Abstract
A flock of Rambouillet sheep was examined because of increased lamb mortality caused by ineffective hemostasis at parturition. Neonatal-affected lambs presented with inadequate hemostasis at the umbilicus, pale mucus membranes, and markedly prolonged activated clotting time. Affected lambs had consistently prolonged 1-stage prothrombin times and activated partial thromboplastin times that supported a defect in the common pathway or defects in both the intrinsic and extrinsic pathway of the coagulation cascade. Decreased activity of vitamin K-dependent procoagulant factors II, VII, IX, and X in male and female lambs suggested either a defect of the hepatic enzyme γ-glutamyl carboxylase, or vitamin K1 2,3 epoxide reductase. Affected lamb hepatic γ-glutamyl carboxylase activity was markedly decreased compared with that of age- and sex-matched control lambs, while vitamin K1 2,3 epoxide reductase and glucose-6-phosphatase activities were similar between an affected and normal lamb. Subcutaneous vitamin K1 supplementation did not increase vitamin K-dependent procoagulant factor activities in 3 lambs administered vitamin K1 daily. These data confirm defective γ-glutamyl carboxylase activity as the cause of impaired coagulation of sheep in this flock. This flock represents the only viable animal model of hereditarily defective γ-glutamyl carboxylase activity.
Introduction
Vitamin K is a cofactor for the posttranslational enzymatic reaction in which γ-glutamyl carboxylase converts glutamic acid (Glu) residues to γ-carboxylated glutamic acid (Gla). 30, 31 Specifically, gamma carboxylation is the addition of a carboxyl group to the gamma carbon of Glu 12 and occurs on the inner surface of the endoplasmic reticulum. 8 Proteins that share this processing before being secreted are referred to as vitamin K–dependent (VKD) proteins. Posttranslational modification occurs for VKD proteins involved in hemostasis, bone metabolism, mineralization, growth control, signal transduction, and cell survival. 4, 6, 13, 24 Gamma carboxylation allows calcium binding and activation of VKD proteins II, VII, IX, X; protein C; protein S; 13 and osteocalcin-hydroxyapatite binding in bone.
The energy for this reaction is provided through the oxidation of vitamin K hydroquinone (K1H2) subsequent to binding the propeptide region of VKD proteins to Gla (Fig. 1). 5, 12 Calcium-binding Gla residues are formed from Glu residues, and K1H2 is converted to vitamin K1 2,3 epoxide. The vitamin K1 2,3 epoxide is reduced to vitamin K1H2, in a 2-step process by vitamin K epoxide reductase, an enzyme sensitive to inhibition by warfarin and other vitamin K antagonists. γ-Carboxylation of coagulation proteins allows them to fold properly and bind receptors to participate in a series of reactions that either regulate or promote the formation of a stable fibrin clot. 11
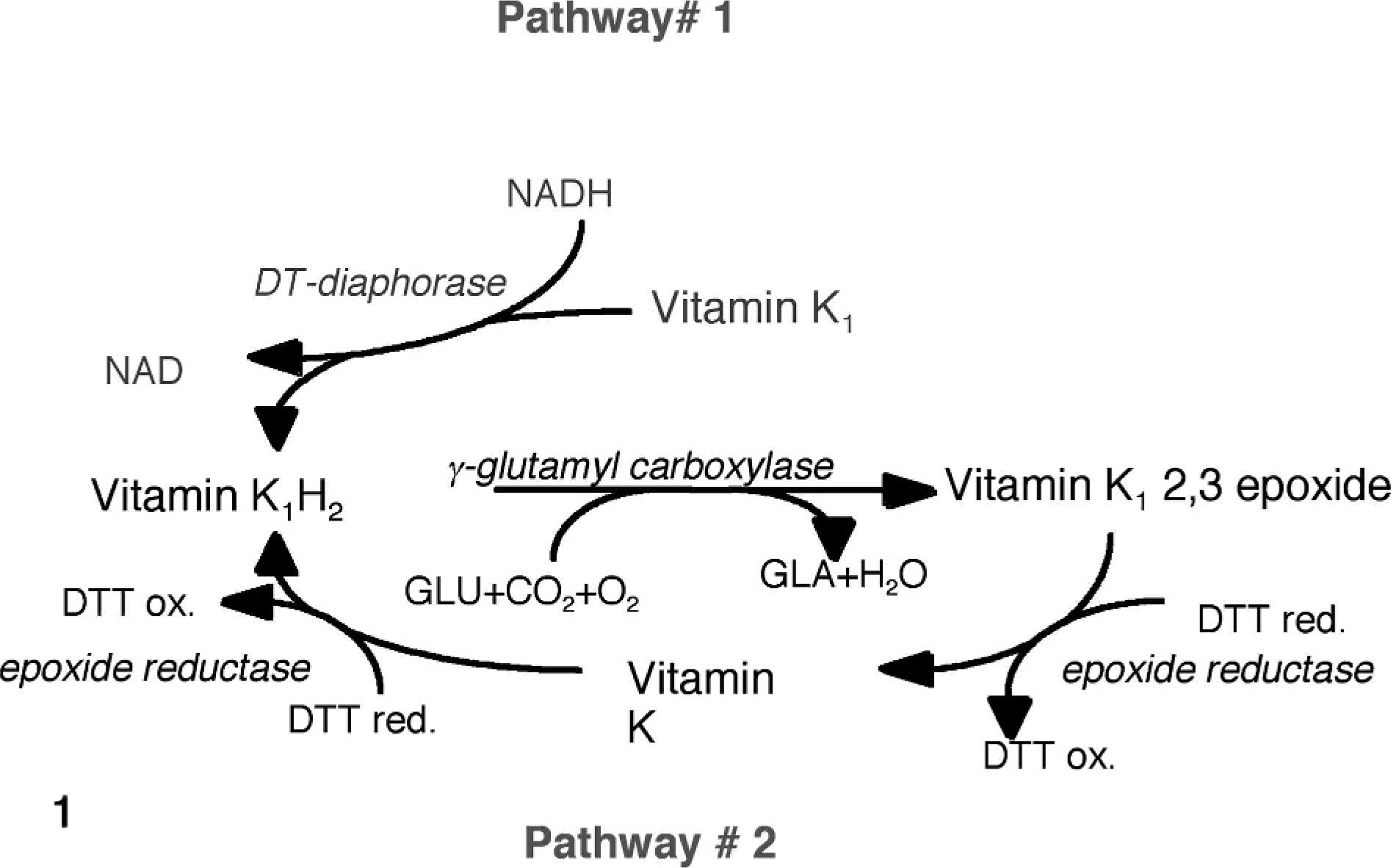
The vitamin K cycle. DTT = dithiothreitol. Glutamic acid (Glu) is carboxylated to γ-carboxyl glutamic acid (Gla), and vitamin K1H2 is oxidized to yield H20 and vitamin K1 2,3 epoxide. Vitamin K1 2,3 epoxide is reduced to vitamin K1H2 by pathway #2 at physiologic concentrations of vitamin K by epoxide reductase possibly by using DTT as an electron donor and by pathway #1 by an NAD(P)H reductase (DT-diaphorase) at supraphysiologic concentrations of vitamin K. 33 Pathway #2 is inhibited by warfarin. Pathway #1 is not.
We report a coagulopathy because of defective Gla activity in Rambouillet sheep similar to that described in human beings. 6, 7, 10, 15, 20, 23, 26 A preliminary report that investigated this sheep flock demonstrated selective reduction of VKD coagulation factor activities and eliminated vitamin K antagonism or the presence of a circulating inhibitor as a cause for bleeding. 1 Affected lambs were born alive but lacked the ability to achieve hemostasis of the umbilical artery and vein. Without intervention, newborn lambs continually bled from the umbilicus and had extensive subcutaneous hemorrhage at sites of previous venipuncture or mild trauma, and body cavity hemorrhage (hemothorax, hemoabdomen, and hemarthrosis), resulting in death. Breeding data indicated the coagulopathy was inherited as an autosomal recessive trait in sheep as in human beings, because approximately equal numbers of male and female affected lambs were observed, and the first report yielded 4 affected lambs of 15 lambs born (approximately 25%) to carrier ewes and a carrier ram.
The purpose of this study was to determine the underlying molecular defect in clinically affected lambs. We examined the relation of vitamin K oxidation and reduction because of the suggestion of altered vitamin K metabolism from the coagulation factor activities in a previous report and what is known about vitamin K (Fig. 1).
Materials and Methods
All reproductively active ewes were synchronized with vaginally inserted progesterone implants (Eazi-Breed CIDR; Pfizer, Groton, Connecticut) followed by removal at 17 days and injection of 500 units of pregnant mare serum (Sioux Biological Sioux Center, IA). The ewes were turned into a pen with a ram known to be heterozygous for the observed coagulation defect and that had been fitted with a marking crayon. All ewes were observed daily for marking by the ram, and delivery dates were calculated from the last known breeding date. Ewes were monitored 24 hours a day near the expected parturition dates to identify, sample, or treat affected lambs immediately after birth.
Coagulation parameters
An activated clotting time (ACT) was determined for each newborn lamb to identify clinically affected bleeding prone lambs. The ACT was performed within 1 hour of birth by using a Vacutainer (Becton Dickinson, Franklin Lakes, NJ) containing 6–10 mg of siliceous earth. Individual tubes were prewarmed to 37°C. Lambs with an ACT longer than 5 minutes were considered affected and were transfused with 120 ml of freshly thawed and prewarmed sheep plasma after blood was collected for evaluation. Affected lambs were sedated with 0.1 mg/kg subcutaneous xylazine, and the jugular vein was catheterized with a 22-gauge Insyte catheter (Becton Dickinson, Franklin Lakes, NJ). Plasma was slowly administered by using a 60-ml syringe attached to an extension set. Blood was collected for evaluation in 3.2% sodium citrate anticoagulant containing tubes for use in coagulation studies immediately before plasma infusion. These plasma samples were either immediately frozen and stored at -80°C or stored on ice, and coagulation studies were performed within 2 hours of blood collection. Prothrombin and partial thromboplastin times were determined with an MLA Electra 700 coagulation analyzer (Pleasantville, NY) and were compared with age-matched control lambs.
Coagulation factor activities
To confirm the bleeding phenotype and relationship to previously observed lambs from this flock, individual coagulation factor activities were determined by plotting respective coagulation times against a standard curve generated for each coagulation factor. Briefly, blood was collected from 20 unrelated adult Rambouillet ewes into 3.2% sodium citrate anticoagulant tubes (Becton Dickinson, Franklin Lakes, NJ). Plasma samples from all 20 ewes were pooled, frozen, and stored at -80°C before testing. Pooled sheep plasma was serially diluted with Owren's buffer (Sigma, St. Louis, MO) at 1 ° 2, 1 ° 4, 1 ° 8, 1 ° 16, 1 ° 32, 1 ° 64, and 1 ° 128. Standard curves for coagulation factors were determined by combining 50 μl of each serial diluted plasma sample from pooled normal sheep plasma with 50 μl of specific factor deficient plasma (George King Biomedical, Overland Park, KS) by using an MLA 700 coagulation analyzer. Times for each dilution of pooled normal sheep plasma were plotted on semilogarithmic paper with 2 cycles × 70 divisions. A straight line was drawn through the linear portion of each curve for each coagulation factor deficient plasma sample and serially diluted pooled normal sheep plasma sample. Plasma samples from affected and age-matched control lambs were appropriately diluted to occur on the linear segment of the standard curve. Activity of coagulation factors were determined by combining 50 μl of each appropriately diluted plasma sample with 50 μl of specific factor deficient plasma by using an MLA 700 coagulation analyzer and were plotted against standard curves for each respective coagulation factor.
Complete blood cell count, clinical chemistry, and necropsy
Blood was collected at the time of parturition from affected and control lambs. A complete blood cell count (CBC) was performed from ethylenediaminetetraacetic acid (EDTA) anticoagulated whole blood with an ADVIA 120 (Bayer, Tarrytown, NY). A differential leukocyte count was determined by manually counting a freshly prepared blood film stained with Wright-Giemsa. A routine clinical chemistry panel was performed from serum collected from a clot tube by using a Hitachi 917 automated chemistry analyzer (Roche Diagnostics, Indianapolis, IN). Two affected and 2 age- and sex-matched control lambs were euthanized with an intravenous overdose of sodium pentobarbital 8 days (R104 and CR102) and 80 days (G159 and CR108) after birth. Liver tissue was snap frozen in liquid nitrogen for evaluation of microsomal enzyme activity. All organ systems were sampled and collected in 10% neutral buffered formalin. Routine 5-μm histologic sections were prepared from paraffin-embedded tissue blocks, stained with hematoxylin and eosin, and evaluated under light microscopy.
Preparation of microsomes and determination of enzyme activities
Hepatocellular microsomes were prepared according to Kotkow et al., 17 using either fresh or snap frozen liver obtained from affected and age-matched control lambs. Briefly, liver samples were divided into ≤1-cm cubes, weighed to approximately 25 g, and combined with 25 ml of homogenization buffer (150 mM NaCl, 50 mM Tris-HCl, 5% [v/v] glycerol, 1 mM EDTA, 1 mM benzamidine, 1 mM phenylmethysufonyl fluoride, pH 7.5). Liver samples in homogenization buffer were disrupted by using a commercial blender and were subsequently homogenized by using a homogenizer fitted with a 7-mm saw-toothed generator (Pro Scientific Inc., Oxford, CT). The homogenate was strained through gauze into a 25 × 89-mm centrifuge tube. Tubes were centrifuged at 4°C for 10 minutes at 10,000 × g. The supernatant was filtered through gauze into a second centrifuge tube and spun at 4°C for 1 hour at 130,000 × g. The supernatant was discarded, and the pellet was resuspended in 3 ml of homogenization buffer. Microsomal protein concentration was determined according to the Lowry Method (Sigma Diagnostics, St. Louis, MO). All microsomal assays were performed at a concentration of 10 mg/ml unless stated otherwise.
Carboxylase activity was determined from standard reaction mixtures (125 μl) containing the following: 250 μg of microsomal protein, 0.8 M (NH4)2SO4, 28 mM 4-morpholine propanesulfonic acid (MOPS) at pH 7.5, 0.5M NaCl, 20 μl 1% (3-[3-(Cholamidopropy1) dimethylammonio]-1-proanesulfonate [CHAPS]), 3.6 mM Phe-Leu-Glu-Glu-Leu, 8 mM dithiothreitol (DTT), 5 μl NaH14CO3, 220 μM of vitamin K hydroquinone (K1H2), and 16 μm proIX. The propeptide activator of γ-glutamyl carboxylase, proIX, was manufactured by Macromolecular Resources (Colorado State University, Ft. Collins, CO), based on residues -18 to -1 of the human proIX sequence. 27 The mixture was incubated at 25°C for 30 minutes in sealed tubes. One milliliter of 10% trichloroacetic acid was added to stop the reactions. Unbound 14CO2 was removed by gently boiling the mixture for 10 minutes or until the volume was reduced by approximately 90%. Total incorporation of 14CO2 was determined by adding 6 ml of Ecolite scintillation cocktail (Fisher Scientific, Hampton, NH) and was counted for 5 minutes/sample on a Beckman LS 1801 scintillation counter. Data are expressed as dpm (degradations per minute) 14CO2 incorporated/250 μg microsomal protein.
Vitamin K epoxide reductase (KO) activity was performed as described previously. 14 Vitamin KO was prepared according to the procedure of Tishler et al. 32 Standard reaction mixtures (125 μl) contained 250 μg of microsomal proteins, 25 mM Tris-HCl pH 7.4, 150 mM NaCl, 0.28% CHAPS, 0.12% phosphatidyl choline, 5 mM NaHCO2, 222 μM vitamin KO, and 6 mM DTT. No exogenous substrates were added. The extraction, purification, and analysis of plasma samples were performed as reported previously. 14
Glucose-6-phosphatase activity was determined according to the methods of Burch et al. 9 Reaction mixtures contained 100 μg of homogenized liver microsomes. Enzyme activity was determined by the reduction of NAD+ in the reserve reaction, where hexokinase converts glucose to glucose-6-phosphate. Spectrophotometer readings were made at time 0 and 15 minutes after the addition of 2 μg/ml hexokinase.
Vitamin K1 administration
Three affected lambs were transfused with ovine plasma, as above, and subsequently were administered 35 mg vitamin K1, subcutaneously every 24 hours. Two lambs (R102 and G160) were administered vitamin K on day 1 after plasma infusion, and 1 lamb (G159) was administered vitamin K beginning day 5 after plasma infusion.
Results
Coagulation parameters
All lambs with an ACT exceeding 5 minutes had markedly prolonged prothrombin and activated partial thromboplastin times and decreased vitamin K–dependent procoagulant factor activities compared with age-matched control lambs (Table 1). These data identified the homozygous affected lambs and confirmed that the defect was the same as previously reported.
Coagulation data of affected and control lambs sampled within 1 hour of birth.
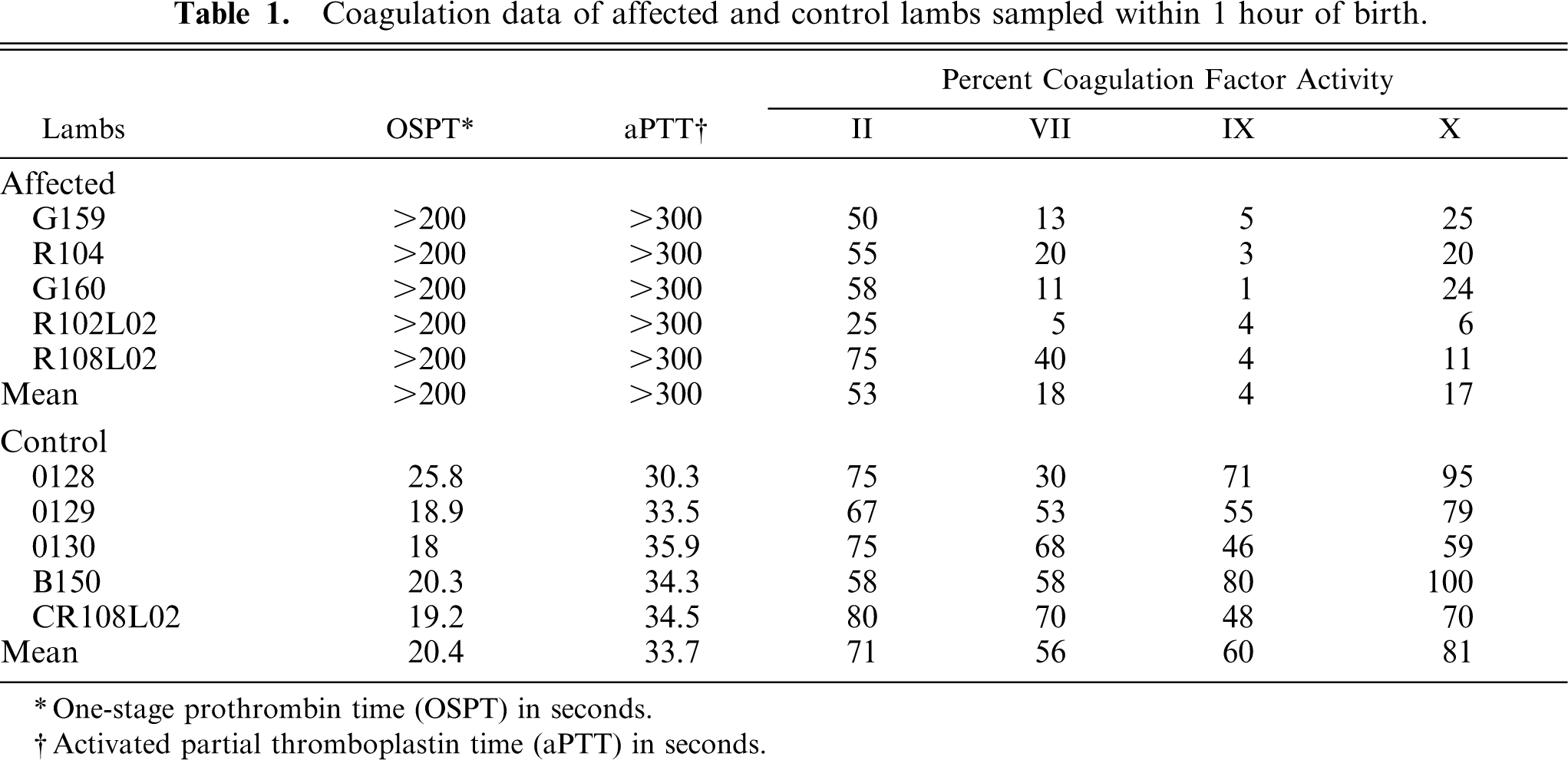
∗ One-stage prothrombin time (OSPT) in seconds.
† Activated partial thromboplastin time (aPTT) in seconds.
Complete blood cell count, clinical chemistry, and necropsy
The results of gross and microscopic examination of clinically affected lambs at necropsy were unremarkable except for a variable degree of subcutaneous, umbilical, and body cavity hemorrhage, most consistently hemarthrosis. Hemorrhage was most notable at sites of previous trauma or venipuncture. Thymic hemorrhage was not present in affected lambs. Serum biochemistry and CBC results, at parturition, were within the reference range for all parameters except for mildly elevated sorbitol dehydrogenase (25–29 IU/l; normal range 10–23 IU/l), which was attributed to mild periparturient hepatocellular injury.
Microsomal enzyme activities
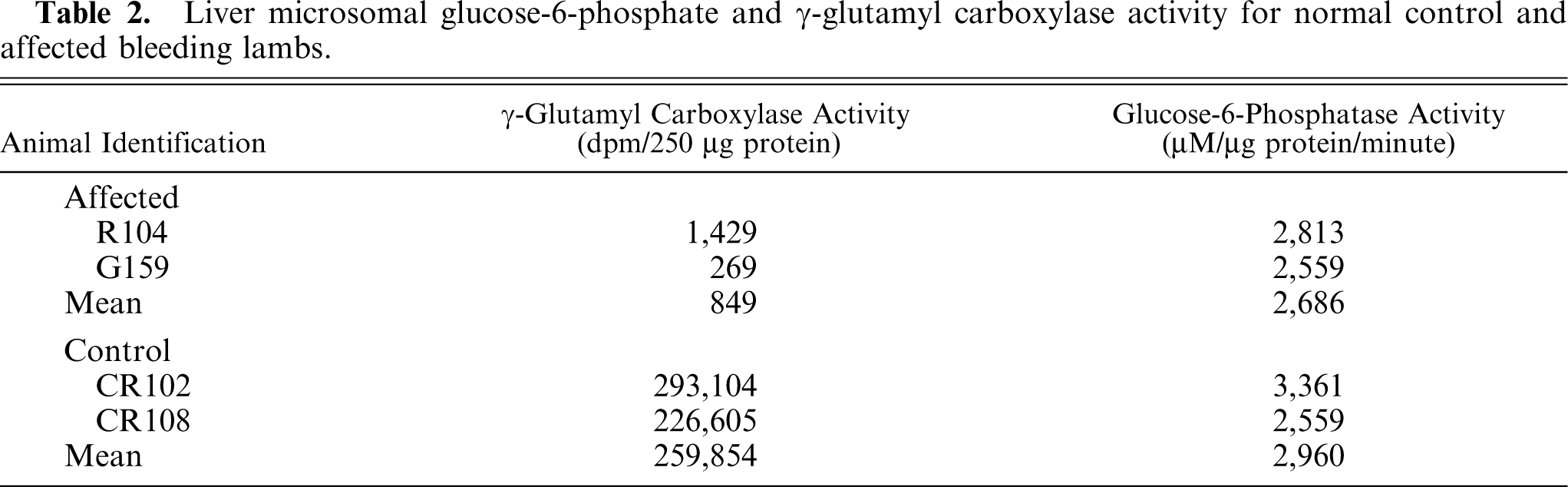
Microsomes prepared from age-matched control lamb liver had incorporation of 14CO2 in the substrate, whereas microsomes from affected lambs had markedly diminished incorporation of 14CO2, indicating severely decreased γ-glutamyl carboxylase activity (Table 2). DTT-dependent epoxide reductase activity (Table 3) and glucose-6-phosphatase activity (Table 2) from affected lambs and age-matched control lambs were equivalent.
Liver microsomal glucose-6-phosphate and γ-glutamyl carboxylase activity for normal control and affected bleeding lambs.
Liver microsomal vitamin K epoxide reductase activity of affected and control lambs.
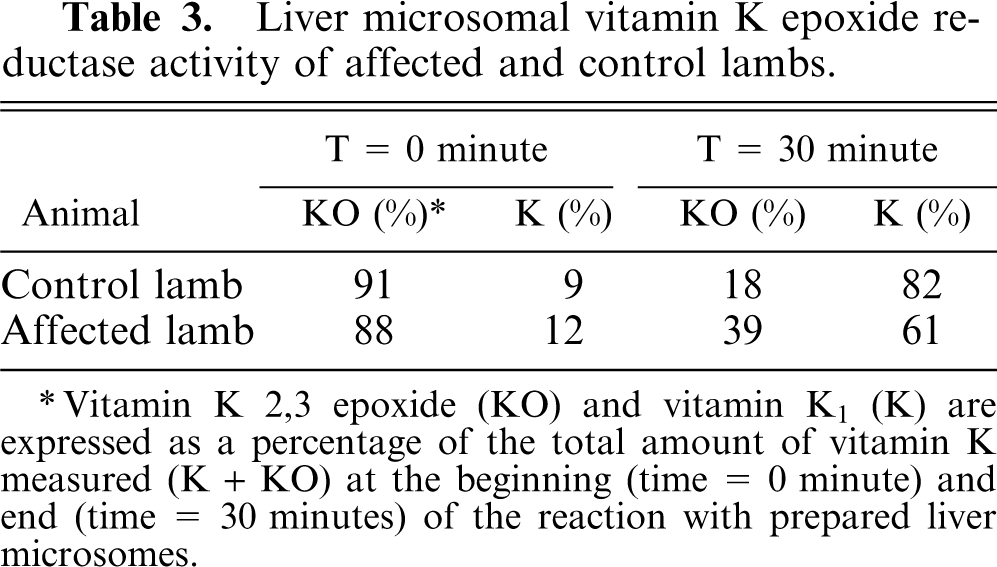
∗ Vitamin K 2,3 epoxide (KO) and vitamin K1 (K) are expressed as a percentage of the total amount of vitamin K measured (K + KO) at the beginning (time = 0 minute) and end (time = 30 minutes) of the reaction with prepared liver microsomes.
Response to vitamin K1 administration
Two lambs were administered 35 mg of Vitamin K1 subcutaneously immediately after the initial plasma transfusion and every 24 hours thereafter or beginning 5 days after the initial plasma infusion (lamb G159) and every 24 hours thereafter. Coagulation factor activities were little changed from 4 to 80 days after initial plasma infusion (Table 4) and daily vitamin K1 administration to affected lambs.
Coagulation factor activities after vitamin K1 administration to affected lambs and factor activity of age-matched normal lambs.
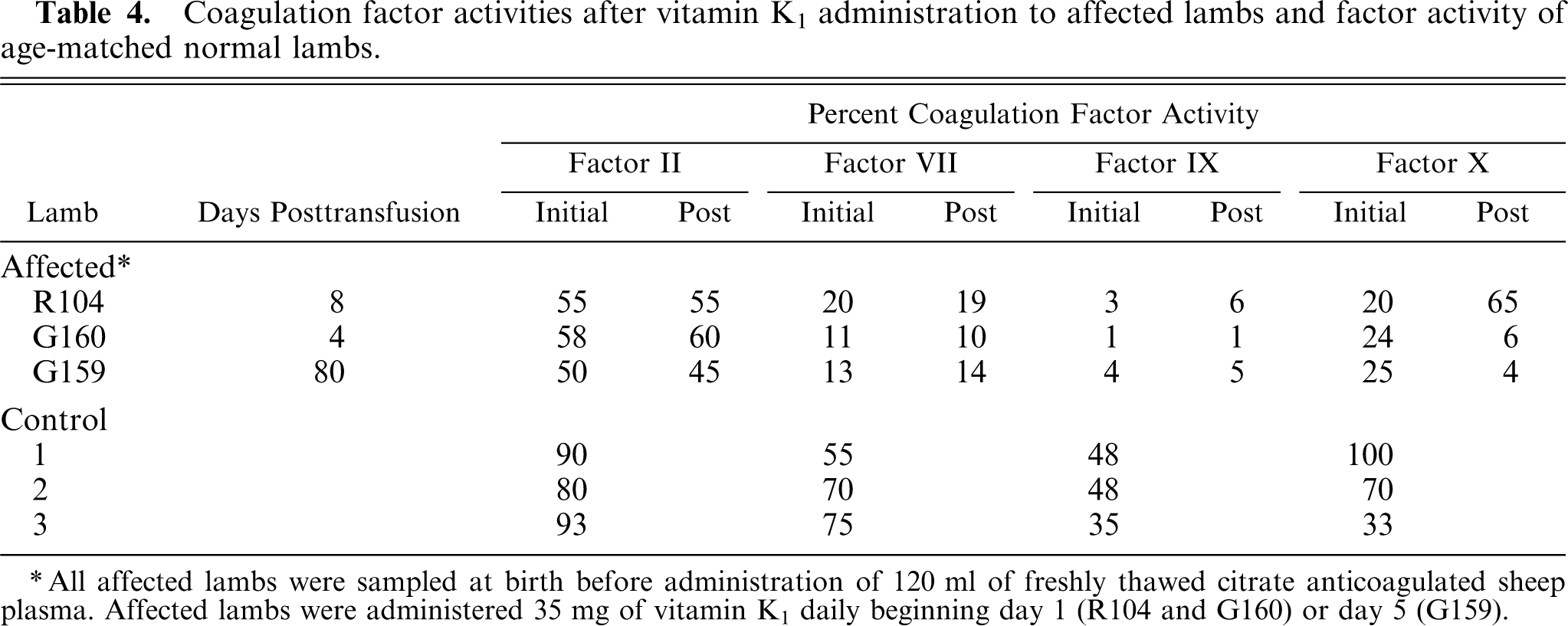
All affected lambs were sampled at birth before administration of 120 ml of freshly thawed citrate anticoagulated sheep plasma. Affected lambs were administered 35 mg of vitamin K1 daily beginning day 1 (R104 and G160) or day 5 (G159).
Defect incidence across multiple seasons
Approximately 10% of the lambs (5 lambs from a total of 51) born into this flock had an ACT exceeding 5 minutes. Three of these lambs were female, and 2 lambs were male.
Discussion
The fatal coagulopathy observed in the affected lambs in this report was the result of significantly reduced levels of vitamin K–dependent coagulation factor activity. Potential causes included aberrant vitamin K metabolism or transport, vitamin K antagonism, or hepatic disease. Investigation excluded hepatic disease and vitamin K antagonism as potential causes of the coagulation defect. 2 We, therefore, analyzed hepatic microsomal enzymes of the vitamin K cycle and demonstrated markedly reduced activity of the enzyme γ-glutamyl carboxylase in enzymatically active microsomes, while activity of epoxide reductase was similar between affected and control lambs. The evidence of enzymatically active microsomes was supported by similar glucose-6-phosphatase activity in control and affected liver microsomes.
The affected lambs described in this report had variably reduced activity of the VKD proteins. While coagulation factors IX and X had minimal activity, the activity of coagulation factors II and VII were consistently higher. Coagulation factor activities in normal sheep relative to humans have been reported and are similar 16 and, therefore, is not considered to be a result of species variation between individual factor activities in affected lambs. Age-matched control lambs were used to eliminate age-associated differences in hemostatic indices reported in humans and horses. 1, 3 In addition, standard curves generated from normal sheep did not demonstrate an inhibitory phase for any of the coagulation factors tested, which excluded the notion of inadequate plasma dilution, contributing to relatively higher activity levels in coagulation factors II and VII. Interestingly, the prothrombin propeptide reportedly has the lowest affinity of the coagulation factors tested 28 yet maintains the highest relative activity in affected lambs. Lin et al. 18 propose that the rate of release of the propeptide appears to be the rate-determining step in substrate turnover; therefore, substrates whose propeptides have relatively low affinities for the carboxylase have increased rates of turnover. This scenario may explain the higher observed activity for factor II. The factor VII propeptide is unique in that it contains a diglutamate pair and appears to act as a glutamate substrate for the carboxylase. We cannot discount the fact that this glutamate pair may interfere with the relative affinity and subsequent activity of the coagulation factor. Unfortunately, the propeptide sequences of the ovine coagulation factors are not known, it is possible that species variation between the respective propeptide sequences may also be contributing to the reported propeptide affinities and influencing the individual factor activity in vivo.
Administration of 35 mg/day vitamin K1 (a dosage exceeding the reported requirement to counteract the effects of warfarin in newborn lambs 22 ) to 3 of the affected lambs did not correct the coagulopathy or improve the VKD coagulation factor activities, and all lambs eventually succumbed to fatal bleeding. Vitamin K administration in some reports of human epoxide reductase deficiency, 21 Devon Rex cats with defective γ-glutamyl carboxylase activity 19, 25 and warfarin intoxication in lambs 22 correct the associated coagulopathy. The molecular defect of γ-glutamyl carboxylase in Devon Rex cats is presumed to be in a region of the enzyme associated with vitamin K and propeptide binding, and increased vitamin K concentration overcomes the decreased binding to γ-glutamyl carboxylase and clinical coagulopathy. 25 The molecular defect of γ-glutamyl carboxylase in affected Devon Rex cats has never been identified.
The inheritance pattern of the bleeding phenotype is suggestive of an autosomal recessive trait. This flock had a lower than expected percentage of affected lambs for such an inheritance pattern. This discrepancy is likely the result of early embryonic loss similar to that reported in γ-glutamyl carboxylase knock-out mice, 35 prothrombin deficient mice, 29 and factor V deficient mice. 34
We determined that the observed coagulopathy in affected Rambouillet lambs is the result of markedly reduced activity of the enzyme γ-glutamyl carboxylase. Determination of the underlying genetic defect will certainly lend insight into the carboxylase active site and expand our understanding of the structure and functional relations of γ-glutamyl carboxylase.
Footnotes
Acknowledgements
We gratefully acknowledge the financial support of the American Heart Association through Grant AHA 9951132Z.
