Abstract
The neuronal ceroid ipofuscinoses (NCL) are a group of heritable, neurodegenerative, storage diseases, typically with an autosomal recessive mode of inheritance. Cytoplasmic accumulation of storage material in cells of the nervous system and, variably in other tissues, characterizes NCL. NCL has been reported in many animal species, but to the authors' knowledge, this is the first report of the disease in a pig. Blindness and seizures are common clinical signs of disease, neither of which was a feature in this pig. The lesions were restricted to the central nervous system, which was diffusely affected, with the most severe lesions in the hippocampus, cerebral cortex, and cerebellum. The histologic lesions included neuronal loss and gliosis, which contributed to mild cerebrocortical and cerebellar atrophy and accumulation of autofluorescent storage material in neurons and glial cells. The storage material had morphologic, histologic, and ultrastructural properties typical of NCL.
The neuronal ceroid-lipofuscinoses (NCLs) are a group of heritable, neurodegenerative, storage diseases, typically with an autosomal recessive mode of inheritance. They are characterized by the cytoplasmic accumulation of autofluorescent material, resembling ceroid and lipofuscin, within cells of the central nervous system (CNS) and variable accumulation in other tissues. The material has been identified as subunit c of mitochondrial ATP synthase (SCMAS) and/or sphingolipid activator proteins (SAPs) A and D. 7, 12, 18 SCMAS is the major component in most forms of the disease, with SAPs comprising a minor portion in some forms. 6, 7 SAPs are the major component in the human infantile form (CLN1), as well as NCL of Miniature Schnauzer dogs and Swedish Landrace sheep. 7, 12, 17, 18
Four clinical human forms of the disease have been described: infantile, late infantile, juvenile, and adult. These have been associated with defects in 8 genes (CLN1–8). CLN1 and CLN2 encode the soluble lysosomal enzymes palmitoyl protein thioesterase 1 and tripeptidyl peptidase 1, respectively, but to the authors' knowledge, the products of the remaining 6 genes have not been identified. 6, 7, 16 Clinical signs of disease include blindness, seizures, progressive psychomotor deterioration, dementia, altered behavior, speech disturbances, and premature death. 6, 7
NCL has been reported in numerous animal species. 3, 5, 12, 18 Reported clinical signs include blindness, seizures, progressive psychomotor deterioration, altered behavior, and premature death. 3, 8, 10, 18 The genetic mutation has been identified in only 1 species—Swedish Landrace sheep—in which there is a mutation in the gene encoding the lysosomal enzyme cathepsin D. 17 In 4 other species, a specific genetic mutation is strongly suspected: a CLN8 mutation in English Setter dogs 9 and mnd mice, 15 and a CLN6 mutation in South Hampshire sheep 3 and Merino sheep. 3 To our knowledge, NCL has not been described in any porcine breed. We describe the clinical and pathologic findings of NCL in a Vietnamese pot-bellied pig.
The pig, whose age was estimated at 2 years, initially manifested minimal hind limb ataxia that did not progress for 3 months. Over the course of the following month, however, the ataxia progressed to tetraparesis, with frequent stumbling and falling. During the subsequent week, the pig's condition deteriorated more rapidly until it could no longer eat or drink on its own and had a staggering gait, falling after 2 to 3 steps. A slight head tilt and intermittent nystagmus were also noted.
The pig was referred to the neurology service at the North Carolina State University Veterinary Teaching Hospital. At the time of presentation, it was nonambulatory tetraparetic with a mild head tilt to the right. Its mentation and attitude were normal. Cranial nerve examination revealed spontaneous vertical nystagmus, but was otherwise unremarkable. When attempting to move, it tended to roll to the right and its head bobbed repeatedly when it became excited. Postural reactions (hopping) and conscious proprioception were absent in all 4 limbs. Withdrawal reflexes were normal. The signs were localized to the brainstem and the cerebellum, and seemed to be more severe on the right side. More diffuse involvement of the spinal cord was suspected on the basis of the severity of its weakness. At that time, the pig was euthanized, and complete necropsy was performed.
There were no grossly apparent lesions at necropsy. Tissue specimens, including those of brain, spinal cord, liver, kidney, spleen, heart, lung, small and large intestine, pancreas, thyroid, and adrenal gland, were fixed in neutral-buffered 10% formalin. Five-micron-thick sections were processed in a routine manner and were stained with hematoxylin and eosin (HE). Sections of the CNS, liver, kidney, small and large intestine, and adrenal gland were stained with luxol fast blue (LFB), Sudan black B, periodic acid–Schiff (PAS), and acid-fast stains. Additionally, sections of cerebellum were stained with Bielschowski's silver stain and immunohistochemically for glial fibrillary acidic protein (GFAP; Z0334, Dako Corporation, Carpinteria, CA). Formalin-fixed specimens of hippocampus were further fixed in 4% formaldehyde/1% glutaraldehyde, followed by 1% osmium tetroxide, then were stained with methanolic uranyl acetate and lead citrate for transmission electron microscopy.
Microscopic lesions were restricted to the CNS. Most neurons and fewer astrocytes and microglia contained variable amounts of golden brown to deeply eosinophilic, granular to homogenous, intracytoplasmic material that often displaced the nucleus or Nissl substance, or both, peripherally (Figs. 1, 2). The material stained magenta with PAS, blue with LFB, black with Sudan black B, and brownish red with acid-fast stains, and exhibited yellow-green autofluorescence by use of fluorescence microscopy (Fig. 2). There was diffuse, mild to moderate microgliosis with satellitosis in regions of the most severe neuronal degeneration.
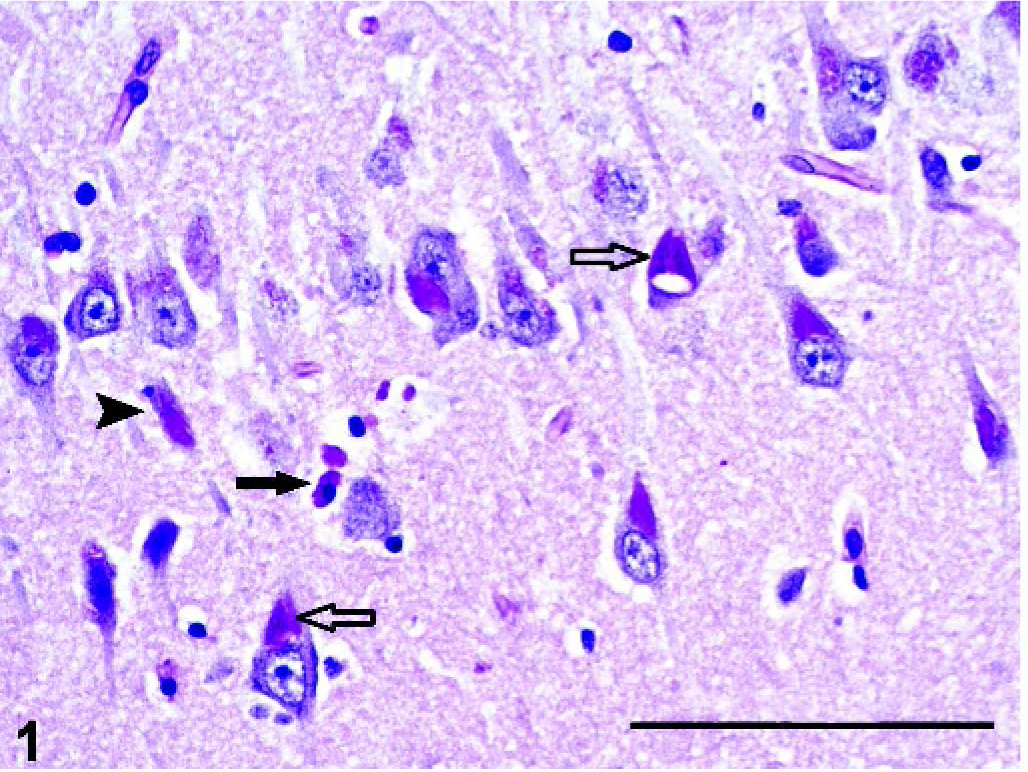
Brain, hippocampus; pot-bellied pig. Accumulation of periodic acid–Schiff (PAS)-positive storage material in neurons (open arrow) and microglial cells (solid arrow); notice necrotic neuron (arrowhead). PAS. Bar = 100 µm.
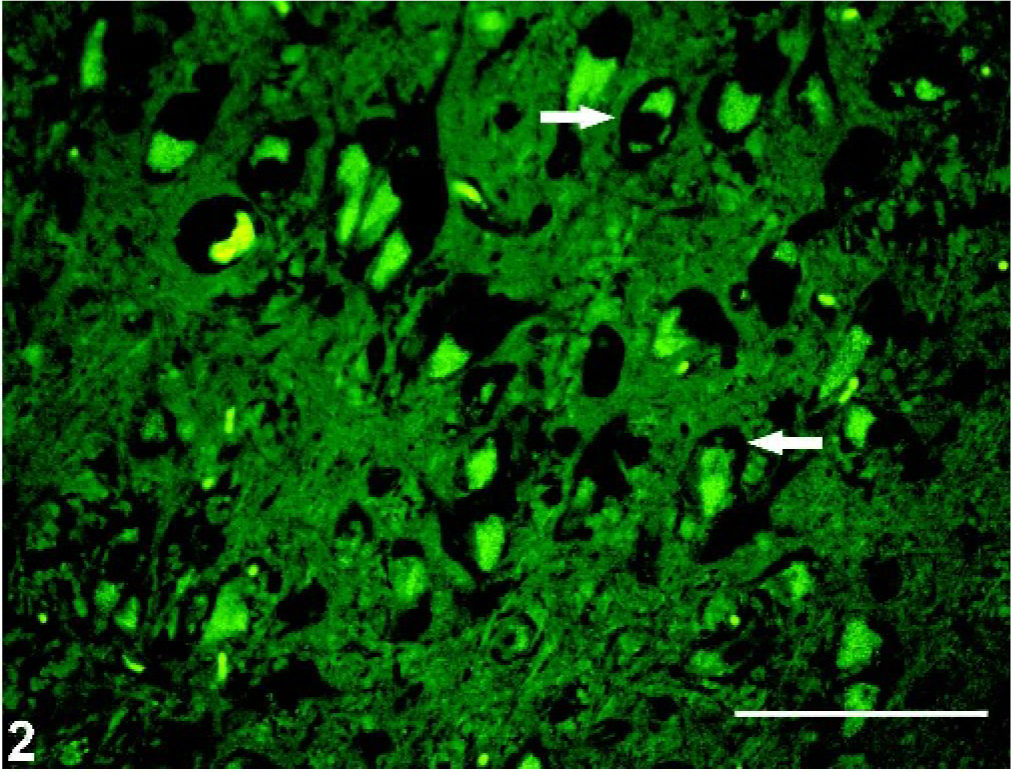
Brain, hippocampus; pot-bellied pig. Autofluorescent storage material within cytoplasm of neurons; notice peripheralization of black Nissl substance (arrows). HE. Viewed under fluorescent microscope. Bar = 50 µm.
The pyramidal cells of the hippocampus were the most severely affected, with moderate neuronal loss and abundant storage material (Fig. 1). The only change noted in the granule cells of the dentate gyrus was the accumulation of minimal amounts of storage material. In the cerebral cortex, the fusiform cells of layer VI contained the most storage material, followed by the pyramidal cells of layers III–V. There was loss of stellate cells in layers II–IV and of the large pyramidal cells in layer V, which contributed to mild cortical atrophy. Loss of cerebellar Purkinje cells was marked (Fig. 3), and axonal distention was occasionally seen (Fig. 4). Only the few remaining Purkinje cells contained small amounts of storage material. The molecular layer was attenuated, and there was mild to moderate, multifocal loss of granular cells, resulting in atrophy of the cerebellar folia (Fig. 3). Bergmann glial cells, positive for GFAP, accumulated at the interface between the molecular and granular layers, with fewer cells scattered throughout the molecular layer (Fig. 5). There was mild to moderate loss of neurons in the cerebellar nuclei, cochlear nuclei, hypothalamus, and ventral horn of the spinal cord, with variable amounts of storage material. Several swollen axons (spheroids) were evident in the hypothalamus and cerebellar nuclei. The larger neurons of the red, oculomotor, and trigeminal nuclei contained moderate amounts of storage material; however, neuronal loss was not evident. Neuronal loss was minimal in the remainder of the brain and the dorsal horn of the spinal cord, with minimal to mild accumulation of storage material. A summary of the histologic lesions is presented in Table 1.
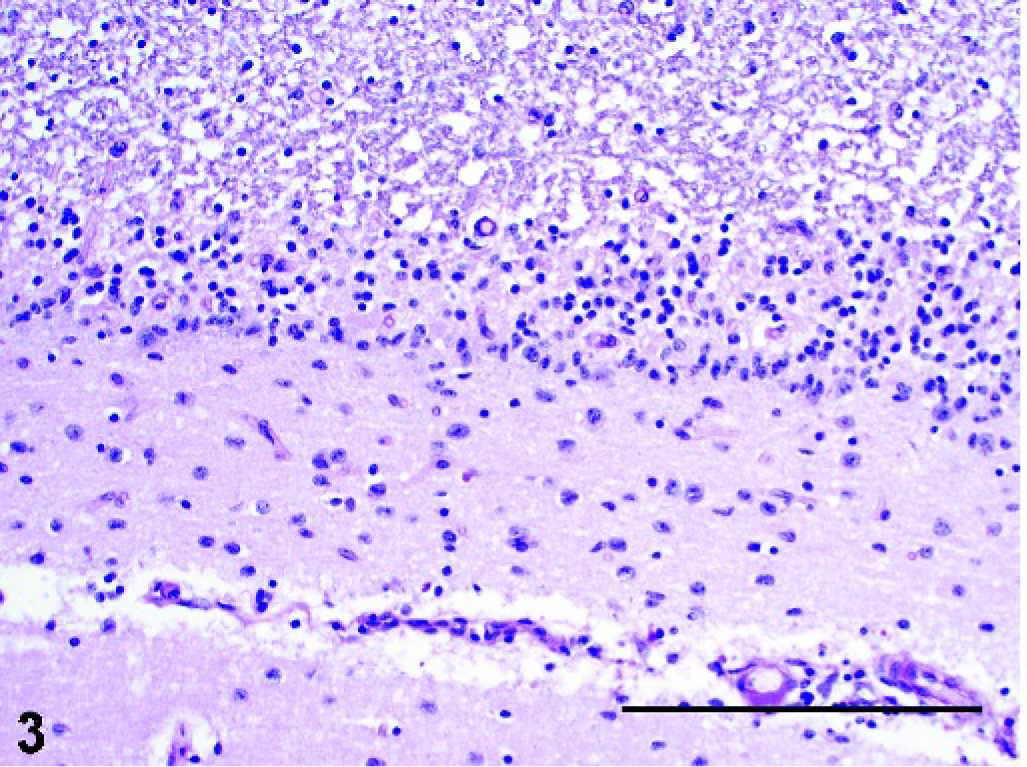
Brain, cerebellum; pot-bellied pig. Loss of Purkinje cells and granule cells with attenuation of molecular and granular layers; Bergmann gliosis in the molecular layer. PAS. Bar = 200 µm.
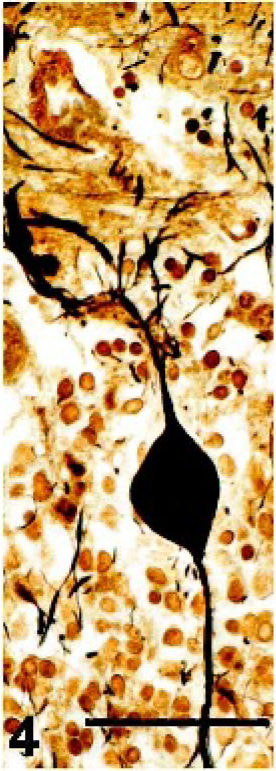
Brain, cerebellum; pot-bellied pig. Purkinje cell with enlargement of the axon in the granular layer. Bielschowski silver stain. Bar = 50 µm.
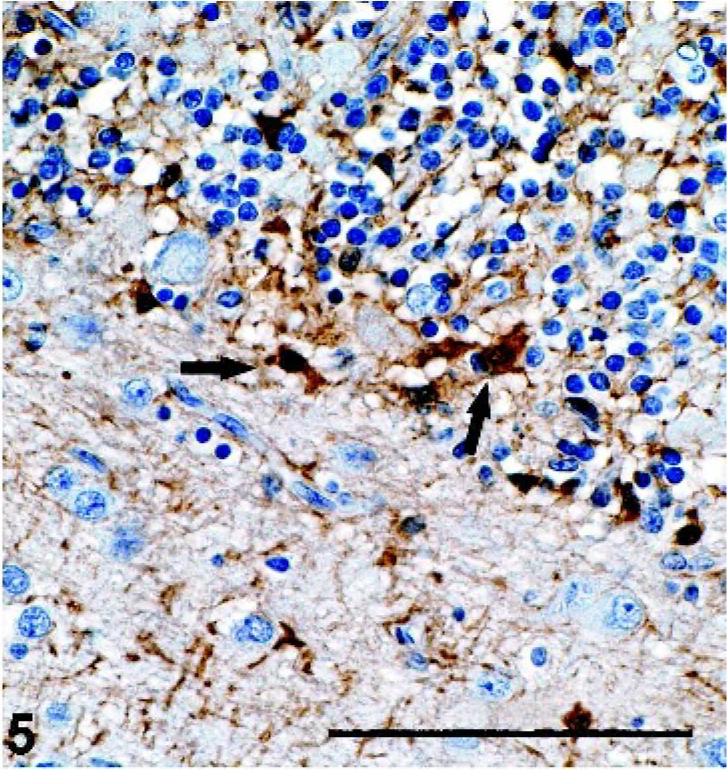
Brain, cerebellum; pot-bellied pig. Interface of molecular and granular layers showing glial fibrillary acidic protein (GFAP)-positive Bergman glial cells (arrows). GFAP immunostain with hematoxylin counterstain. Bar = 100 µm.
Lesion distribution of neuronal ceroid lipofuscinosis (NCL) in a pot-bellied pig.∗
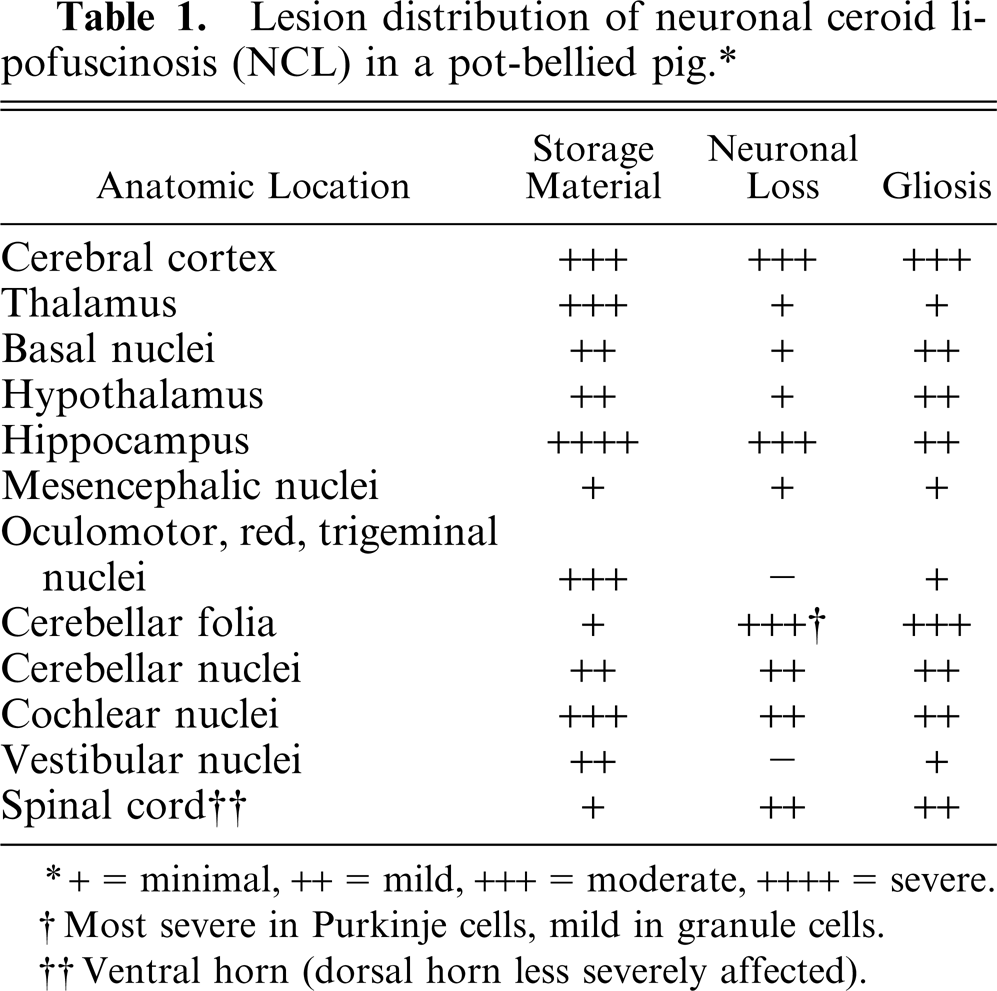
+ = minimal, ++ = mild, +++ = moderate, ++++ = severe.
Most severe in Purkinje cells, mind in granule cells.
Ventral horn (dorsal horn less severely affected).
Ultrastructural findings included membranous material forming multilamellar profiles of up to 5 layers, fewer curvilinear profiles in a granular osmophilic matrix, and a few granular osmophilic deposits (GRODs; Fig. 6), typical of NCL. 6, 7, 10, 18 GRODs have been associated with SAPs, whereas membranous profiles have been associated with SCMAS. 7, 18 The ultrastructural patterns in this pig were consistent with the accumulation of mainly SCMAS, with smaller amounts of SAPs.
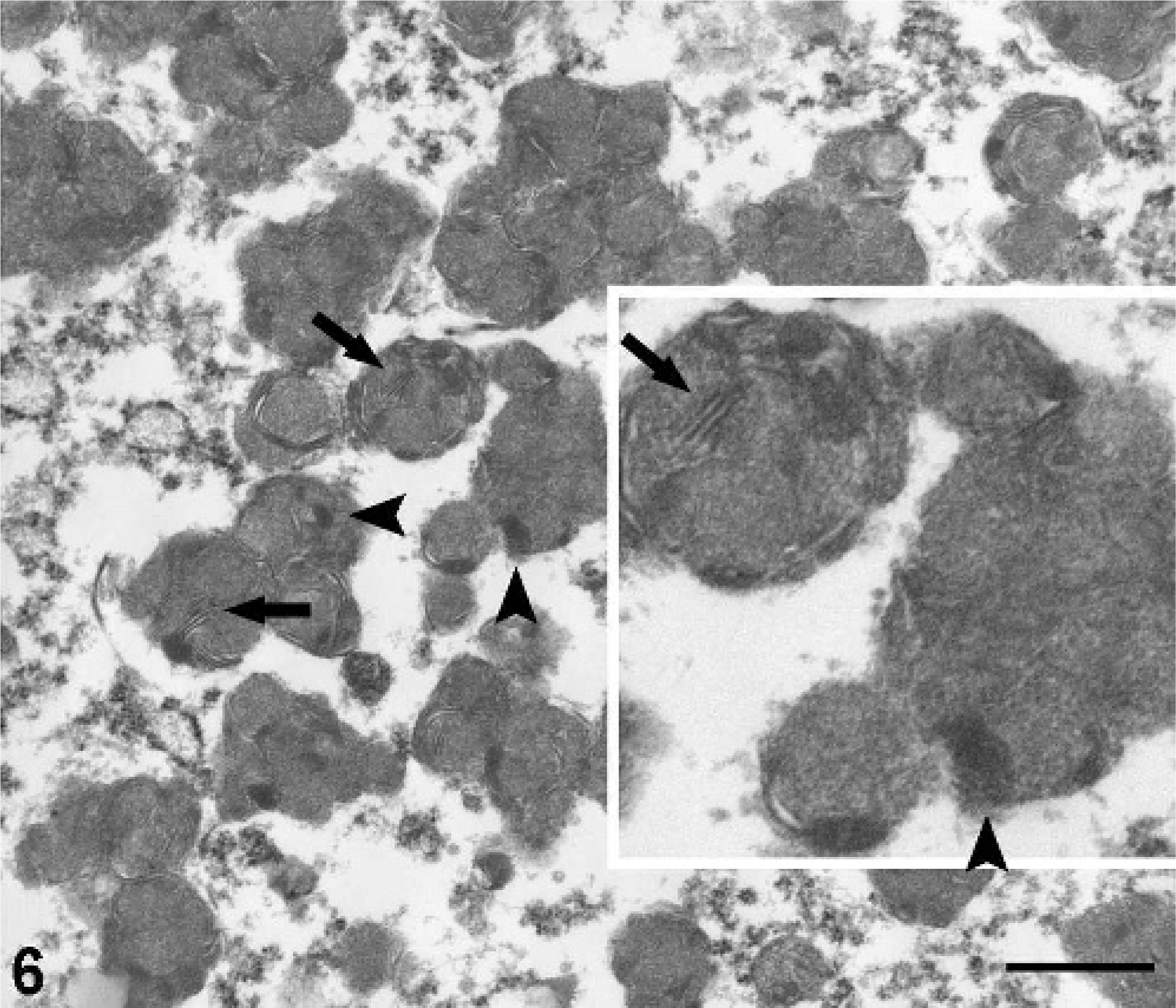
Brain, hippocampus; pot-bellied pig. Transmission electron micrograph of granular, osmophilic deposits (GRODS; arrowheads) and multilamellar profiles (arrows) of neuronal storage material. Methanolic uranyl acetate and lead citrate. Bar = 600 nm.
There was little correlation between accumulation of storage material and neuronal loss (see Table 1). This is consistent with human forms of NCL and supports the theory that neuronal loss is not solely attributable to the accumulation of storage material, but that other factors (e.g., excitotoxicity) may play a role. 16 This is also supported by the consistent observation that only the cells of the CNS are destroyed, even when storage material accumulates in cells outside the CNS. 6, 7, 10
The absence of seizures, visual deficits, and involvement of cells outside the CNS is unusual for NCL. The lack of involvement outside the CNS has been reported in Rambouillet 4 and Swedish Landrace sheep, 17 a mixed-breed terrier, 8 a Chihuahua, 11 and a domestic cat. 20 Blindness is an important feature in humans, developing in all forms except CLN4 (adult-onset NCL). 6, 7 Visual deficits were also absent in Swedish Landrace sheep, 17 Nubian goats, 5 a Cocker Spaniel dog, 12 a mixed-breed terrier, 8 and 2 domestic cats. 13, 20 Seizures occur in all forms of human NCL except a subset of CLN4. 6, 7 Seizures are far less common in animals, reported in only 3 species: Merino sheep, 3 English setter dogs, 10 and 1 domestic cat. 20 Though these 3 features may have developed later in the course of the disease (had the pig survived long enough), an alternative, perhaps more likely scenario given the severity of the CNS lesions, is that these features were not characteristics of the disease in this pig.
The most striking lesion was found in the hippocampus, followed by the cerebral cortex and cerebellum. Hippocampal lesions are prominent in most human cases of NCL where the distribution of lesions within the hippocampus varies with the genetic defect. 16 Though the pyramidal neurons are principally affected, in the most severe forms (CLN1 and CLN2), the neurons of the dentate gyrus also are destroyed. 16 In other human forms, there is mild to moderate accumulation of storage material in the neurons of the dentate gyrus, but, except for CLN5, there is typically no neuronal loss. 16 This is in contrast to NCL in animals, in which the hippocampus is not usually as severely affected. In fact, of the over 25 reports reviewed, the hippocampus is mentioned in only 10, and is listed among the most severely affected regions in 6. 1, 8, 11, 14, 17, 19 Similar to the condition in humans, the pyramidal neurons are principally affected, but the neurons of the dentate gyrus are typically spared, as they were in this pig. The spinal cord is also involved in most cases of NCL in animals and may be one of the most severely affected sites. 2, 5, 12 Similar to that in this pig, the ventral horn is often more severely affected. 2, 5
This report of NCL in a Vietnamese pot-bellied pig provides an opportunity to make others aware of the disease in this species. Future studies should aim at identifying families and siblings with this condition, which will allow characterization of the storage material and determination of the genetic basis of this important disease.
Footnotes
Acknowledgements
The authors would like to thank James Mangum for invaluable assistance in preparing the illustrations. We would also like to thank the histologists at North Carolina State University and the National Institute of Environmental Health Sciences for slide preparation, and Dr. Michael Dykstra and Laura Reuss in the Laboratory for Advanced Electron and Light Optical Methods at North Carolina State University College of Veterinary Medicine for their expertise in electron microscopy. This work was supported in part by the intramural research program of the National Institute of Environmental Health Sciences, a division of the National Institutes of Health.
