Abstract
Gross and histologic lesions of paratuberculosis were studied in water buffaloes. Small intestines and associated mesenteric lymph nodes of 405 water buffaloes were examined. Of these, 20 animals having visible changes of intestinal thickening, mucosal corrugations, and enlargement of mesenteric lymph nodes exhibited histologic alteration characteristics of mild to moderate granulomatous inflammation. The histologic lesions observed in these animals were classified into 3 grades on the basis of type of cellular infiltration, granuloma formation, and presence of acid-fast bacilli. Grade-1 lesions observed in 8 animals were marked by the presence of scattered epithelioid macrophages amid large number of lymphocytes in the intestinal villi and in the paracortical regions of the associated mesenteric lymph nodes. Another 8 animals classified under grade-2 revealed microgranulomas, infiltration with a larger number of epithelioid macrophages besides lymphocytes in the intestinal villi, and granulomas in the mesenteric lymph nodes. Grade-3 lesions observed in 4 animals were characterized by the presence of epithelioid granulomas and giant cells in the intestines and the mesenteric lymph nodes. The Ziehl-Neelsen's stained tissue sections revealed acid-fast bacilli in grade-3 and −2 animals and acid-fast granular debris in grade-1 animals. Among these 20 buffaloes, 14 (70%) were positive in the IS900 specific polymerase chain reaction and 6 (30%) were positive in the bacterial culture.
Paratuberculosis (Johne's disease) is a chronic progressive bacterial disease of ruminants caused by Mycobacterium avium subsp. paratuberculosis (MAP) affecting mainly intestinal tract and regional lymph nodes. 9, 33 The disease has been reported both in the domestic and wild ruminants including cattle, sheep, goat, bison, species of camelides and cervids, bighorn sheep, rocky mountain goat, deer, and tule elk. 4, 5, 9, 37 The animals acquire infection during early calfhood and the clinical disease appears in the adults after 2–4 years of long incubation passing through various stages of early and subclinical infection. 9 Economic losses in subclinical infection are caused by reduced feed efficiency, productivity decline, decreased milk protein and fat, premature culling, reduced slaughter weight at culling, increased incidence of mastitis, and reduced fertility. In cattle, the clinical disease is characterized by intermittent and incurable diarrhea, decrease in milk production, and chronic weight loss that terminate into death of animals. Pathologic manifestations include chronic granulomatous enterocolitis and regional lymphangitis and lymphadenitis. 9
The pathology of natural 4 and experimental 13, 24, 31 paratuberculosis with varying degrees of gross and histologic lesions has been described in bovines. Buergelt et al. 4 described mild, moderate, and advanced lesions in cattle based on the severity of lesions and pattern of cellular infiltration. In bison, the natural infection was characterized by mild to moderate lesions. 5 The types of histologic lesions at different stages of mycobacterial infections have been found to be determined mostly by the local immune responses elicited by the host. 27 The lesions in the early and subclinical stages have been associated mainly with cell-mediated immune response and advanced lesions with marked humoral immune response. 7 In the subclinical infection, animals shed few or no bacteria in feces, whereas detectable bacteria are shed in the clinical and advanced stages of infection. 7
Buffaloes are not immune to paratuberculosis, but are considered comparatively resistant. 18 In India, paratuberculosis is widely prevalent in cattle, 2, 15, 18 sheep 16, 26 and goats. 32 But in buffaloes, which are often reared side by side with cattle, generally subclinical infections have been reported on the basis of Johnin skin test and tissue smear examination. 2, 15, 18, 33, 35 Convincing studies comprising histologic evaluation and/or confirmation of the infection by bacterial culture or bacterial genome detection have not been undertaken in water buffaloes despite their economic importance. India possesses more than 55% (167 million) of the total world buffalo population, contributing more than 50% of the total milk yield in the country. 12 Being a major domesticated ruminant in Asia, its importance with regard to control and eradication of paratuberculosis cannot be underestimated. Because buffaloes belong to a separate genus, Bubalus, the pathology of paratuberculosis infection in cattle 4, 13, 24, 31 and other ruminants 5, 10, 16, 25, 26, 28, 34, 37 is not likely to be similar.
The objective of the present study was to describe the gross and histologic lesions of naturally occurring paratuberculosis in water buffaloes.
Materials and Methods
Animals and tissues
Approximately 1,000 buffaloes in poor physical conditions brought for slaughter at Bareilly municipal slaughterhouse, Uttar Pradesh State, India, were examined for visible abnormalities in the small intestine and associated mesenteric lymph nodes (MLN) during October 2002 and March 2003. Buffaloes brought to the slaughterhouse generally belong to small dairy units in the villages of Bareilly and 4 surrounding districts, where they are maintained with cattle. Tissue samples were collected from 400 animals (mostly females) showing any gross abnormalities in the small intestines, such as congestion, hemorrhage, and mucosal thickening or corrugations. Approximately 10-cm pieces of the jejunum and ileum, a section of ileocecal junction, and associated MLN were collected in labeled specimen bags and transported in ice to the laboratory within an hour. Tissues were also collected from 5 buffaloes necropsied in the postmortem room of the institute, 1 of which was suspected for paratuberculosis infection on the basis of mucosal corrugation in the small intestine and enlarged MLN.
Histopathology
In the laboratory, tissue samples were processed using sterile instruments. The excess fat and mesenteric attachments were trimmed off, and intestinal contents were flushed out for the examination of intestinal mucosa. Impression smears, prepared by scraping intestinal mucosa and cut surfaces of MLN, were stained by Ziehl-Neelsen's (ZN) method. 1 Sections (0.5-cm thickness) of intestines and lymph nodes were preserved in 10% neutral buffered formalin, and remaining tissues were stored frozen at −20°C until used. The formalin-fixed tissue samples were dehydrated through graded ethanol and embedded in paraffin blocks. Sections of 4–5-µm thickness were cut from paraffin blocks and routinely stained with hematoxylin and eosin (HE). The adjacent sections were stained by ZN method. The selected sections were stained with Gram's and Periodic Acid–Schiff methods to rule out other bacterial or fungal infections. 1
Buffaloes showing granulomatous enteritis and lymphadenitis, as well as other evidences of infection such as demonstration of acid-fast bacilli (AFB) in tissue smears and histologic sections, bacterial culture, and positive tissue polymerase chain reaction (PCR) were diagnosed as cases of paratuberculosis. The histologic lesions were graded into 3 categories according to preponderance of cellular components in granulomatous lesions. Grade-1 lesions revealed occurrence of a few epithelioid macrophages occasionally with acid-fast granular debris in diffusely infiltrated lymphocytes. Grade-2 lesions consisted of focal granulomatous changes containing AFB or acid-fast granular debris. Grade-3 lesions had diffuse granulomatous changes consisting of epithelioid granulomas and multinucleated giant cells containing AFB. The presence of lymphocytes along with epithelioid macrophages and giant cells has already been taken as criterion for histomorphologic confirmation of paratuberculosis lesions in sheep and goats. 10, 25 Thus, we considered the severity and pattern of lymphocyte infiltration in description of lesions of preclinical paratuberculosis in buffaloes.
Bacterial culture
The procedure for the bacterial culture was used as described previously. 32, 36 Briefly, 2 g of intestinal mucosa and associated lymph node tissue were homogenized in 10 ml of sterile distilled water. The suspension was kept undisturbed for 30 minutes and then 5 ml of supernatant was collected in a 50-ml centrifuge tube containing 25 ml of 0.9% hexadecylpyridinium chloride for overnight decontamination. The decontaminated samples were inoculated on Herrold's egg yolk medium (pH 7.4) with and without mycobactin-J and incubated at 37°C for 16 weeks. The tubes were examined every 4 weeks for growth, and the presence of at least 1 colony consisting of strongly acid-fast bacteria in tubes with mycobactin J was recorded as positive. 32, 36
Bacterial purification by centrifugation
Bacterial purification from intestinal mucosa was performed in 12 out of 20 animals showing histologic lesions. Because bacterial purification was carried out retrospectively (i.e., after examination of histologic sections), it was performed only on those cases in which sufficient frozen tissues were available for collection of mucosal homogenate. For bacterial purification, 8 the mucosal homogenate was collected from the frozen intestine by deep scraping of the mucosa using a sterile glass microscopic slide, and approximately 5 g of mucosal scraping was subjected to several steps of centrifugation by suspending it in 0.2-M and 0.3-M sucrose and 1.5-M potassium chloride solutions as differential and density gradient medium. Finally, bacterial pellets were washed with sterile phosphate buffered solution, and smears were prepared for the demonstration of AFB by ZN method.
Polymerase chain reaction
The DNA isolation from tissues was carried out by the method described previously. 29 Amplification of 626 base pair (bp) fragments from IS900 sequence was carried out in a 50-µl reaction mixture containing 1× PCR buffer; 2-mM MgCl2; 200-µM dATP, dGTP, dCTP, and dTTP; 0.5 U Taq polymerase (MBI Fermantas, MD); 1 µM each of both primers (BN1: 5′GTT ATT AAC GAC GCC CAG C-3′; BN2: 5′ACG ATG CTG TGT TGG GCG TTA G-3′), and 0.5-µl purified genomic DNA solution. The PCR conditions consisted of an initial denaturation at 94°C for 4 minutes, 30 cycles of denaturation (94°C for 1 minute), annealing (60°C for 1 minute), synthesis (72°C for 1 minute), and final elongation at 74°C for 4 minutes. For each round of PCR, positive (DNA isolated from pure culture) and negative (DNA isolated from intestine of paratuberculosis-negative animals) controls were included. DNA template was replaced with sterile distilled water to serve as PCR control. The amplified PCR products were run in 1.5% agarose gel stained with ethidium bromide (5 µg/ml) and visualized under ultraviolet light. The specificity of PCR product was confirmed by restriction endonuclease analysis and by sequencing the cloned PCR products. Sequences were compared with published sequence of IS900 (Accession No: X16293).
Results
The results of gross and histologic examination of tissues, demonstration of AFB in stained tissue sections and smears, bacterial culture, bacterial purification, and bacterial genome detection by PCR are given in Table 1. Twenty (2%) animals out of 1,000 buffaloes examined had granulomatous lesions compatible with paratuberculosis infection. Of these, 8 animals each (40%) had grade-1 and grade-2 lesions, and 4 (20%) had grade-3 lesions. Both gross and histologic changes were milder in the grade 1 than the grade 2, which was milder than the grade-3 lesions. Histologic lesions observed in remaining buffaloes were congestion, hemorrhage, nonspecific enteritis, and parasitic infestations.
Summary of results of various test employed on intestinal tissues of buffaloes showing histologic lesions of paratuberculosis.
1 = thickening of intestinal mucosa; 2 = mucosal corrugations; 3 = lymphadenopathy; 4 = prominent lymphatics.
1 = grade-1 lesion; 2 = grade-2 lesion; 3 = grade-3 lesion.
G = acid-fast granular debris; Pb = paucibacillary; Mb = multibacillary; ND = not done.
Thickening of the intestinal mucosae in 4 (20%), mucosal corrugations in 16 (80%), and lymphadenopathy in 12 (60%) buffaloes were observed. Prominent serosal lymphatics were observed in 2 animals. Congestion and petechial hemorrhages were found on the borders of the mucosal corrugations in 5 animals with grade-2 histologic lesions. Prominent transverse corrugations were observed in the intestinal mucosae of animals with grade-3 lesions (Fig. 1). Mesenteric and ileocecal lymph nodes were generally enlarged and edematous. The distinction between cortex and medulla was less marked, and cut surfaces oozed cream-colored fluid when MLNs were incised.

Small intestine; buffalo No. 16. Mucosal corrugations evident in grade-3 lesion.
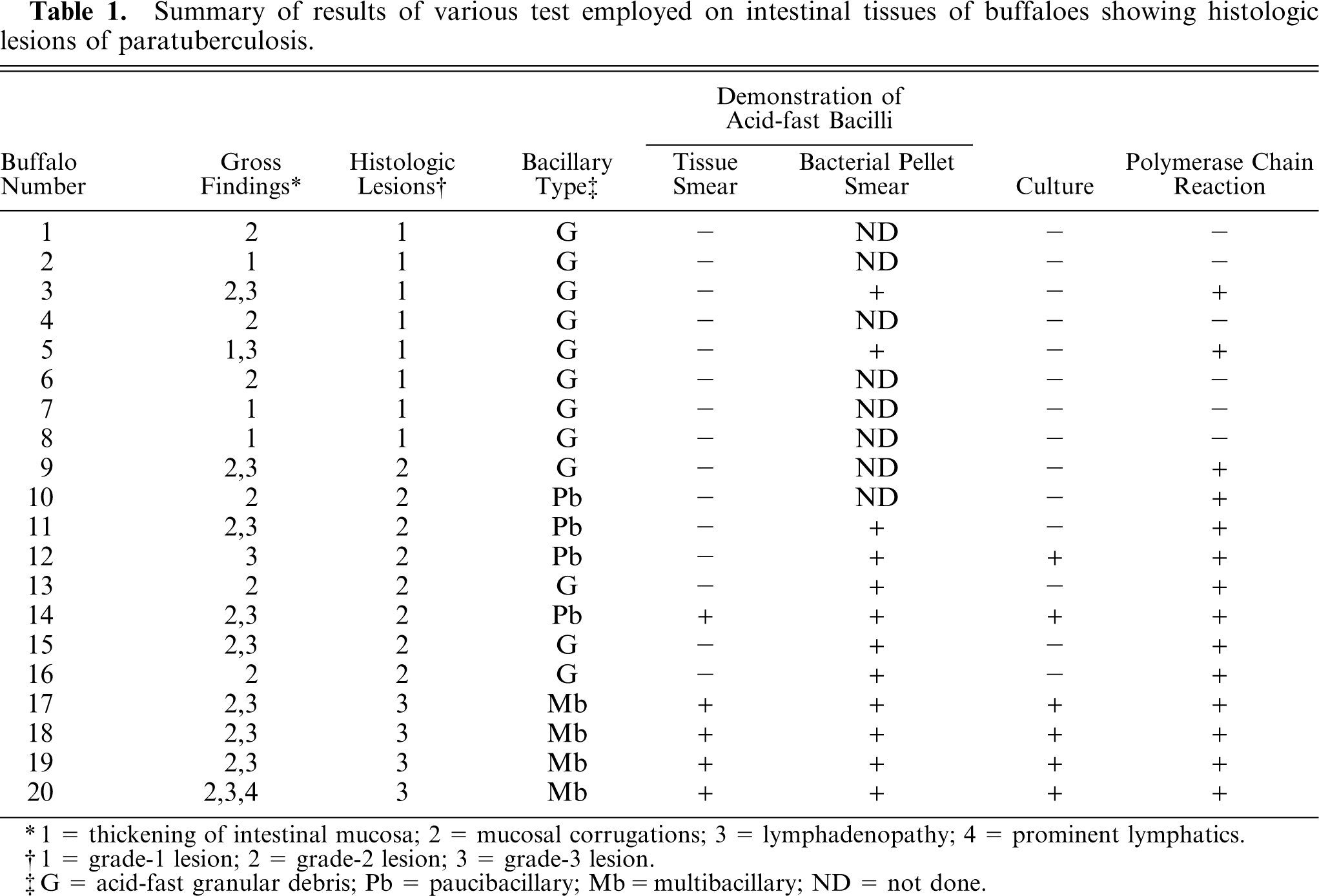
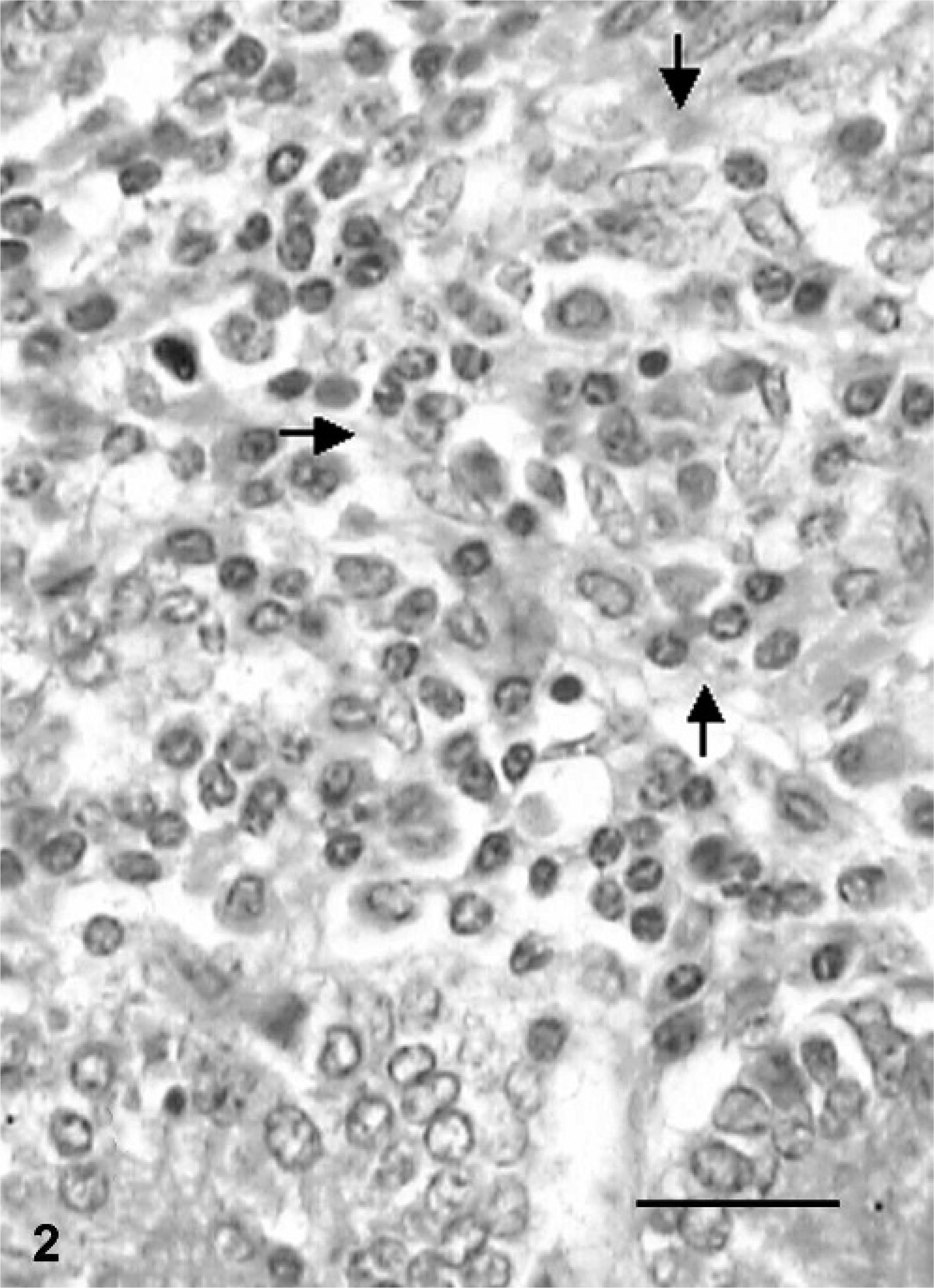
Grade-1 lesions were characterized by the infiltration of scattered macrophages among a large number of lymphocytes in the lamina propria of intestinal villi. Preponderance of macrophages was also seen at the periphery of lymphoid follicles of Peyer's patches. The paracortical regions in the MLN had a greater number of epithelioid macrophages. Multinucleated giant cells were rarely observed in either the small intestine or the MLN. Buffaloes with grade-2 lesions had several small aggregates of epithelioid macrophages in the form of microgranulomas in the villi and crypt regions of the small intestine (Fig. 2) and granuloma in the paracortical regions of the MLN (Fig. 3). Microgranulomas were neither encapsulated nor surrounded by multinucleated giant cells. The infiltration with globule leukocytes in the lamina propria of the small intestine was observed in two animals.
Small intestine; buffalo No. 11. Microgranuloma (arrows) consisting of epithelioid macrophages in crypt region in grade-2 lesion. HE. Bar = 40 µm.
Mesenteric lymph node; buffalo No. 14. Granuloma consisting of epithelioid macrophages in grade-2 lesion. HE. Bar = 40 µm.
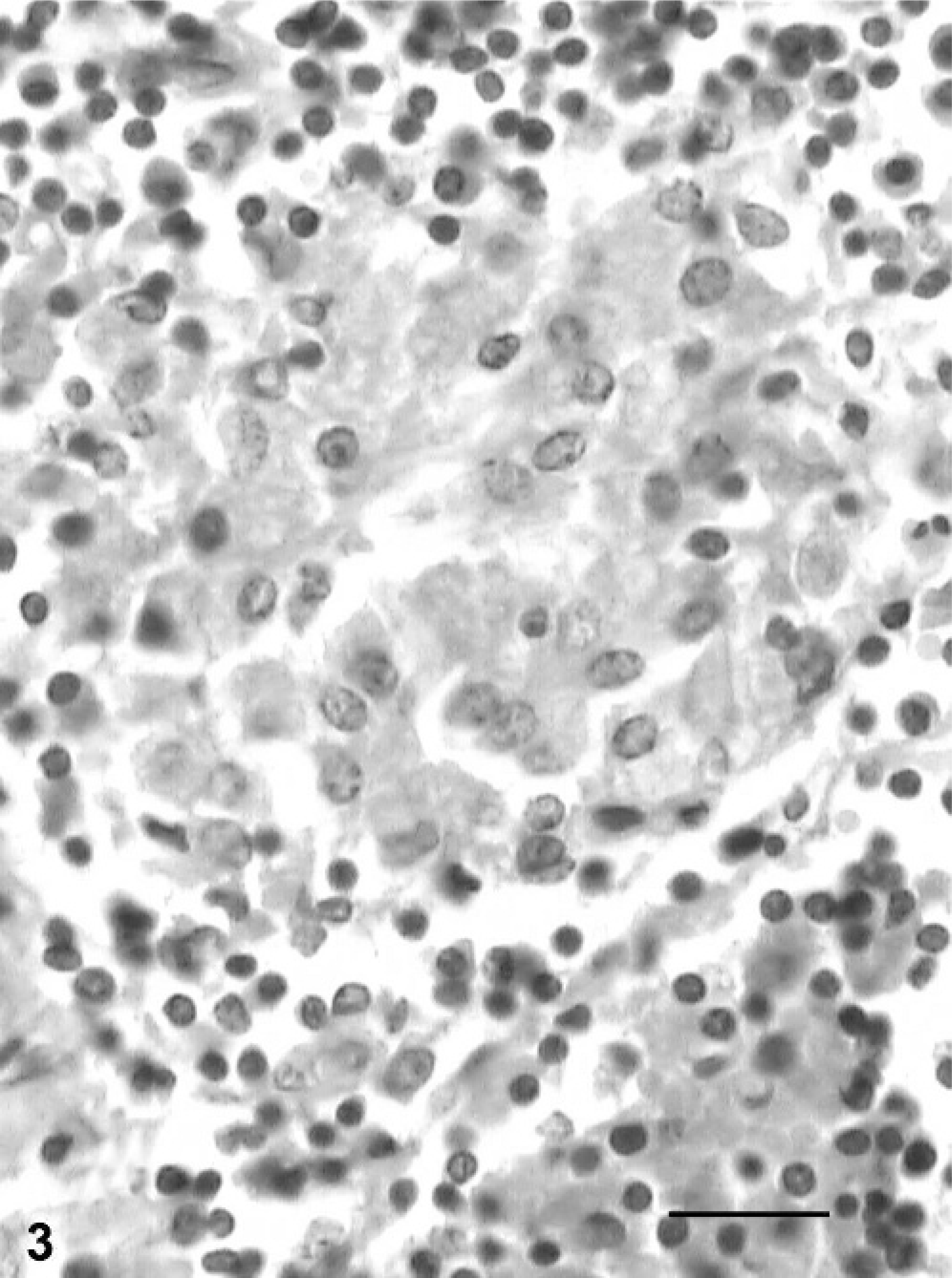
Typical granulomatous changes consisting of epithelioid macrophages and multinucleated giant cells were observed in the small intestines of buffaloes classified under grade-3 lesions. Necrotic changes in granulomas of the intestine and MLN were observed in 1 animal. In this case, both Langhan's and foreign-body giant cells were observed in the MLN. In 1 animal, submucosa was quite thickened because of the presence of granuloma and foreign-body giant cells. The prominent granulomatous changes consisting of multiple granulomas and multinucleated giant cells were observed in the MLN (Fig. 4). The capsule of the lymph nodes was thickened, and lymphatics were dilated with cellular infiltrates.
Mesenteric lymph node; buffalo No. 20. Multinucleated giant cells in the paracortex in grade-3 lesion. HE. Bar = 40 µm.
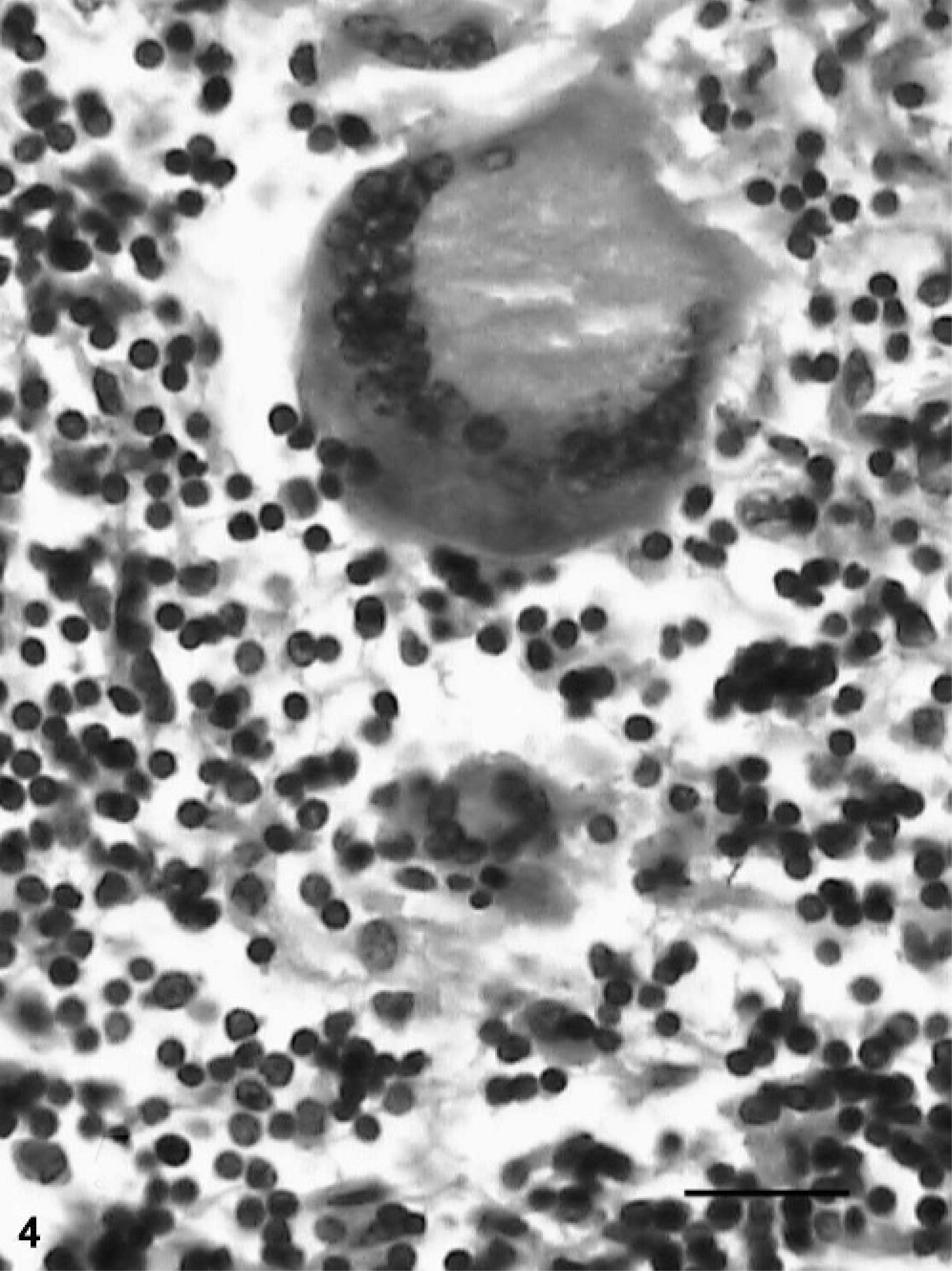
The acid-fast staining of tissue sections revealed AFB compatible with MAP in all animals with grade-3 lesions (Fig. 5) and in 4 of 8 animals (50%) with grade-2 lesions. The sections from remaining animals showed few macrophages containing acid-fast granular debris that were diffusely stained spherical- or irregular-shaped acid-fast material. Routinely prepared and stained tissue smears revealed AFB in 5 animals. The smears prepared from the purified bacterial pellets were more revealing, with 7 more animals being found positive. Six (30%) and 14 (70%) buffaloes were positive in the bacterial culture and IS900 PCR, respectively. Sequence analysis of cloned PCR products of buffalo isolates (Accession No: AY660657) showed around 98% of sequence homology with known sequence of IS900 gene.
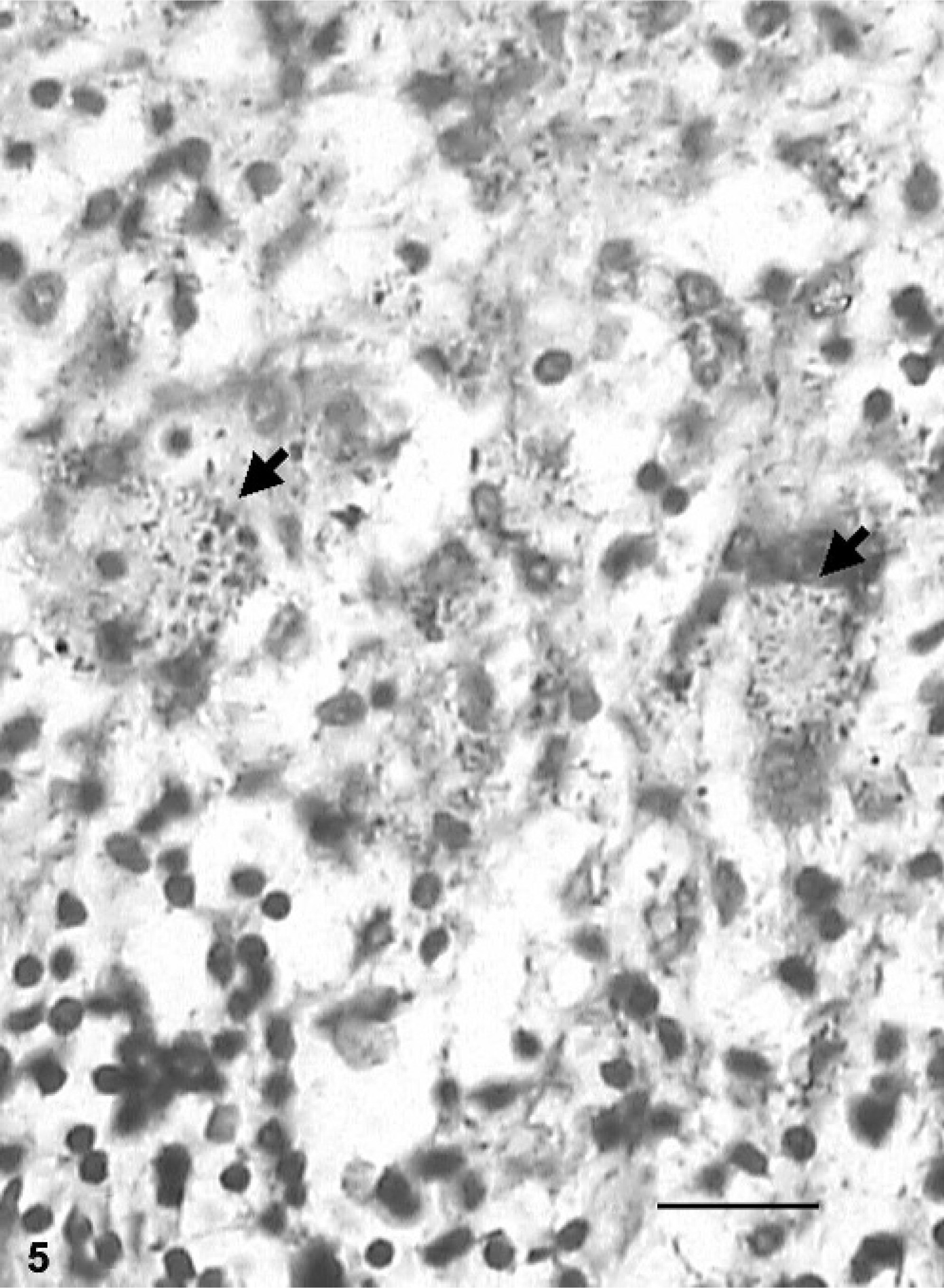
Small intestine; buffalo No. 20. Acid-fast bacilli (arrows) within macrophages in grade-3 lesion. ZN method. Bar = 40 µm.
Discussion
The water buffalo is an important domestic animal for milk and meat production and draft power in India and other Southeast Asian countries, contributing significantly to their agricultural economy. The present study describes the pathology of paratuberculosis in water buffaloes and conclusively confirms the infection in these animals. The 2% infection occurrence in buffaloes observed in this study is relatively higher than previous reports (0.1 to 0.5%) 2, 15, 18, 35 probably because the study was conducted on a selected population of animals in poor condition. The previous studies also involved fewer and less refined methods (smear examination and Johnin skin testing) employed for detecting infection. The reliability of the allergic test is largely unknown in paratuberculosis in buffaloes.
The gross and histologic lesions of paratuberculosis observed in buffaloes had a close resemblance to those described in domestic cattle. The grade-3 lesions observed in 4 (20%) animals represented classical granulomatous inflammatory changes of paratuberculosis. The lesions had diffuse infiltration of epithelioid macrophages and multinucleated giant cells in the small intestine and MLN, similar to those described in bovines previously. 4, 31 Loose aggregates of epithelioid macrophages were more common in the lamina propria in intestinal villi, unlike well organized granulomas seen in tuberculosis, 27 suggesting that they could be characteristic of paratuberculosis. 4, 31 Focal necrosis and mineralization, although rare in bovines, has been frequently reported in paratuberculosis lesions in the small ruminants with or without parasitic infection. 37 The observation of caseation and necrosis in the intestine and lymph node of 1 buffalo indicated uncommonality of lesions in buffalo paratuberculosis. 31 Like Mycobacterium avium subsp. avium, the potentiality of MAP in causing caseation and necrosis cannot be ruled out. 16, 17 Although this buffalo had revealed Langhan's and foreign-body giant cells, the concurrent bacterial, fungal, or parasitic infections were ruled out by appropriate staining of tissue sections. 28
Grade-2 lesions had several microgranulomas composed chiefly of epithelioid macrophages without giant cells. In grade-1 lesions, epithelioid macrophages were not aggregated in the form of granulomas, and they were found scattered among lymphocytes. Lymphocyte response to infection as also seen in the present study marked the lesions in preclinical stage of paratuberculosis and has already been reported in sheep and goat. 10, 25 This was further substantiated as all grade-2 and at least 25% of grade-1 buffaloes were confirmed to be infected with MAP on the basis of IS900 PCR and purified bacterial smears. Studies on experimental and natural cases of paratuberculosis in sheep and goat 10, 20, 25, 34 have indicated preponderance of lymphoid cells to epithelioid macrophages and giant cells in the early and paucibacillary cases, in which bacteria could not generally be demonstrated. Diffuse lymphocytic infiltration has been reported in the early stages of other mycobacterial infections and correlated with strong cell-mediated immune (CMI) response mediated by different subpopulation of lymphocytes such as helper, cytotoxic, and suppressor cells. 11, 22 The observation of mild lesions in adult buffaloes, akin to those in the younger animals, 2 could be attributed to several reasons including strong CMI response resisting progression of infection 3, 13 and latency or persistence of infection. 16, 18, 23, 25 Six animals with grade-1 lesions, which were not confirmed by PCR, bacterial culture, or demonstration of AFB in sections, could better be categorized as suspected for paratuberculosis infection until more sensitive molecular techniques such as in situ PCR and hybridization on tissues are developed.
Absence of AFB in all buffaloes with grade-1 lesions and in a few with grade-2 lesions has already been observed in subclinical paratuberculosis. 5, 13, 24 In these cases, macrophages contained acid-fast granular debris, which could be partly degraded, or dead bacilli similar to those reported previously in the tuberculoid lesions of paratuberculosis in sheep. 14, 16, 26 In the present study, tissue PCR (70%) showed greater sensitivity than bacterial culture (30%) for detecting MAP in the infected tissues, suggesting the importance of testing tissues by PCR for diagnosis of subclinical infections. The PCR assay has been reported to be more sensitive on tissue samples than other types of clinical samples like feces. 30 The inability to demonstrate AFB in sections and to isolate MAP in the culture from tissues showing microgranuloma with positive PCR results in certain grade-1 and grade-2 buffaloes could have been due to the occurrence of a scant number of viable bacilli, nonculturable spheroplastic form of bacteria, or degraded bacteria in tissues. 6, 7, 19, 21 The density and viability of bacteria is known to vary in lesions in different parts of the intestine and MLN, influencing the results of bacterial culture. 17, 19 In water buffaloes, information on these aspects is not yet available.
The histologic lesions observed in buffaloes indicate the prevalence mostly of subclinical infection in the Bareilly region of Uttar Pradesh State, India. Studies from other parts of India corroborate this finding in buffaloes. 18, 35 Prevalence of subclinical infection, however, cannot mitigate the importance of buffaloes in the epidemiology of paratuberculosis infection. Rather, this species may be more important in the propagation of infection in the environment and yet be escaping detection, possibly because of harboring subclinical infection and being an intermittent or nonfecal shedder. The partial sequencing of IS900 gene of bubaline isolates showed 98% homology with known sequence of IS900 gene and caprine isolates of Indian origin suggesting that buffalo strains may not be very genetically different from other strains circulating in the country. However, molecular characterization of buffalo isolates of MAP and their similarities and differences with other isolates of ruiminants would reveal actual epidemiologic significance of paratuberculosis infection in water buffaloes.
It is deduced from this study that pathomorphologic lesions in paratuberculosis in buffaloes closely resembled those in bovine paratuberculosis, and it was easier to detect MAP and its DNA in animals with more advanced lesions.
Footnotes
Acknowledgements
We are thankful to Dr. M. P. Yadav and Dr. O. P. Paliwal for providing facilities to carry out the work. We thank Dr. N. S. Parihar, principal scientist and head (retired), Division of Pathology, for useful corrections in the manuscript.
