Abstract
Brains from 100 horses, aged 2-25 years, were systematically examined by histopathology at 46 different neuroanatomical sites. The horses were sourced from a slaughterhouse (group A, n = 57), from a kennel that collected dead animals, and from 2 diagnostic laboratories (group B, n = 43). All horses from group A and 26 horses from group B were examined by a veterinarian in the period before death. None of the horses were known to exhibit clinical signs suggestive of neurologic disease. Among the main changes identified were vacuolation in the neuropil (n = 73), neurons (n = 32), white matter (n = 31), and focal perivascular lymphoid cell infiltrates (n = 35). Spheroids were frequently seen (n = 91), and 10 horses each had more than 10 spheroids in the cuneate or gracile nucleus. Statistically significant age-related changes noted included intraneuronal (n = 97) and glial or extracellular lipofuscin deposition (n = 41), hemosiderin deposition around blood vessels (n = 60), and calcium depositions (n = 24). One horse had low-grade nonsuppurative meningoencephalitis; Alzheimer type II cells were detected in the brains of 2 horses. Hyalinized vessel walls in the cerebellum were observed in 1 horse. It was concluded that some histopathologic changes are a frequent feature in equine brains, which has implications for the pathologists involved in equine neurology and disease surveillance.
Knowledge of common background histopathologic changes in the equine brain is critically important for veterinary pathologists who are required to interpret and evaluate these changes. Equally, an understanding of the spectrum of background pathologic change to be expected in the equine brain is important when undertaking neurosurveillance studies, as well as in the identification of emerging or remerging diseases. Recent studies documented incidental neuropathologic changes in sheep, 17 cattle, 13, 21 and pigs. 20 Vacuolation, spheroids, and diffuse inflammatory changes were the main features noted in these species. To date, there are few studies of the background changes in equine brains, with most of these studies focused on a single change. 2, 8, 19, 22, 28 Kikuchi 22 noted the presence of age-related changes in 25 equine brains, including lipofuscin, hemosiderin, and calcium accumulations, whereas Hurst 19 described the occurrence and distribution of calcium deposits in 14 of 18 equine brains examined. Additional studies identified cerebrovascular hemosiderin depositions in the brains of 25 healthy horses. 28 Further, a comparative study reported on cerebrovascular disease in various animal species, including horses. 8 In the latest study, the occurrence of neuroaxonal dystrophy in the accessory cuneate nucleus of 37 horses with neurologic disease was compared with 34 clinically normal horses. 2
The aims of the present study were to record neuropathologic changes in brains from 100 horses of different age groups, none of whom were known to have had neurologic disease and to test their occurrence in relation to the ages of horses involved.
Material and Methods
Brains from 100 horses varying in age from 2 to 25 years old were obtained from a meat factory (group A, n = 57) and 2 disease investigation facilities (n = 26), as well as a kennel (n = 17) where dead horses were received for purposes of feeding to hounds (group B, n = 43). Therefore, group A consisted of healthy horses all of which had passed antemortem inspection immediately before slaughter and who were considered fit for human consumption. Group B included horses who had been diseased and/or had died for various reasons unrelated to neurologic disease. It contained 26 horses who had been under veterinary care. Detailed postmortem findings were available for only 19 of the group B horses. The age of each horse in the study was estimated by examination of the occlusal surface of the incisors of the lower jaw. 25 The horses from both groups A and B were grouped into 4 arbitrary age-range categories to facilitate the determination of age-related changes: 2 to 6 years (n = 21), 7 to 9 years (n = 31), 10 to 15 years (n = 22), and older than 15 years (n = 26). The horses were Standardbred, Thoroughbred or Irish Draft. However, no clinical signs suggestive of neurologic signs or behavioral dysfunction had been reported in any of the horses from either group.
All the horses from group A were shot by a free bullet. Ten group B horses were also shot, and the remainder either died of natural causes or had been euthanized by lethal injection. Brains from group A horses were all removed within a few hours of death; in the case of horses from group B, brains were removed within 24 hours of death. Heads from all of the horses in the study were disarticulated from the carcasses and brought to the laboratory, where the brains were removed either by sagittal sectioning of the skull by using a band saw or by removing the top of the cranium with a hand saw. Brains were fixed in 10% formol buffered saline solution for a minimum of 14 days before processing.
Coronal sections (3 mm) of the cerebrum at the level of the corpus striatum, the thalamus, the mesencephalus at the nucleus ruber, the medulla oblongata at the level of the pons and the obex, and a sagittal section of the cerebellum were taken from each brain, placed in cassettes, and processed to paraffin blocks by using standard histopathologic methods. Sections from each paraffin block cut at 4 μm were stained with Gill-2 hematoxylin and eosin (Thermo, Sheridan, UK; HE). Where appropriate, additional sections were stained by using Perl's blue for hemosiderin, van Kossa for calcium deposits, Periodic acid-Schiff (PAS) for lipofuscin and Congo red for amyloid. Immunohistochemical staining for glial fibrillary acidic protein (GFAP) was performed by using protease (Sigma, St. Louis, MO) 0.05% in Tris buffered saline (TBS) pretreatment and 30-minute antibody (Dako A/S, Glostrup, Denmark) exposure at a dilution of 1 ° 1,000. The streptavidin-horse radish peroxidase (HRP) method was used as the detection system (Dako A/S).
All brains were screened systematically at 46 different neuroanatomical locations, and changes within each location were documented (Table 1). Vacuoles within each anatomical area were enumerated for descriptive purposes and were classified as either neuronal, neuropil, or white matter associated. Where spheroids were present, the number was counted within a particular neuroanatomical area and then was described as singular, less than 10 per area, or multiple, 10 or more per area. Lipofuscin deposits were classified as intraneuronal, intraglial, or extracellular. For purpose of quantification, lipofuscin deposition was arbitrarily graded from grade 1, one third of neurons affected to grade 3 where all neurons affected per anatomical area. The presence of calcium deposits within the blood-vessel walls; gray matter; or white matter; and the presence and locations of any gliosis, satellitosis, perivascular cuffing, or degenerative changes of neurons or vascular structures were also recorded.
∗ The anatomic terminology used here was taken from the Nomina Anatomica Veterinaria, 4th ed., published by the International Committees on Veterinary Gross Anatomical Nomenclature. Ithaca, NY, 1994.
† dors. = dorsales; lat. = laterales; ventr. = ventrales; rostr. = rostralis; tr. = tractus; n. = nervi; med. = medialis; caud. = caudalis.
The findings for the 2 groups and for each of the age categories were compared by using statistical techniques. Results were classified as significant at the 95% level (P ≤ .05) and as highly significant at the 99.9% level (P < .001). For each of the histopathologic changes, chi-square analysis was used to assess the significance of differences noted in the findings for the different age groups. Here, the analysis was based on the number of anatomical areas that were found to have a change present and not on the intensity of the change. A univariate analysis was also performed by using an unpaired Student's t-test to investigate possible associations between the age of the animals and the quantitative presence of lipofuscin depositions. Differences in the findings for groups A and B were also assessed by using the unpaired Student's t-test. The analysis was carried out by using Statistix 8 (Analytical Software, Tallahassee, Florida, USA).
Results
The average age of the horses in this study was 11 (± 6) years. Twenty-one horses were between 2 and 6 years of age, 31 horses were between 7 and 9 years of age; 22 were between 10 and 15 years old, and 26 horses were older than 15 years. The age distribution was similar for group A horses from the meat factory and group B horses from 2 disease-investigation facilities and the kennel. The only macroscopic change was seen in those horses euthanized by shooting, where hemorrhage in the mid brain or the brainstem was consistently present.
Neuropil vacuolation was common, and it was seen in 73 of 100 horses (Table 2). More than 2 areas of the central nervous system (CNS) were affected in 31 horses. The vacuoles were variable sized, numbered 1 to 5 per affected anatomical area and were most frequently observed in the nucleus motorius n. oculomotorii (52/73) (Fig. 1). Significantly more horses in group B than in group A were found to have neuropil vacuolation (P < .05). Neuronal vacuoles were observed in 32 of 100 horses and were most commonly present in the Purkinje's cells (10/32) and in the large neurons of the nucleus tr. mesencephali n. trigemini (10/32) (Fig. 2). White matter vacuoles, seen in 31 of 100 horses, were primarily found in the capsula interna (17/31). Neuronal and white matter vacuoles generally affected only 1 or 2 anatomical sites per horse (28/32 and 26/31, respectively). Frequencies of the different types of vacuolation were unrelated to age (Table 2).
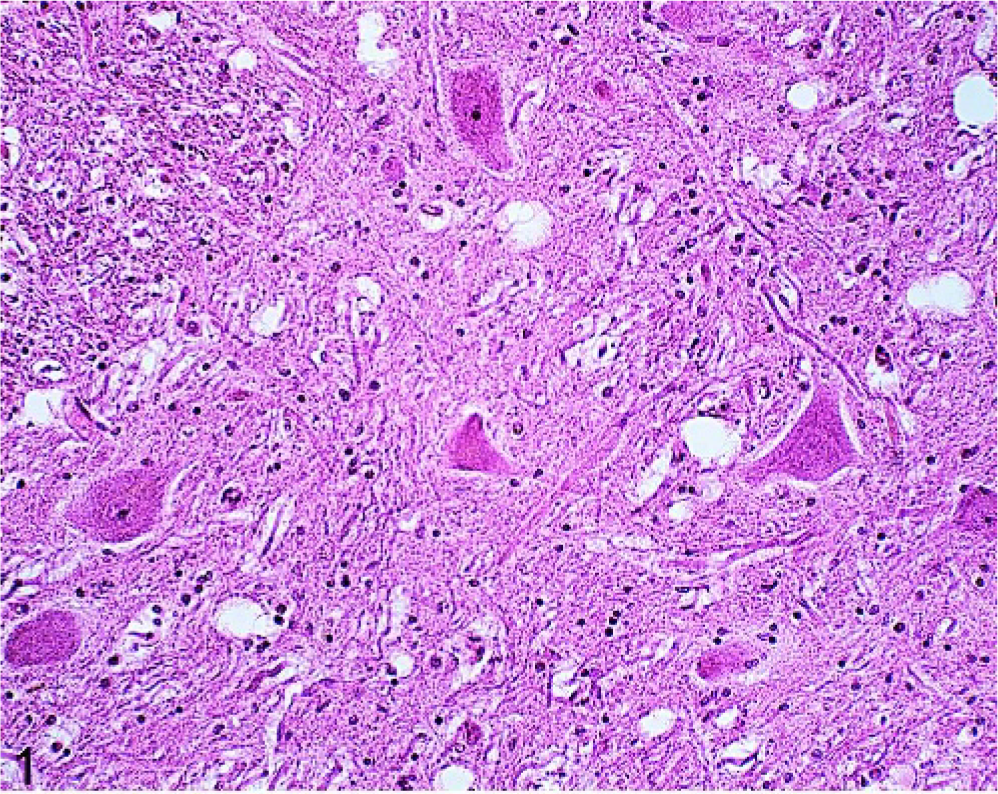
Brain; oculomotor nucleus; 18-year-old horse (No. 60). Discrete neuropil vacuolation with more than 10 vacuoles per nucleus. HE.
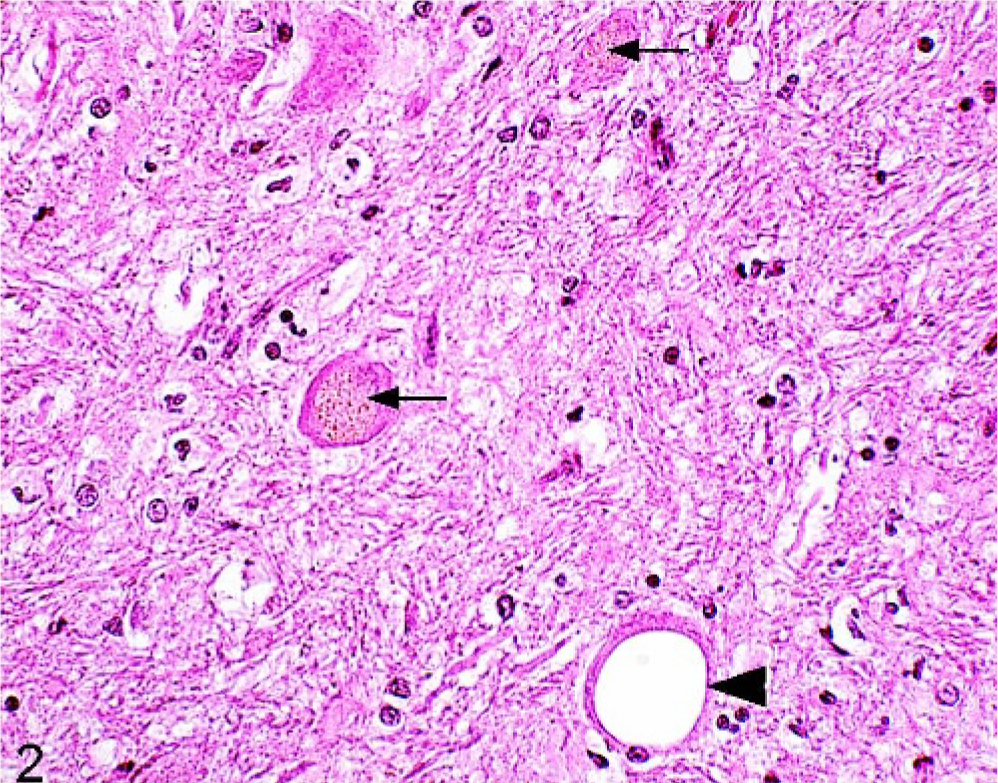
Brain; raphe at the level of the obex; 20-year-old horse (No. 73). One vacuolated neuron (large arrow) and intraneuronal lipofuscin depositions (small arrows). HE.
Histopathologic findings in the brains of 100 horses with no histories of neurologic disease. The horses were obtained from a meat plant(n = 57) and from routine submissions to diagnostic laboratories (n = 43). The age ranged from 2 years to old age.

∗ n. = nervi; dors. = dorsalis; tr. = tractus; trig = trigemini; lat. = lateralis; ret = reticularis; corp. = corporis; No = not significantly related to age; yes = significantly related to age; ND = not analyzed.
Spheroids, representing swollen axons, were seen in nearly all the horse brains (91/100). In general, spheroids were more common in younger horses, but this finding was not statistically significant. However, where multiple spheroids, more than 10 per anatomical area, were found, horses had a higher average age. Spheroids were common in the brainstem particularly in the nucleus funiculus lat. (55/91). Nevertheless, they generally occurred as single spheroids in different anatomical areas per affected brain (66/91). Neuroaxonal dystrophy 2 was found in 10 affected brains. This was consistent with multiple spheroids, more than 10 per area, in either the nucleus gracilis or the nucleus cuneatus. In some instances, the spheroids were associated with coalescing vacuoles in the surrounding gray and white matter, and some spheroids were also vacuolated. Where multiple spheroids were present, gliosis and brown pigment depositions was also a feature in the affected nuclei (Fig. 3).
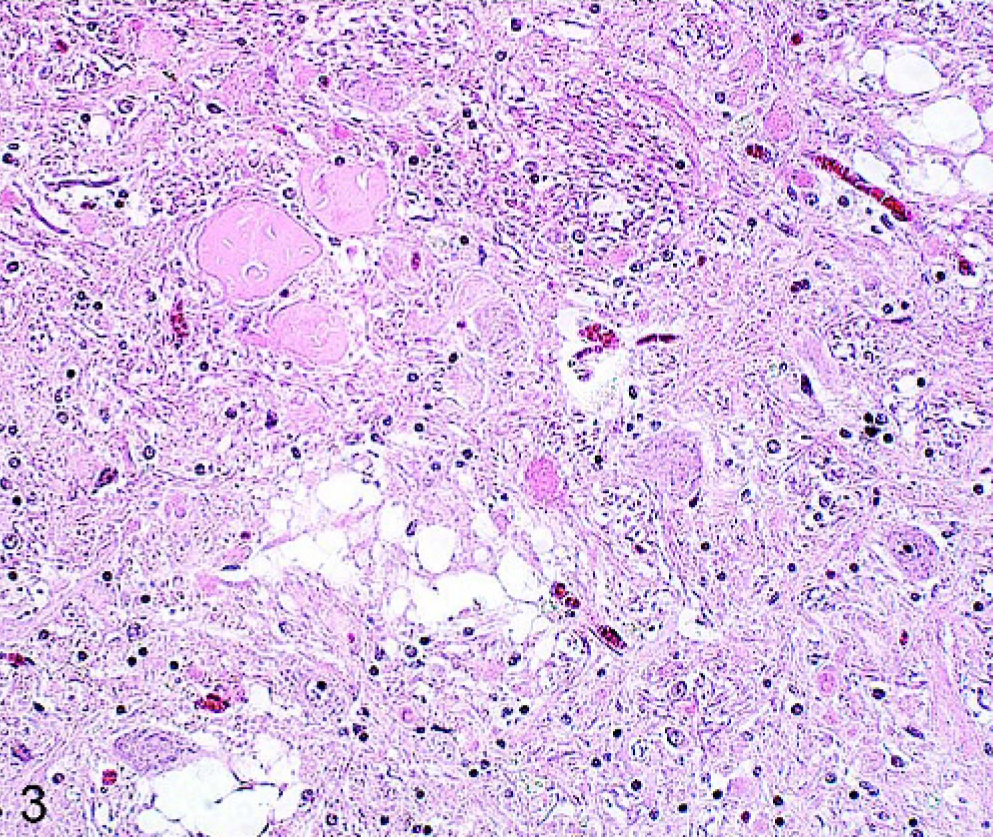
Brain; cuneate nucleus; 21-year-old horse (No. 39). Multiple spheroids accompanied by vacuoles and pigment. HE.
Lipofuscin deposition was common in both neurons and neuropil (97/100 and 41/100, respectively) (Fig. 2). The most common sites for neuronal lipofuscin deposition were the nucleus tr. mesencephali n. trigemini (85/97) and the nucleus ruber (75/97). Lipofuscin deposited in the neuropil was either in the cytoplasm of glial cells or more frequently occurred as free granular accumulations that stained weakly positive for PAS. Overall, the number of areas affected by lipofuscin depositions increased significantly with age (P < .002), as did the intensity of the deposits (P < .001). The 4 horses unaffected by lipofuscin depositions were between 2 and 4 years of age.
A red granular intraneuronal pigment was seen in 6 of 100 horses. The pigment, which did not stain with PAS, has not been reported in previous studies. 3, 8, 21, 27 It was mainly observed in the midbrain (4/6), in the substantia nigra, and in a lateral nucleus. In 2 cases, it was seen in the thalamus (Fig. 4). The nature of the pigment was not identified.
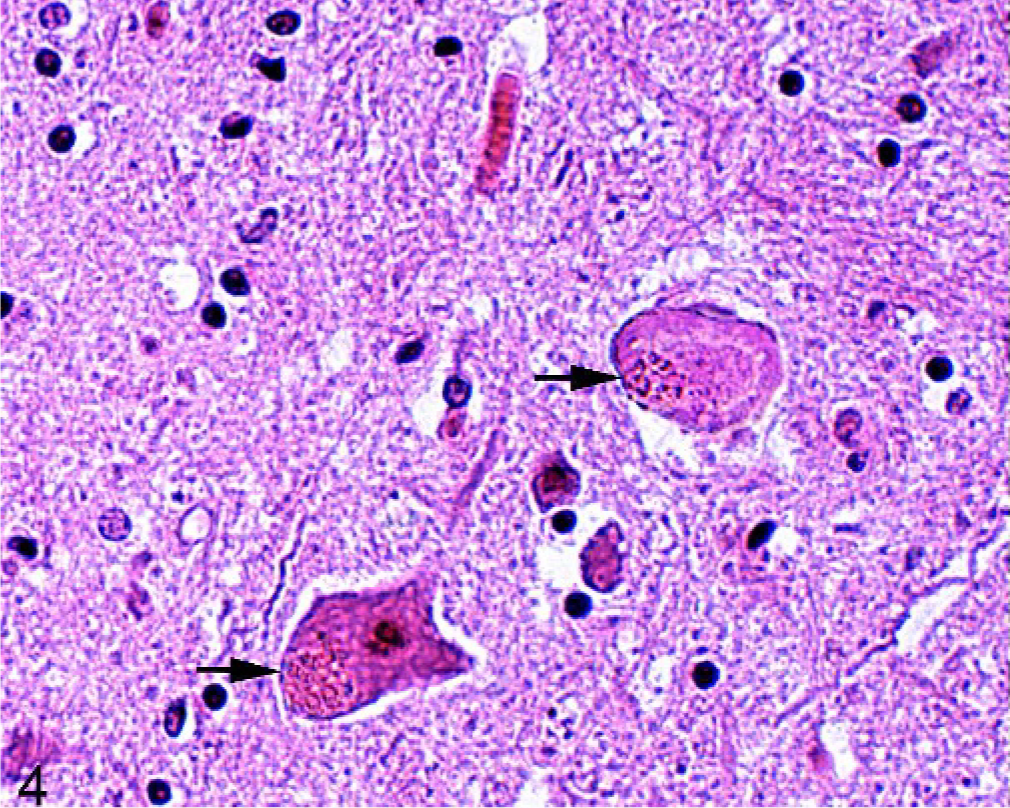
Fig. 4. Brain; lateral aspect of the midbrain; 4-year-old horse (No. 36). Two neurons with introcytoplasmic red pigment (arrows). HE.
Hemosiderin was noted in 60 of 100 animals and was found in macrophages (Fig. 5). Deposits were noted primarily in perivascular spaces in the white matter of the tel- and diencephalon. Significant more of the older horses (P < .001) and horses from group A were affected (P < .05).
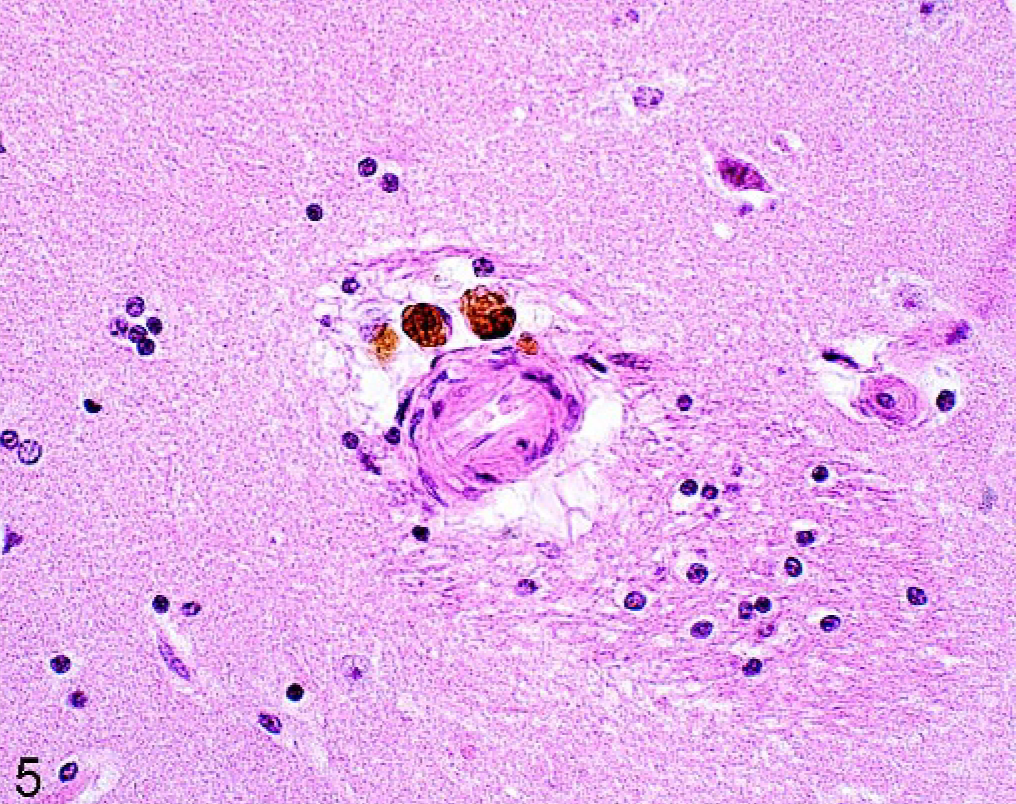
Brain; caudate nucleus; 20-year-old horse (No. 49). Hemosiderin as brown pigment in macrophages in the perivascular space. HE.
Heavy calcium accumulations were predominantly seen in the rostral part of the brain in the region of the capsula interna (7/24) and externa (7/24). Significantly more calcium accumulations were observed in older horses (P < .05) and in horses from group A (P < .05). Calcium deposits were seen mainly in vessel walls and in adjacent gray and white matter (24/100) (Fig. 6). The degree of vessel-wall involvement ranged from small globular areas within the tunica media of the vessel wall to the obliteration of the entire vessel by coalesced aggregates of calcium. The severity and the extent varied from very low, with only 1 vessel affected, to cases where larger areas of the brain were involved and at least 10 vessels in a particular brain area were affected. In 1 horse (No. 8), calcification was observed in the blood vessels of the ependyma. Calcium was also found as single deposits in gray and white matter, without any vessel involvement. These accumulations varied in size and occasionally coalesced.
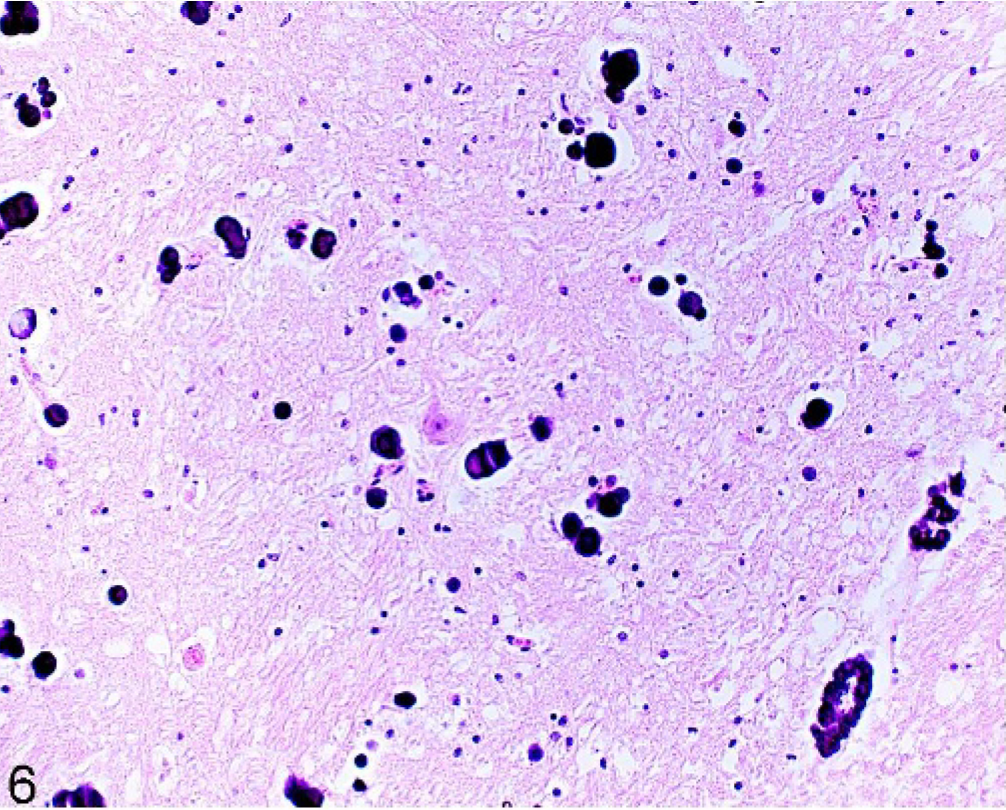
Brain; globus pallidus in crus cerebri, thalamus; 25-year-old horse (No. 95). Circular globular calcium deposition in vessel walls and neuropil. HE.
Perivascular cell infiltrations and gliosis were seen in 35 of 100 and 29 of 100 horses, respectively. These changes were most common in the cerebrum. The majority of horses had small localized lymphocytic perivascular accumulations in areas unassociated with gliosis. One horse (No. 35) from group A had nonsuppurative meningoencephalitis. Here, variable-size foci consisting of thick mononuclear perivascular cuffs were accompanied by marked gliosis, including multinucleated cell accumulations in gray and white matter throughout the brain. The most severe lesions were in the nucleus caudatus (Fig. 7). Mononuclear cell infiltrations in the meninges mainly associated with larger meningeal vessels were seen in 4 horses. In these cases, leucocyte margination was noted in meningeal vessels.
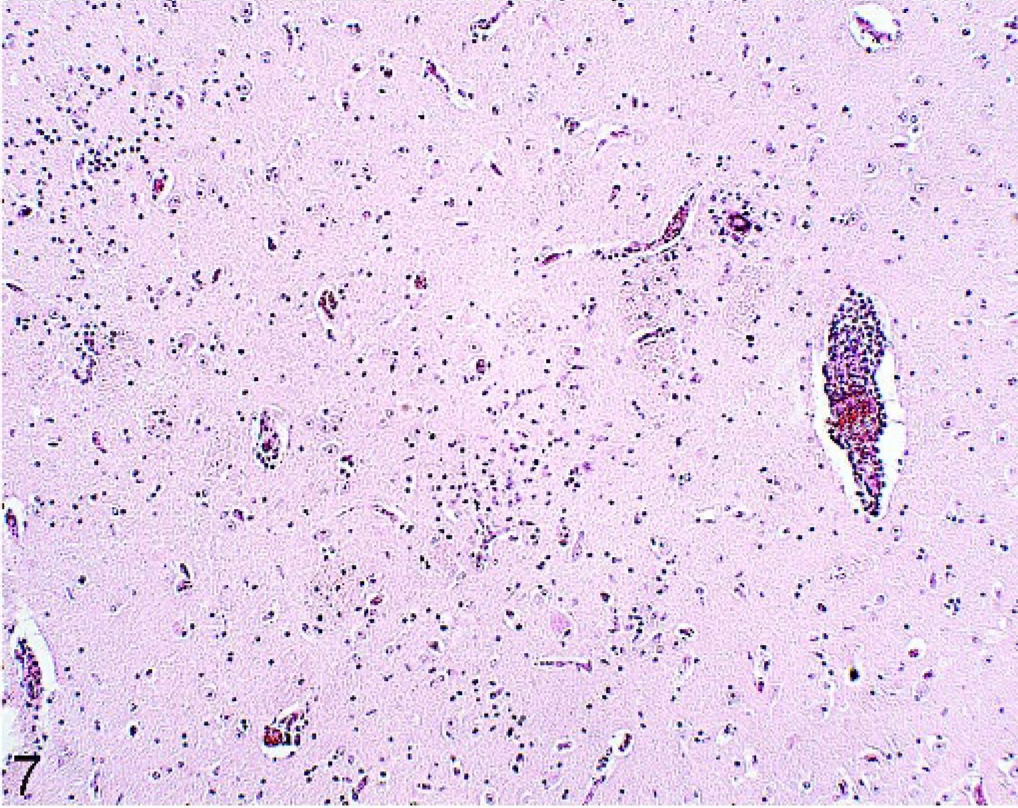
Brain; caudate nucleus; 17-year-old horse (No. 35). Perivascular cuffing and gliosis. HE.
Swollen astrocytes with a clear, sometimes indented, nucleus and hyperchromatic margins, typical of Alzheimer type II cells (Fig. 8) were present in 2 horses (Nos. 93, 96) from group B. Although the Alzheimer type II cells could be found singly, they were mainly in groups of 2 to 6 predominantly in the cerebral cortex. In 1 animal (No. 93), they were also noted in small numbers in the corpus striatum and thalamus, while in the other affected horse (No. 96), they could be detected throughout the gray matter of the entire brain. Mild edema and gliosis were also present in the cerebral cortex of the latter horse. GFAP staining of the astrocytes in the affected horses was considerably reduced in the gray and white matter compared with a normal horse (No. 51). Horse No. 96 was 5 years of age and had suffered from severe peritonitis and necrosis of portion of the wall of the small intestine. The second horse (No. 93) was 10 years of age and had a history of previous colic surgery and chronic peritonitis. Neurologic signs were not reported in either horse.
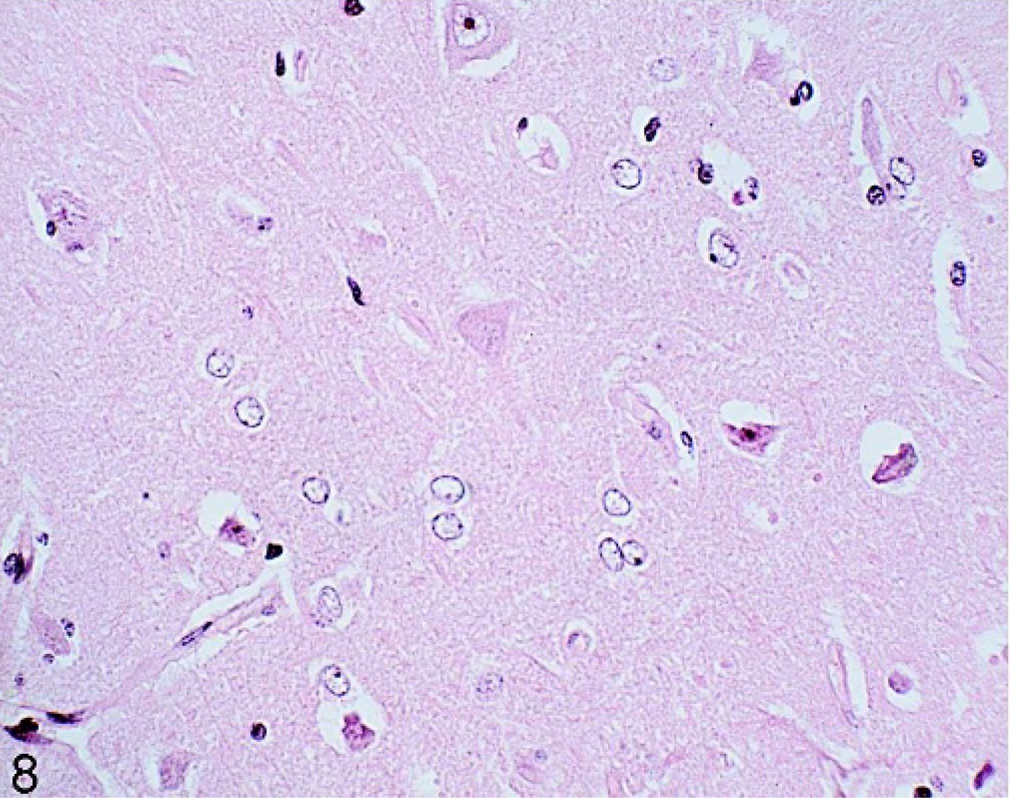
Brain; frontal cortex; 5-year-old horse (No. 96). Astrocytes with swollen vacuolated nuclei and chromatin dispersed to the periphery, Alzheimer type II cells. HE.
Light pink homogenous amorphous material was observed in the wall of capillaries in the stratum moleculare and granulosum of the cerebellum (Fig. 9) in 1 horse (No. 14). The lesions were focal and involved about 6 foliae. The basal membrane of affected vessels was intact, and the endothelial cells were hyperplastic. The amorphous material stained negative for amyloid with Congo red and was negative on PAS stain. The molecular layer was also infiltrated with lymphocytes and glial cells, but the vessel walls were spared. Extensive hemosiderin deposits were seen in the molecular and granular layer in close vicinity to the affected vessels. The Purkinje's cell layer was present but in the affected areas was disrupted. There was no clinical history available for this 15-year-old horse, which was collected from the kennel.
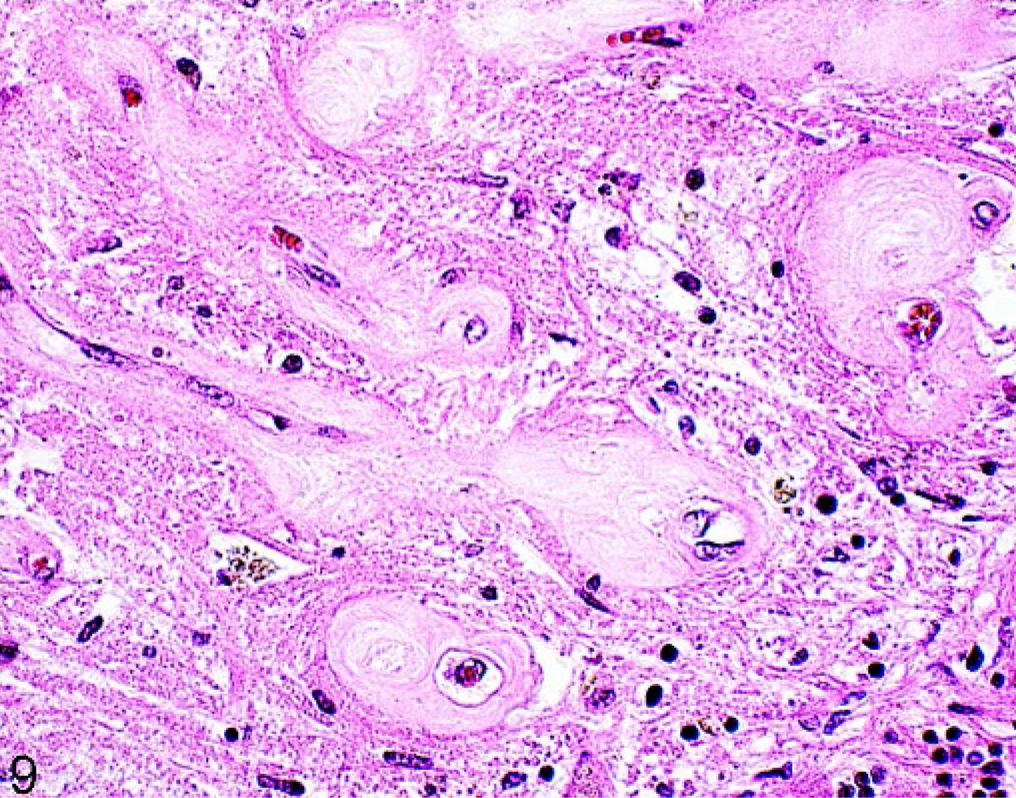
Brain; cerebellum; 15-year-old horse (No. 14). Pale pink amorphous material in the wall of vessels in the molecular layer of the cerebellum, brown hemosiderin deposition present. HE.
A single layer of neuroepithelial cells on the surface of the stratum moleculare of the cerebellum, resembling the fetal external granular layer, was observed in animals up to 8 years of age.
Discussion
The main non-age–related changes included vacuolation in neurons, neuropil, and white matter, as well as the presence of spheroids. The age-related changes noted consisted of intraneuronal and neuropil lipofuscin deposition and deposits of hemosiderin and calcium. Infiltrates of lymphoid cells in perivascular spaces, nonsuppurative meningoencephalitis, Alzheimer type II cells, and degenerative vascular changes were occasional observations.
Pathologic changes, some of which were age related, were identified in the brains of all 100 horses throughout group A and group B. Eighty-three of the horses had been under veterinary care before death and did not exhibit neurologic disease. The changes observed in the brains of the 17 horses without a clinical history were similar to the 83 examined. Therefore, the findings of this study are thought to be representative for horses without clinical neurologic disease.
There were no consistent differences in the distribution and frequency of non-age–related findings in horses that were collected from the slaughterhouse (group A) and that were considered healthy and the horses collected from disease investigation centers and a kennel (group B), which were presumed to have underlying diseases other than neurologic disease. The fact that the non-age–related changes were very common throughout both groups A and B presents compelling evidence that such changes are present in equine brains, regardless of clinical history. In addition, our material and methods compare well with previous studies on incidental and age-related neuropathologic changes in a variety of species. The examined populations consisted of apparently healthy slaughtered animals, such as horses, 22, 28 pigs, 20 sheep 29 or cattle. 35 Other investigators examined populations of horses, 19, 22 sheep, 17 or dogs 4 from disease investigation centers or groups of apparently healthy cattle, 13 sheep, 37 and dogs. 26 All of these animals were considered free of neurologic signs, although no specific neurologic examination had been performed.
The statistical analysis of the histopathologic changes in relation to age was based on the presence or the absence of the change per anatomical area and not on the intensity of the change in an area, because the quantification of the finding per area was confounded by the varying sizes of the nuclei when sectioning the brains manually. Quantities were, therefore, for descriptive purposes only.
Neuronal, neuropil, and white matter vacuolation have not been previously reported in horses, despite the frequency with which they were seen in the present study as a non-age–related finding. 2, 8, 22, 28 Establishing the location and the extent of neuronal vacuolation in animal populations is of extreme importance in the light of surveillance of all species for transmissible spongiform encephalopathies. Neuronal vacuolation is frequently found in apparently healthy cattle, 13 in sheep, 37 and in pigs, 20 but they have not been documented in horses before this study. 2, 8, 22, 28 The findings for the horses studied here was very similar to that seen in sheep, where the number of vacuoles per anatomical area is usually very low. 36 Interestingly, the anatomical distributions of what are presumably incidental neuronal vacuolation differ in the different species. In the horse, the main areas involved were the mesencephalic trigeminal nucleus, the Purkinje's cells, and the neurons in the raphe at the level of the obex. In contrast, in cattle neuronal vacuolation was observed mainly in the red nucleus, the habenula, and the formatio reticularis 13 ; in the medulla in sheep 37 ; and in the dorsal vagal nucleus in pigs. 20 Hooper 17 reported that increasing prevalence of neuronal vacuoles in sheep was age related. In contrast, in the present study of horses, no age relation could be found.
The oculomotor nucleus was the main site affected by neuropil vacuolation in horses. Neuropil vacuolation occurs frequently in the rostral colliculus of adult pigs 20 and is more randomly distributed in cattle 13 and sheep 29 brains from clinically normal animals. It is unclear whether neuropil vacuolation contributes significantly to clinically detectable reduced neural function. However, neuropil vacuoles were significantly more often seen with horses in group B, in which there would have been a delay in removing the brain rather than with horses from group A. This suggests a link of vacuolation with autolysis, though not exclusively, and in part supports the theory that autolysis may predispose brain tissue to artifactual vacuolation. 31 However, it was also suggested that suboptimal fixation may contribute to neuropil vacuolation, 31 all brains studied here were fixed in adequate volumes of 10% formol saline solution for at least 14 days.
White matter vacuolation in the horses reported in the present study occurred mainly in the internal capsule of the basal ganglia. Similar findings have been reported in cattle 13 and in pigs, 20 but these investigators reached no conclusion as to their significance. Vacuolation of white matter has been associated with congenital and acquired degenerative diseases, but the most commonly attributed reasons include hepatoencephalopathy and uremia, where the vacuolation is distributed at the junction of gray and white matter. 15, 30, 32 However, white-matter vacuoles in uremia often occur in large numbers not confined to any specific location within the CNS. 18
Spheroids are swollen axons and can result from diverse insults, including trauma, hypoxia, intoxications, nutritional deficiencies, and storage disorders. 33 In the present study, these usually occurred singly in the gray matter of brainstem nuclei. No changes were observed that could give any indication of the origin of the swollen axons. They have been reported as frequent incidental findings in sheep, 17 cattle, 13 dogs, 4 pigs, 20 man, 5 and horses 2 with no history of clinical neurologic disease. In the present study, single spheroids were more common in younger horses, and multiple spheroids were more common in older horses. However, this difference was not statistical significant. Similarly, Beech 2 was unable to relate the occurrence of multiple spheroids in the cuneate nucleus in 34 normal horses to aging. Nevertheless, in studies on dogs 4, 33 and humans, 10 the occurrence of spheroids is apparently related to increasing age.
The multiple spheroids, which were observed in 10 horses, were confined to the cuneate and gracile nucleus. These 2 nuclei receive stimuli through the spinal cord by long dorsal fibers from the areas of the hind limbs and forelimbs. 3 They have been associated with neuroaxonal dystrophy, without causing neurologic signs in humans, 10 dogs, 26 and horses. 2 In contrast, the lesion in this location was also found in horses exhibiting neurologic signs such as ataxia from vitamin E deficiency, 1 equine degenerative myeloencephalopathy 24 and in association with gait abnormalities in Morgan horses 2 and in Suffolk sheep. 6
Intraneuronal lipofuscin depositions were significantly observed in almost every horse in the present study and the quantity of the pigment and the number of areas affected increased with increasing age. Similar observations were made previously in horses 22 and dogs. 4, 34 In young horses in the present study, intraneuronal lipofuscin was largely confined to the mesencephalic trigeminal nucleus, the red nucleus, and the vestibular nucleus. This observation contrasts with findings in other equine studies, 22 where the olivary nucleus and the pyramidal cells of the cortex were the regions affected in young horses. In dogs, the neurons of cranial nerve nuclei and the pyramidal neurons appeared to be the earliest affected. 4 Although, it was suggested that the anatomical distribution of lipofuscin is related to the activity of the nuclei, 34 the differences seen between this study, and the others are most likely because of the variations in methodologies.
Age-related glial and neuropil lipofuscin depositions were confined to the more rostral part of the brain. 22 Lipofuscin depositions are caused by an intracellular accumulation of cell debris after autophagic lysosomal degeneration. 7 Finding this pigment in neuropil may be caused by occasional exocytosis. 7 Another theory is that glial cells containing lipofuscin pigment become pyknotic and die, and afterward only the extra cellular deposit can be observed. 22 The finding in the present study, that lipofuscin depositions unassociated with cells were only observed in horses over 4 years of age, is consistent with this latter theory.
The presence of a PAS-negative intraneuronal granular red pigment in the midbrain and the thalamus has not been previously reported in horses. 2, 8, 22, 28 These may be similar to the small intraneuronal eosinophilic inclusions that have been described in normal human brains in the substantia nigra, the locus coerulus, and the thalamus. 16 It was suggested that these structures consisted of distended elements of the rough endoplasmatic reticulum and were related to aging. 16
Hemosiderin was apparent frequently within macrophages in perivascular spaces, particularly in the cerebral white matter tracts. This distribution is similar to that reported by others. 23, 29 These investigators found that hemosiderin was common in brains from clinically normal horses but was rarely seen in the brains of other domestic animals. 29 In the present study, it is very likely that the hemosiderin originated from microhemorrhages, although no changes in the vessel walls were observed.
Cerebrovascular calcium depositions occur most commonly in horses 8 but are also found in cattle, cats, mice, and dogs. 8, 13, 23, 35 In the present study, such deposits were observed mainly in the rostral part of the brain in vessel walls and in the adjacent tissue. This finding is similar to studies in horses and cattle. 8, 19, 22, 35 In cats, however, Mandara et al. 23 reported foci of calcification throughout the brain. The pathogenesis of the calcified foci seen in the present study is not entirely clear, because vessel-wall changes were not observed. Although calcium depositions were detected in horses as young as 2 years of age, there was a significant age relation in this study. In cattle, calcium depositions were also often seen in older animals. 13, 35
Only 1 case of meningoencephalitis, with no apparent neurologic signs, was found in a 17-year-old horse collected from the slaughter house. The cause of these multifocal changes could not be determined, but the nature and the distribution of the lesions are somewhat suggestive of a protozoal infection.
In other cases, sporadic perivascular nonsuppurative lymphoid cell infiltrates were mainly seen in the frontal cortex, a finding that is also common in cattle, 13 in sheep, 17 and in adult pigs 20 without any history of clinical neurologic disease.
In the present study, Alzheimer type II cells were observed in the brains of 2 horses. They have been reported in horses affected by renal failure, 9 liver disease, 32 and gastrointestinal dysfunction. 14, 27 The cells are characterized by a reduced GFAP expression, 32 as was the case here. However, the cause of these changes could not be found.
The pathogenesis of the hyalinized vessel walls in 1 horse in the cerebellum could not be established. Similar vascular cerebellar lesions in the horse have not been reported previously, either as a congenital defect or as an acquired disease. However, age-related vascular changes have been described in humans consisting of thickening of vessel walls, fibrosis, and hyalinization. 12
Interestingly, in the present study, some horses up to the age of 8 years frequently had a single layer of neuroepithelial cells on the cerebellar surface. The fetal external granular layer, which is located immediately subjacent to pia, is depleted in humans in the first year of life. 11 This has also been documented as a feature of the brains of young animals 32 but not as a reminiscent feature in the adult animal, as seen in the horses here.
The examination of a large number of horses in the present study identified an extensive range of histopathologic changes. As a result, some of these common changes can now be categorized as non-age–related findings in the horse. It was concluded that some histopathologic changes are a frequent feature in brains from clinically normal horses. The location and frequency of such lesions constituted important knowledge required by neuropathologists, particularly when involved in disease surveillance and the emergence of new-disease entities.
Footnotes
Acknowledgements
We would like to thank Donal Minihan for assisting with the statistical analysis. We thank Ursula Fogarty, Irish Equine Centre, Naas for the provision of horses' heads and their clinical history. We also thank the kennels in Kildare and the meat factory in Straffan for their cooperation. Professional and technical assistance was given by Anne Marie O'Donoghue, Brian Cloak, and the staff in the Histopathological Section in the Central Veterinary Research Laboratory, Abbotstown, Dublin. The study was funded by the Irish Department of Agriculture and Food with special thanks to Dr. Michael Gunn.

