Abstract
Although pure granular cell tumors have been reported in various sites in the dog, only one tumor has been reported in the central nervous system. Two dogs presented with neurologic signs had brain lesions detected by magnetic resonance imaging in the area of the olfactory bulbs and frontal cortex. In both dogs, a clinical diagnosis of a granular cell tumor was made from tissues obtained from stereotactic biopsies guided by computed tomography. Surgical removal of the tumors was followed by histopathologic, ultrastructural, and immunocytochemical characterization. Although not conclusive, these studies indicated that the granular cells were not of leucocyte origin but may have been derived from the meninges. One dog died 12 months after surgery, and the other was alive 4 months later.
In humans, granular cell tumor (GCT) is a descriptive term for a histogenetically heterogeneous group of tumors occurring both within the nervous system and extraneurally. 2,8,21,26 In the central nervous system (CNS) of animals, GCTs have been reported in the rat and in one ferret and one dog. 13,16,19,22,28 In the rat, the GCT is the most common primary CNS tumor and is thought to be of meningeal cell derivation. 28 One canine meningioma had a granular cell component. 20 In humans, most GCTs of the CNS occur within the infundibulum or neurohyophysis. 2,26 Immunocytochemical and ultrastructural findings suggest that these tumors are derived from specialized astrocytes. 2,26 A granular cell component has also been described as part of some human astrocytomas and oligodendrogliomas. 2,5,10 In all these tumors, the granules appear ultrastructurally to be comprised of similar single membrane-bound autophagic vacuoles. 9,10,26 Despite immunocytochemical studies, the histogenesis of these cells remains controversial. 14,19–21,24 The much less common human GCTs of intracranial or peripheral nerves maybe derived from Schwann cells. 3,4,89,23,24 The human extraneural GCTs reported in the skin, tongue, breast, gastrointestinal tract, and biliary tree probably arise from within peripheral nerves; a similar histogenesis is suspected for some extraneural tumors in the dog, cat, horse, and cockatiel. 8,9,20,23 Here, we describe salient neuroimaging, clinical, and pathologic features of two canine GCTs of the CNS.
Case history
Dog No. 1
A 12-year-old castrated male Golden Retriever was presented with a history of several generalized seizures. A neurologic examination revealed only proprioceptive placing deficits in all four limbs. Computed tomography (CT) and magnetic resonance (MR) imaging of the brain demonstrated a well circumscribed, diffusely enhancing mass within the left olfactory bulb and frontal cortex (Fig. 1). There appeared to be extension ventrally into the sphenopalatine sinus. A CT-guided stereotactic biopsy and subsequent transfrontal craniotomy were done, and the mass was almost completely resected. The dog survived for 12 months postoperatively. A CT scan 1 month prior to death did not detect any tumor regrowth, although the dog died of complications from seizures.
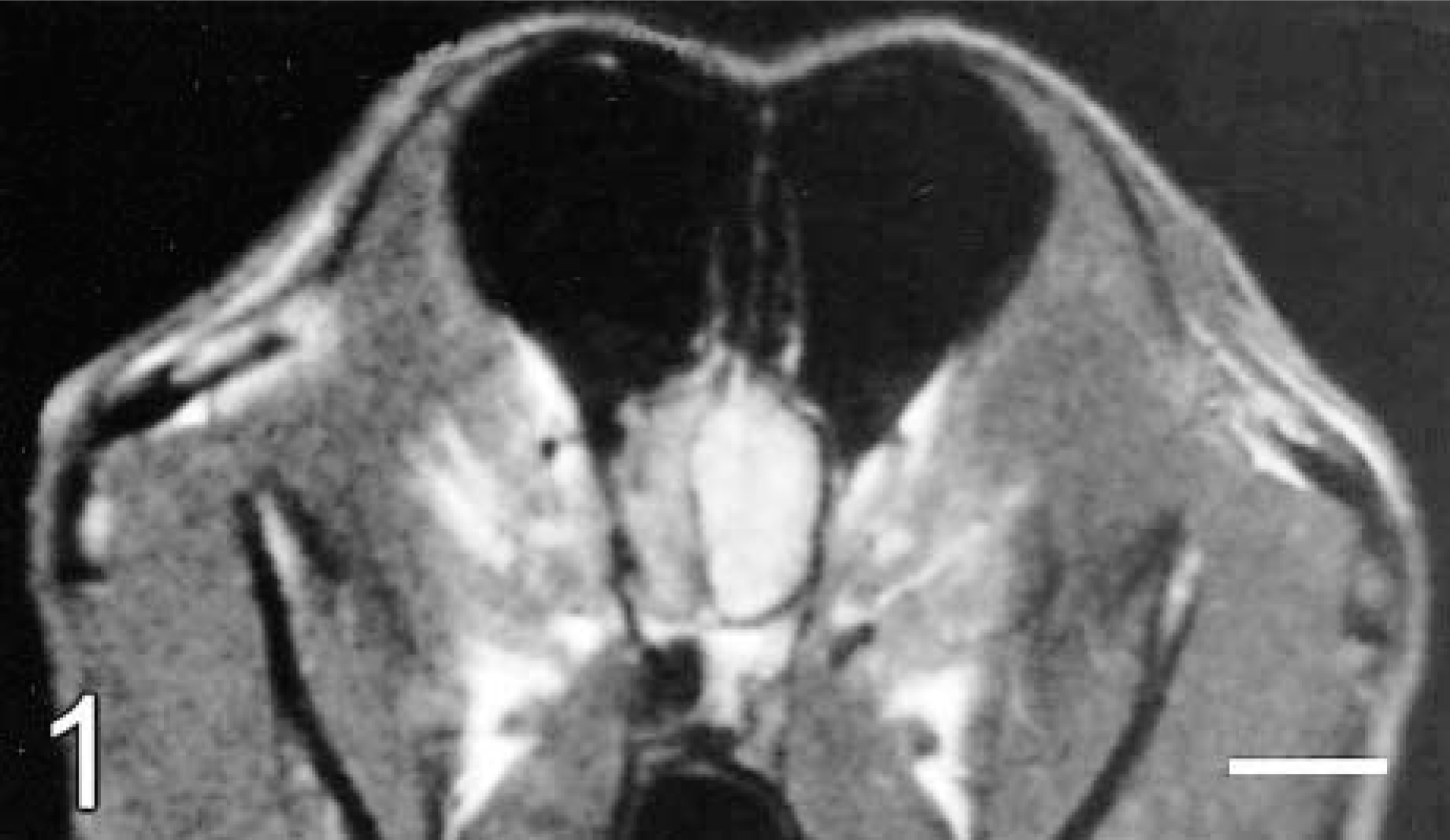
Brain; dog No. 1. Transverse T1-weighted MR image at the level of the olfactory lobes following gadolinium contrast administration. There is a diffusely enhancing, well-demarcated mass within the left olfactory region, with transcalvarial extension into the sphenopalatine sinus.
Dog No. 2
A 12-year-old castrated male Australian Shepherd dog was presented with a 4-week history of seizures. Neurologic examination revealed changes in mentation, decreased to absent conscious proprioception in both pelvic limbs, and absent conscious proprioception in the left thoracic limb. Elevated hepatic enzymes and hepatomegaly on ultrasound were considered to be the effects of prior cortisone and phenobarbital therapy. MR imaging confirmed a mass in the left rostral frontal lobe with a right deviation of the falx cerebri (Figs. 2, 3). A CT-guided stereotactic biopsy and a subsequent transfrontal craniotomy for surgical removal of the mass was done. Four months later, the dog is neurologically normal and being maintained on anticonvulsant therapy.
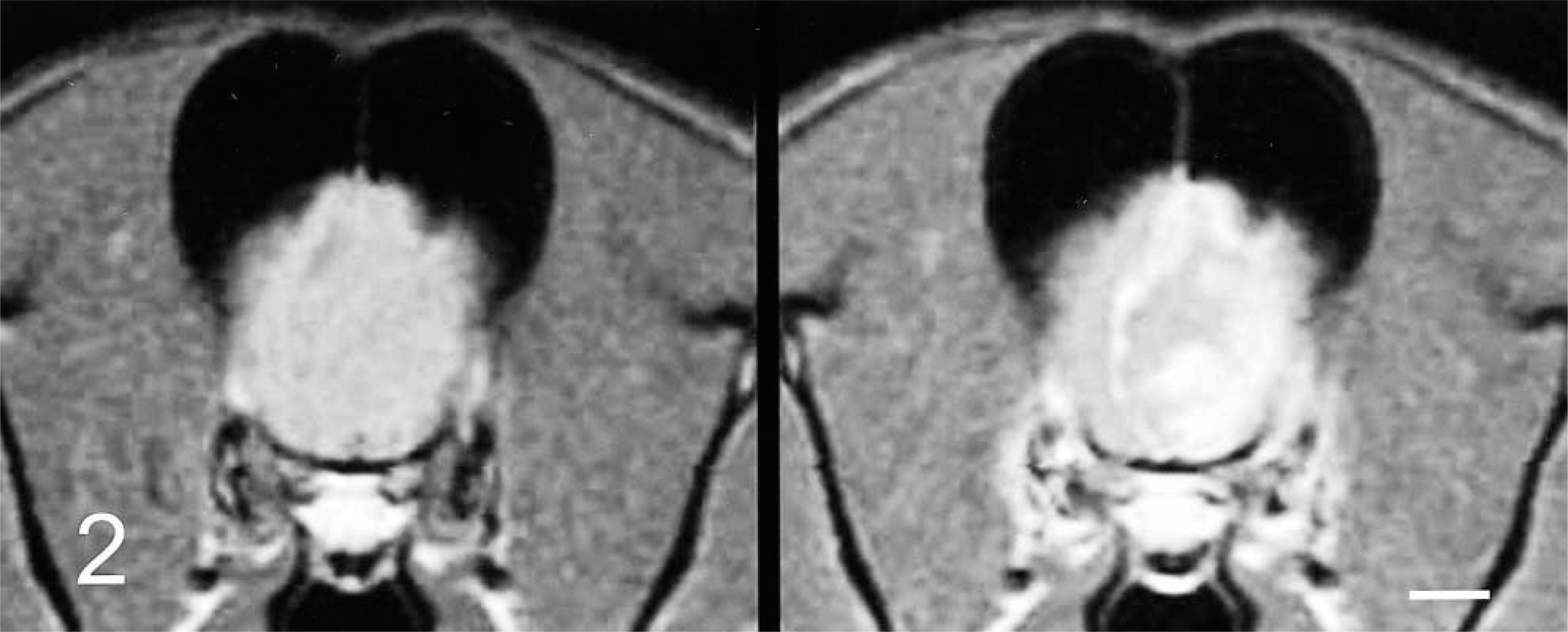
Brain; dog No. 2. Transverse T1-weighted MR images at the level of the frontal lobe. Precontrast image (left) demonstrates a right deviation of the falx cerebri. Postcontrast image (right) illustrates an irregular contrast–enhancing mass with enhancement of the lateral and falcine meninges.
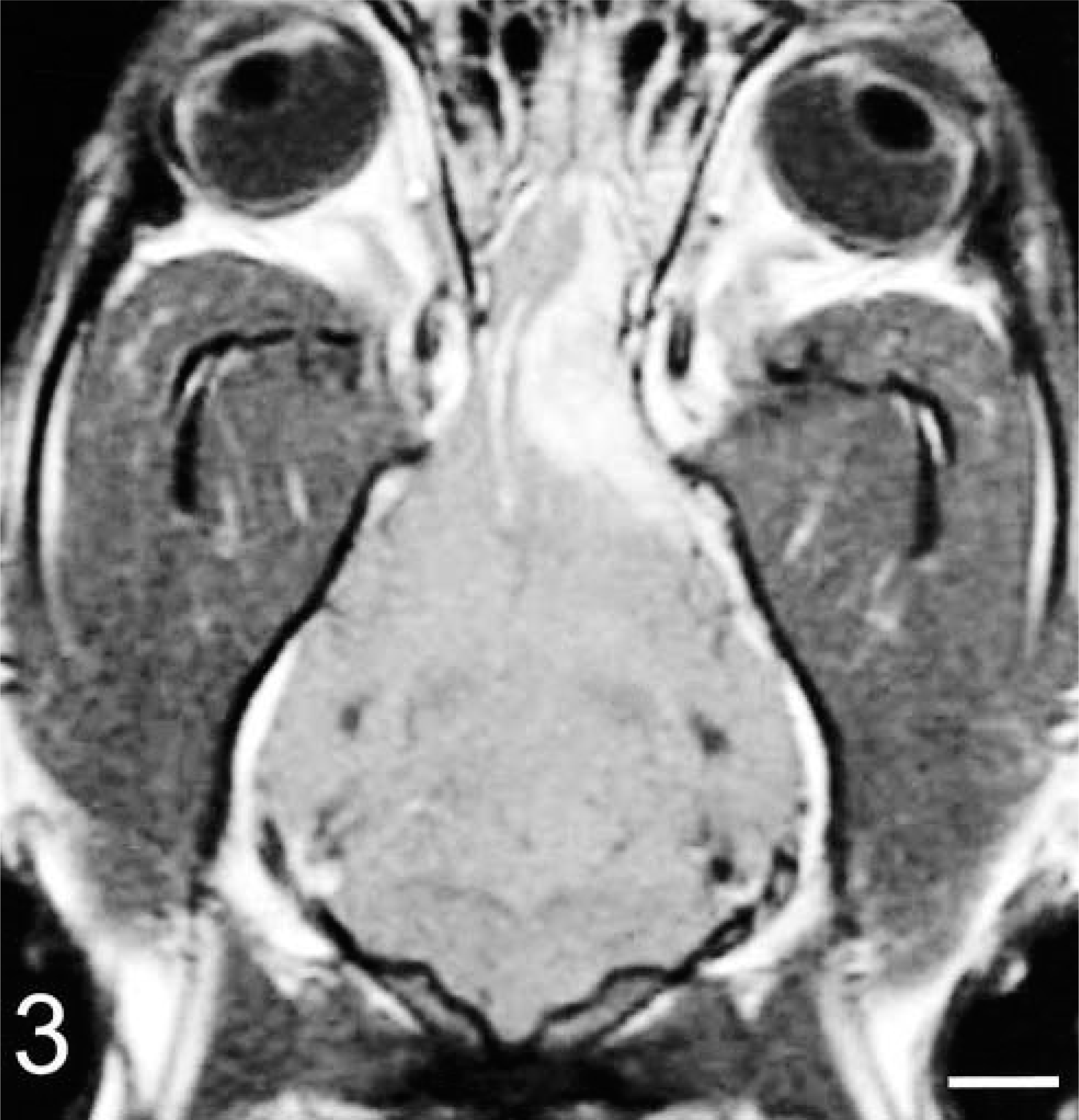
Brain; dog No. 2. A dorsal T1-weighted postcontrast MR image of the brain illustrating the broad-based meningeal involvement of the mass.
Materials and Methods
From tissue specimens of the brain masses obtained by both CT-guided stereotactic biopsy and later surgical removal, a smear preparation and hematoxylin and eosin (HE)-stained sections from both frozen and formalin-fixed, paraffin-embedded tissue of both dogs were examined. From fresh tumor tissue obtained at craniotomy from both dogs, samples were immersion fixed in either 10% buffered formalin or 2.5% glutaraldehyde in 0.1 M cacodylate buffer, pH 7.4, and then routinely processed for evaluation by histopathology and immunocytochemistry or by transmission electron microscopy (TEM), respectively. 7,17 Histochemical stains included diastase-resistant periodic acid-Schiff (PAS) and Masson's trichrome. Immunocytochemical staining was done on selected sections with primary mono- or polyclonal antibodies, used as recommended, for antigen visualization in either frozen or formalin-fixed, paraffin embedded tissues (Table 1). Primary antibodies to glial fibrillary acidic protein (GFAP) 1:2,000 (Dako Corp., Carpinteria, CA); vimentin 1:200 (Dako Corp.); MIB-1 1:50 (Innovex Biosciences, Richmond, CA); ubiquitin 1:1,000 (Novocastro Laboratories, Newcastle upon Tyne, UK); α-1-antitrypsin 1:250 (Novocastro Laboratories); α-1-anti-chymotrypsin 1:200 (Novocastro Laboratories); S-100 protein 1:400 (Novocastro Laboratories); pancytokeratins (Lu-5) 1:100 (Biocare Medical, Walnut Creek, CA); CD1a, b, and c; CD3∊, CD11a, b, c, and d (αD); CD18; and CD45 (P.F. Moore, UC Davis, Davis, CA); and CD 1b (C. Howard, Compton, UK) were used as previously described. 17 Tissue sections were stained using a previously described unlabeled streptavidin–biotin immunohistochemical technique. 7 Appropriate positive and negative control tissues, either frozen or formalin fixed and paraffin embedded, that are routinely used in this laboratory were included in all staining procedures. The functional specificities of the antibodies used for the canine leucocyte immunophenotyping studies were interpreted as described. 17
Summary of results of immunocytochemical staining of granular cells in two canine tumors.
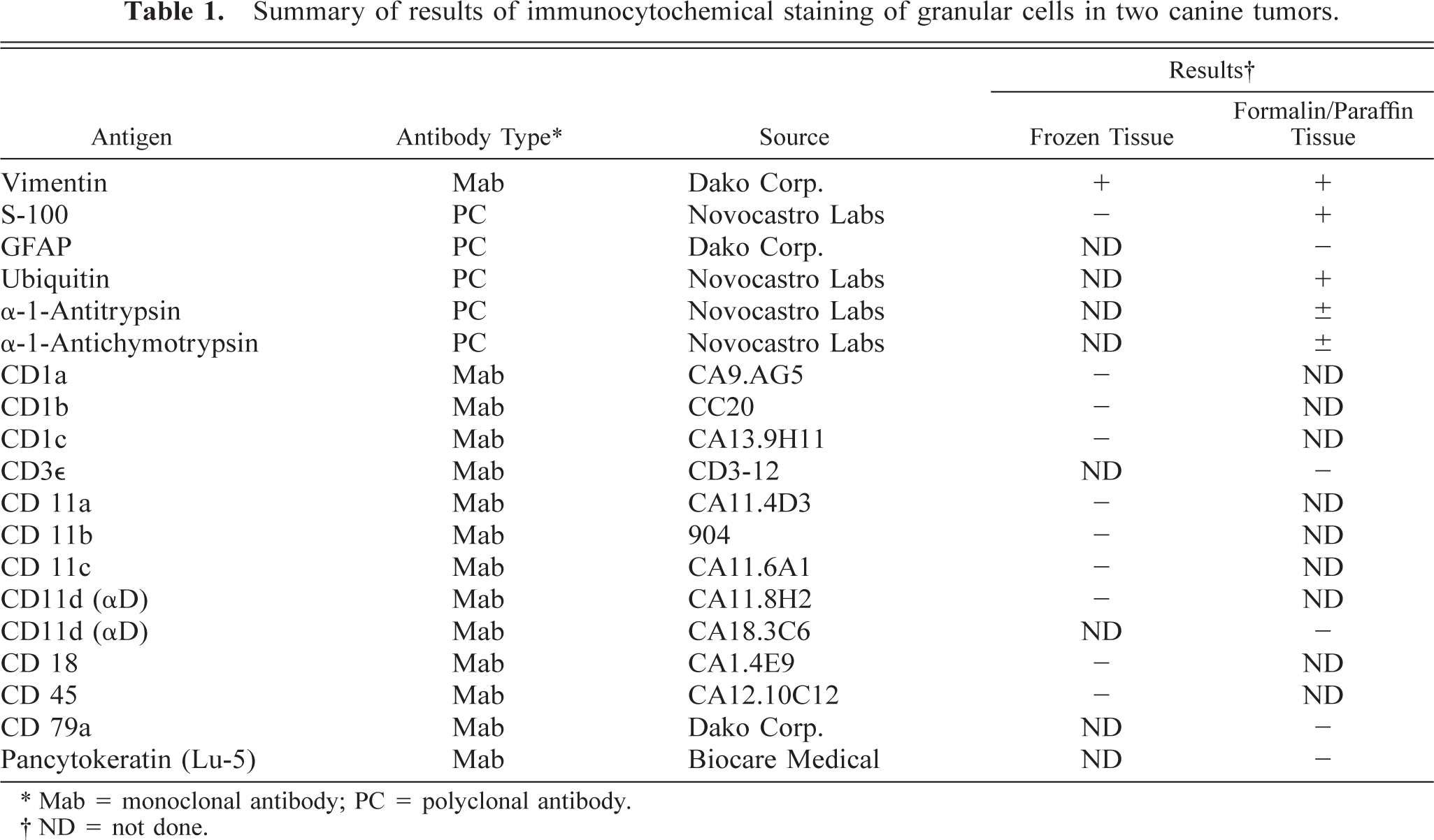
∗ Mab = monoclonal antibody; PC = polyclonal antibody.
† ND = not done.
Results
In dog No. 1, the precontrast CT images indicated only a right-side shift of the falx cerebri, but the postcontrast images revealed a well-circumscribed, uniformly enhancing mass with well-defined smooth margins in the left olfactory region. On MR images, the mass showed diffuse postgadolinium contrast enhancement. This lesion was also hyperintense on T1- and T2-weighted MR images and slightly hyperintense on proton density (PD) images (Fig. 1). There was also apparent transcalvarial extension into the sphenopalatine sinus (Fig. 1).
In dog No. 2, precontrast T2-weighted MR images indicated a hyperintense mass within the left frontal lobe and olfactory bulb (Fig. 3). The mass was isointense on PD and T1-weighted images (Fig. 2, left). Following contrast administration, there was plaquelike enhancement of the meninges of the falx cerebri and left frontal region (Fig. 2, right).
Impression smears examined immediately from tissue obtained by CT-guided stereotactic biopsy demonstrated the distinctive appearance (Fig. 4) of granular tumor cells in dog No. 1 as previously described. 12,25 In dog No. 2, the smear preparation revealed reactive astrocytosis, normal neurons, some macrophages, and a few lymphocytes and plasma cells, but no granular cells were seen. The smear was interpreted as a reactive response to an adjacent lesion. Multiple biopsy cores were then taken from different sites by the CT-guided stereotactic procedure and were submitted for histologic evaluation.
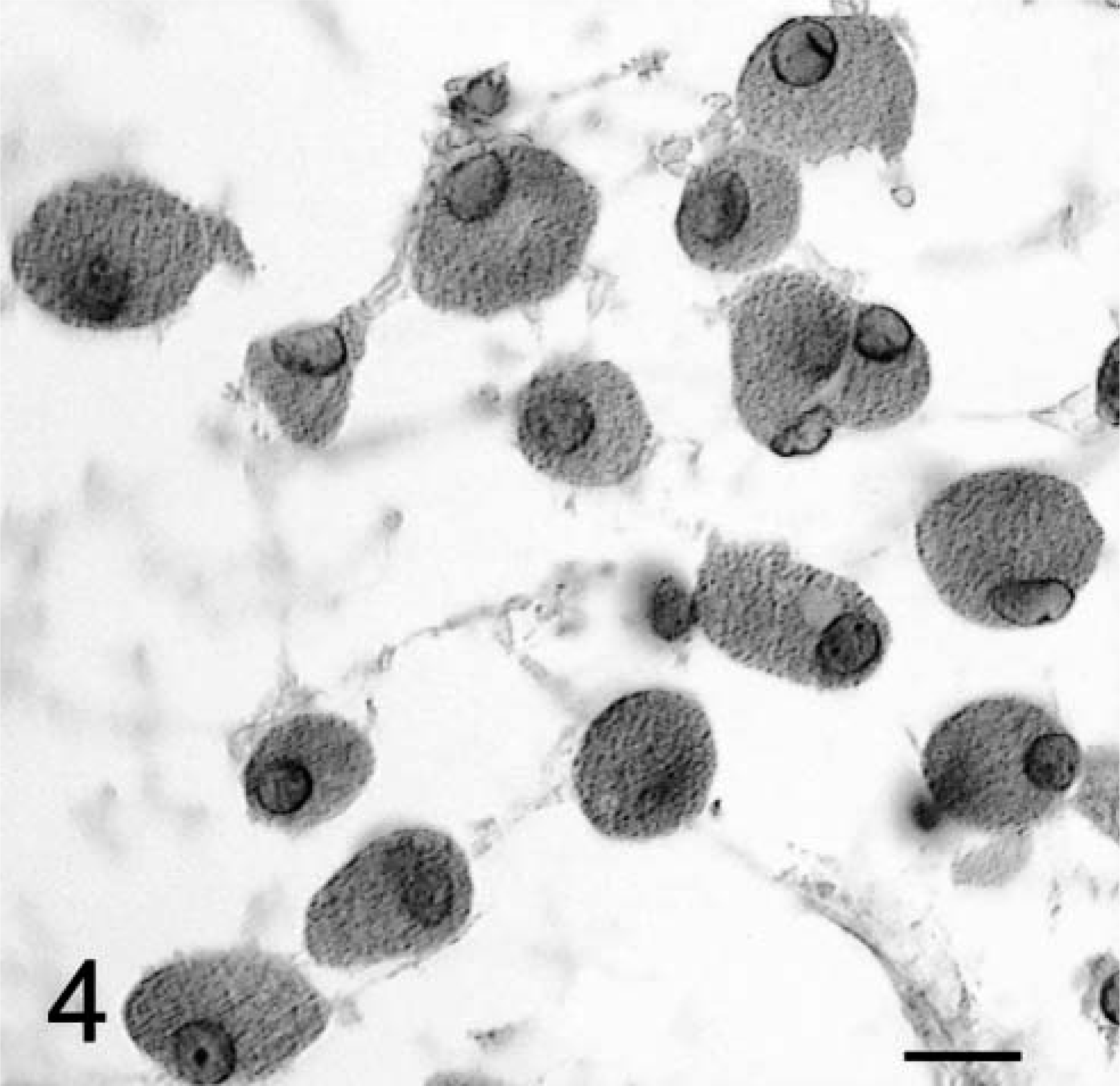
Brain tumor; dog No. 1. Impression smear made from a CT-guided stereotactic biopsy. Note the large, rounded, and separated granular cells of various size. The cells have an eccentrically placed round nucleus with granular eosinophilic cytoplasm and a sharp cytoplasmic border. HE. Bar = 3 µm.
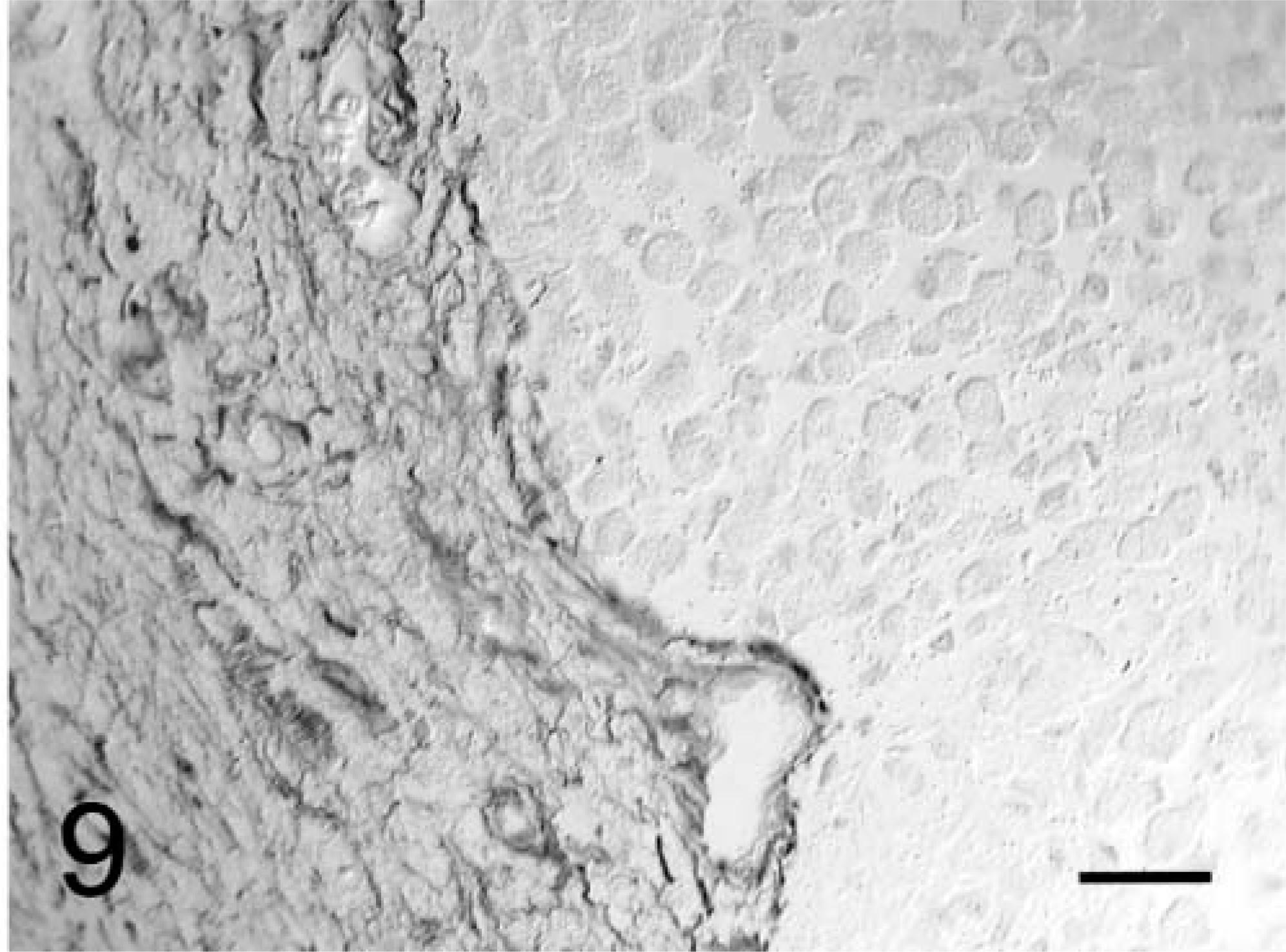
In both dogs, the tumors formed diffuse sheets of granular tumor cells of various sizes interspersed between blood vessels (Fig. 5). Tumor cells were very distinctive because of their cytoplasmic distension by a finely granular, slight vacuolar eosinophilic, diastase-resistant, PAS-positive material. The cytoplasm was rounded to elongate, with a very sharp cytoplasmic border and evenly dispersed granules. Occasional smaller clusters of cells had much less cytoplasm. Nuclei were irregular with a single nucleolus and a hyperchromatic margin and tended to be situated towards the periphery of the cell (Figs. 4, 5). Mitotic figures were rare, and MIB-1 staining revealed a proliferative index of up to 2% within both tumors. The margins of the tumor from dog No. 1 were poorly defined, with individual cells infiltrating the surrounding thickened meninges on one surface. Brain tissue from the interface with the mass was not submitted for examination. In dog No. 2, granular cells emanated from sharply demarcated multiple collagenous foci associated with meninges overlying the brain (Fig. 6). Spindle-shaped cells in the meninges were embedded in an homogenous collagen-positive matrix. These cells either occurred singly or in small clumps of three or four cells whose cytoplasm became larger towards the periphery of the solid tissue, where there was an abrupt transition into an accumulation of large granular cells (Fig. 7). In both tumors, the cytoplasm of spindle and large granular cells was strongly positive for PAS and immunocytochemically strongly positive for ubiquitin (Fig. 8), variably positive for S-100, α-1-antichymotrypsin, α-1-antitrypsin, and vimentin, and consistently negative for GFAP (Fig. 9), pancytokeratins, and an established panel of canine cell-specific markers for subpopulations of canine leukocytes and macrophages (Table 1). Between the granular tumor cells were some CD11d-positive and to a lesser degree CD18-positive infiltrating cells but never any positive staining of the spindle or granular tumor cells. There was also staining of isolated infiltrating CD3+ T lymphocytes and CD45-positive cells but no positive staining of B cells (Table 1).
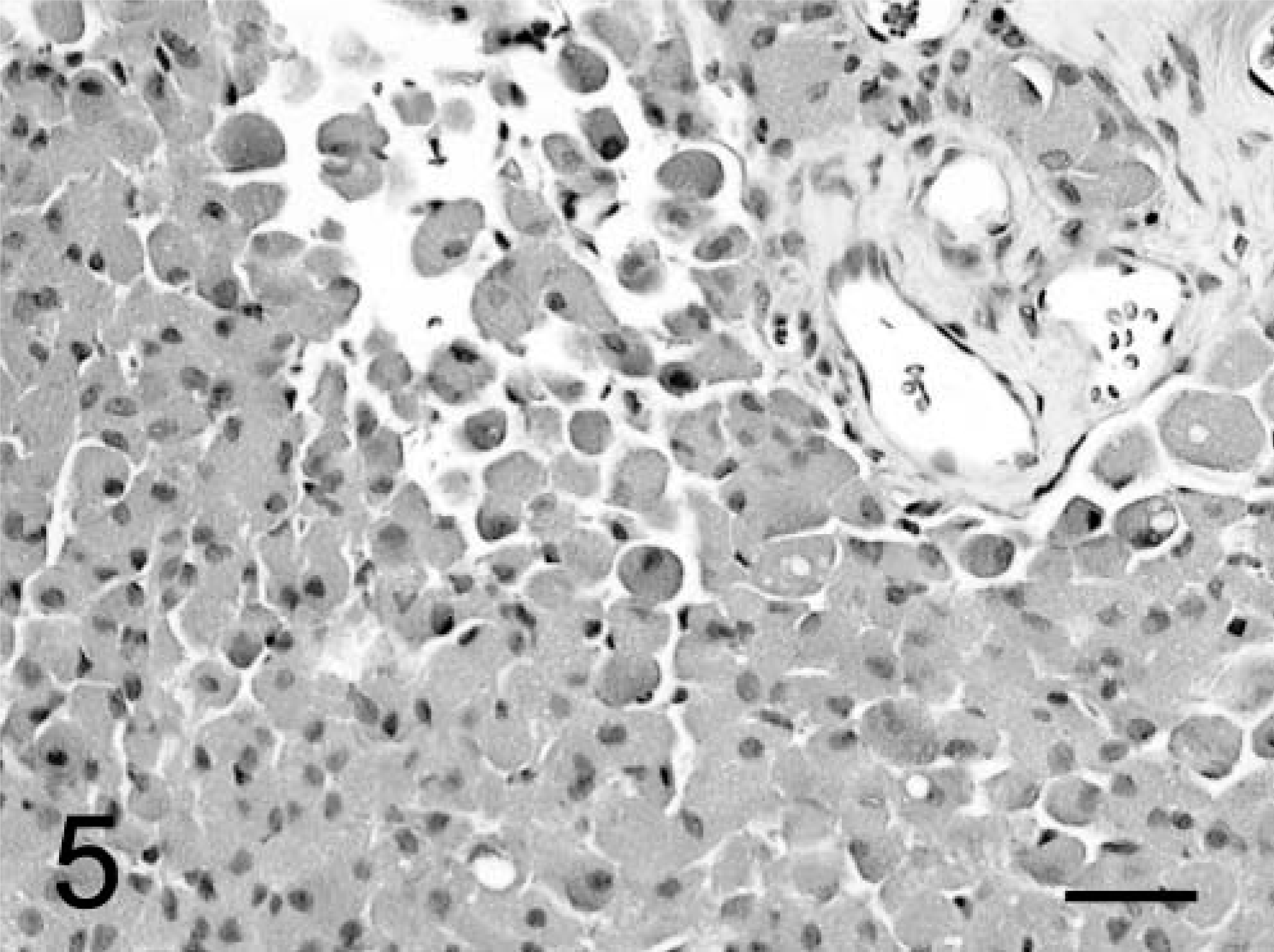
Brain, granular cell tumor; dog No. 1. Note confluent sheets or aggregates of granular cells, which contain prominent eosinophilic granules and eccentrically placed nuclei. HE. Bar = 31 µm.
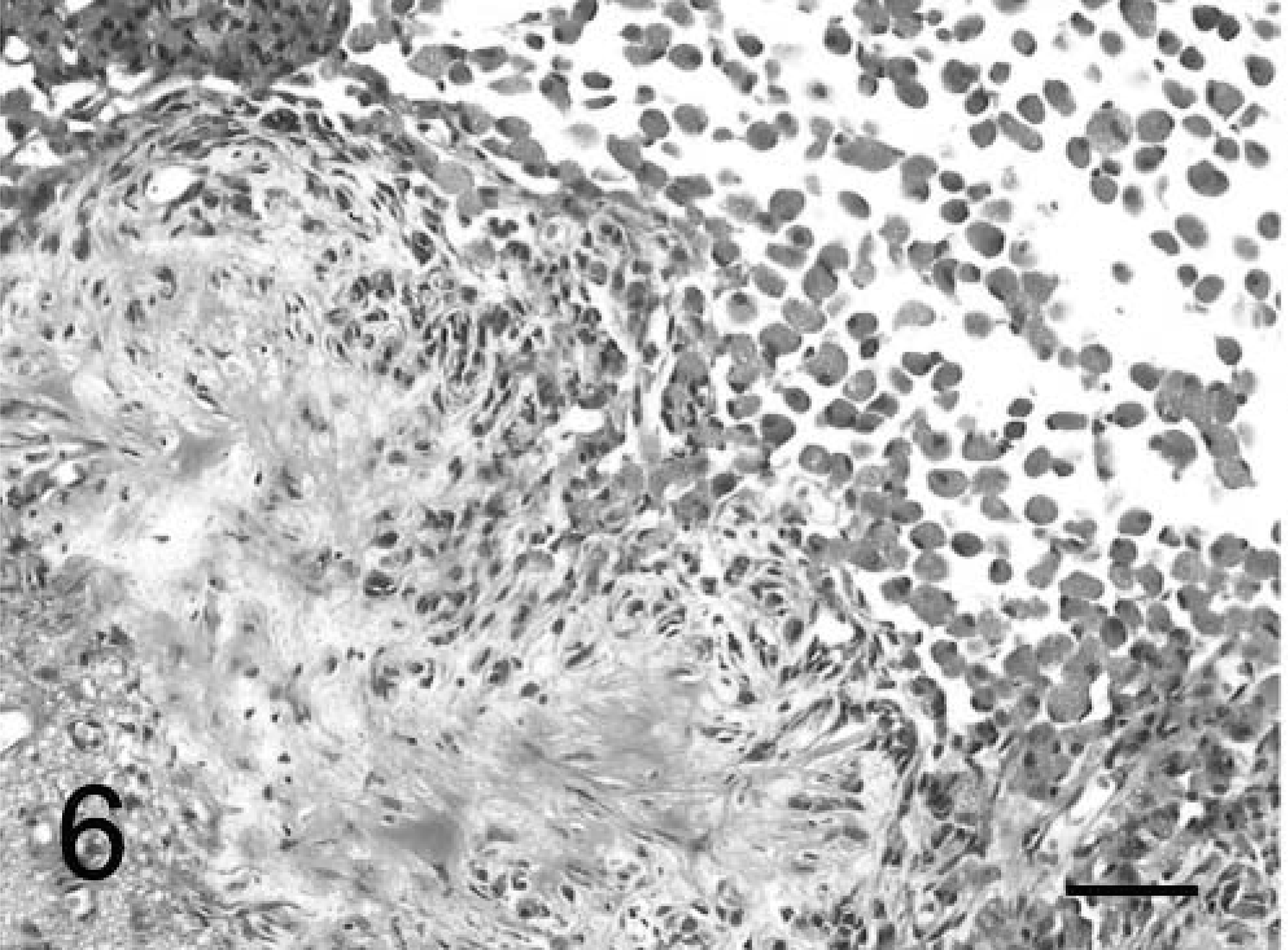
Meninges and brain parenchyma; dog No. 2. Note the proliferation of granular cells from meningeal collagenous foci into the brain. Masson's trichrome. Bar = 42 µm.
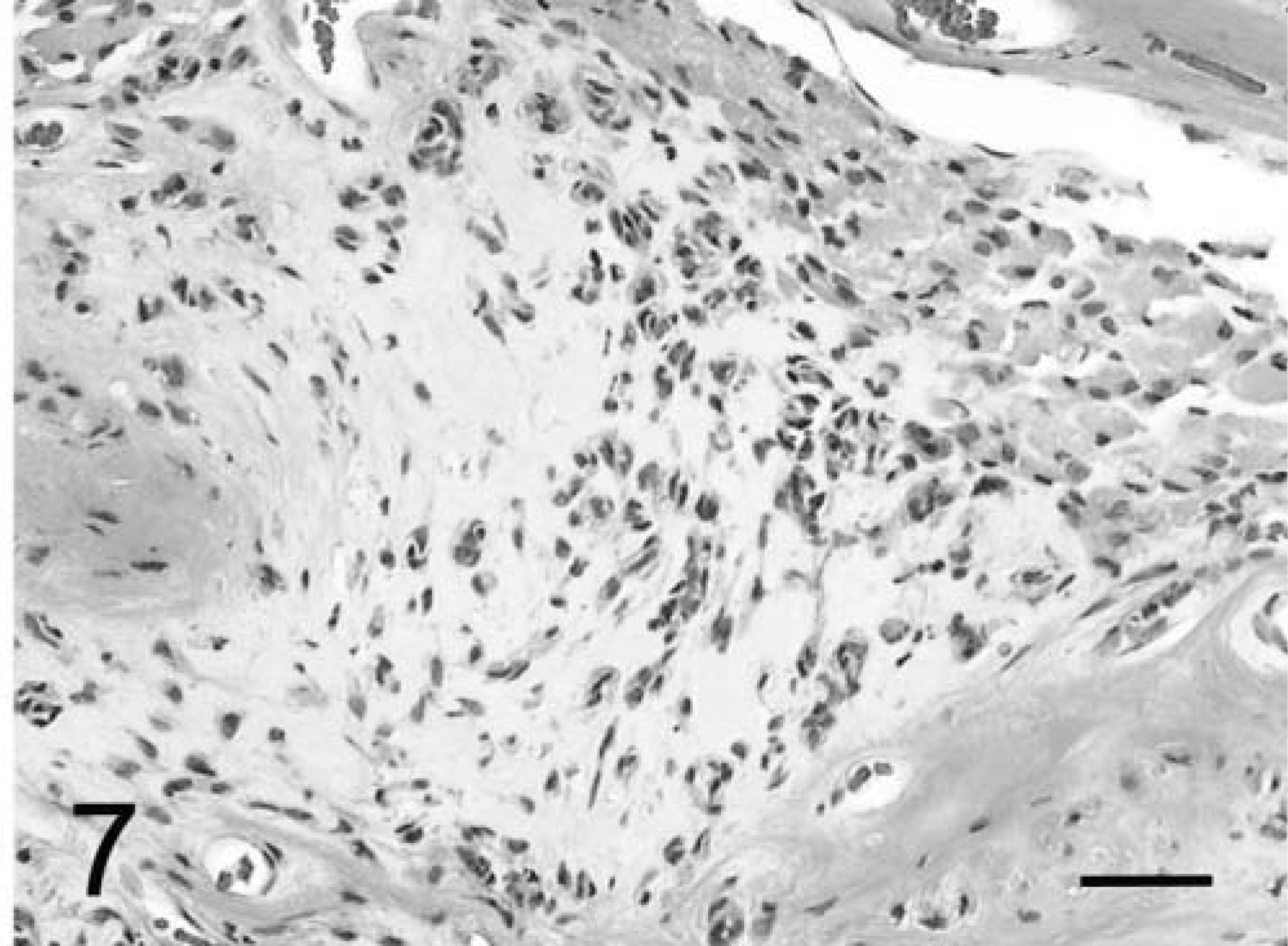
Meninges; dog No. 2. Spindle cells within meninges collagenous tissue are transitioning into large granular cell aggregates. Masson's trichrome. Bar = 53 µm.
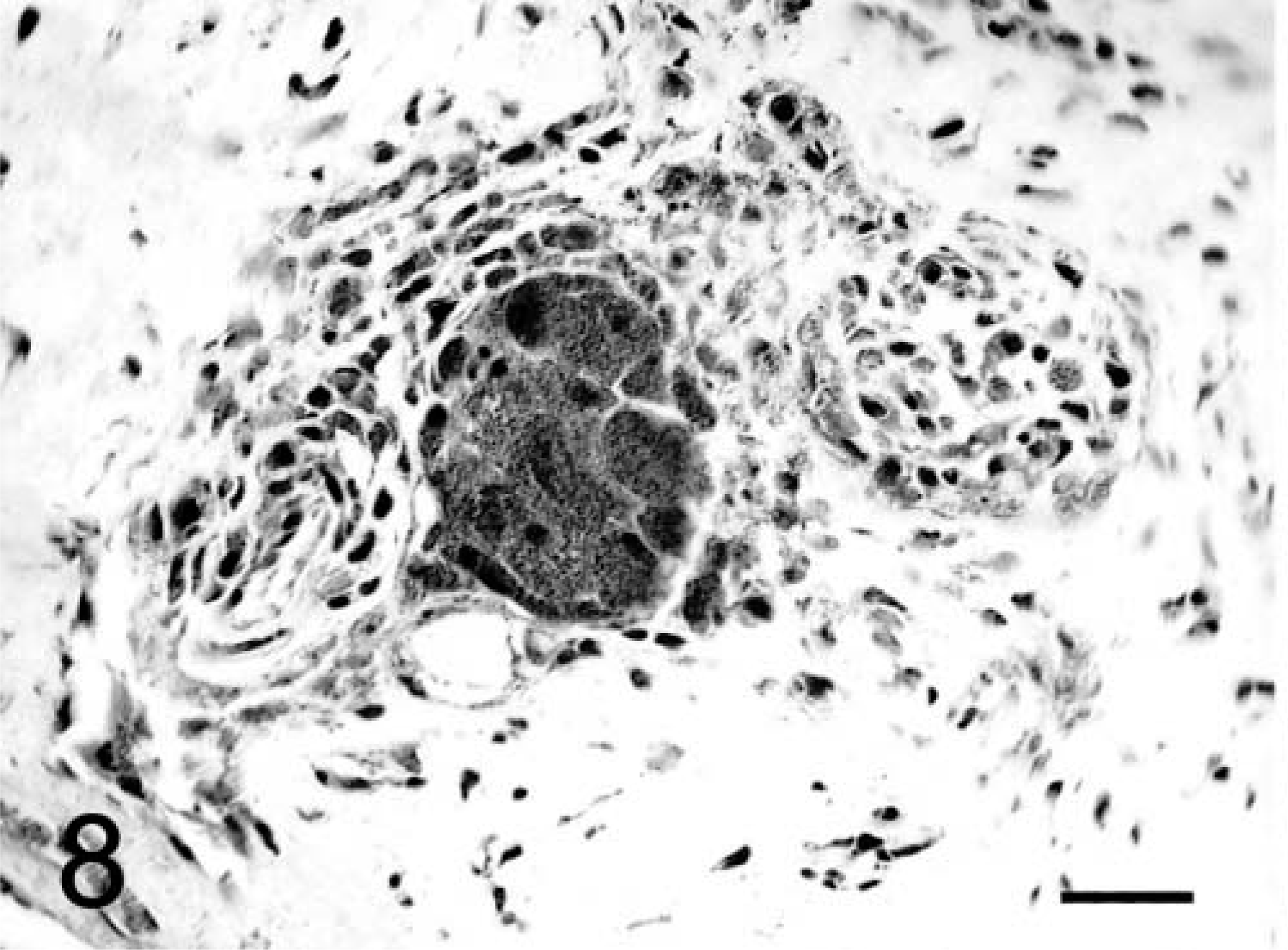
Brain granular cell tumor; dog No. 2. The cytoplasm of both the spindle and large granular cells in the meninges stains strongly positive for ubiquitin. Streptavidin–biotin immunocytochemistry. Bar = 25 µm.
Brain, granular cell tumor; dog No. 2. Border of the tumor with adjacent brain shows strongly positive GFAP staining of reactive astrocytes with the uniformly negative staining granular cells. Streptavidin–biotin immunocytochemistry. Bar = 36 µm.
Ultrastructurally, the cytoplasm of the large granular cells in both cases was packed with autophagosomes containing various types of structures, including residual bodies, large dense irregular granules, sheets of small membrane-bound granules with a dense core and outer halo, multivesicular bodies, empty vesicles, and a few membranous myelin-like whorls (Fig. 10). These single membrane-bound intralysosomal structures were often separated by large empty spaces (Fig. 10). Mitochondria were elongate, bulbous, and sometimes branched and were dispersed with other organelles between the phagolysosomes. The peripherally located nuclei had prominent marginated chromatin. Occasional desmosomal junctions connected adjacent cells. Cytoplasmic membranous pinocytotic vesicles were often numerous. In dog No. 2, the spindle-shaped cells had prominent pinocytotic vesicles and cytoplasmic interdigitations with occasional desmosomal attachments. There were some scattered lysosomes containing mainly myelin-like whorls or zebra-like bodies, some dense bodies, and empty vesicles. There were many extracellular filaments attached to the cytoplasmic surface. Extracellularly, the matrix was composed of these intermediate filaments interspersed with cross-banded collagen. There was no basal lamina around these cells (Fig. 10).
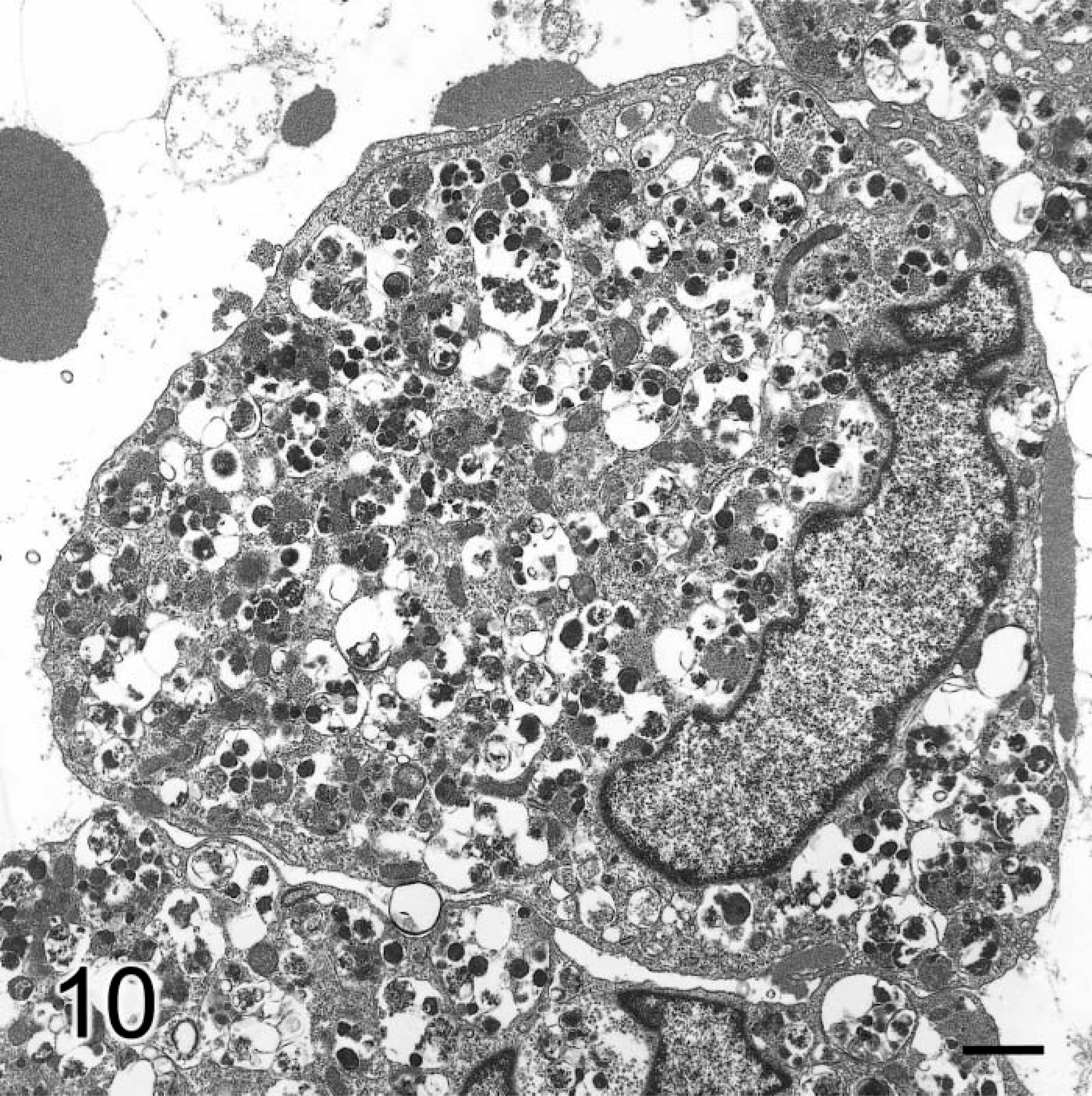
Brain, granular cell tumor; dog No. 1. Electronmicrograph. Granular cell has eccentric nucleus and large numbers of autophagosomes containing multiple dense bodies and other structures. Bar = 1 µm.
Discussion
These two canine CNS GCTs were very similar histologically and ultrastructurally to those intracranial GCTs described in rats, humans, one dog, and one ferret. 2,13,15,19–21,26 The CT and MR images also shared some similarities with those of human GCTs in the CNS. 1,18 However, these canine tumors appear to be of meningeal cell origin and not of pituicyte, oligodendroglial, or astrocytic derivation as in most of the human CNS tumors. 2,10,15,21,26
Both diffuse and heterogeneous contrast enhancement patterns are seen with CT in a few cases of nonhypophyseal intracerebral human tumors. 1 The features of CT and MR images of a neurohypophyseal GCT have also been described. 18 Common findings with T1-weighted MR images include isointense masses with heterogeneous, punctiform contrast enhancement. 1,18 The T-weighted images vary from isointense to hypointense. 1,18 The canine tumors of this report share these features, except that they were hyperintense on T2-weighted MR images. The spectrum of these changes illustrates the difficulty of making a specific tumor diagnosis based on neuroimaging features alone particularly with the small numbers of human and canine tumors examined.
The histogenesis of these canine tumors was not convincingly determined despite the histologic, immunohistochemical, and ultrastructural studies. In humans, similar studies suggest that these GCTs represent a phenotype expressed by a wide variety of cells. 21,24 Most of the human intracranial GCTs occur in the neurohypophysis, often with suprasellar spread, and are thought to be derived from pituicytes. 2,10,26 Clearly this was neither the location nor cell of origin of these canine tumors. However, some human intracranial astrocytomas are composed in part or entirely of granular cells. 11 These canine tumors were not of astrocytic derivation, based on the uniformly negative staining for GFAP expression and on their ultrastructural features. 2,26,27 Both tumors had substantial involvement of the meninges, and the second tumor had multiple foci of spindle-shaped tumor cells that were embedded in a collagen-positive matrix, findings also not consistent with an astrocytoma. Although human oligodendrogliomas with a large granular cell component also have been described, there was no histologic or ultrastructural evidence to support that diagnosis in these canine tumors. 2,5,26
In the second dog, an intriguing finding regarding tumor histogenesis were the clusters or single PAS-positive spindle-shaped cells embedded in islands of collagen, which were separated by sheets of large granular cells. The histochemical staining and ultrastructure of the spindle cells suggest that they progressively change towards large granular cells once the latter are free of the collagenous extracellular matrix. A similar but less obvious pattern was also seen in the meninges of the first tumor. Such a maturation or transition of precursor cells towards large granular cells with ultrastructural features similar to those in these canine tumors has been described in some human CNS and extraneural GCTs. 15,23 In the rat, there is convincing gross, histologic, and ultrastructural evidence suggesting that intracranial GCTs originate from meningeal arachnoid cells and that some of these tumors may also contain mixtures of both cell types. 28 The major meningeal involvement in both these canine tumors suggests that, as in the rat, they may be derived from a cell type forming the leptomeninges. Other support for this hypothesis is provided by a canine meningotheliomatous meningioma in which a substantial granular cell component has been described. 20 Also, the previously reported canine GCT was probably of meningeal origin. 19 Because ultrastructurally the spindle cells had some interdigitating processes, occasional desmosomal junctions, and pinocytotic vesicles as seen in the rat, it is possible that histogenetically they may be of similar meningothelial cell origin. 27,28 There have been only two reports of a suspected meningeal origin for these human tumors, one in the spinal cord and the other in a basilar suprasellar site. 10,15 Some human GCTs have been found within cranial nerves and are suggested to be of Schwann cell origin. 3,4,23 However, in these canine tumors, the lack of any clinical or anatomic association with cranial nerves, of any positive S-100 staining, and of basal lamina formation excludes Schwann cells as the cells of origin. 8,14,23,24
An important finding from this study was that the uniform lack of expression of many canine leucocyte markers by these spindle-shaped cells or large granular cells suggests that these cells are not of macrophage/histiocytic lineage despite their appearance and autophagosomal contents. 17 This evidence supports the hypothesis that the granular cells found in many different types of primary brain tumors reflect some nonspecific metabolic transformation of each tumor cell type. 21 The intense uniform positive staining for ubiquitin in these cells may indicate activation of ubiquitin-dependent protein degradation by cytosolic proteosomes. 6 Alternatively, lysosome-mediated breakdown of proteins can be facilitated by protein–ubiquitin conjugates within the endosome–lysosome system. 6 The precise subcellular location of ubiquitin would require further immuno-TEM examination. In some human GCTs, the variable staining of granules with the anti-proteinases α-1-antitrypsin and α-1-antichymotrypsin has been interpreted as evidence of their histiocytic origin. However, such activity has been found in many other cell types and is therefore of limited cell specificity. 21,24
A major challenge arising from both human and animal GCTs is to understand the basis for the implied difference in histogenesis between the cytologically pure GCT and those various primary CNS and peripheral nervous system tumors with a GCT component.
Footnotes
Acknowledgements
We thank Mr. S. Maslowski for his excellent assistance with the CT-guided stereotactic biopsy procedures and the MR imaging studies, Ms. D. K. Naydan for her immunocytochemical expertise, and Dr. W. G. Ellis for his help with the ubiquitin staining.
