Abstract
Pulmonary arteriopathy (PA) is the pathologic hallmark in human medicine of diffuse constrictive (medial and intimal remodeling) or multifocal complex (plexiform and dilatative lesions) arterial lesions, or both, that lead to irreversible obliteration of the arterial lumen. Clinically, PA leads to pulmonary arterial hypertension (PAH), of which idiopathic (IPAH) is one of the 5 subsets, and ultimately, to right-sided heart failure (RHF). Clinical and pathologic findings from 6 dogs with diagnosis of IPAH and PA were reviewed. These dogs were of various pure (5/6, 83%) and mixed (1/6, 17%) breeding, 5 months to 9 years (mean 5.2 years) old, and predominantly female (4/6, 67%) and reproductively intact (4/6, 67%). Doppler echocardiography (n = 5) indicated increased pulmonary arterial pressures during systole (70-135 mm Hg, mean 98 mm Hg) and diastole (35-80 mm Hg, mean 58 mm Hg). All 6 dogs had right ventricular pressure overload, right ventricular eccentric hypertrophy, and RHF. Histologic examination confirmed the clinical diagnosis of IPAH in all dogs, revealing PA characterized by 1 of the 4 main human histologic subsets: 1) isolated medial hypertrophy (1/6, 17%); 2) medial hypertrophy-intimal thickening without the plexiform lesion (1/6, 17%); 3) medial hypertrophyintimal thickening concurrent with the plexiform lesion, which often was regionally clustered and situated near branching points of the respiratory artery, the poststenotic dilatation lesion, and vasculitis (4/6, 66%); and 4) isolated arteritis (1/6, 17%). Ancillary lesions similar to those in humans also complicated the PA (5/6, 83%). The complex lesions and ancillary exudative alveolitis seemed to be important indicators of severe, likely rapidly progressive and fatal, IPAH.
Keywords
Pulmonary arteriopathy (PA), previously known as plexogenic pulmonary arteriopathy, is the pathologic hallmark of a small group of diseases in human medicine with a similar, multifactorial pathobiology. It involves complex interactions of major genes, modifying genes, and epigenetic factors that result in pulmonary arterial hypertension (PAH), a devastating, life-threatening condition that is difficult to diagnose in the early stages. 2, 13, 20, 22 Histologically, PA is a constellation of constrictive and complex, obstructive, and proliferative vascular lesions that are localized to the pre- and intra-acinar pulmonary arteries and lead to the irreversible obliteration of the vascular lumen. These morphologic changes lead to progressive vascular resistance, increased right ventricular afterload, right ventricular eccentric hypertrophy, right atrial dilatation, and ultimately, right-sided heart failure (RHF).
Although the cause and pathogenesis of pulmonary hypertension (PH) remain poorly understood, attendees of the Third World Symposium on PAH revised the clinical and pathologic classification to incorporate recent advances in the understanding and clinical management of PH and to provide a more unified approach to diagnosis and therapy. 22, 24 Accordingly, within PH, PAH constitutes one category. Although previous clinical descriptions separated primary from secondary forms, current classifications subdivide PAH into 5 forms: idiopathic (IPAH), familial (FPAH), associated with risk factors or conditions (APAH), persistent pulmonary hypertension of the newborn, and associated with substantial venous or capillary involvement. 24 The latter 2 subdivisions do not manifest histologically as PA and, thus, will not be considered further. 22
Idiopathic pulmonary hypertension is a rare, sporadic disease with a predisposition for postpubescent females and mean age at diagnosis of 36 years. 19 Familial pulmonary hypertension develops in approximately 6% of cases of PAH as an autosomal dominant disorder with incomplete penetrance and genetic anticipation. 20 Pulmonary arterial hypertension associated with substantial venous or capillary involvement can be linked to the ingestion of certain drugs or chemicals, such as anorexigenic drugs or pyrrolizidine alkaloid-containing plants, and select diseases, such as certain congenital systemic to pulmonary shunt conditions, portal hypertension, collagen vascular disease, hematologic conditions, genetic or metabolic diseases, or infection with human herpesvirus 8 or human immunodeficiency virus. 5, 24
It is not certain whether IPAH, FPAH, and APAH are molecularly and, thus, mechanistically distinct processes; however, recent research suggests that all forms are based on genetic perturbations changing the phenotype of pulmonary cells. 13 An imbalance in the control of genes involved in cell proliferation and apoptosis apparently predisposes to cell damage by epigenetic factors and subsequent misguided angiogenesis. Implicated cellular pathways include impaired potassium channel and growth factor receptor function, altered serotonin transport regulation, increased oxidant stress, enhanced matrix production, and altered synthesis of nitric oxide, prostacyclin, and endothelin.
The revised pathologic classification of PH replaces a previous sequential grading scheme with a broad spectrum of arterial, capillary, and venous lesions. 12, 22 Within PH, PA includes lesions that are limited to the pre- to intra-acinar pulmonary arteries and are classified as constrictive (medial and intimal remodeling) or complex (plexiform and dilative lesions). 22 Pulmonary arteries are defined by the level of the accompanying respiratory tract structure, such that pre-acinar arteries are found proximal to the terminal (or tertiary) bronchiole, terminal arteries are found at the terminal bronchiole, and intra-acinar arteries are found from the level of the respiratory bronchiole distally. PA then is characterized as: 1) isolated medial hypertrophy; 2) medial hypertrophy and cellular to fibrous intimal thickening in a concentric laminar, concentric nonlaminar, or eccentic nonlaminar pattern; 3) plexiform and/or dilatation lesions or arteritis; and 4) isolated arteritis. These lesions can develop independently, concurrently with each other, and concurrently with venous-to-venule medial hypertrophy–intimal thickening. Adventitial thickening is an ill-defined change that usually is not involved in PA.
Constrictive lesions (Nos. 1 and 2) have a diffuse distribution and can affect hemodynamic function upon loss of vasorelaxant properties or production of vasoconstrictors. 22 Complex lesions (Nos. 3 and 4) have a multifocal distribution and are the hallmark of severe or rapidly progressive PAH, or both. The vessels in the dilatation lesion may rupture and predispose to various alveolar exudative lesions. 12, 22 Other ancillary, nonspecific changes commonly associated with PA include: thrombotic lesions, acute, organized, or recanalized; elastic arteries with neointimal or atheromatous plaques, medial hypertrophy, and elastin degeneration; bronchial vessel medial hypertrophy-intimal thickening; arterial calcification; foreign body emboli; and organized infarct with perivascular lymphocytic infiltrates. 22
Evaluation of PA by histologic examination has several important considerations and limitations. Several histologic sections that range from pre- to intra-acinar arteries are required, because the corresponding lesions are not necessarily distributed uniformly, and apparently normal vessels may exhibit plexiform lesions further downstream or upstream. 6, 22 Additionally, the revised pathologic classification advocates a nonsequential descriptive approach rather than a sequential grading system as was used previously. 12, 22 This revised approach reflects current research indicating that the characteristic plexiform lesion may not necessarily be an end-stage phenomenon, but can develop as a solitary lesion independent of medial hypertrophy and intimal thickening, which previously were believed to precede its development. 6, 22 Finally, all forms of PH, regardless of the cause, have some common pathologic features including medial hypertrophy of elastic and muscular arteries, dilatation and intimal plaques of elastic arteries, and right ventricular eccentric hypertrophy. 22 Still other diseases with no direct relationship to the presence or absence of PH, such as pulmonary interstitial disease or tumors, can have isolated or localized medial and intimal thickening of pulmonary arteries. Thus, the identification of PAH and PA is based on the localization of defined constrictive and complex lesions to pre- to intra-acinar pulmonary muscular arteries.
In dogs, PH characterized by PA with constrictive and complex lesions has been documented as naturally acquired in a 12-week-old, reproductively intact female, mixed-breed Chihuahua-type dog with left-to-right patent ductus arteriosus; in a 21-month-old, reproductively intact male Pembroke Welsh Corgi dog with right-to-left patent ductus arteriosus; in a 2-year-old, female (neuter status not provided) Labrador Retriever dog with a large, congenital, ventricular septal defect; and in an 8-year-old, male (neuter status not provided) Labrador Retriever dog without an underlying condition. 7, 9, 16, 26 It has been induced experimentally in 12- to 16-week-old, male (neuter status not provided) Beagle dogs by intracardiac injection of monocrotaline pyrrole, a pyrrolizidine alkaloid. 11 To the authors' knowledge, the incidence of PA in PAH associated with systemic-to-pulmonary shunt conditions has not been reported, largely because of lack of or incomplete histologic examination findings. 14 Clinical cases of PH associated with risk factors in dogs include alveolar hypoxia, increased pulmonary venous pressure, and pulmonary vascular obstruction. These conditions, similar to those in humans, have not been associated with PA; however, in many instances, the diagnosis was based on results of clinical tests, and detailed histologic examination was not pursued or described. 8, 14
The retrospective review of archived necropsy cases from the Pathology Service, Veterinary Medical Teaching Hospital (VMTH), University of California at Davis (UC Davis), California, reported here is the first documentation of multiple dogs presenting with various clinical stages and pathologic classifications of IPAH and PA. The purpose of this retrospective study was to discern whether the antemortem diagnosis of IPAH correlated with the histologic features of PA on postmortem examination, as defined by the pathologic classification used in human medicine.
Materials and Methods
Cases that were necropsied at the VMTH between 1983 and 2003 and having diagnosis of pulmonary vasculopathy were retrieved from the hospital database. Cases were included if they at least had antemortem imaging studies (preferably Doppler echocardiography) and complete necropsy had been performed. Cases with an antemortem diagnosis of idiopathic or primary PH were selected for review of the echocardiographic reports and archived echocardiographic images by a cardiologist (FEC) without knowledge of the necropsy results, and for detailed review of the pathologic findings by selected pathologists (DWW, TSZ). Clinical records were consulted to document breed, age, sex, and neuter status, duration and features of the presenting complaint(s), clinical course of disease, results of diagnostic procedures, and clinical diagnosis.
Two-dimensional (2-D) and Doppler echocardiographic studies were performed, using an Acuson 128XP/10 (n = 3; Siemens Medical Solutions Inc., Malvern, PA) or an HP Sonos 5500 (n = 3; Hewlett Packard Andover, MA), by a board-certified cardiologist or a cardiology resident under the former's supervision. Standard echocardiographic views were obtained. 25 Pulmonic stenosis was excluded in all dogs by observation of normal structure and motion of the pulmonic valve, lack of poststenotic pulmonary arterial dilatation on 2-D echocardiography (n = 6), and laminar pulmonary flow with normal velocity <1.5 m/s on pulsed wave Doppler echocardiography (n = 5). 4 Color flow mapping routinely was used to guide the alignment between the regurgitant jet and the Doppler signal. Optimal alignment with regurgitant flow was identified by a solid envelope on continuous wave Doppler echocardiography, from which peak tricuspid or pulmonic regurgitant jet velocity was measured. Systolic right ventricular-to-right atrial and diastolic pulmonary artery-to-right ventricular pressure gradients were calculated by application of the modified Bernouli equation (pressure gradient = 4 × [velocity in meters per second] 2 ) to the peak tricuspid or pulmonic regurgitant jet velocity, respectively. To give estimated pulmonary artery pressures in systole (SPAP) and diastole (DPAP), these pressures were added to the estimated pressure in the right atrium of 5 mm Hg for dogs not in heart failure or 10 mm Hg for dogs in heart failure. 23 The PH of each dog was categorized on the basis of estimated SPAP, such that mild equated to <50 mm Hg, moderate equated to between 50 and 75 mm Hg, and severe equated to >75 mm Hg. 15
Subjective echocardiographic assessment of right ventricular chamber size and wall thickness was made by comparison with left ventricular dimensions in the right parasternal long-axis view. 15 Normal right ventricular chamber size was identified if it was less than half the chamber dimension of the left ventricle. Moderate right ventricular dilatation was reported if the right ventricular chamber size was 50–100% of left ventricular chamber size. Severe right ventricular dilatation was reported if the right ventricular chamber was larger than the left. Normal right ventricular free-wall thickness was identified as less than half of the ventricular free-wall thickness. Moderate right ventricular hypertrophy was reported when wall thickness was between 50 and 150% of that of the left ventricular free wall. Severe right ventricular hypertrophy was reported when wall thickness exceeded 150% of that of the left ventricular free wall. RHF was identified on physical examination by the presence of ascites, a palpable abdominal fluid wave, or palpable hepatomegaly, and by dilated hepatic veins on abdominal ultrasonography.
Pathologic characterization of each case included a review of the documented findings on gross examination, evaluation of histologic slides, and application of immunohistochemical (IHC) analysis with special staining of select sections of lung tissue. All tissues from each case were immersed in neutral-buffered 10% formalin, processed in routine manner, and embedded in Tissue Prep (Fischer Scientific, Fairlawn, NJ). Five-micron-thick sections were cut and stained with hematoxylin and eosin (HE). Select serial sections of lung were stained with Masson's trichrome for collagen and smooth muscle and, in some instances, with acid orcein-Giemsa for elastin, Mallory's phosphotungstic acid hematoxylin for fibrin, and Okajima's stain for hemoglobin. 1, 27 They also were examined by IHC analysis using the following antibodies (all antibodies are mouse monoclonal anti-human unless otherwise specified): α-smooth muscle actin (SMA), clone 1A4 (1 ° 200, BioGenex, San Ramon, CA); anticanine CD45 (leukocyte common antigen [LCA]), clone IOC12 (1 ° 50, Dako Corp., Carpenteria, CA); Ki-67, clone MIB-1 (1 ° 75, Dako Corp.); and rabbit polyclonal von Willebrand factor VIII (F8), clone A0082 (1 ° 50, Dako Corp.); and in some instances with pancytokeratin, clone Lu-5 (1 ° 200, Biocare, Walnut Creek, CA), antibovine vimentin, clone 3B4 (1 ° 200, Dako Corp.), and CD31, clone JC/70A (1 ° 50, Dako Corp.). Immunohistochemical analysis was performed using a standard streptavidin–biotin–horseradish peroxidase technique as described, with similar cited modifications. 17, 21 Normal canine tissue and normal intrapulmonary structures within the case section were used as positive external and internal controls, respectively. Omission of the primary antibody served as a negative control. Leukocytes and epithelial cells were not included in the assessment of mitotic activity by immunoreaction to Ki-67.
In each instance, the following criteria were evaluated: disruption of normal venous or arterial architecture, or both; the level and distribution (diffuse or multifocal) within the vascular tree at which this disruption occurred; the features that characterized this disruption; the presence of each feature relative to others; and the presence, absence, and character of concurrent nonvascular-oriented pathology. Subsequently, each case was assessed relative to the revised human pathologic classification for PAH (Table 1). 12, 22 Differentiation between small arterioles and venules was guided by the presence of an accompanying airway or the occurrence within the alveolar septum, respectively. The severity of muscular hypertrophy and intimal thickening was based on the degree of occlusion of the vascular lumen, such that mild, moderate, and severe equated to minimal, partial, and complete luminal occlusion, respectively. Severity of the plexiform lesion was based on its frequency per 4× field, such that mild, moderate, and severe equated to ≤1 per three 4× fields, 1 per two 4× fields, and ≥1 per 4× field, respectively. The latter assignment was used to approximate the chance of finding the lesion before death in a tru-cut biopsy procedure that includes at least 3 biopsy specimens from 1 biopsy procedure and at least tertiary bronchi.
Case summary of canine idiopathic pulmonary arterial hypertension and pulmonary arteriopathy.
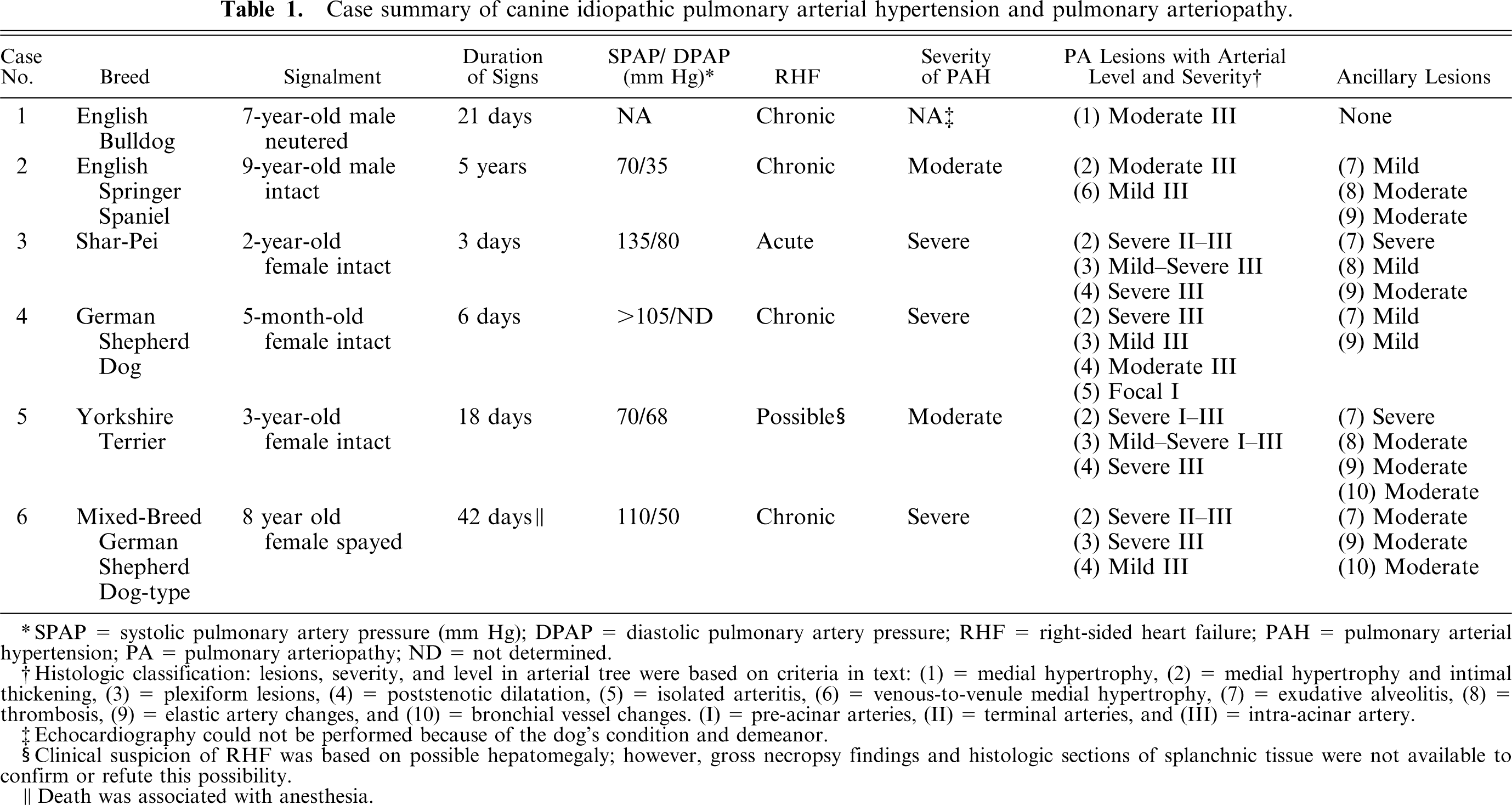
SPAP = systolic pulmonary artery pressure (mm Hg); DPAP = diastolic pulmonary artery pressure; RHF = right-sided heart failure; PAH = pulmonary arterial hypertension; PA = pulmonary arteriopathy; ND = not determined.
Histologic classification: lesions, severity, and level in arterial tree were based on criteria in text: (1) = medial hypertrophy, (2) = medial hypertrophy and intimal thickening, (3) = plexiform lesions, (4) = poststenotic dilatation, (5) = isolated arteritis, (6) = venous-to-venule medial hypertrophy, (7) = exudative alveolitis, (8) = thrombosis, (9) = elastic artery changes, and (10) = bronchial vessel changes. (I) = pre-acinar arteries, (II) = terminal arteries, and (III) = intra-acinar artery.
Echocardiography could not be performed because of the dog's condition and demeanor.
Clinical suspicion of RHF was based on possible hepatomegaly; however, gross necropsy findings and histologic sections of splanchnic tissue were not available to confirm or refute this possibility.
Death was associated with anesthesia.
Results
Summary of case series
Archival review yielded 6 cases from 1992 to 2003 that fulfilled the specified criteria for pulmonary vasculopathy and idiopathic PH (Table 1). The cohort was composed of various purebred dogs and 1 mixed-breed dog, 4 females (3 reproductively intact, 1 spayed) and 2 males (1 reproductively intact, 1 neutered), and age ranging from 5 months to 9 years (mean 5.2 years of age). The duration of clinical signs of disease ranged from 3 days to 5 years (mean 320 days). Clinical signs commonly were characterized by progressive dyspnea or tachypnea, or both, and progressive lethargy, weakness, or exercise intolerance or intermittent collapse, or both, usually temporally associated with stress, exercise, or excitement.
Pulmonary artery pressures were estimated from continuous wave Doppler echocardiographic examination of tricuspid and pulmonic regurgitant jet velocity in 5 dogs. Estimated SPAP ranged from 70 to 135 mm Hg (mean 98 mm Hg; normal range 15–25 mm Hg), and DPAP ranged from 35 to 80 mm Hg (mean 58 mm Hg; normal range 10–15 mm Hg). 10 Accordingly, moderate PH was identified in dog Nos. 2 and 5, severe PH was identified in dog Nos. 3, 4, and 6, and incomplete echocardiographic examination precluded objective measurement of regurgitant jet velocity in dog No. 1 (Table 1). Echocardiography of all 6 dogs revealed paradoxic motion of the interventricular septum, indicative of right ventricular pressure overload, and moderate right ventricular hypertrophy. Also, the right ventricle was either moderately (dog Nos. 1 and 5) or severely dilated (dog Nos. 2–4, 6). Right-sided heart failure was identified in dog Nos. 1–4 and 6 and was suspected in dog No. 5. All dogs arrested spontaneously, except for dog No. 6, in which death was associated with routine anesthesia for a lung biopsy.
In all dogs, postmortem evaluation revealed pulmonary changes consistent with the pathologic classification of PA in humans, as well as variable degrees of the aforementioned compensatory changes associated with acute and chronic PH, and subsequent RHF. 22 By use of histologic examination, RHF was acute in dog No. 3 and chronic in dog Nos. 1, 2, 4, and 6. In dog No. 5, the suspected RHF on clinical examination could not be confirmed by postmorten examination, because a gross pathology report was not available and splanchnic viscera were not submitted for evaluation. Evidence of cardiac malformations or gross structural abnormalities in the great vessels was not found in any of the remaining dogs. On histologic review of lung sections from all dogs, all 4 histologic subsets of PA and several ancillary lesions were identified. Representative lesions and their distribution within the pulmonary arterial tree are described under the subsequent headings of isolated medial hypertrophy, medial hypertrophy–intimal thickening, complex and constrictive lesions, and ancillary lesions. A general overview and unique features of each case are described under the heading of individual case summaries.
Isolated medial hypertrophy
For this lesion, mild-to-moderate (nonocclusive) medial hypertrophy developed diffusely throughout the intra-acinar arteries, but rarely involved alveolar capillaries. Medial hypertrophy was characterized by increased size and numbers (hyperplasia) of smooth muscle cells in the tunica media, with extension into nonmuscularized intra-acinar arteries (Fig. 1). This alteration was accompanied by increased deposition of collagen and elastic fibers, as confirmed by positive IHC results and trichrome staining for smooth muscle cells and collagen fibers. Plump endothelial cells lined the tunica intima. The lesion did not contain cells immunoreactive to LCA. Rarely, affected intra-acinar arteries had endothelial cells immunoreactive to Ki-67.
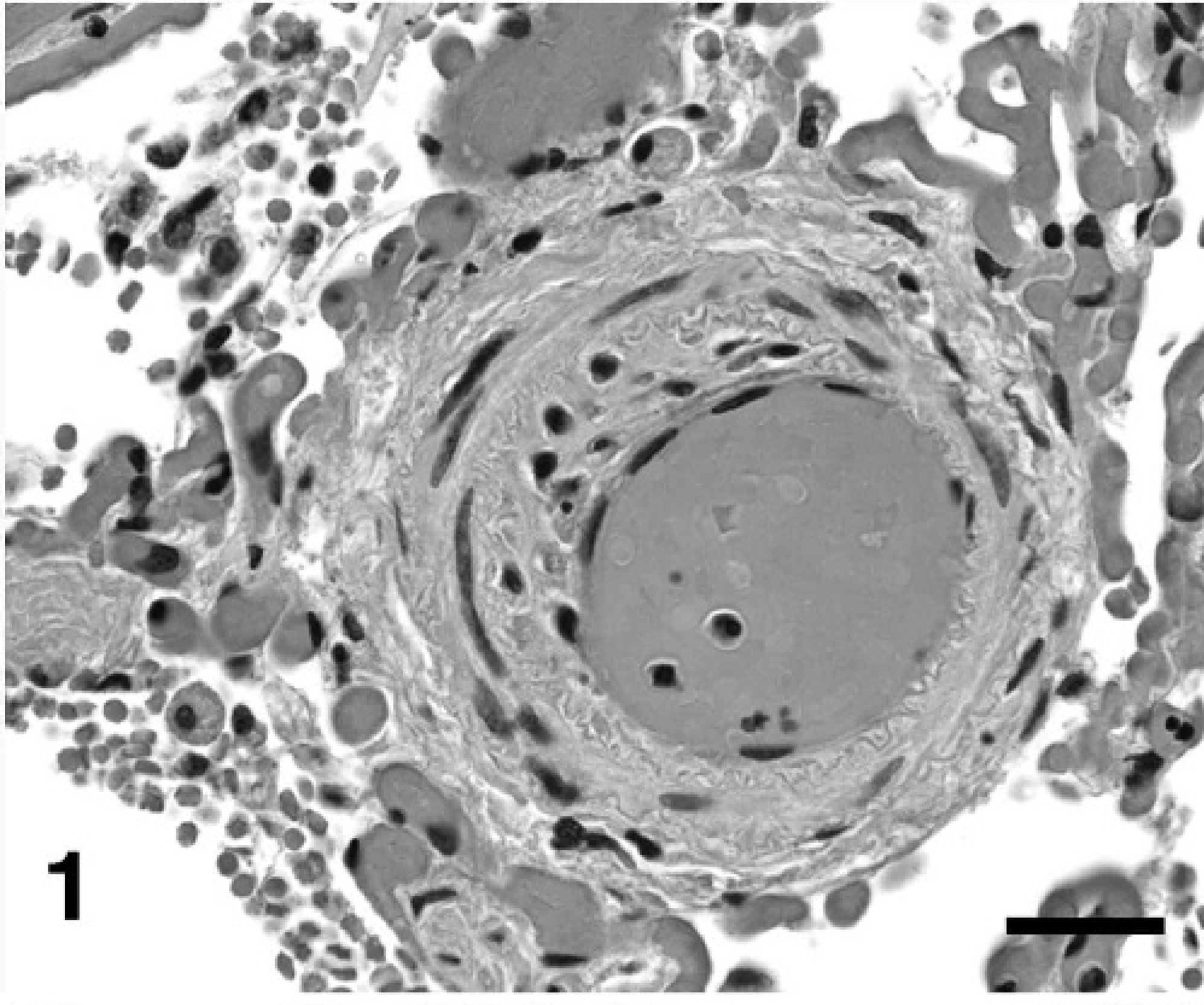
Lung, dog No. 3: Eccentric nonlaminar medial hypertrophy and intimal thickening of an intra-acinar artery causing moderate stenosis is characterized by compromise of the internal elastic membrane, intimal migration, and proliferation of vasoformative cells, and increased extracellular matrix. HE. Bar = 20 µm.
Medial hypertrophy–intimal thickening
In this lesion, terminal and intra-acinar arteries had medial hypertrophy, often intimal thickening by cells, and rarely, fibrous connective tissue. At the level of the terminal respiratory artery, medial hypertrophy was mild to moderate (nonocclusive) and usually not accompanied by intimal thickening. Distally, however, medial hypertrophy was moderate to severe (occlusive) and was accompanied by intimal thickening as well as medial hypertrophy of small venules.
Medial hypertrophy was characterized by alterations similar to those described previously. Intimal thickening was characterized by disruption of the inner elastic membrane and narrowing of the vascular lumen by the proliferation and migration of plump, spindle- to oval-shaped cells and deposition of eosinophilic, fibrillar-to-hyalinized or pale basophilic, wispy extracellular matrix in a concentric laminar, concentric nonlaminar, or infrequently, an eccentric nonlaminar pattern (Figs. 1, 2). This alteration caused partial to complete luminal occlusion. (Fig. 3). These vasoformative cells were immunoreactive to SMA and, in the innermost layer, were immunoreactive to F8. The extracellular matrix rarely stained for collagen by trichrome and contained few elastin fibers (Fig. 4). The lesion did not contain cells immunoreactive to LCA. Cells immunoreactive to Ki-67 were not evident.
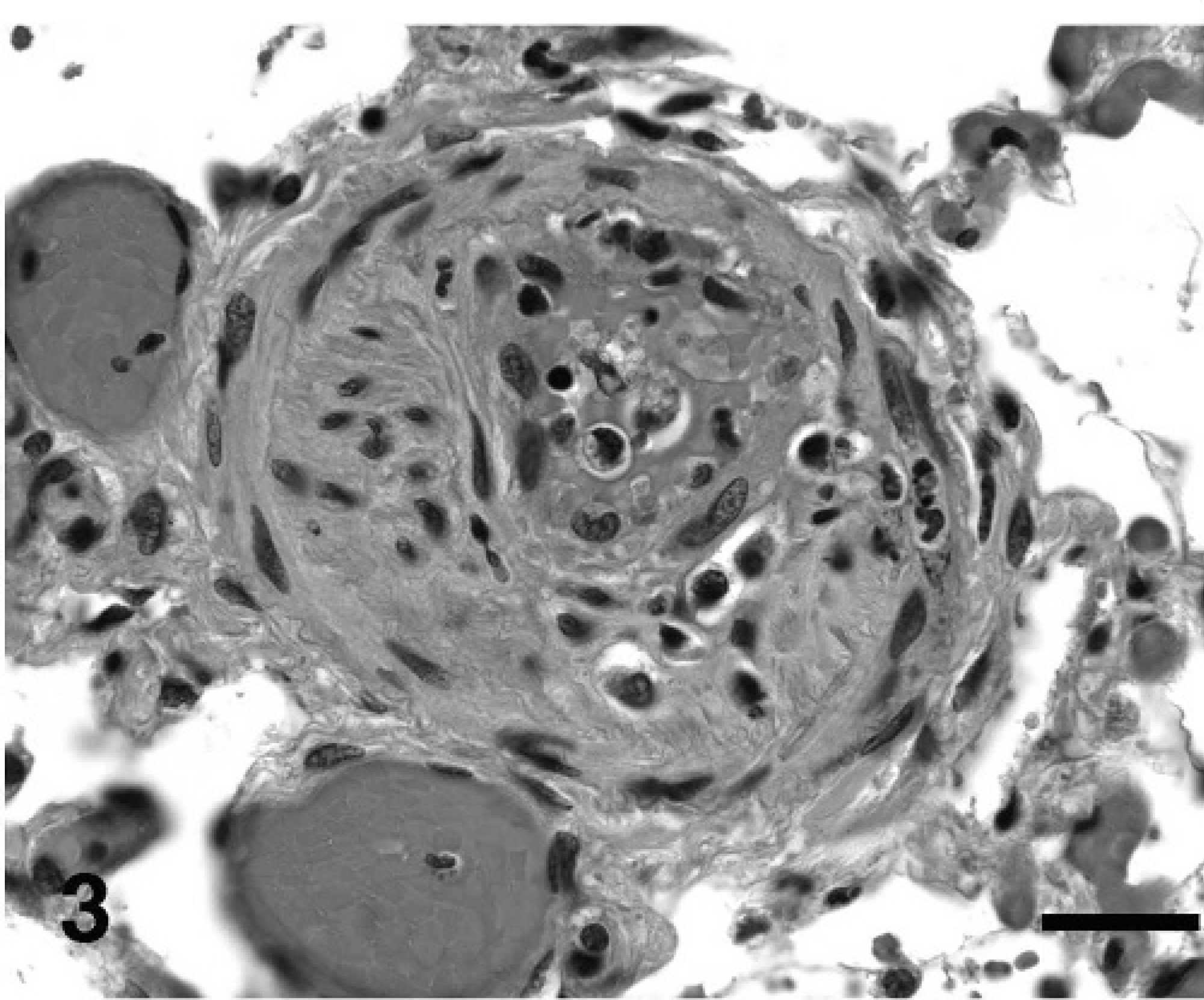
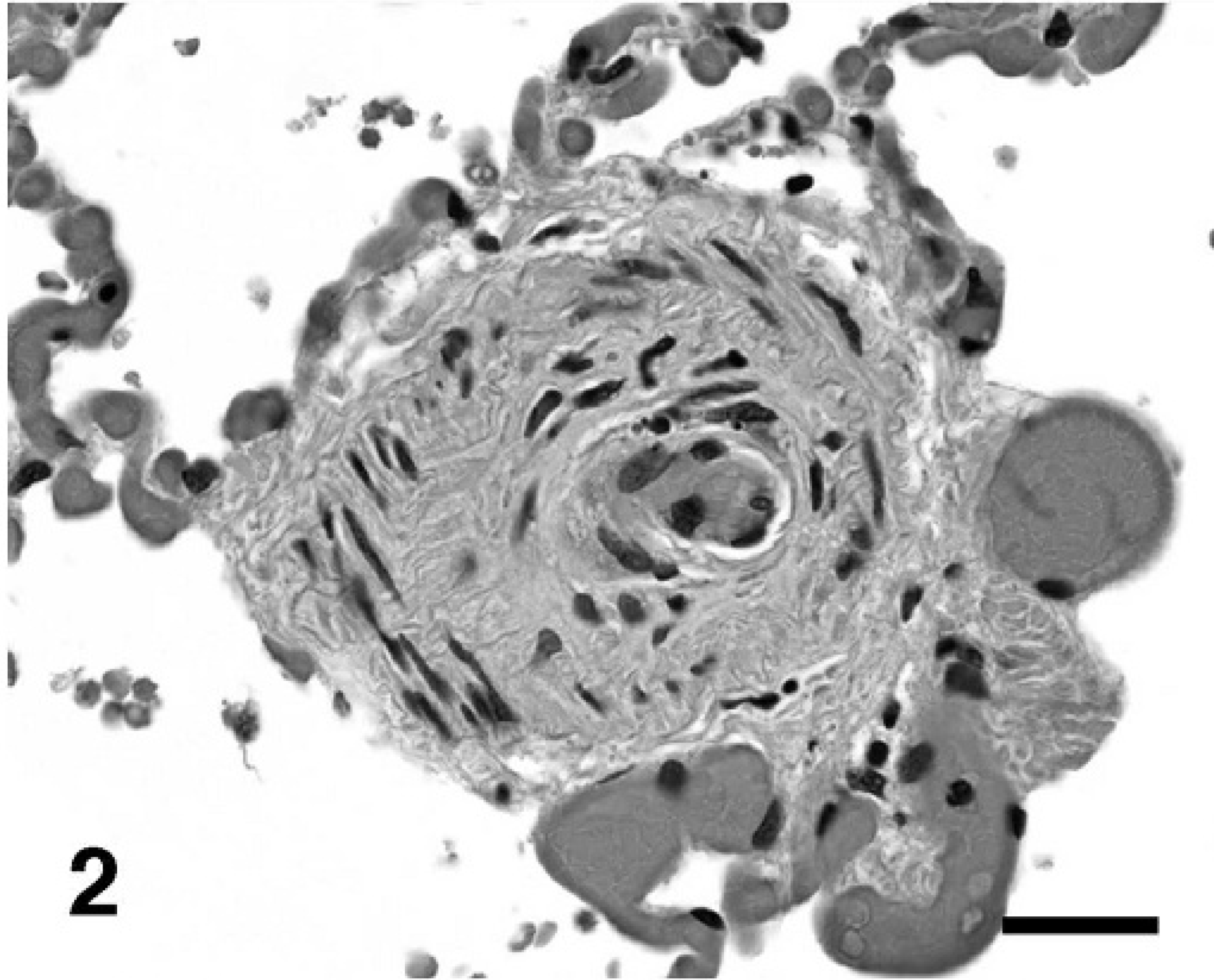
Lung, dog No. 3: Early plexiform lesion in an intra-acinar artery with active fibrinoid necrosis of the vascular wall admixed with vasoformative cells and variable extracellular matrix that occludes the lumen and partially destroys the vascular wall to extend into the perivascular tissue. There is moderate poststenotic capillary dilatation. HE. Bar = 20 µm.
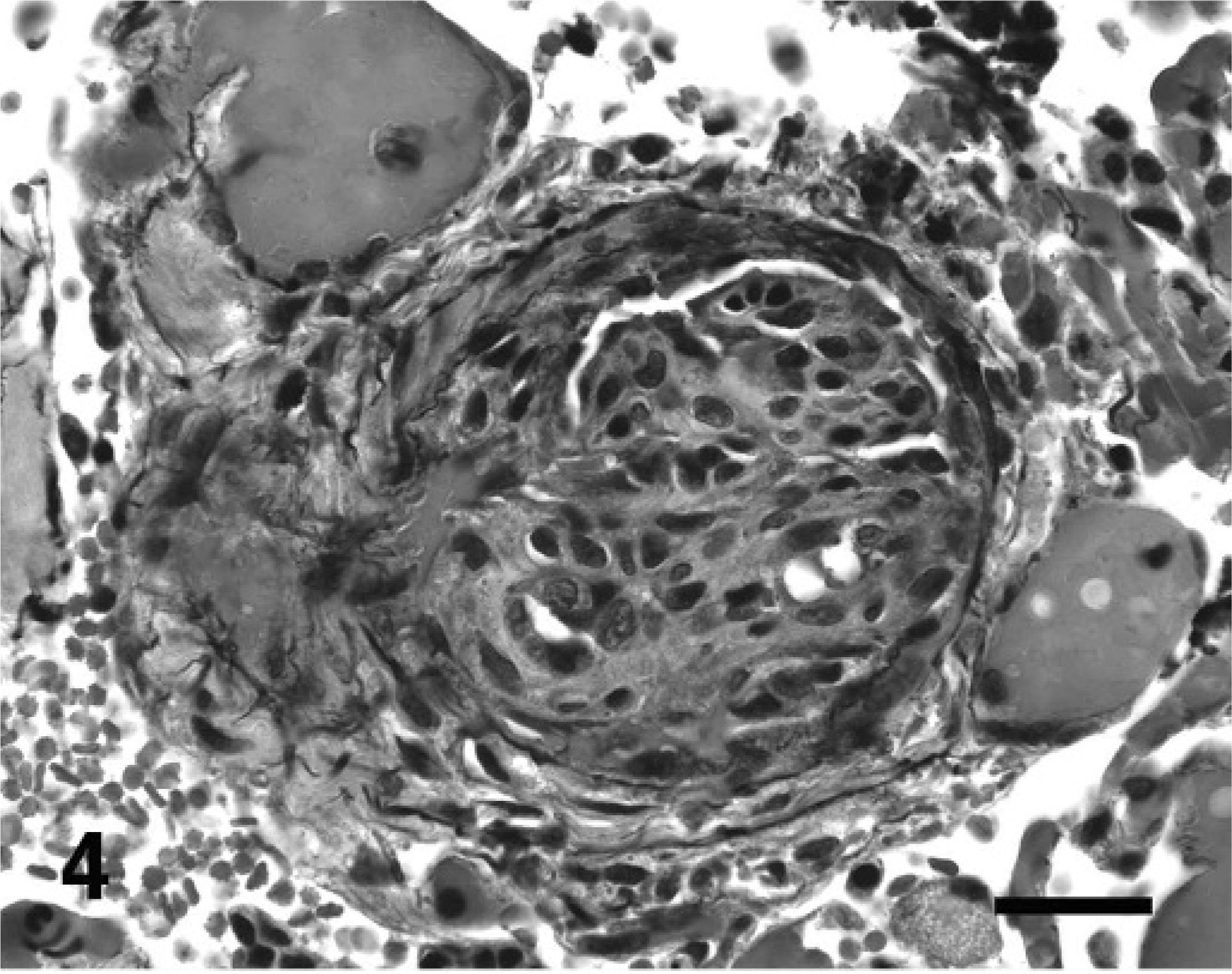
Lung, dog No. 3: A plexiform lesion stained for elastin (black) highlights compromised internal and external elastic membranes and the increase in elastin as a component of extracellular matrix deposition. Acid-orcein-Giemsa. Bar = 20 µm.
Complex and constrictive lesions
For this lesion, pre- to intra-acinar arteries had diffuse constrictive lesions and multifocal complex lesions, including plexiform and dilatation lesions, often with associated fibrinonecrotic to leukocytic arteritis. The constrictive lesions tended to develop at least one level higher in the pulmonary arterial tree than did the complex lesions, but were most severe at or below the level of the complex lesions and did not involve the venous system. Plexiform lesions often had a clustered distribution, such that in some regions, there was 1 structure per three 4× fields, and in other regions, there were up to 4 structures per 4× field. The plexiform lesion developed at branching points, predominantly of terminal to alveolar duct arteries and infrequently of pre-acinar secondary arteries or the most distal ramifications of intra-acinar arteries (Fig. 5). The dilatation lesion involved the distal ramifications of the artery containing the plexiform lesion; however, this spatial relationship was not always apparent in the section examined. In one dog, there also was focal, moderate, nonsuppurative arteritis affecting a secondary artery that had no other alterations and was removed from constrictive and complex lesions (i.e., isolated arteritis). Plexiform and dilatative lesions also were associated occasionally with exudative lesions (Fig. 6).
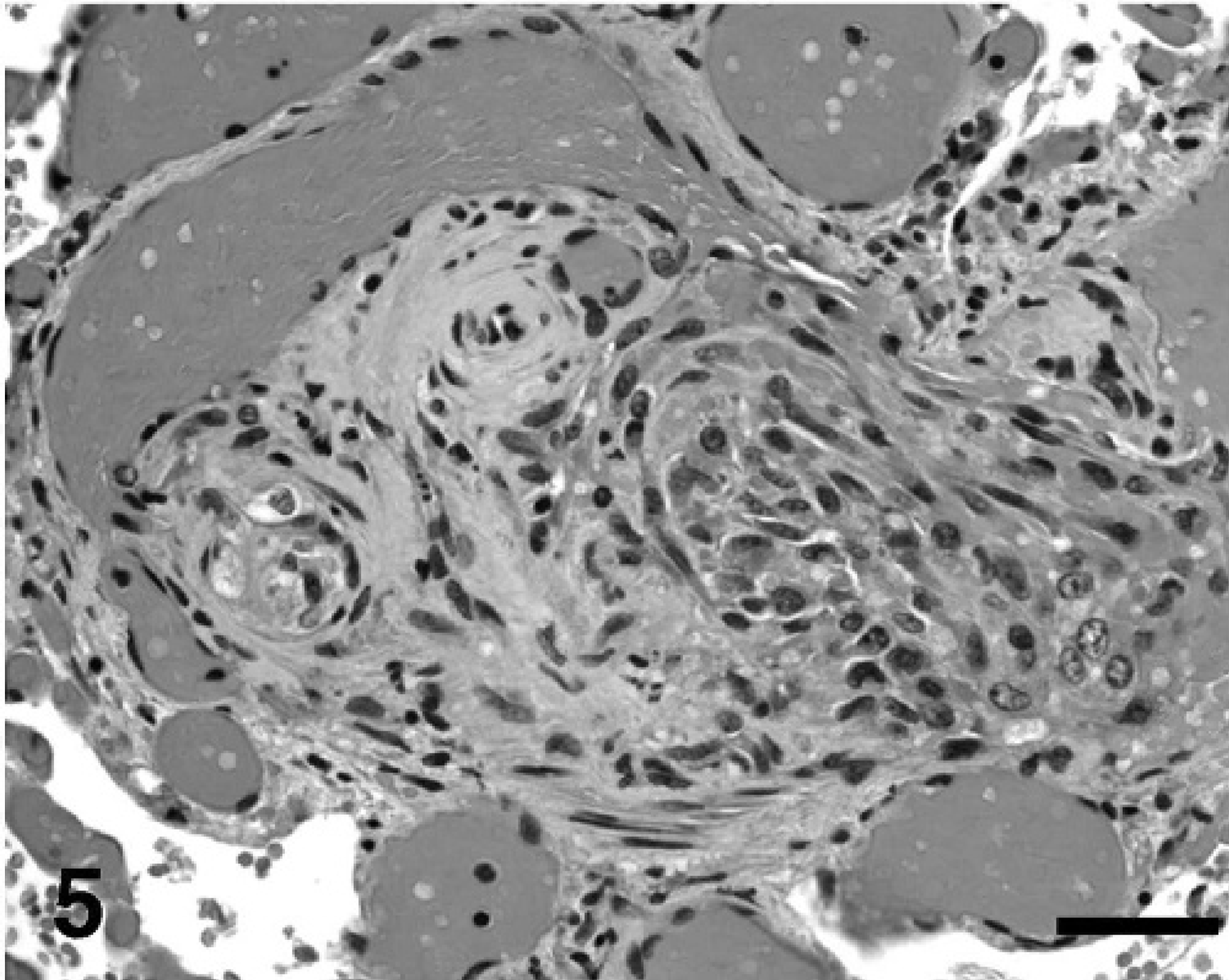
Lung, dog 3: Mature plexiform lesion in an intra-acinar artery, and the proximal ramification showing concentric laminar hypertrophy and intimal proliferation. The plexiform lesion has remnant fibrinoid necrosis of the vascular wall admixed with vasoformative cells and various extracellular matrix that expand and occlude the lumen and form small vascular clefts. The lesion partially destroys the vessel wall and extends into the perivascular tissue. Poststenotic capillary dilatation almost assumes an angiomatoid appearance. HE. Bar = 20 µm.
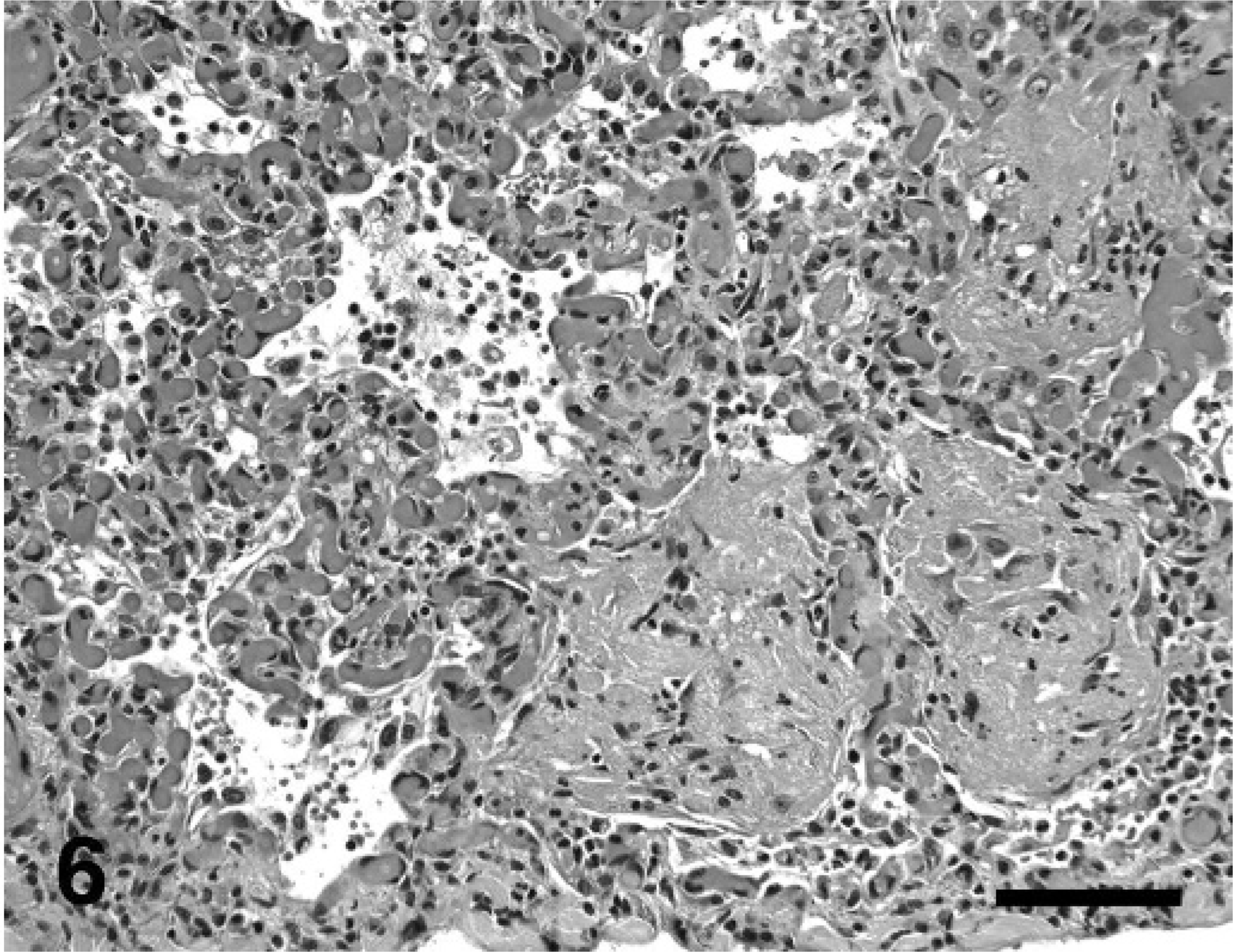
Lung, dog No. 3: Exudative alveolitis develops predominantly along the subpleura in association with marked vascular dilatation. Exudative material fills and replaces alveoli and is composed of coalescing lakes of proteinaceous material admixed with few to moderate numbers of neutrophils, histiocytes, and (myo)fibroblasts and is accompanied by marked hyperplasia of type-II pneumocytes. HE. Bar = 200 µm.
Intimal thickening was similar to that described previously, with 2 exceptions. The cellular and matrix deposition was observed more often in the eccentric nonlaminar pattern (Fig. 2) as well as in the concentric laminar and concentric nonlaminar patterns (Fig. 3). Second, affected arteries infrequently had fibrinonecrotic to leukocytic arteritis. When present in section, the artery located proximal to the plexiform lesion often had medial hypertrophy and fibrous to cellular intimal thickening in a concentric laminar pattern.
Lung, dog No. 3: Concentric nonlaminar medial hypertrophy and intimal thickening of an intra-acinar artery causing severe stenosis and mild poststenotic capillary dilatation. HE. Bar = 20 µm.
The plexiform lesion had a dynamic appearance in size, structure, and composition (Fig. 5). In affected arteries, active or remnant fibrinoid necrosis of the vascular wall was admixed with tightly packed vasoformative cells and various extracellular matrix that expanded and occluded the lumen and formed small vascular clefts. These lesions partially destroyed the vessel wall and extended into the perivascular tissue. The perivascular tissue also contained small to moderate numbers of histiocytes and lymphocytes, and fewer neutrophils and plasma cells that infrequently were present within the plexiform lesion, as confirmed by IHC analysis to be LCA. Plump, oval cells, which were immunoreactive to F8, lined or were proximate to vascular clefts (Figs. 7a, b). Depending on the plexiform structure, the remaining cells were variably immunoreactive to SMA (Fig. 7c) or only vimentin, and similarly stained for smooth muscle or collagen with trichrome. Cells immunoreactive to Ki-67 were confined to plexiform lesions and varied in number among structures, sometimes being absent.
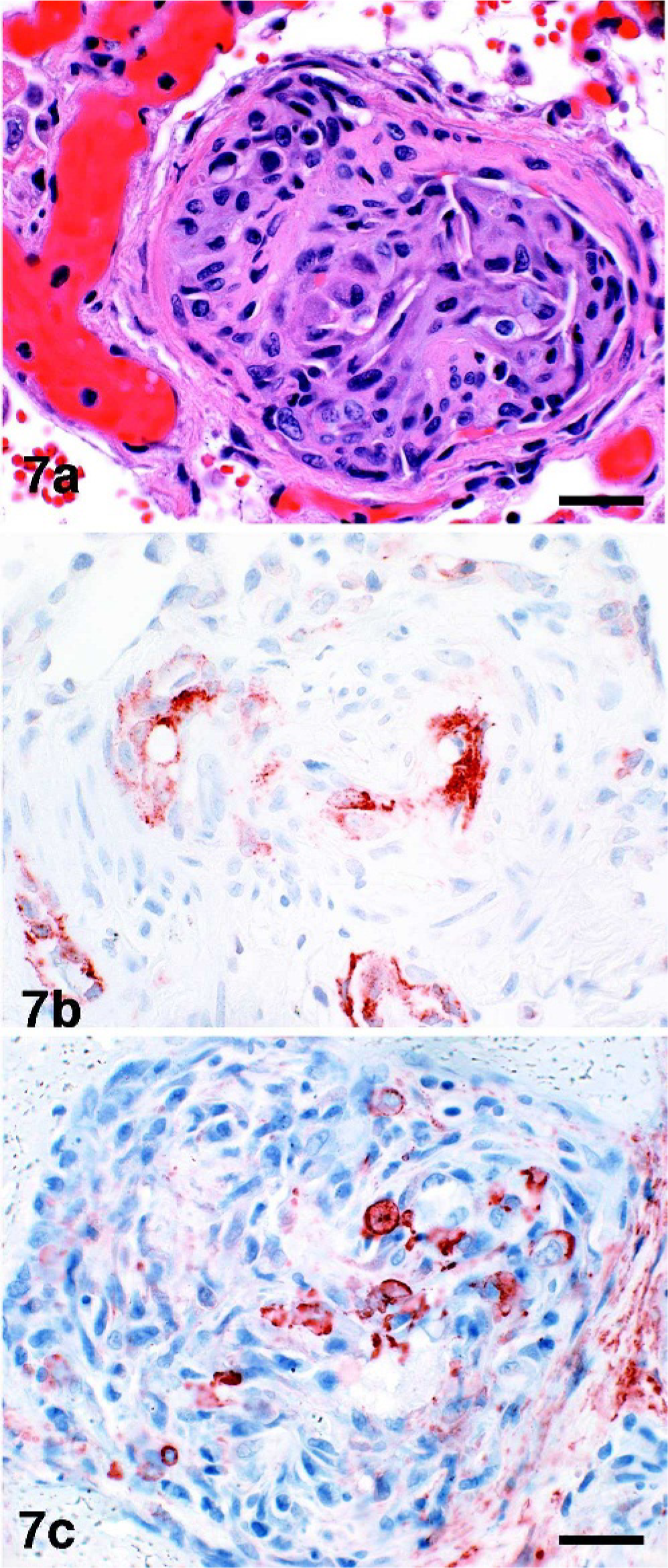
Lung dog No. 5: Immunoreactivity of plexiform lesion.
The dilatation lesion developed where an artery and its ramifications located distal to the plexiform lesion were thin walled and markedly dilated with blood. These vessels sometimes assumed an angiomatoid appearance. The wall of some dilated vessels had segmental fibrinoid necrosis.
Ancillary lesions
In addition to constrictive and complex arterial lesions, some dogs had evidence of thrombosis, exudative alveolitis, or changes in elastic pulmonary or bronchial arteries, or all three. In thrombotic lesions, pre-acinar arteries and fewer terminal or intra-acinar arteries had, with mild to moderate frequency, occlusive to nonocclusive acute, organized, or recanalized fibrin thrombi that sometimes were associated with focal fibrinoid necrosis of the underlying vascular wall. A mild exudative alveolitis surrounded some thrombosed vessels. More-extensive, exudative lesions were associated with dilated vessels and were observed predominantly along the subpleura, as well as in proximity to plexiform and occasionally constrictive lesions (Fig. 6). The exudative process filled and replaced alveoli. It was composed of coalescing lakes of amorphous to granular, eosinophilic material admixed with small to moderate numbers of neutrophils, histiocytes, and (myo)fibroblasts and was accompanied by marked hyperplasia of type-II pneumocytes, which often were hypertrophied and pleomorphic, and spanned remnant, damaged alveolar septa. The associated capillary bed infrequently contained acute fibrin thrombi. Staining with PTAH for fibrin, trichrome for collagen, and IHC for LCA, epithelial cells (pancytokeratin), mesenchymal cells (vimentin), and smooth muscle cells (SMA) confirmed the composition of the exudate, cellular infiltrate, and thrombi. Alterations in larger elastic arteries were characterized by mild to moderately thick, noncircumferential-to-circumferential neointimal plaques that sometimes were associated with underlying medial hypertrophy and elastin degeneration. Finally, bronchial arteries infrequently had mild medial hypertrophy.
Individual case summaries
Although most dogs had limited gross evidence of pulmonary vascular disease, the lung from dog No. 3 (2-year-old, reproductively intact female Shar-Pei dog) had distinctive changes reflective of the histologic lesions. Small to medium-sized luminal structures (interpreted as blood vessels) had increased prominence attributable to a white-to-gray rim effect, which corresponded histologically to PA with constrictive and complex lesions. The subpleural region of all lung lobes had multifocal to coalescing, irregular, sharply demarcated, depressed, gray to dark-red foci. Occasionally, similar foci were scattered within the parenchyma. Histologically, these regions corresponded to the exudative lesion.
An individual case summary of the histologic appearance of PA, including the nature, severity, and distribution within the pulmonary arterial tree, is presented in Table 1. Dog No. 1 had isolated medial hypertrophy. Dog No. 2 had medial hypertrophy–intimal thickening without the plexiform lesion, but with concurrent venule medial hypertrophy. Dog Nos. 3–6 had variations of the plexiform lesion and medial hypertrophy–intimal thickening. Dog No. 4 also had focal, isolated, nonsuppurative arteritis. No dog had the plexiform lesion independent of medial hypertrophy–intimal thickening. Ancillary lesions included exudative alveolitis (dog Nos. 2–6), vascular fibrin thrombosis (dog Nos. 2, 3, 5), and changes in elastic arteries (dog Nos. 2–6) and bronchial arteries (dog Nos. 5, 6). Clinical examination of dog No. 1 revealed edema of the soft palate, which subsided with 1 intravenously administered dose of dexamethasone. This dog also had moderate alveolar edema with histiocytosis. No dog had arterial calcification, foreign body emboli, or organized infarction with perivascular lymphocytic infiltrates, as has been described in some human cases. 22
In dog No. 4 (5-month-old, reproductively intact female German Shepherd Dog), the pulmonary vasculature generally had a subtle infantile appearance. Within PA, the morphology, immunophenotype, and distribution in the pulmonary arterial tree of constrictive lesions and complex lesions were similar to those of dog No. 3, with a few deviations. The constrictive lesion predominantly affected intra-acinar arteries. The plexiform structures were smaller and uniformly less frequent. The dilatation lesion was less severe and without angiomatoid formation. Dissimilar to all other cases, there was isolated nonsuppurative arteritis affecting 1 secondary artery that had no other structural changes.
In dog No. 5 (3-year-old, reproductively intact female Yorkshire Terrier dog), the PA was generally similar to that in dog No. 3; however, the constrictive and complex lesions were more pervasive throughout the pulmonary arterial tree, and the complex lesions developed higher in the pulmonary tree and were of slightly different morphology and composition. The medial hypertrophy–intimal thickening lesion extended from the pre-acinar arteries, including the elastic arteries, to the intra-acinar arteries, but without extension into the capillary bed. The plexiform lesion developed predominantly at branching points of secondary, terminal, and respiratory arteries. The plexiform lesion contained a greater number of F8-positive endothelial cells and a consistent predominance of smooth muscle cells and matrix other than collagen, as indicated by IHC analysis for SMA and trichrome staining. Similar to that in dog No. 3, however, the plexiform lesion had a clustered distribution with up to 3 structures in some 4× fields, and a marked poststenotic dilatation lesion, sometimes assuming an angiomatoid appearance.
In dog No. 6, (8-year-old, spayed female, mixed-breed German Shepherd Dog-type), the constrictive and complex lesions of the PA had morphology and immunophenotype similar to that of the lesions in dog Nos. 3 and 5, respectively; however, the distribution was more uniform throughout the lobes and extended lower into the pulmonary arterial tree than that in dog Nos. 3–5. The medial hypertrophy–intimal thickening lesion had similar morphology, immunophenotype, and distribution to that of the lesion in dog No. 3, but also extended into the capillary bed. In contrast to that in dog Nos. 3–5, the plexiform lesion was consistently more frequent rather than clustered, with up to 4 structures per 4× field, and developed predominantly at branching points of terminal and intra-acinar arteries, including the most distal ramifications. Dog No. 6 also had elastic arteries with morphologic alterations similar to those described in dog No. 5, but intimal plaques sometimes were atheromatous.
Discussion
This retrospective study of 6 dogs with ante- and postmortem diagnoses of PAH and PA provides a histologic description of a constellation of lesions that appear similar to those described in the human pathologic classification of PA. 22 It also indicates several possible trends, for which a larger study population is required for confirmation. Ante- and postmortem evaluations did not reveal a predisposing cause for PAH, such as those identified for APAH in human medicine and described or hypothesized in veterinary medicine. 7, 8, 11, 15, 16, 22 Thus, this canine series represents IPAH or perhaps FPAH.
In this small cohort of dogs, 67% were reproductively intact, 67% were female, and mean age at diagnosis was 5.2 years (middle-aged). This possible trend is interesting because the VMTH sees mostly neutered pets, and in humans with IPAH, postpubescent females are predisposed, and 36 years (middle-aged) is the mean age at diagnosis. 19 On the basis of results of this study and the previous case report in a Labrador Retriever dog, there currently is no identifiable breed predisposition.
Estimation of right heart pressure gradients from continuous wave Doppler echocardiographic recordings of tricuspid and pulmonic regurgitant velocities is widely used in human and veterinary patients to assess the presence and magnitude of PH. 15, 18 In the current study, a trend was observed between the severity of PH assessed by echocardiographically estimated SPAP and the severity of the histologic lesions, with the possible exception of dog No. 5. One explanation may be that SPAP was underestimated in this dog because of suboptimal alignment of the Doppler signal with regurgitant flow. 3
Histologic examination of HE-, special-, and IHC-stained sections of lung from multiple regions, which ranged from large elastic pulmonary arteries to capillaries, indicated that these dogs with IPAH had PA characterized by the same constellation of constrictive and complex lesions as described in the human pathologic classification of PA. Similar ancillary lesions also complicated the PA. All 4 of the main histologic subsets were identified, including isolated medial hypertrophy (dog No. 1), medial hypertrophy–intimal thickening without the plexiform lesion but with venule medial hypertrophy (dog No. 2), variations of the plexiform lesion and medial hypertrophy–intimal thickening (dog Nos. 3–6), and isolated arteritis (dog No. 4). No dog had the plexiform lesion independent of medial hypertrophy–intimal thickening. Ancillary lesions included exudative alveolitis (dog Nos. 2–6), fibrinonecrotic or leukocytic arteritis of vessels having complex and sometimes constrictive lesions (dog Nos. 3–6), vascular fibrin thrombosis (dog Nos. 2, 3, 5), and changes in elastic arteries (dog Nos. 2–6) and bronchial arteries (dog Nos. 5, 6). No dog had arterial calcification, foreign body emboli, or organized infarction with perivascular lymphocytic infiltrates, as was evident in humans with PA. 22
In humans and dogs, constrictive lesions have a diffuse distribution at the level of the affected artery, and the complex lesions have a multifocal distribution. 22 Additionally, the plexiform lesion may be regionally clustered and apparently normal vessels may exhibit plexogenic changes further downstream or upstream, as indicated in dog Nos. 3–5. 6, 22 This possibility makes the examination of multiple lung sections that range from pre-acinar to intra-acinar arteries imperative for a correct diagnosis. Also, plexiform lesions located distal to the respiratory artery can be more difficult to identify.
Human studies indicate that the diffuse constrictive lesions can affect hemodynamic function upon loss of vasorelaxant properties or production of vasoconstrictors. 22 The complex lesions are important indicators of severe or rapidly progressive PAH, or both. Within the complex lesions, the plexiform lesion indicates severe occlusion of the affected vessel along its entire length and, thus, permanent alteration in vascular morphology and communication with the capillary bed. The dilatation lesion predisposes to various exudative lesions. 6, 22 On the basis of this small study, the complex lesions seem to have the same connotation in dogs as in humans. Dog No. 2 had a 5-year clinical course of IPAH and PA with medial hypertrophy–intimal thickening, whereas dog Nos. 3–6 had a 3- to 42-day clinical course (mean 17 days) of IPAH and PA with complex lesions. Furthermore, among the last series of cases, dog No. 6 had the longest clinical course of 42 days and PA with the least extensive dilatation lesion and least exudative alveolitis. Inconsistent with this trend, dog No. 1 had a 21-day clinical course and PA with isolated medial hypertrophy. One explanation for this discrepancy may be that respiratory distress was complicated by laryngeal edema. In light of the pulmonary condition, response to therapy, and the proximal respiratory conformation in brachycephalic breeds, laryngeal edema could be considered a sequela and confounding factor rather than a cause for the PA. This dog also had moderate alveolar edema, for which the pathogenesis was uncertain. The latter was not associated with left-sided heart failure, as confirmed by echocardiography, and was not predisposed by the PA having complex, dilatative, or exudative lesions.
Medial hypertrophy in humans and dogs connotes hypertrophy and hyperplasia of smooth muscle fibers, increased deposition of connective tissue and elastic fibers in the tunica media, and extension of smooth muscle into nonmuscularized intra-acinar arteries. 22 Human studies indicate that medial hypertrophy represents not only a numerical increase but also a phenotypic change of smooth muscle cells in the tunica media toward matrix secretion. Phenotypic studies in dogs are needed to further define the similarity of this change to those in humans.
Intimal thickening in humans and dogs can be cellular or fibrous and deposited in a concentric laminar, eccentric nonlaminar, or concentric nonlaminar pattern which, in humans, depends on the cell type and stimulus. 22 In both species, the concentric laminar pattern usually is found just proximal to the plexiform lesion. 4 Even though the vasoformative cells may display immunophenotypic and, in humans, ultrastructural features of fibroblasts, myofibroblasts, and smooth muscle cells, human studies indicate that they actually originate from the localized proliferation and transformation of pluripotential endothelial cells or intimal fibroblasts, or both. 22 In humans and dogs, eccentric or concentric nonlaminar thickening are composed predominantly of (myo)fibroblasts and various types of connective tissue. Human studies suggest that they originate from the localized proliferation of intimal fibroblasts, perhaps in response to hemodynamic stresses and local release of growth factors. Interestingly, in dogs with plexiform lesions, intimal thickening in the eccentric nonlaminar pattern was observed more frequently in constrictive lesions than in those from dogs that did not have concurrent plexiform lesions. Again, similar studies in dogs are required to further define the similarity of this change to those in humans.
The plexiform lesion in humans and dogs generally arises at normal branching points or the origin of a supernumerary artery, suggesting that hemodynamic alterations are associated with the pathogenesis of this lesion. 6 In humans, the level of the plexiform lesion within the pulmonary arterial tree is dictated partly by the inciting cause. 22 For example, PAH associated with a cardiovascular shunt condition tends to develop in 100- to 200-µm-diameter arteries, and IPAH tends to develop in arteries <100 µm in diameter. These dogs with IPAH had PA with plexiform lesions that usually developed near branching of the intra-acinar respiratory artery. The distribution, however, varied among dogs, developing most proximal in dog No. 5 to involve branching points at pre-acinar secondary arteries and furthest in dog No. 6 to involve the most distal ramifications of intra-acinar arteries. Three-dimensional morphologic studies are needed to determine whether any of these vessels actually were supernumerary.
The plexiform lesion in humans and dogs is a glomoid structure composed of a dilated artery containing active or remnant fibrinoid necrosis of the parent vessel wall, tightly packed vasoformative cells that often form small vascular clefts, and a variable extracellular tissue matrix. 6, 22 This appearance should not be confused with thrombosis and recanalization, which is an ancillary lesion. In humans, the vasoformative cells variably have been designated endothelial cells, fibrillary cells, smooth muscle cells, and myofibroblasts, but recent research suggests they are predominantly proliferating immature (central core) and quiescent mature endothelial cells (cells lining vascular clefts). 6 As the structure evolves, they become admixed with connective tissue components, myofibroblasts, and inflammatory cells, of which the latter more often are within the periphery. Thus, depending on the stage of development and differentiation, the plexiform lesion in humans and dogs has a dynamic appearance in structure and cellular composition. The structural variation is demonstrated by the notably smaller size of plexiform structures in dog No. 4 versus dog Nos. 3, 5, and 6, and by the less severe and extensive dilatation lesion in dog No. 6 versus dog Nos. 3–5. The variable composition is demonstrated by the lesions in dog Nos. 5 and 6, in which there was consistent composition of cells immunoreactive to SMA, and F8 versus those in dog Nos. 3 and 4, in which there was more variable composition, including cells immunoreactive only to vimentin.
The dilatation lesion in humans and dogs is located distal to the plexiform lesion and indicates a poststenotic, aneurysmal or vein-like dilatation of the arterial branch and its ramifications. 22 Sometimes, it can assume an angiomatoid appearance, as demonstrated in dog Nos. 3 and 5. In humans, these changes predispose to alveolar edema and various exudative lesions. This sequela seems true in dogs, because dog Nos. 3 and 5 had the most extensive and severe dilatation lesion as well as the most extensive and severe exudative alveolitis. The exudative alveolitis was characterized by alveolar accumulation of dense proteinaceous material, infiltration of alveolar macrophages and other inflammatory cells, and proliferation of type-II pneumocytes. It tended to have a subpleural distribution, which may be related to the increased shear force sustained by more-peripheral blood vessels. Less frequently, the exudative lesion was scattered throughout the parenchyma and was associated with mural fibrinoid necrosis of plexiform and sometimes constrictive and ancillary lesions. Overall, the presence of the dilatation lesion and perhaps the exudative alveolitis suggests the concurrent presence of the plexiform lesion, whether it is apparent in the histologic section examined.
Isolated arteritis in humans and dogs (dog No. 4) develops concurrently with, but not independent of other complex lesions. 22 In humans, there is conjecture of its true relation and pathologic relevance to PA. The lesion usually affects larger-caliber arteries that are of no topographic relation to the origin of the vessel containing other complex lesions. It contains variable degrees of the fibrinoid necrosis that is characteristic of complex and sometimes constrictive lesions. Similarly, in dog No. 4, the focal nonsuppurative arteritis had no topographic relation to the complex and constrictive lesions and did not contain fibrinoid necrosis.
In human medicine, progress in understanding the pathobiology of IPAH has been hindered mostly by the paucity of information regarding the natural history and development of the integral vascular changes and by the lack of an animal model that fully replicates the vascular pathology. 11 The findings from this case series suggest that dogs of various breeds, ages, and sex and neuter status, exhibit the clinical and pathologic development of IPAH and PA as described in human medicine, and thus may provide a suitable animal model to help elucidate the pathobiology of this condition. Important limitations to this case series and to such a conclusion include: the small number of cases in this series and of previously reported cases with concurrent detailed histopathologic findings; the lack of viral testing to detect inapparent infections; the lack of genetic testing to differentiate IPAH from FPAH; and the lack of more-extensive genotyping and phenotyping of cells in these lesions to help discern the pathophysiology. Nonetheless, the clinical course of disease, antemortem diagnostic test results, and concurrent histologic findings suggest that PA is a manifestation of IPAH or perhaps FPAH in dogs, and that the human pathologic classification characterize the canine lesions as well.
In conclusion, the clinical diagnosis of IPAH in dogs can be confirmed by histologic examination of multiple lung sections that include the range of pre-acinar to intra-acinar arteries. The morphologic diagnosis should include one or more of the 4 main histologic subsets of PA as well as ancillary lesions, as described in the human pathologic classification and this canine study. Similar to that in humans, the plexiform lesion in these dogs often had a regionally clustered distribution among lung lobes and although slightly variable, had a tendency to develop near branching of the intra-acinar respiratory artery. Development of dilatation and exudative lesions and perhaps of constrictive lesions with mural fibrinoid necrosis should increase suspicion of the presence of plexiform lesions that may be inapparent in the section examined, lying upstream or downstream from even normal-appearing vessels. In these dogs, the presence of complex lesions versus only constrictive lesions seemed to be important indicators of severe, likely rapidly progressive and fatal IPAH. Exudative alveolitis also seemed to carry the same connotation and, thus, is an important ancillary lesion to include in the pathologic classification and morphologic diagnosis.
Footnotes
Acknowledgements
We thank the West Coast Subdivision of the C. L. Davis DVM Foundation for the opportunity to present this work during the 21st Annual Meeting of the West Coast Veterinary Conference, Asilomar Conference Grounds, Pacific Grove, California, 13–14 May, 2004. The authors thank Dr. Robert A. Fairley for additional review. This research was supported in part by R01-HL48411 from the National Institutes of Health.
