Abstract
Cardiac mesothelioma with granular cell features was diagnosed in a 10-year-old Golden Retriever presenting with pericardial and abdominal effusions. The diagnosis was based on gross, morphologic, and immunohistochemical features. The immunohistochemical profile of the neoplasm was pancytokeratin positive, vimentin positive, and S-100 negative; most gross and morphologic features were consistent with both mesothelioma and granular cell tumor. To the authors' knowledge, the prognosis for either primary cardiac mesothelioma or granular cell tumor in the dog is unknown. At 4 months after thoracotomy, pericardectomy, and mass excision, this dog was alive and without clinical evidence of pericardial or abdominal effusions. We describe a granular morphologic variant of cardiac mesothelioma in a dog.
To our knowledge, we present a case of canine cardiac mesothelioma with granular cell features for the first time, but we believe that this entity has been described as canine intrathoracic granular cell tumor 4 and primary cardiac granular cell tumor. 7 Although it is rare in all species, the most common papillary neoplasm in the thorax is mesothelioma. Dogs with thoracic mesothelioma commonly present with respiratory distress and thoracic and pericardial effusions. 3, 6, 9 Previous case reports of granular cell tumors in the thorax of dogs describe cell morphology typical of granular cells, but do not address mesothelioma as a possibility in the differential diagnosis. 4, 7 The morphologic features of the neoplastic cells described in those reports also were found in the dog of this report, but in addition, we describe cell-to-cell junctions, cell surface structures, and immunohistochemical cytokeratin positivity consistent with mesothelial cells. It is unclear whether these later features were absent or were not observed in previous cases.
A 10-year-old, spayed female Golden Retriever was presented to a referring veterinarian because of anorexia of 10 days' duration. Complete blood count results were unremarkable. Serum biochemical analysis revealed increased alkaline phosphatase (ALP) activity of 322 IU/liter (reference interval 23–212 IU/liter), increased alanine transaminase (ALT) activity of 102 IU/liter (reference interval 10–100 IU/liter), and low albumin concentration of 2.4 g/dl (reference interval 2.7–3.8 g/dl). Urinalysis revealed low specific gravity of 1.006 (reference interval 1.015–1.045). An enlarged cardiac silhouette was noted on a right lateral thoracic radiographic view. Loss of abdominal contrast and hepatic silhouette enlargement were identified on a lateral abdominal radiographic view. The dog was referred for further evaluation.
On physical examination, the dog was normothermic and panting, but was not in respiratory distress. On auscultation, muffled heart sounds were heard, with normal heart rate of 108 beats per minute. The abdomen was distended and mildly painful on palpation. Abdominal mass lesions were not palpable.
Lead-II electrocardiography revealed sinus rhythm with intermittent supraventricular premature complexes. Initial echocardiography revealed a large amount of pericardial effusion with no overt mass lesions. Therapeutic pericardiocentesis yielded 330 ml of sanguineous fluid with packed cell volume of 14% and total solids concentration of 1.6 g/dl. The fluid was not submitted for cytologic analysis or cell counting. Abdominal ultrasonography revealed moderate hepatomegaly with substantial hepatic venous distention, and there was a moderate amount of noncomplex abdominal fluid. Follow-up echocardiography 24 hours after pericardiocentesis did not reveal a cardiac mass lesion.
The clinical finding of abdominal distention in this dog was attributed to the presence of pericardial effusion causing increased right-sided cardiac pressure and subsequent hepatic venous congestion. The mild increase of ALP and ALT activities, and the decrease in albumin concentration also were attributed to hepatic venous congestion. The differential diagnosis for pericardial effusion in this patient included idiopathic pericarditis and neoplasia. The dog was discharged, and the owner was instructed to return for repeat echocardiography in 1 week.
The dog was presented 18 days after discharge for re-evaluation. It was reported to be doing well at home, but abdominal distention had been worsening over the preceding 24–48 hours. On echocardiographic re-evaluation, a 1- to 2-cm mass lesion was identified at the craniolateral aspect of the right atrium, as well as a small amount of pericardial effusion with no evidence of tamponade.
The dog was returned 3 days later for planned thoracotomy. Repeat echocardiography was not performed on the day of surgery. Right fourth intercostal space thoracotomy was performed. The pericardial sac was incised to remove 350 ml of serosanguineous fluid. Two 1- to 2-cm, friable, papillary masses were identified. One of the masses extended from the auricular appendage of the right atrium and the other from the left dorsal surface of the aorta. The auricular appendage was removed. The aortic mass was debulked, but resection was precluded by its invasion into the aortic wall as well as the inaccessibility of the mass from the right lateral thoracotomy. Subtotal pericardectomy was performed. Patient recovery was unremarkable, and the dog was discharged on the fourth postoperative day.
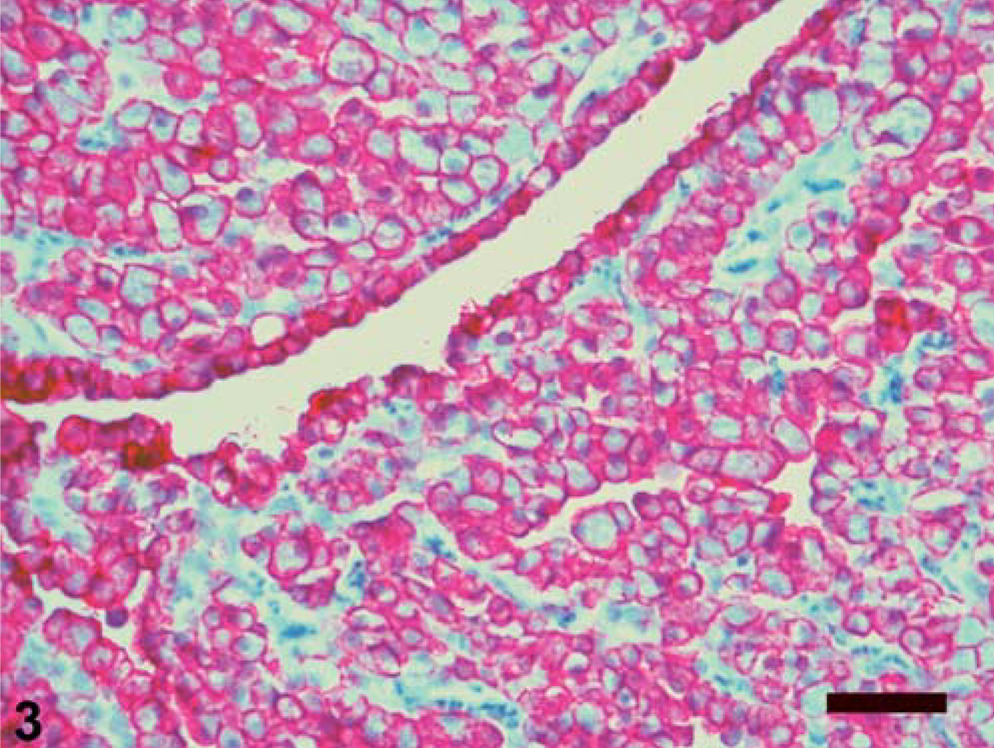
The right atrial and aortic root masses and pericardial sac were submitted for histologic examination. The differential diagnosis included aortic body tumors (chemodectomas), aberrant thyroid and parathyroid tumors, hemangioendothelioma, adenocarcinoma, mesothelioma, and granular cell tumor (on the basis of the aforementioned case reports). Histopathologic features included fibrovascular papillary projections supporting sheets of globoid cells, which extended from the surfaces of the atrial and aortic root samples (Fig. 1). In the atrial sample were clusters of cells within the subepicardial connective tissue. The cells comprising the center of the sheets had abundant eosinophilic granular cytoplasm and often were surrounded by a single peripheral layer of morphologically similar cells containing less cytoplasm (Fig. 2). The nuclei were round, central in the smaller cells and often eccentric in the larger cells where they were compressed by the cytoplasmic contents. Only one mitotic figure was identified, and there were small numbers of cells with pyknotic nuclei. Neoplastic cells were not identified in the pericardial sac. Components of the differential diagnosis based on gross and histologic findings were mesothelioma and granular cell tumor. Additional histochemical stains, immunohistochemical staining, and electron microscopy were used to further differentiate the tumor. The peripheral cells were moderately periodic acid–Schiff (PAS) positive, and the larger central cells were minimally PAS positive. The cells had strong, peripheral, pancytokeratin (Fig. 3) and vimentin immunohistochemical staining, and S-100 staining was negative. Electron microscopy revealed abundant lysosomes with variably electron-dense content, peripherally displaced nuclei, and rare cytoplasmic organelles (Fig. 4). The cells also had abundant intercellular junctions (Fig. 5a), some of which were desmosomes (Fig. 5b), and numerous, long, tortuous, surface microvilli. The microvilli did not have rootlets or a glycocalyx (Fig. 5c). The diagnosis of mesothelioma with granular cell morphology was made on the basis of these morphologic and immunohistochemical features.
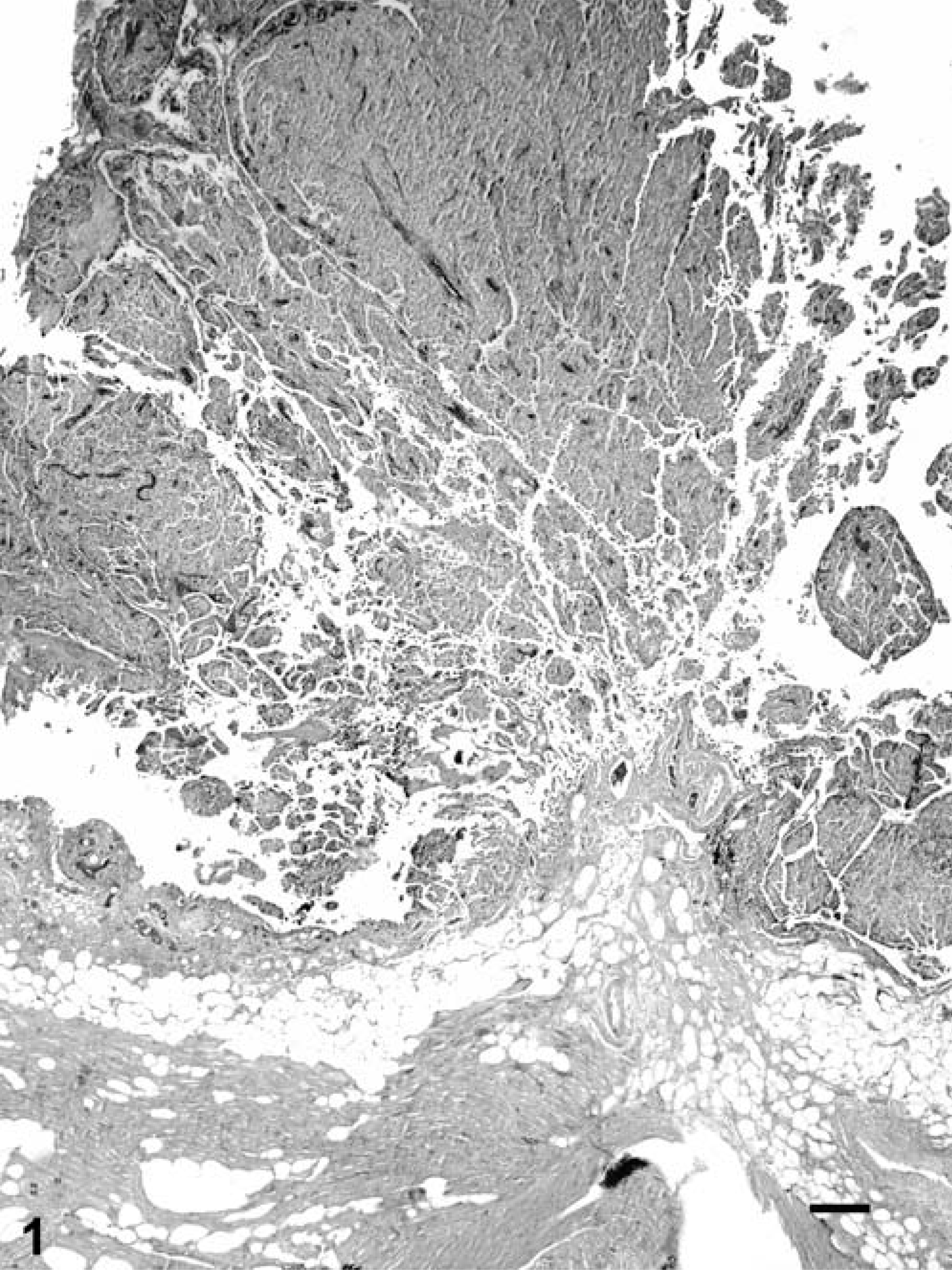
Right atrial auricular appendage; Golden Retriever. A papillary mass composed of globoid cells extends from the epicardial surface of the atrium. HE. Bar = 200 µm.
Right atrial auricular appendage; Golden Retriever. Sheets of cells have abundant granular cytoplasm centrally, and a more compact appearance peripherally. HE. Bar = 100 µm.

Right atrial auricular appendage; Golden Retriever. Globoid neoplastic cells have strong peripheral immunolabeling for pancytokeratin. Ventana Enhanced Alkaline Phosphatase Red Detection Kit, hematoxylin counterstain. Bar = 100 µm.
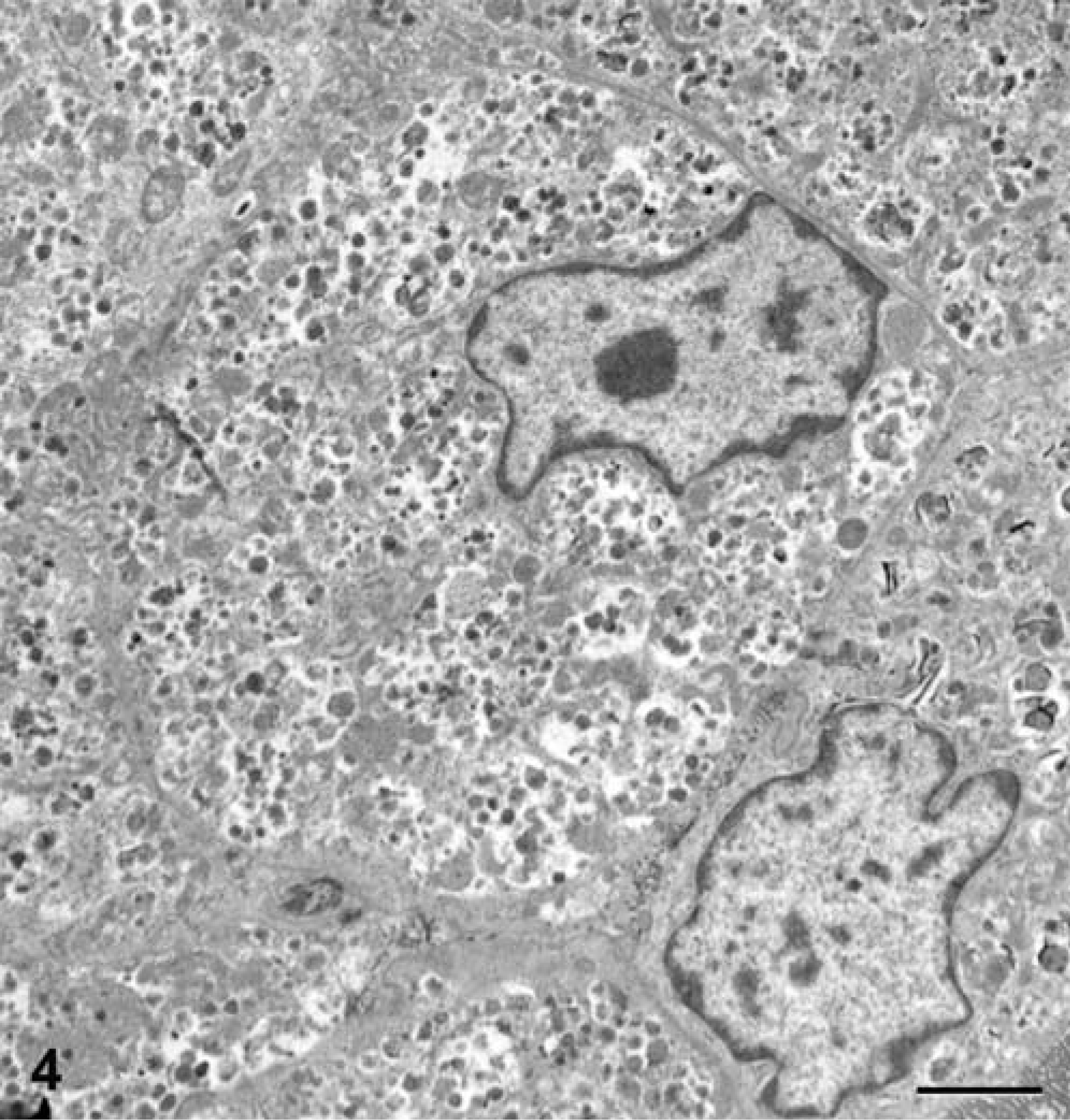
Right atrial auricular appendage; Golden Retriever. Electron microscopy reveals neoplastic cells with eccentric nuclei and abundant lysosomes that contain variably electron-dense material. Karnofsky's fixative, uranyl acetate, and lead citrate. Bar = 2 µm.
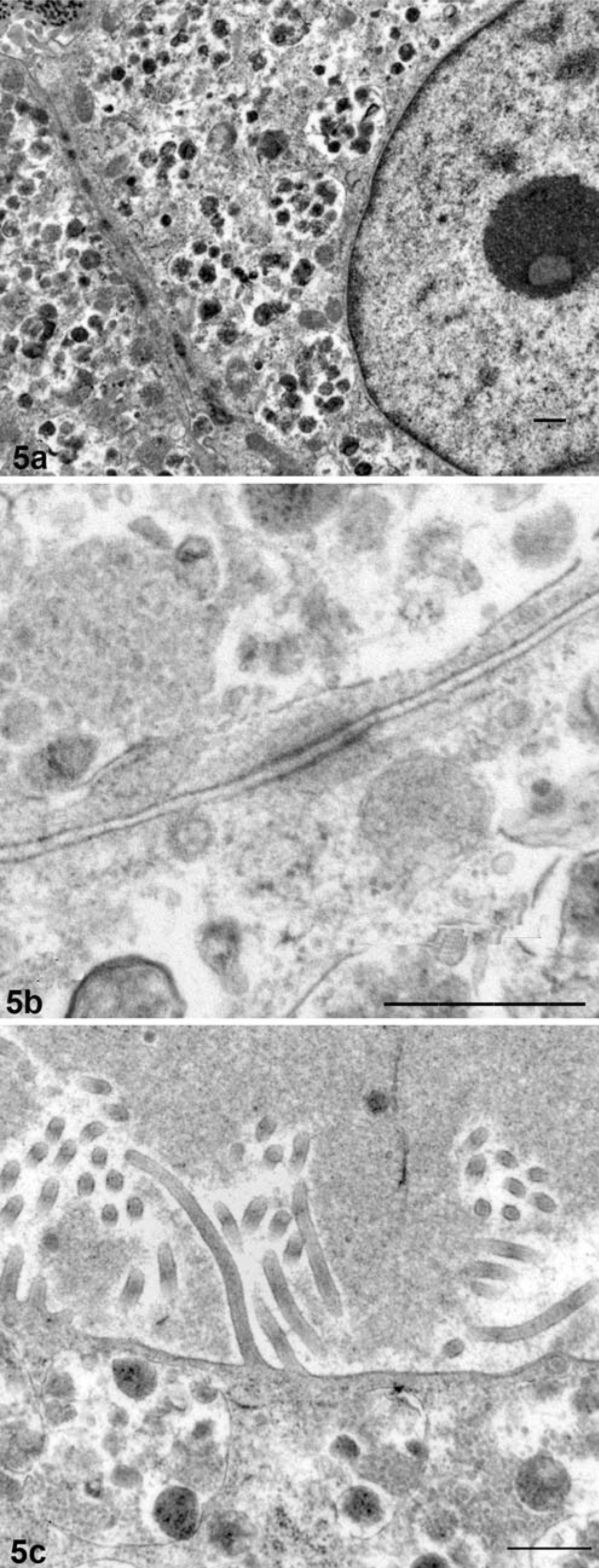
Right atrial auricular appendage; Golden Retriever. Electron microscopy reveals neoplastic cells with numerous intercellular junctions
A single layer of mesothelial cells cover the epicardial surface of the heart. This layer acts as the visceral layer of the pericardium and, among a growing list of functions, produces serous fluid. Although epicardial mesothelioma is rare, atriocaval mesothelioma, a specific entity described in humans and rats, bears a strong resemblance to the tumor we have described. In rats, these tumors develop in and at the base of the atrioventricular septum, the wall of the caudal vena cava, and the outer wall of the right atrium. 2
Granular cell tumors tend to arise in species-dependent anatomic locations. Canine granular cell tumors develop predominantly on the tongue, where they have a benign behavior. 5 Reported histochemical and immunohistochemical staining patterns have been inconsistent for granular cell tumors, 1 and these inconsistencies underlie a continuing dispute over the histogenesis of this tumor. The best supported case for a single cell of origin suggests the Schwann cell, 4, 5 but evidence of multiple lineages for these tumors also has been presented. 10
On the basis of the findings in this case and the similarities with previous reports, we question the existence of intrathoracic granular cell tumors in dogs. We suggest that a granular morphologic variant of mesothelioma develops in the canine thorax, and that this morphology could easily result in the misdiagnosis of granular cell tumor. The distinction is important because diagnosis of granular cell tumor in the dog implies benign behavior, whereas thoracic mesothelioma is often complicated by spread and the effusions these tumors can generate. The morphology of the tumor of this report was benign, but the location, focal invasion, and extension along the epicardial surface raised some concerns about potential tumor regrowth and effusion recurrence. In a retrospective report regarding mesothelioma-related pericardial effusions, the median survival was 60 days, and no dog survived longer than 120 days without adjunct chemotherapy. 8 Four months after surgery, the dog of this report remains free of clinical complications.
