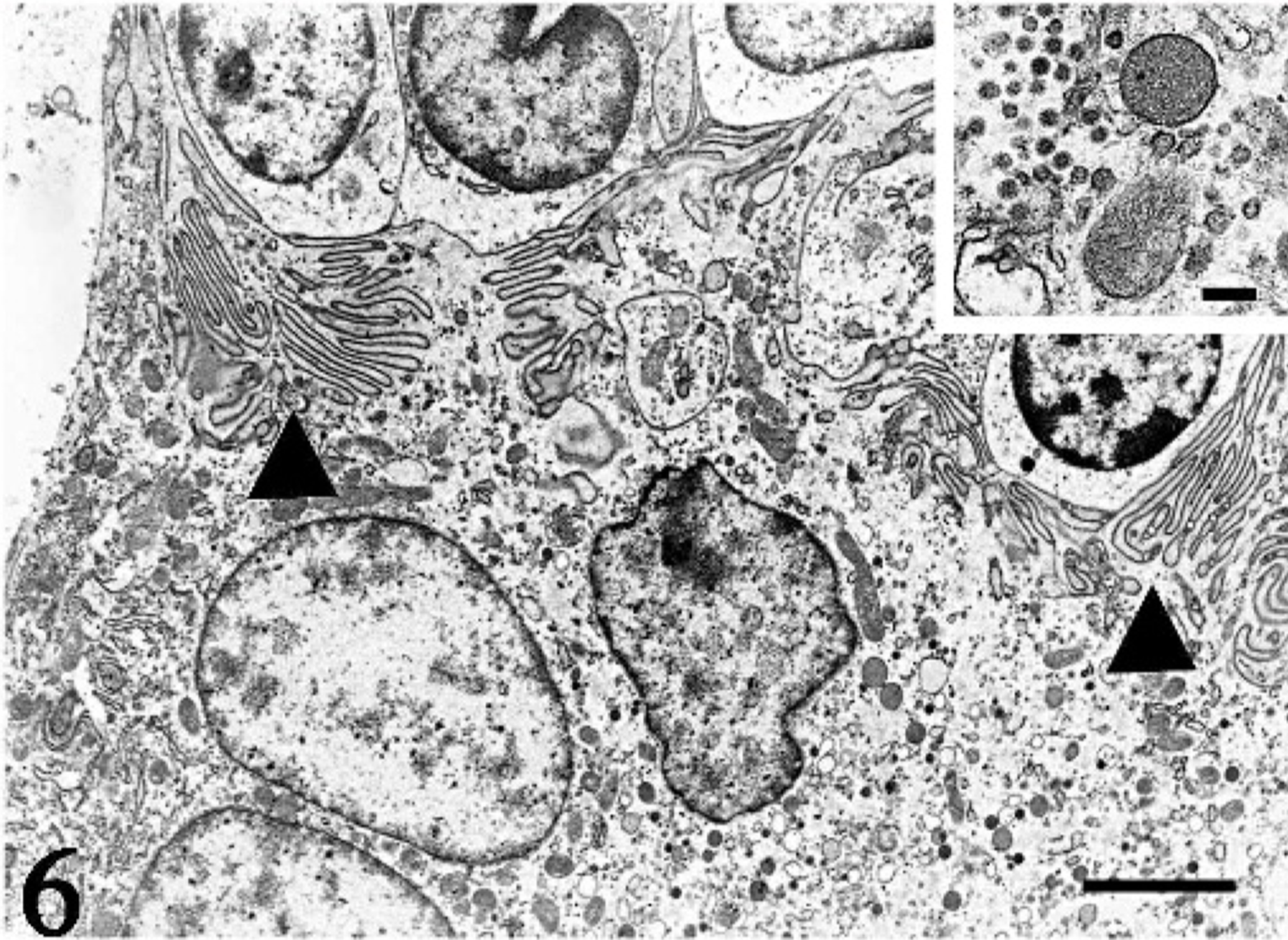
Thymus; pig No. 2. A multinucleated giant cell is rich in organella and attaches to adjacent cells by membrane interdigitation (arrowheads). Electron microscopy. Bar = 3 μm. Inset: There are numerous coated vesicles and fine granules in the cytoplasm. Electron microscopy. Bar = 0.5 μm.
PCR for the detection of PCV-2 revealed amplified positive template but no amplification of samples from affected thymus and lymph nodes (data not shown).
Systemic granulomatous lesions in pigs have been reported in cases of mycobacterial infection, 7, 9 brucellosis, 3 mucormycosis, 10 and PMWS. 2, 4, 6, 8 However, no etiologic agents suggesting mycobacterial infection, 7 brucellosis, 3 mucormycosis, 10 and PMWS 2, 4, 5, 8 were detected in the present cases. Sarcoidosis in humans is a disease characterized by systemic granulomatous lesions without caseous necrosis. 1, 11 Schaumann conchoidal bodies and/or Asteroid inclusion bodies were ultrastructurally detected in the large epithelioid cells and multinucleated giant cells appearing in lesions of sarcoidosis. However, in the present pig cases no such inclusions were observed by electron microscopy. Even though histologic and molecular detection of pathogens proved unsuccessful, infection by a certain intracellular pathogen is still suspected in the present case because ultrastructural findings of the cells in the lesions were similar to those of mycobacterial infection. 7 There are a few reports 2, 6 describing thymic granulomatous lesions in PCV-2–infected pigs. Histologic findings of the lesions often are similar to those in the present cases. However, neither PCV-2 antigen nor sequence was detected in the present lesions. Therefore, it is suspected that the granulomatous lesions are formed by mechanisms other than PCV-2 infection in the present and even in the previous cases.
Granuloma of the thymus without pathogen has not been reported so far in pigs, and this may be the first report of idiopathic thymic granulomatous lesions in pigs.
Footnotes
Acknowledgments
We wish to thank Dr. Kenji Kawashima at the National Institute of Animal Health for his practical support in carrying out experiments to detect PCV-2, and Dr. Takuya Hirai at the Nippon Institute for Biological Science for his meaningful advice.
