Abstract
Primary chondrosarcoma was found in the quadrate lobe of the liver of a 6-year-old, intact, male Golden Retriever. At 6 months after partial hepatectomy, recurrence in the liver occurred. The dog died of its systemic metastases 10 months thereafter. Histologically, the hepatic mass revealed neoplastic chondrocytes with abundant chondroid matrix, and there were few myxoid areas where the cellularity and pleomorphism of the neoplastic cells were more prominent. The neoplastic cells were positive for periodic acid-Schiff and were immunohistochemically positive for vimentin and S-100 protein; the matrix was deeply stained for alcian blue and was metachromatic for toluidine blue stain. This tumor might be derived from pluripotent mesenchymal cells in the connective tissue of the liver. To the best of our knowledge, in all mammalians, including humans, this is the first report of extraskeletal chondrosarcoma primarily arising in the liver.
Most chondrosarcomas develop from the skeletal cartilage, whereas some occur in extraskeletal tissues that possess preexisting cartilage tissues. On the other hand, extraskeletal chondrosarcomas occur in the soft tissue or parenchymal organs, which lack preexisting cartilage. 6, 8 Although extraskeletal chondrosarcoma is rare in animals, it has been reported in dogs 6, 7, 9 and a cow. 13 In dogs, some extraskeletal chondrosarcomas occurred in the heart and lung, including cartilage, 9 whereas others were seen in the omentum 9 or spleen, 6 where no cartilage originally existed. This is the first report on extraskeletal chondrosarcoma that occurred primarily in the liver of a dog.
A 6-year-old, intact, male Golden Retriever was presented with a primary complaint of vomiting 3 months in duration. Initial laboratory database revealed increases in white blood cells (WBCs) and serum alkaline phosphatase (ALP) value. Three months later, anorexia and abdominal distension accompanied by elevated values of WBCs and ALP developed. Radiographic and ultrasonographic examinations confirmed a large hepatic mass in the quadrate lobe but no abnormalities in the bones. Immediately, the lobe with the mass was excised en bloc surgically. No gross lesions were found in any other abdominal organs at surgery. The removed hepatic mass, which measured 16 × 15 × 10 cm, was histopathologically examined and diagnosed as extraskeletal conventional chondrosarcoma. After 6 months of liver lobectomy, the dog was presented with anorexia, abdominal distension, and the laboratory abnormalities included peripheral leukocytosis and elevated ALP value. Furthermore, repeat abdominal ultrasonography revealed recurrent hepatic masses. At that point the anticancer therapy was instituted with oral administration of cyclophosphamide. The daily dosage was 50 mg at 10-day intervals for the first 3 months. However, since the clinical condition was not improved, it was increased by 100 mg at 5-day intervals and maintained for 7 months until death. Notwithstanding repeated anticancer treatment, the dog died eventually after 16 months of liver lobectomy.
At necropsy there was a large volume of bloody peritoneal fluid and multiple masses of varied sizes in all lobes of the liver (Fig. 1). Furthermore, the tumor masses were located in the lung, diaphragm, omentum, peritoneum, spleen, adrenal glands, kidneys, and abdominal wall and on the serosa of the gastrointestinal tract. The largest two masses, 17 × 12 × 8 and 14 × 9 × 7.5 cm, were observed in the liver and the omentum, respectively. The cut surfaces of all tumor masses had a lobular pattern of cartilage with its translucent gray or white color. Hemorrhagic necrosis and gelatinous areas were found sometimes, especially in the larger masses. Other gross lesions were thickened cardiac valves, enlarged parathyroids, and nodular cortical hyperplasia of the adrenals.
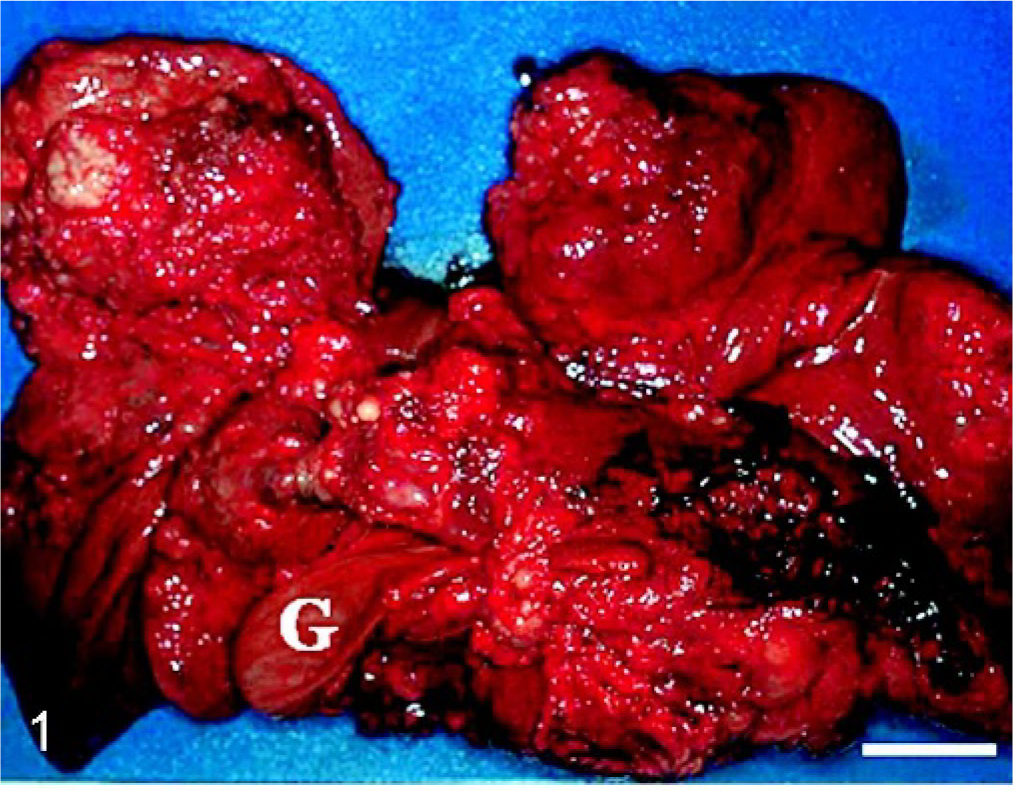
Liver (visceral face); dog. Varied sized masses are seen in the all lobules. G = gallbladder. Bar = 5 cm.
Tissue samples from all major organs and tumor masses were fixed in 10% buffered formalin, embedded in paraffin wax, and sectioned at thickness of 4 μm. Deparaffinized sections were stained with hematoxylin and eosin (HE), alcian blue (pH 2.5), toluidine blue (pH 2.5 and 4.1), or periodic acid–Schiff (PAS). Some sections were pretreated with diastase before PAS reaction for demonstration of glycogen. In order to give more convincing proof of chondrosarcoma, additional sections also were subjected to immunohistochemistry using rabbit polyclonal antibody against cow S-100 protein (DAKO, Glostrup, Denmark), mouse monoclonal antibody against vimentin V9 (DAKO), or mouse monoclonal antibody against cytokeratin AE1/AE3 (DAKO). Labeled streptavidin-biodin (LsAB) technique was employed. In brief, after reacting with primary antibodies, sections were incubated with biotinylated goat anti-rabbit immunoglobulin G (IgG) antibody or anti-mouse IgG antibody (DAKO) and peroxidase-conjugated streptavidin (DAKO). Finally, the reaction with each antigen was visualized by addition of diaminobenzidine tetrahydrochloride and was counterstained with hematoxylin.
Histopathologic examination of the tumor masses in the liver and other organs revealed irregularly shaped cartilaginous lobules that were well demarcated by vascularized fibrous bands from the hepatic parenchyma (Fig. 2). Generally, at the central areas of the lobules the tumor cells were located within lacunae surrounded by amorphous basophilic chondroid matrix (Fig. 3). The cellularity was greater at the edges of the lobules. The tumor cells had a round or polygonal appearance, varied in size, and contained moderate volumes of faintly eosinophilic cytoplasm. Round or oval nuclei with plural nucleoli were seen commonly. Binucleate cells sometimes were present, although mitotic figures were rare. On the other hand, there were a few lobules showing poor cartilaginous differentiation characterized by higher cellularity and myxoid matrix. In such areas, the tumor cells exhibited more prominent pleomorphism and nuclear atypia compared with those in other well-differentiated chondroid areas (Fig. 4). Two to three mitotic figures per 400× field were visible. Regardless of whether the area was myxoid or chondroid, intercellular matrices were stained with alcian blue (Fig. 5) and metachromatic with toluidine blue. The tumor cells contained glycogen granules, which were diastase digested and PAS positive. No osteoid differentiation was seen. Based on these histologic findings, extraskeletal chondrosarcoma was diagnosed. Immunohistochemically, the tumor cells were consistently positive for vimentin and S-100 protein (Fig. 6). Generally, there were no differences in histology between the surgically removed hepatic mass and the masses sampled at necropsy, except that the former mass was devoid of poorly differentiated myxoid areas.
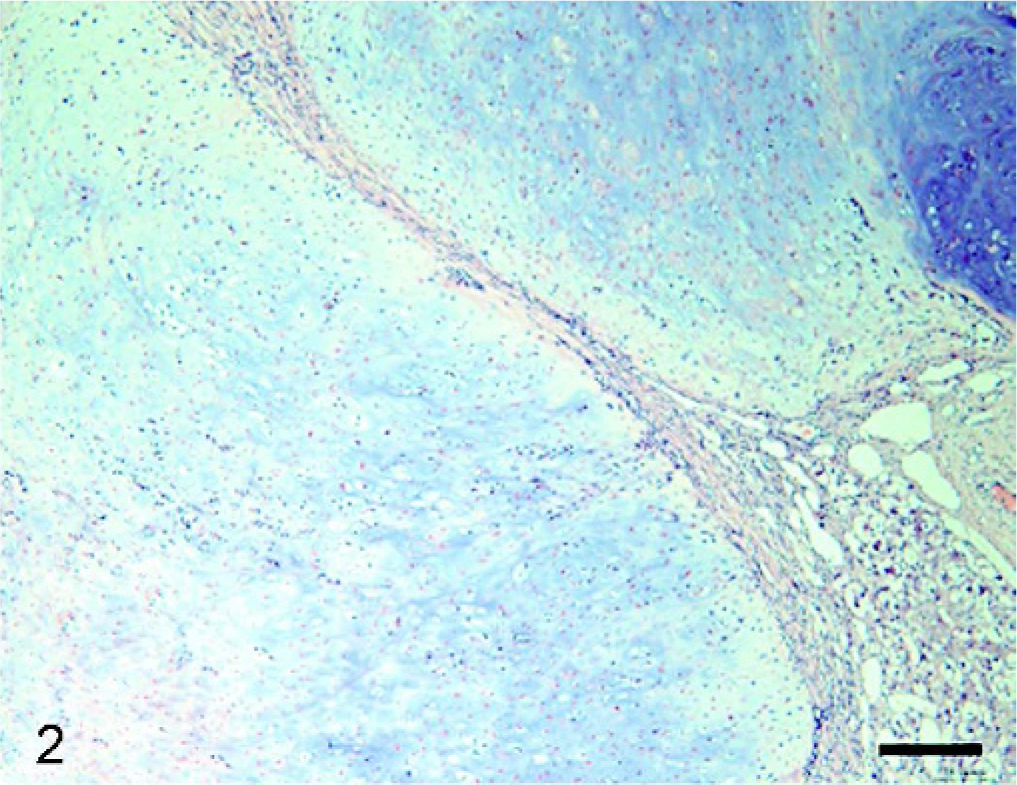
Liver mass; dog. Cartilaginous lobules are well demarcated by vascularized fibrous bands. HE. Bar = 100 μm.
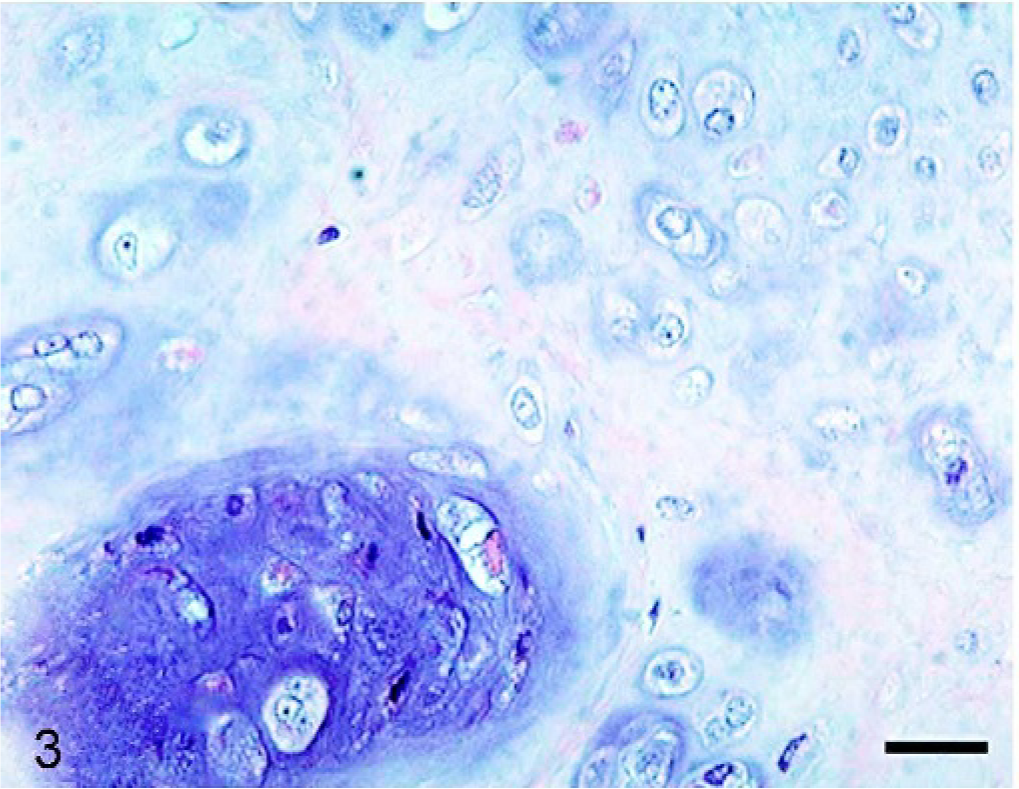
Liver mass; dog. Well-differentiated chondroid area where tumor cells are located within lacunae surrounded by hyaline cartilage matrix. HE. Bar = 25 μm.
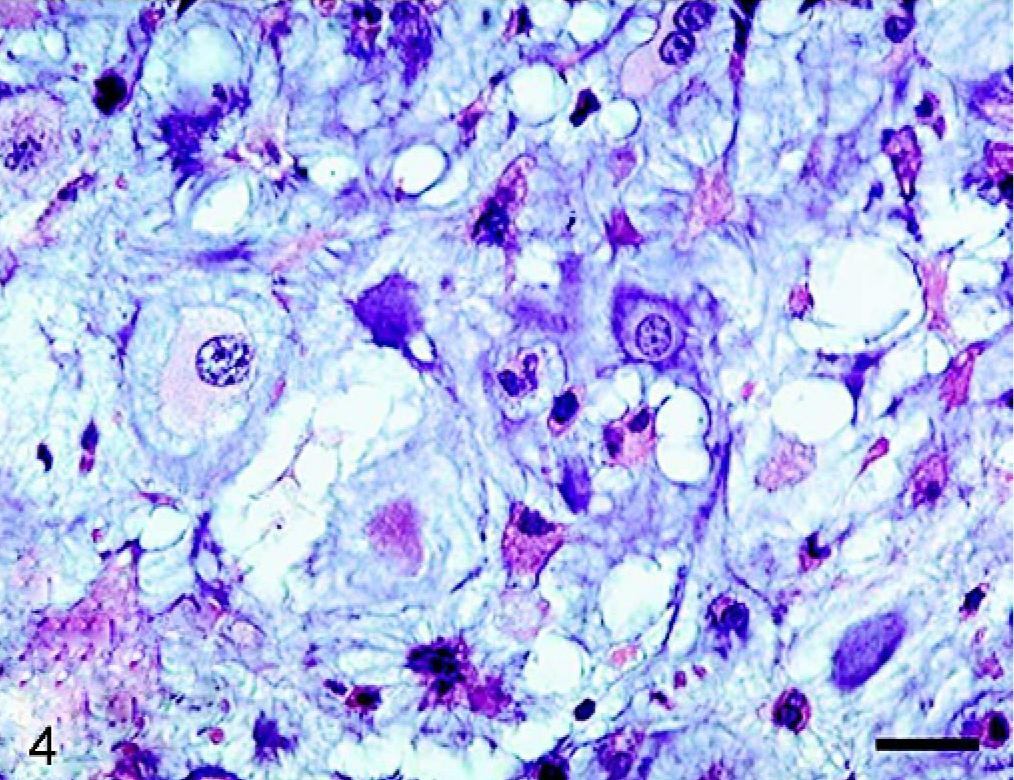
Liver mass; dog. Poorly differentiated myxoid area where tumor cells show strong pleomorphism. HE. Bar = 25 μm.
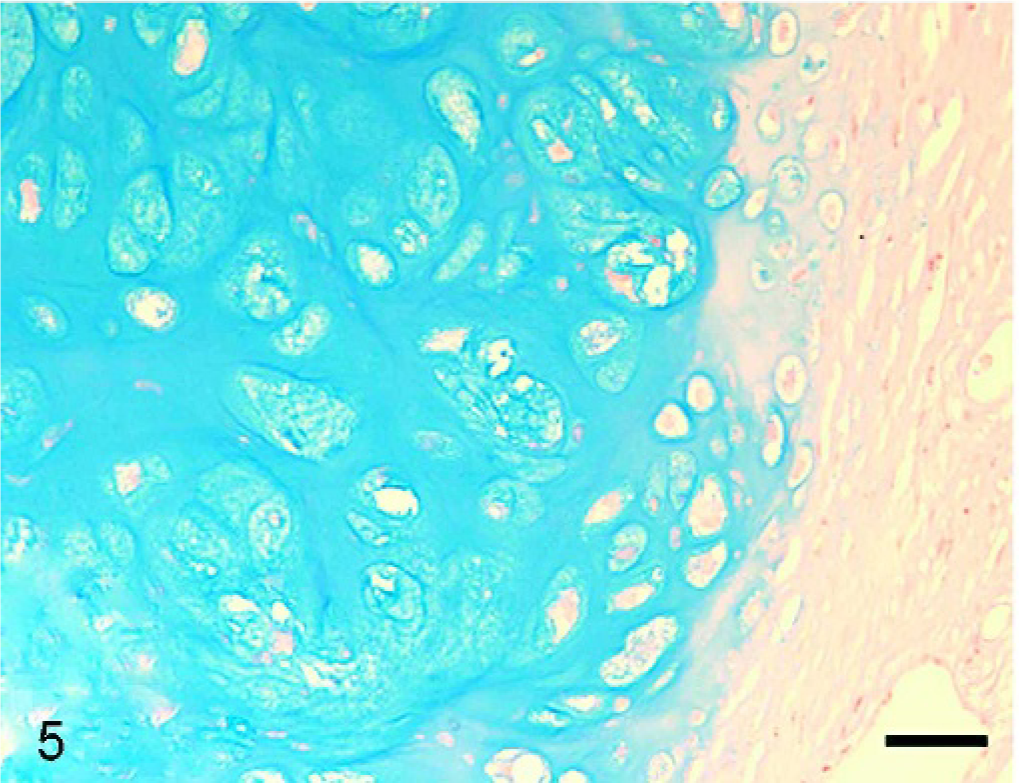
Liver mass; dog. Cartilage matrix stained deeply with alcian blue. Bar = 25 μm.
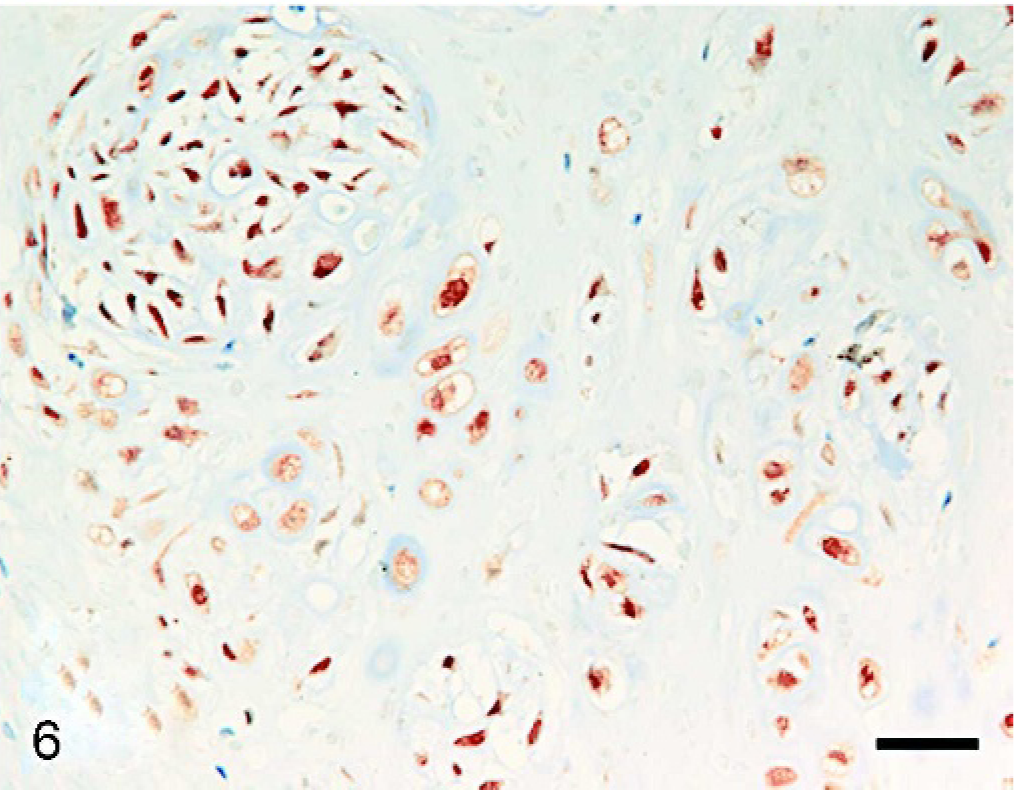
Liver mass; dog. Almost all tumor cells are positive for S-100 protein. LsAB method. Bar = 25 μm.
Based on the cartilaginous differentiation of tumor cells and matrix, the present case was diagnosed as extraskeletal chondrosarcoma. Moreover, the intercellular matrix was stained with both toluidine blue and alcian blue, which corresponded to normal cartilaginous tissue. This case could be diagnosed easily as malignant tumor in that metastases to many organs occurred. As for human extraskeletal chondrosarcomas, 3 subtypes (conventional, myxoid, and mesenchymal) have been reported. 3, 7 The current tumor was characterized by well-developed chondroid lobules, although poorly differentiated myxoid areas coexisted. The myxoid area can be recognized as representing a lower stage of differentiation and a more malignant phenotype compared with the chondroid area.
Chondrosarcoma in the liver should be distinguished from other candidate tumors with cartilaginous differentiation, such as myxoid liposarcomas 12 or malignant mesotheliomas. 14 Although both chondrosarcoma and liposarcoma are positive for S-100 protein and vimentin, 1, 5 chondrosarcoma could be distinguished from liposarcoma in that neoplastic chondrocytes did not include lipid droplets in the cytoplasm. On the other hand, mesothelioma is sometimes positive for cytokeratin but is consistently negative for S-100 protein. 14 Thus, an application of immunohistochemistry for S-100 protein, cytokeratin, and PAS reaction is helpful for precise differential diagnosis of chondrosarcoma.
As for the cell origin of extraskeletal chondrosarcoma, the sarcoma cells may be considered to be undifferentiated mesenchymal cells with cartilaginous differentiation in the connective tissues of the liver, although we could not find a clue to explain why malignant transformation occurred in this case. This might also be supported by our observations regarding the liver of a dog with congenital portosystemic shunts in which cartilage metaplasia was seen incidentally (unpublished data). With respect to histogenesis of extraskeletal osteosarcoma, it also is presumed that undifferentiated mesenchymal cells with osteogenic potential are present in the extraskeletal tissue. The continued persistence of cytokines and growth factors can stimulate their differentiation into bone-forming (metaplastic) tissue and can provide a fertile environment for malignant transformation. 11
In general, liver lobectomy is known to improve survival times for massive hepatocellular carcinoma (HCC) in dogs 10 and for extraskeletal osteosarcoma in a cat. 2 In a retrospective study 10 on 48 dogs with massive HCC, the median survival time of dogs treated by liver lobectomy was >48 months, which was significantly longer than that of conservatively treated dogs (9 months). En bloc excision of the liver with extraskeletal osteosarcoma in a cat gave rise neither to clinical signs of metastasis nor hepatic disease over 42 months. 2 The present case survived 16 months after liver lobectomy, which appeared shorter when compared to other lobectomized animals. This might be explained by the different timing of institution of anticancer therapy after surgical treatment. The cat was treated with the chemotherapeutic agent carboplatin 4 weeks after initial surgery, when there was no evidence of local recurrence or distant metastasis. 2 In the present case, anticancer therapy using cyclophosphamide was instituted 6 months after initial surgery, when local recurrence in the liver occurred.
