Abstract
This study investigated the potentiation of porcine circovirus 2 (PCV2)-induced postweaning multisystemic wasting syndrome by porcine parvovirus (PPV) and found it was associated with excessive production of tumor necrosis factor-α (TNF-α). Colostrum-deprived conventional pigs were inoculated intranasally with PCV2 or PPV alone or in combination (PCV2 and PPV). In vitro assay of TNF-α, obtained from alveolar macrophages coinfected with PCV2 and PPV, showed a significant increase in TNF-α compared to single infection of macrophages with either PCV2 or PPV alone (P < 0.05). All pigs inoculated with PCV2 and PPV developed severe postweaning wasting syndrome, whereas clinical signs (e.g., weight loss) were present but perhaps less severe in either PCV2- or PPV-inoculated pigs. Compared to the pigs inoculated with PCV2 or PPV alone, pigs inoculated dually with PCV2 and PPV showed significantly (P < 0.05) increased levels of TNF-α. Levels of TNF-α in the sera were reversely correlated with the body weight in pigs experimentally infected with dual inoculation of PCV2 and PPV (rs = -0.92, P < 0.001). These data suggest that a potentiation of PPV in PCV2-induced PMWS is associated with the excessive production of TNF-α.
Keywords
The porcine circoviruses (PCV) are members of the genus Circovirus, family Circoviridae, which are the smallest nonenveloped, single-stranded, circular DNA viruses that replicate autonomously in mammalian cells. 24, 33 The virion DNA, encapsulated by a single viral protein, is a single-stranded negative sense circular molecule of roughly 1,800 bases that has 6 open reading frames (ORFs). 13, 24, 26 Two types of PCV have been characterized and were subsequently named PCV1 and PCV2. 26
Postweaning multisystemic wasting syndrome (PMWS) is now well established as a wasting disease caused by PCV2 and is a major economic concern in all pig-producing areas of the world. 6 A hallmark of its clinical signs is progressive weight loss. 6 Porcine parvovirus (PPV) has been used as a model coinfecting agent with PCV2 to induce PMWS in experimental studies. 1, 12, 19, 20 Pigs experimentally infected with PCV2 display mild clinical signs and lesions typically associated with this virus, but coinfection of pigs with PCV2 and PPV may result in wasting disease. 1, 12, 19, 20, 23 However, the mechanisms by which PPV potentiates progressive wasting in PCV2-associated PMWS are not known.
Tumor necrosis factor-α (TNF-α) is a macrophage-derived factor originally isolated in the course of studies of the pathogenesis of cachexia in chronically infected animals. 4 TNF-α suppresses lipoprotein lipase in isolated adipocytes and is involved in the pathogenesis of viral wasting disease such as acquired immunodeficiency syndrome. 5, 14, 31 The present study was undertaken to test the hypothesis that potentiation of progressive wasting in the pigs with PMWS by PPV is associated with excessive production of TNF-α. The objectives of this study were 1) to compare the level of TNF-α produced by macrophages in vitro after dual inoculation of PCV2 and PPV with those after inoculation of each virus alone and 2) to compare the body weight and the level of TNF-α in pigs after dual inoculation of PCV2 and PPV with those after inoculation of each virus alone. This would afford a better understanding of the relationship between PCV2 and PPV in pigs with PMWS.
Materials and Methods
In vitro infection
Porcine alveolar macrophages were obtained from pigs that were seronegative for PCV, porcine reproductive and respiratory syndrome virus, and swine influenza virus as previously described.
3
Alveolar macrophages were washed with complete medium (RPMI 1640 medium containing 10% fetal calf serum, 2-mM
The absence of endotoxin in virus stocks, cell culture supernatants, and media was assessed with the Limulus amebocyte lysate assay (Sigma Chemical Co., St. Louis, MO). The sensitivity of the assay, with a standard of lipopolysaccharide from Escherichia coli O55 ° B5 (Sigma Chemical Co.) was in the range of 2 to 6 pg/ml.
Experimental design
Tissue-culture-propagated PCV2 (strain SNUVR000463) and PPV (strain SNUVR000464) were used as the sources of viral inocula. 15 For inoculation, a PCV2 pool containing a 1.2 × 105 tissue culture infective dose 50 (TCID50)/ml and a PPV pool containing a 1.3 × 105 TCID50/ml was prepared as previously described. 20 Thirty-two colostrum-deprived conventional 28-day-old pigs were randomly divided into 4 groups. All pigs were seronegative for PCV, PPV, and porcine reproductive and respiratory syndrome virus. 8 pigs were inoculated intranasally with 1 ml of a 1 ° 20 dilution of the PCV2 pool, and another 8 pigs were inoculated intranasally with 1 ml of a 1 ° 20 dilution of the PPV pool. A further 8 pigs were inoculated intranasally with a mixture (2 ml) of an equal volume of a 1 ° 20 dilution of both the PCV2 and PPV pools. Eight negative control pigs were inoculated with PCV-free PK-15 cell lysates. All experimental groups were held in separate isolators (two pigs per isolator). Blood samples were collected, and body weight was measured at 0, 4, 7, 10, 14, 17, 21, 24, 28, 31, and 35 days post-inoculation (dpi). All pigs from each group were euthanized at 35 dpi.
Blood sampling protocol
Blood was drawn into sterile vacuum tubes without additives. Tubes were immediately immersed in melting ice, allowed to clot, and centrifuged. The levels of TNF-α in the serum samples were measured immediately. Serial samples from a given pig were run at the same time.
Quantitation of TNF-α in culture supernatants and sera
Porcine TNF-α protein levels in cell culture supernatants and sera were measured with an enzyme-linked immunosorbent assay (ELISA, Endogen, Woburn, MA) according to manufacturer's instructions. Recombinant porcine TNF-α was used as the standard, and the assay sensitivity was 38 pg/ml. ELISA assay was performed in triplicate.
In situ hybridization
A 547 base pair (bp) DNA fragment from ORF 2 was used as a PCV2 probe. The forward and reverse primers were 5′-CAGTTCGTCACCCTTTCCC-3′ (nucleotides 939 to 957) and 5′-GGGGGACCAACAAAATCTCT-3′ (nucleotides 1466 to 1485), respectively. 16 For the PPV probe, a 330 bp DNA fragment from viral protein 2 was used. The forward and reverse primers were 5′-CATACACTGGACAATCACAACAAA-3′ (nucleotides 3451 to 3474) and 5′-GCCTAATTGCTGTTGCTTCTG-3′ (nucleotides 3760 to 3780), respectively. 16 The 291 bp cDNA fragments from a genomic library were used as probe for TNF-α. The forward and reverse primers for TNF-α were 5′-GCTGTACCTCATCTACTCCC-3′ and 5′-TAGACCTGCCCAGATTCAGC-3′, respectively. 35
Polymerase chain reaction (PCR) products were purified with a 30-kD cut-off membrane ultrafiltration filter. The nucleotide sequences of the purified PCR products were determined by means of BigDye chemistry with the ABI Prism Sequencer (Applied Biosystems, Foster City, CA). Sequencing was performed on the purified PCR products before they were labeled by random priming with digoxigenin-dUTP (Boehringer Mannheim, Indianapolis, IN), according to the manufacturer's instructions.
In situ hybridization was performed on the lymph nodes of infected and noninfected pigs to confirm the infection of PCV2 and PPV and determine the expression of TNF-α, as previously described. 9, 15
Statistical analyses
The response over time of TNF-α production between different groups and within each group was analyzed. The Mann-Whitney U-test was used to compare the treatment and control groups at each point in time. The Wilcoxon matched signed rank test was used to compare TNF-α concentration to that at the previous time point within each treatment group. In the in vivo experimental study, the Pearson correlation analysis was used to assess the relationship between TNF-α concentration and body weight in all pigs of the 4 groups throughout the experiment. A value of P < 0.05 was considered significant.
Results
In vitro concentration of TNF-α
In comparison of the groups at each time point, significant differences were not detected in TNF-α concentration between the 3 treatments and 1 control group at baseline (0 hour). However, significant differences were detected at 2, 4, 8, 12, 16, 20, 24, and 48 hpi. At 2, 4, 8, 24, and 48 hpi, there were no significant differences in TNF-α concentration between the 3 treatment groups. At 12, 16, and 20 hpi, TNF-α concentration was significantly higher in the dual PCV2- and PPV-coinfected group than in either the PCV2- or PPV-infected group (P < 0.05). At 16 and 20 hpi, TNF-α concentration was significantly higher in the PPV-infected group than in the PCV2-infected group (P < 0.05) (Fig. 1).
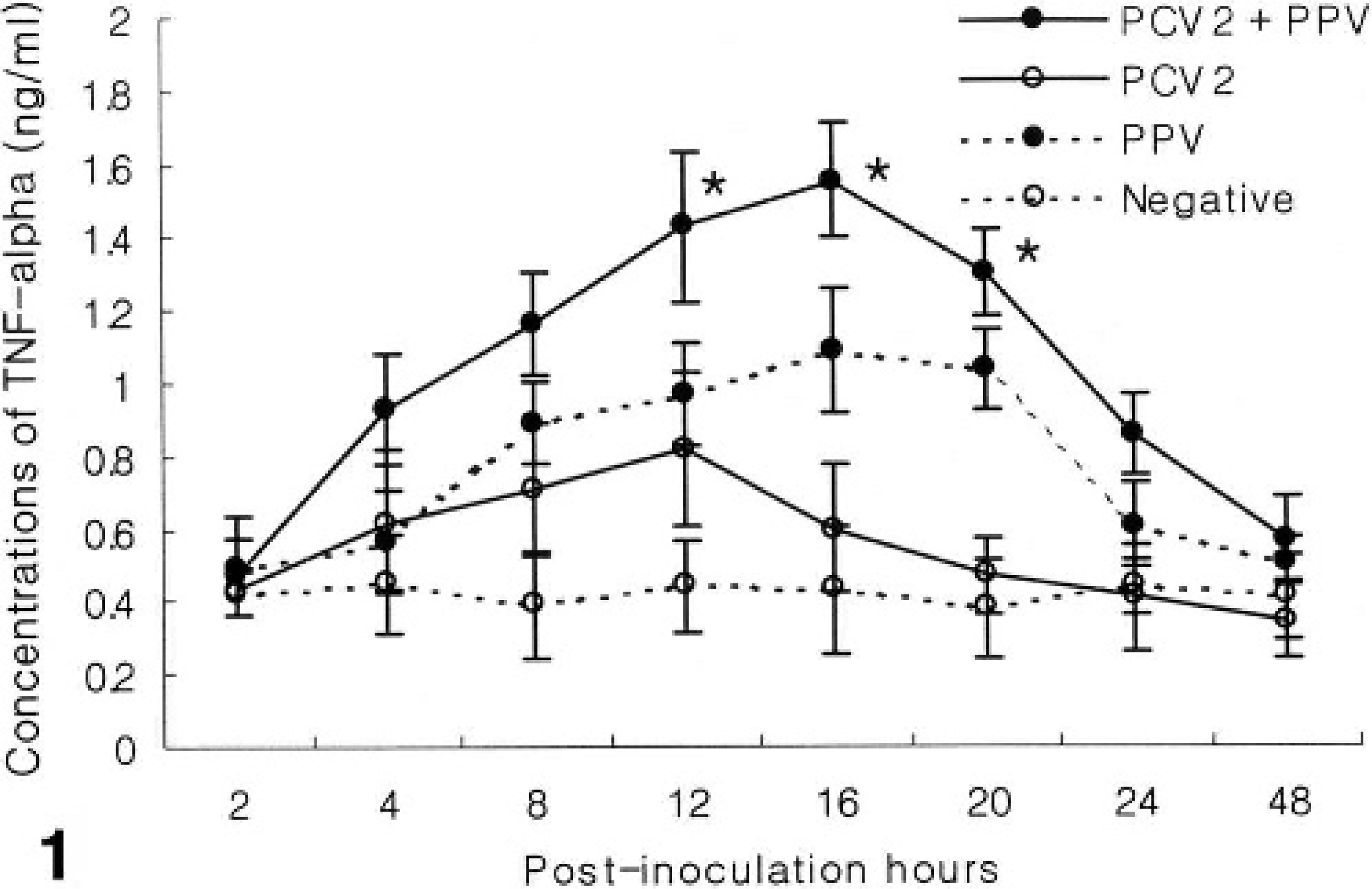
TNF-α production in alveolar macrophages experimentally infected with PCV2 and PPV alone or in combination (PCV2 and PPV). TNF-α concentration was significantly higher in the dual PCV2- and PPV-coinfected group than in either the PCV2- or PPV-infected or control group at 12, 16, and 20 hpi (∗P < 0.05). Values are mean ± SD.
Clinical signs
All pigs inoculated with uninfected cell lysates or single virus inocula (PCV2 or PPV) remained clinically normal, whereas clinical signs of PMWS were observed in all pigs inoculated with PCV2 and PPV. Pigs inoculated with PCV2 and PPV became lethargic at 14 dpi. Five pigs were seen to be thin and unthrifty at 17 dpi, 7 became anorectic at 20 dpi, and 3 developed icterus at 24 dpi.
Microscopic lesions
Tissues samples from uninfected control pigs were histologically normal. Minimal histopathologic changes were seen in pigs inoculated with PCV2 or PPV alone. Mild lymphoid hyperplasia and germinal center formation were observed in pigs inoculated with PCV2 alone.
Histologic lesions in the pigs inoculated dually with PCV2 and PPV were severe. The most striking lesions consisted of multifocal granulomatous inflammation of the lymph nodes, ileal Peyer's patches, tonsil, and spleen. These were associated with infiltrates of epithelioid macrophages and multinucleated giant cells. There were intracytoplasmic and intranuclear amphophilic inclusion bodies in epithelioid macrophages and multinucleated giant cells. Granulomatous inflammation was observed in the lymph nodes of all pigs. Lymph nodes were depleted of mature lymphocytes and contained pyknotic basophilic nucleic and karyorrhectic debris, and the germinal centers were reduced or absent. The thymus in 3 pigs showed granulomatous lesions. A mild multifocal histiocytic interstitial inflammatory infiltrate was also seen in the liver of 3 pigs. The kidney and myocardium, aside from rare focal histiocytic infiltrates, were normal.
Body weight
Significant differences in body weight were not detected between the 3 treatment groups and the uninfected control group at baseline (0 dpi). However, body weight in the PCV2-infected group was decreased significantly compared to PPV-infected and the uninfected control group at 35 dpi (P < 0.05). Body weight in the dually PCV2- and PPV-coinfected group was decreased significantly compared to the other 2 treatment groups and the uninfected control group at 35 dpi (P < 0.05) (Fig. 2).
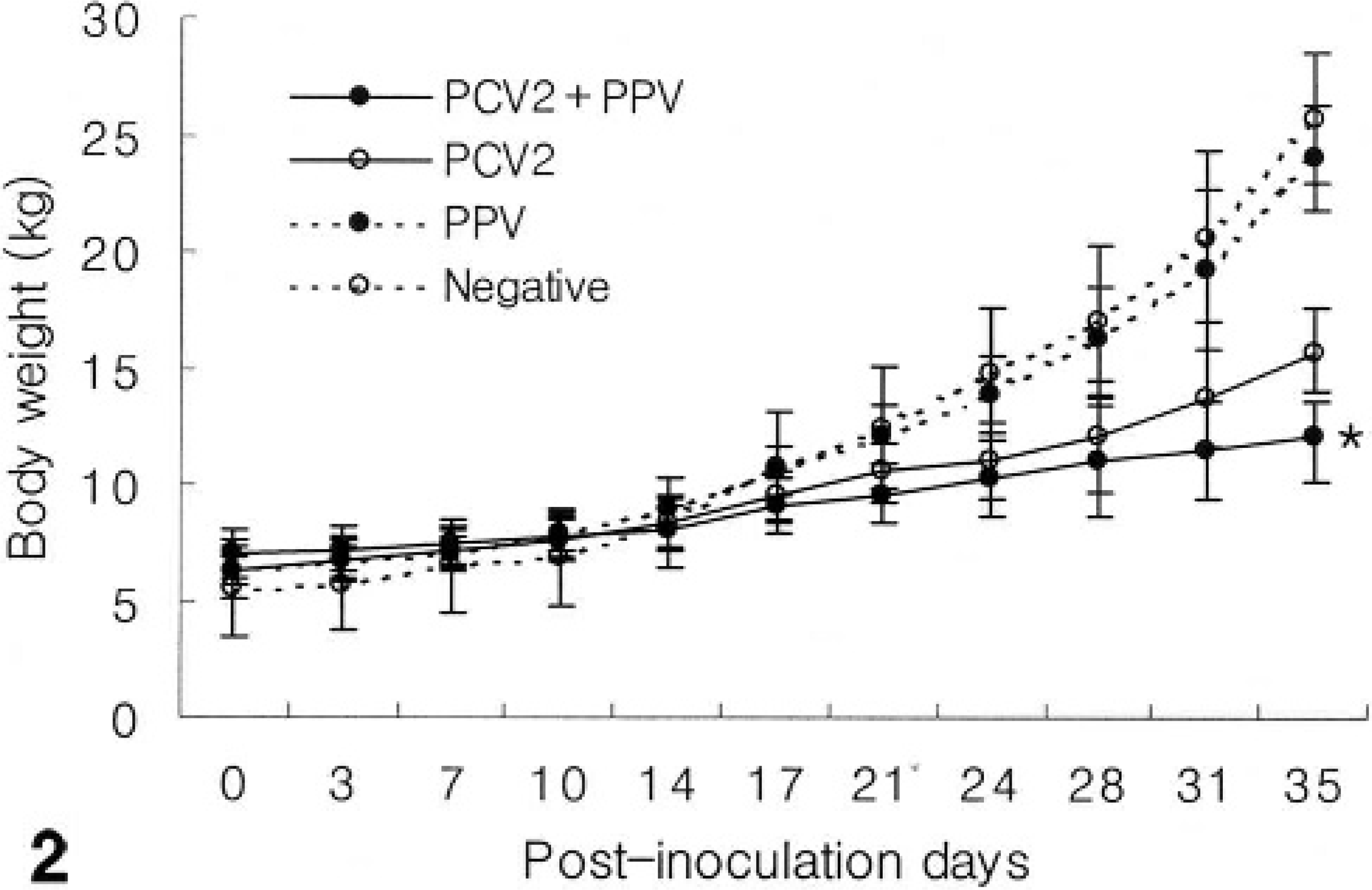
Body weight in pigs experimentally infected with PCV2 and PPV alone or in combination (PCV2 and PPV). Body weight in the dual PCV2- and PPV-infected groups were decreased significantly compared to the other 2 treatment groups and the negative control group at 35 dpi (∗P < 0.05). Values are mean ± SD.
Levels of TNF-α in serum of infected pigs
Levels of TNF-α in the sera of animals experimentally infected with dual inoculation of PCV2 and PPV were compared to those after inoculation of each virus alone. Before virus inoculation, TNF-α levels in serum were below the detection levels of ELISA (38 pg/ml). Compared to the pigs inoculated with PCV2 or PPV alone, pigs inoculated with both PCV2 and PPV showed significantly (P < 0.01) increased levels at 4, 7, 10, 14, 17, and 21 dpi. At 24, 28, and 31 dpi, TNF-α levels in the sera of animals experimentally infected with both PCV2 and PPV were significantly more increased than in either the PCV2- or PPV-infected group (P < 0.05). Levels of TNF-α in the sera of animals experimentally infected with either PCV2 or PPV alone were significantly higher than those in the uninfected control pigs at 4 and 7 dpi (P < 0.05) (Fig. 3). Levels of TNF-α in the sera were reversely correlated with the body weight of pigs experimentally infected with dual PCV2 and PPV inoculation throughout the experiment (rs = -0.92, P < 0.001).
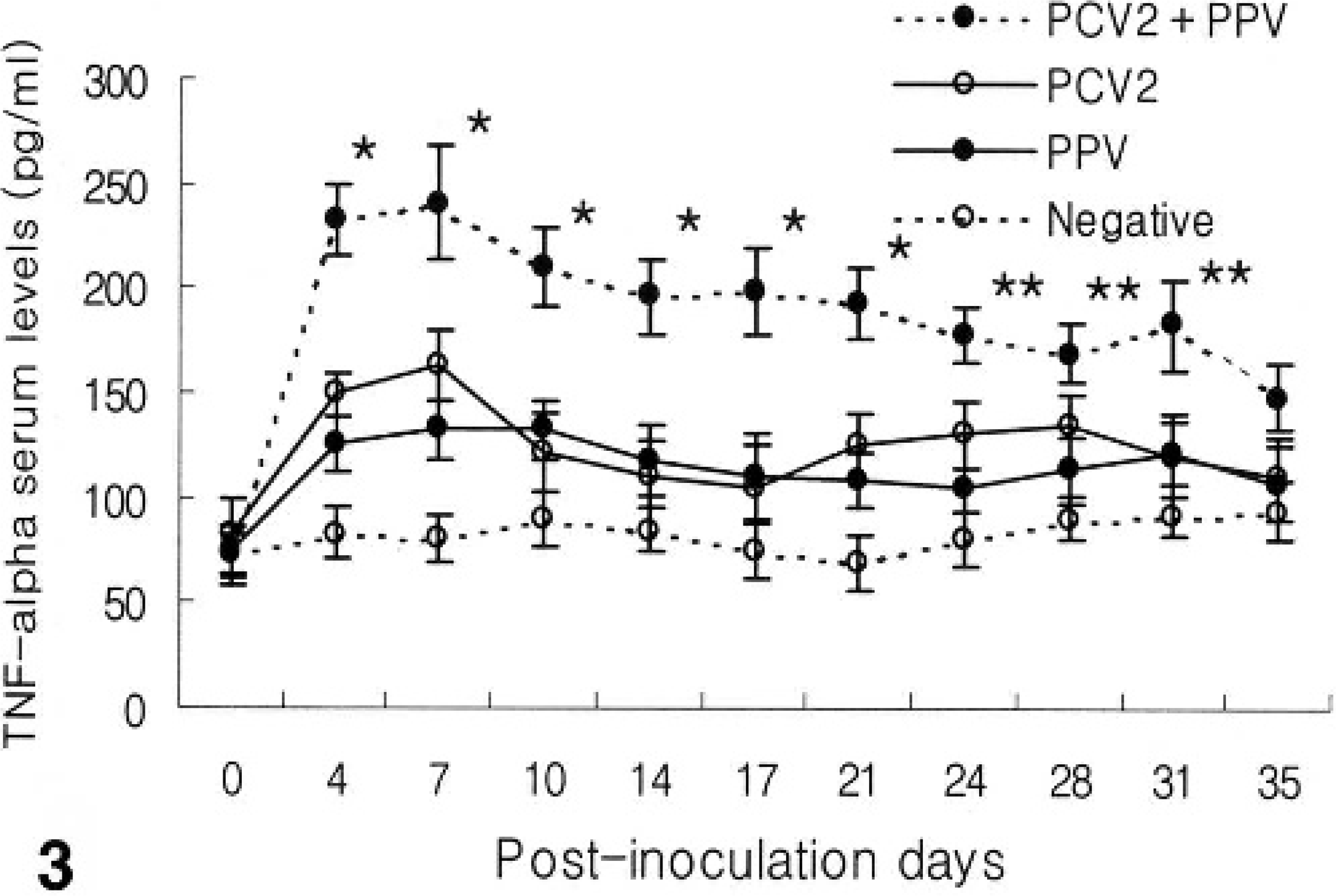
TNF-α production in pigs experimentally infected with PCV2 and PPV alone or in combination (PCV2 and PPV). At 4, 7, 10, 14, 17, and 21 dpi, TNF-α concentration was significantly higher in the dual PCV2- and PPV-coinfected group than in either the PCV2- or PPV-infected group or control group (∗P < 0.01). At 24, 28, and 31 dpi, TNF-α concentration was increased significantly in the dual PCV2 and PPV-coinfected group than in either the PCV2- or PPV- infected group or control group (∗∗P < 0.05). Values are mean ± SD.
In situ hybridization
Hybridization signals for PCV2 and PPV were detected in the lymph nodes from pigs inoculated dually with PCV2 or PPV. Hybridization signals for either PCV2 or PPV were detected in the lymph nodes from pigs inoculated with either PCV2 or PPV, respectively. However, PCV2 DNA was not detected in the lymph nodes from pigs inoculated with PPV and vice versa. Positive cells typically exhibited a dark-brown reaction product, mainly in the cytoplasm but occasionally in the nucleus, without background staining. Hybridization signal for PCV2 (Fig. 4) and PPV (Fig. 5) was detected within histiocytes in lymph nodes. Pretreatment with DNase A eliminated hybridization signal for PCV2 and PPV. Sections from uninfected control pigs showed no hybridization signal for PCV2 or PPV.
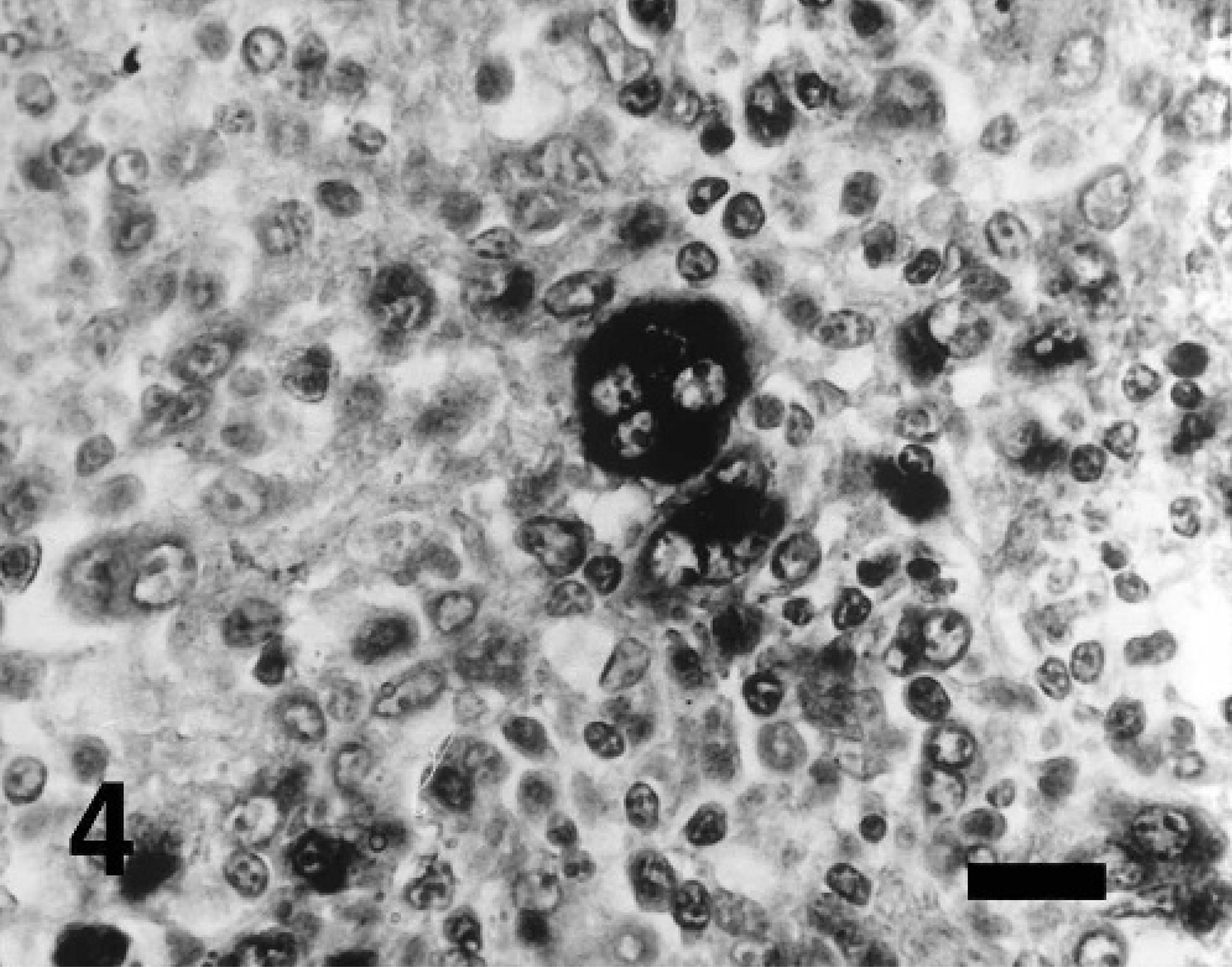
Lymph node; pig experimentally coinfected with PCV2 and PPV at 35 dpi. PCV2 DNA was detected in multinucleated giant cells. In situ hybridization; DNA probe; nitroblue tetrazolium/5-bromocresyl-3-indolylphosphate, methyl green counterstain. Bar = 55 μm.
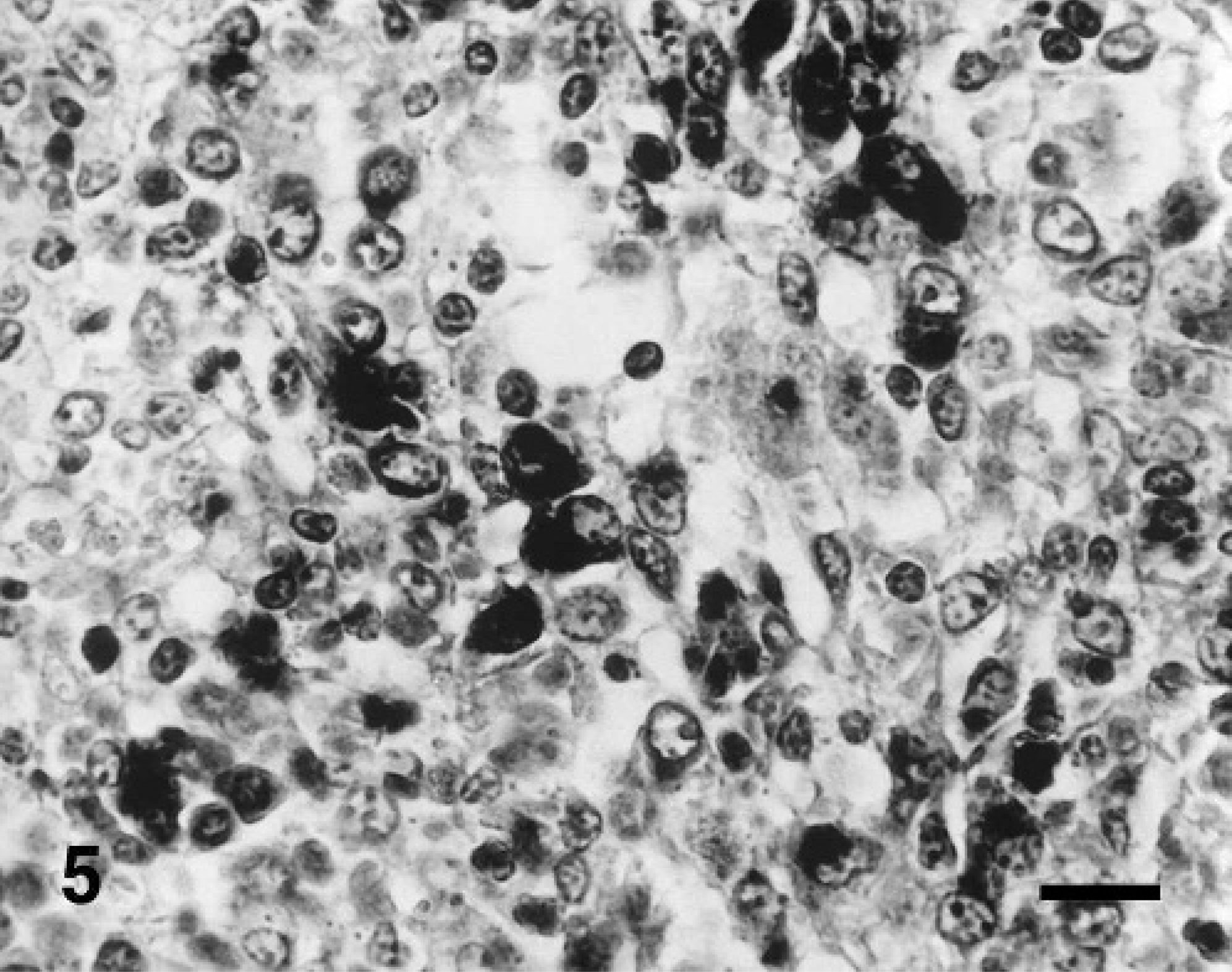
Lymph node; pig experimentally coinfected with PCV2 and PPV at 35 dpi. PPV DNA was detected in macrophages. In situ hybridization; DNA probe; nitroblue tetrazolium/5-bromocresyl-3-indolylphosphate, methyl green counterstain. Bar = 55 μm.
Pigs inoculated with PCV2 or PPV alone had weak hybridization signal, whereas pigs inoculated dually with PCV2 and PPV had strong hybridization signals for TNF-α in the lymph nodes. The morphology of host cells was preserved despite the relatively high temperature required during the incubation procedure. Positive cells were identified as containing a dark-brown or black reaction product in the cytoplasm without background staining. A strong hybridization signal for TNF-α was seen in epithelioid macrophages and multinucleated giant cells of lymph nodes from pigs inoculated dually with PCV2 and PPV (Fig. 6). Pretreatment with RNase A eliminated hybridization signals for TNF-α. Sections from uninfected control pigs showed no hybridization signal for TNF-α.
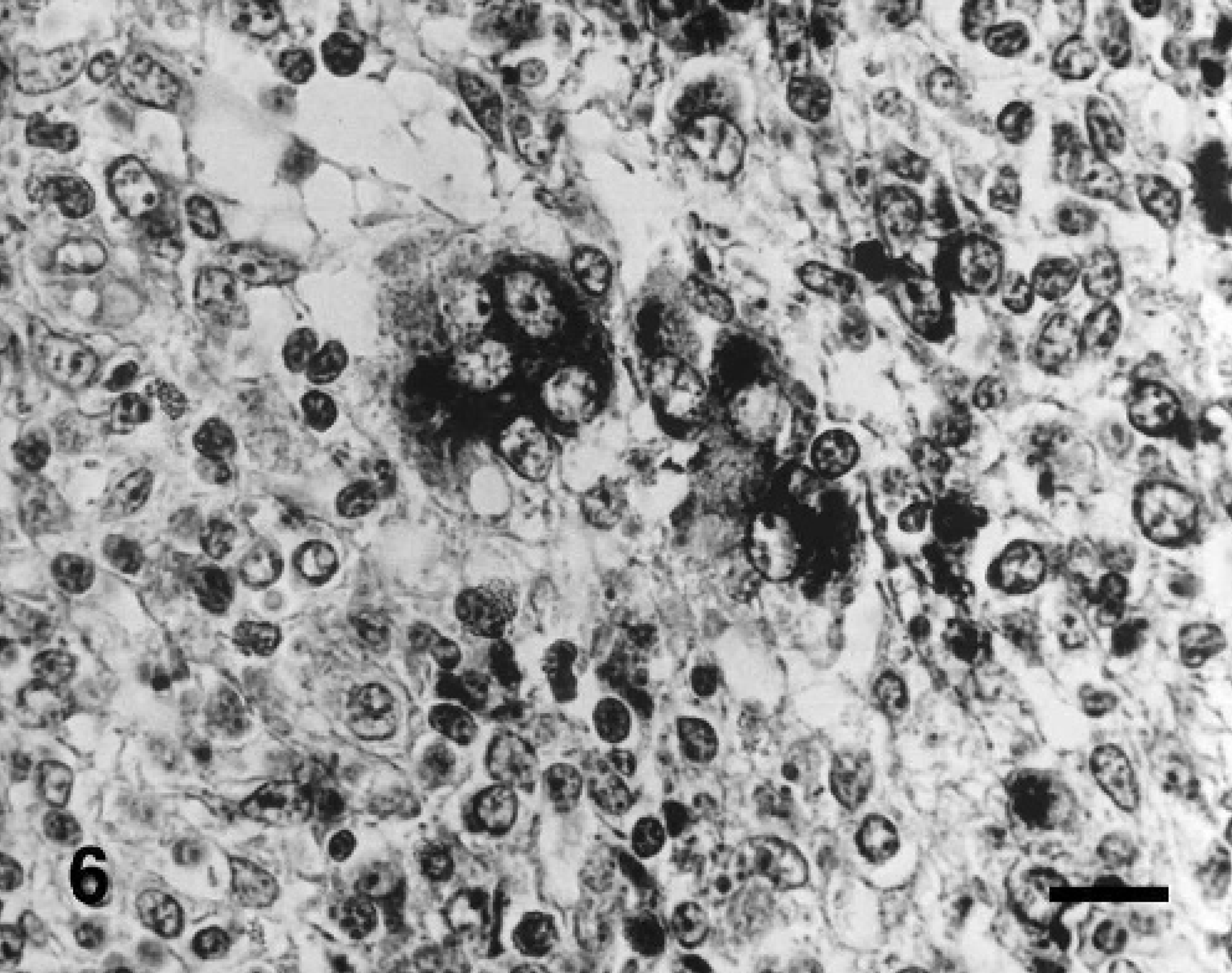
Lymph node; pig experimentally coinfected with PCV2 and PPV at 31 days postinoculation. Tumor necrosis factor-α RNA was detected in multinucleated giant cells. In situ hybridization; cDNA probe; nitroblue tetrazolium/5-bromocresyl-3-indolylphosphate, methyl green counterstain. Bar = 55 μm.
Discussion
The results of this study indicate that PPV potentiate the maximal production of TNF-α in pigs with experimental PMWS and suggests a possible contribution of this cytokine to the major clinical manifestation of the disease. Macrophages are the main targets for PCV2 and PPV, 7, 8, 15 and this infection causes TNF-α secretion, which is linked to a variety of disease symptoms. The present study provides direct evidence that TNF-α is elevated in serum and selectively expressed at the site of granulomatous lesions, providing further support for the role of this cytokine in pathogenesis of PMWS. Although conditions for blood collection and storage may clearly influence the measured TNF-α levels, it is believed that with endotoxin-free blood collection tubes, separation of serum within 1 hour, and storage at 4°C before separation (routine in the present study), the TNF-α concentrations measured reflected circulating serum levels. 2
The mechanisms by which coinfection of PPV and PCV2 produce maximal secretion of TNF-α are unknown. Secretion of TNF-α in the macrophages coinfected with PPV and PCV2 appears to be an additive effect of the viral inoculum of the individual viruses presented to the macrophages in a similar way that adenovirus-stimulated macrophage can release the TNF-α in the dose-dependent manner.36 In addition, live PPV and PCV2 were more effective for secretion of TNF-α than the equivalent dose of an ultraviolet-inactivated preparation (data not shown). Therefore, additional mechanisms requiring intracellular viral replication and/or triggered by viral gene products seem to be required for enhanced TNF-α synthesis.
Because PPV alone did not induce lesions in this and previous studies, 20 the mechanisms by which lesions could be exacerbated by PPV still need to be resolved. Immune activation is a key component of the pathogenesis of PCV2-associated PMWS in swine. 21 These studies suggests that new DNA synthesis, mitosis, and proliferation of macrophages induced by immune stimulation provides the optimal conditions for PCV2 replication. Both PCV2 and PPV are nonenveloped DNA viruses and both are dependent upon host cell DNA synthesis for efficient viral replication. 32 In this study, PPV can potentiate the maximal secretion of TNF-α in PCV2-infected macrophages. It has also been demonstrated that single macrophages may be coinfected with both PCV2 and PPV. 15 Therefore, PPV infection of macrophages could promote high levels of replication of PCV2 during the preclinical phase of dual infection by inducing TNF-α secretion.
Intracellular cytokine levels as measured by flow cytometry revealed that TNF-α expression was not increased in feeder pigs suffering from natural PMWS. 30 Although the duration of PMWS in feeder pigs is not known, these feeder pigs could be late stage because mean body weight of the PMWS-affected animals was 17.0 kg, whereas reference pigs displayed a mean weight of 41.4 kg. 35 Moreover, in the present study, TNF-α level is decreased gradually by the progression of PMWS and could not express in the late stage. It is possible to explain the difference in the TNF-α expression between natural and experimental PMWS, which could be caused by a difference in the duration of infection or the different coinfection of virus and bacteria.
Little is known about the clinical relationship between TNF-α and PMWS. Progressive weight loss in pigs with PMWS is considered to develop through one of the general mechanisms. TNF-α may interfere with lipoprotein lipase activity 4, 5, 14 and other aspects of lipid metabolism. 29, 34 Interestingly, TNF-α also induces cachexia in experimental animals. 4, 28 It is tempting to propose that one mechanism of weight loss in pigs with PMWS could be related to an altered metabolism attributed to prolonged TNF-α production. The immune system eliminates invading pathogens through an inflammatory response that is sometimes harmful to host cells and tissues. Our findings suggest that the clinical manifestations of PMWS, such as retardation of growth may be explained by the effects of high local concentrations of TNF-α, perhaps with release of TNF-α into the blood-stream. However, there is significantly reduced weight gain in pigs infected with PCV2 only at 35 dpi, although the level of TNF-α was not increased. Further study is needed to define the alternative mechanisms for the weight loss.
Granulomatous tissues constantly evolve from loose cell aggregates into highly organized structures that may have considerable cell turnover within them. 10 TNF-α has been shown to have an important role in granulomatous formation and maintenance in human granulomatous tissues. 18, 27 In the present study, hybridization signals of TNF-α were selectively found in macrophages forming the granulomatous inflammation, whereas only a few positive cells were recognizable in the surrounding tissues. This might indicate that TNF-α is essential for the maintenance of granulomatous tissue in pigs.
Multinucleated giant cells are highly stimulated inflammatory cells of macrophage lineage at a terminal stage of differentiation and a common cellular component of granulomatous inflammation. 22 In the current study, multinucleated giant cells were found to contain a considerable amount of hybridizable TNF-α nucleic acid. They should not therefore be regarded as nonfunctioning cells, 25 but are likely to be actively synthesizing many products. The finding that the multinucleated giant cells investigated in this study contain TNF-α nucleic acid supports the view that they may also be important sources of TNF-α.
In conclusion, the results of this study demonstrate overproduction of TNF-α following dual inoculation of PCV2 and PPV and suggest that this cytokine makes an important contribution to some of the major clinical manifestations of PMWS, such as progressive weight loss. PCV2/PPV coinfection has been demonstrated in a significant numbers of cases of PMWS in pigs in Korea and Western Canada. 11, 17 This study suggests mechanisms by which PCV2 and PPV may interact and lead to the development of PMWS as is often seen in the field.
Footnotes
Acknowledgements
The research reported here was supported by Ministry of Agriculture, Forestry and Fisheries-Special Grants Research Program (MAFF-SGRP) and Brain Korea 21 Project, Republic of Korea.
