Abstract
Multisystemic eosinophilic epitheliotropic disease was diagnosed histologically in a 17-year-old Quarter Horse intact mare that was presented with a chronic history of respiratory distress. At necropsy, the lungs were poorly collapsed and the pulmonary parenchyma contained innumerable, discrete, spherical nodules in a miliary pattern. A few similar nodules were scattered in the liver and the renal lymph nodes. Histologically, these nodules consisted of fibrosing eosinophilic granulomas. Based on histologic findings and the absence of an etiologic agent, a diagnosis of multisystemic eosinophilic epitheliotropic disease was made.
Keywords
Multisystemic eosinophilic epitheliotropic disease (MEED) occurs primarily in young horses, ranging in age from 3 to 13 years. 8 The disease is histologically characterized by eosinophilic and lymphoplasmacytic infiltrates and the formation of eosinophilic granulomas in different organs, including the pancreas, salivary glands, gastrointestinal tract, biliary and bronchial epithelium, and skin. 12 The clinical signs vary according to the organs affected. Horses usually present with severe weight loss. The prognosis of horses with MEED is invariably poor. 6 However, attempted clinical management includes treatment with hydroxyurea and dexamethasone. 7
Multisystemic eosinophilic disease has been reported in human beings (referred to as hypereosinophilic syndrome [HES]), 19 dogs, 1 cats, 11 and ferrets. 3 There are no specific diagnostic tests for this disease. In animals, MEED remains a diagnosis of exclusion. In humans, there are three features for defining HES. First, the patient must have sustained blood eosinophilia of greater than 1500/µl present for longer than 6 months. Second, the other etiologies for eosinophilia must be absent. Third, the patient must have signs and symptoms of organ involvement. 5 The etiology for this syndrome is not known, although parasitic, allergic, autoimmune, viral, and toxic causes and production of eosinophilopoietic cytokines from activated or neoplastic T-lymphocytes have been suggested in horses. 8, 12 Several mechanisms are hypothesized for dysregulated overproduction of eosinophils in HES. There may be overproduction of eosinophilopoietic signals by abnormal T-cell clones. Alternatively, the signals produced may be abnormal, resulting in enhanced or prolonged biologic activity. There can be alteration in the signal transduction mediated by the cytokine receptors, resulting in constant stimulation of eosinophil production. Finally, the suppressive regulatory steps in either the eosinophils or in the cells generating eosinophilopoietic signals may be defective. 13
Here we report a case of MEED in a horse that is somewhat unusual because of the severity of pulmonary disease, age of the horse, lack of skin lesions, and clinically insignificant intestinal involvement. A 17-year-old Quarter Horse mare was presented to the Oklahoma State University Boren Veterinary Teaching Hospital for the evaluation of respiratory distress. The mare had a 4.5-month history of cough that was unresponsive to two previous 14-day courses of antimicrobial treatment. No treatment had been administered in the last 6 weeks. No nasal discharge was noted previously. The mare was reported to have a good appetite, but had been losing weight. Vaccinations were not current. The mare had been dewormed two times over the previous 4.5 months.
The mare was thin and afebrile and had increased heart (48/minute) and respiratory (32/minute) rates. Increased inspiratory effort was present, as well as bilateral harsh lung sounds on thoracic auscultation. Brief attempts to utilize a rebreathing bag for auscultation caused respiratory distress. Although the history of chronic cough was consistent with recurrent airway obstruction, the mare's clinical signs were not. Arterial blood gas analysis revealed hypoxia (PaO2 = 85.5 mm Hg), hypocapnia (PaCO2 = 38.7 mm Hg), and respiratory alkalosis (pH = 7.480). The increased alveolar arterial oxygen gradient (20.8) was consistent with significant pulmonary dysfunction due to either ventilation perfusion mismatch or diffusion impairment. Thoracic radiographs revealed a diffuse homogeneous miliary interstitial pattern throughout all lung fields (Fig. 1). Differential diagnoses included silicosis, neoplasia, and fungal pneumonia.
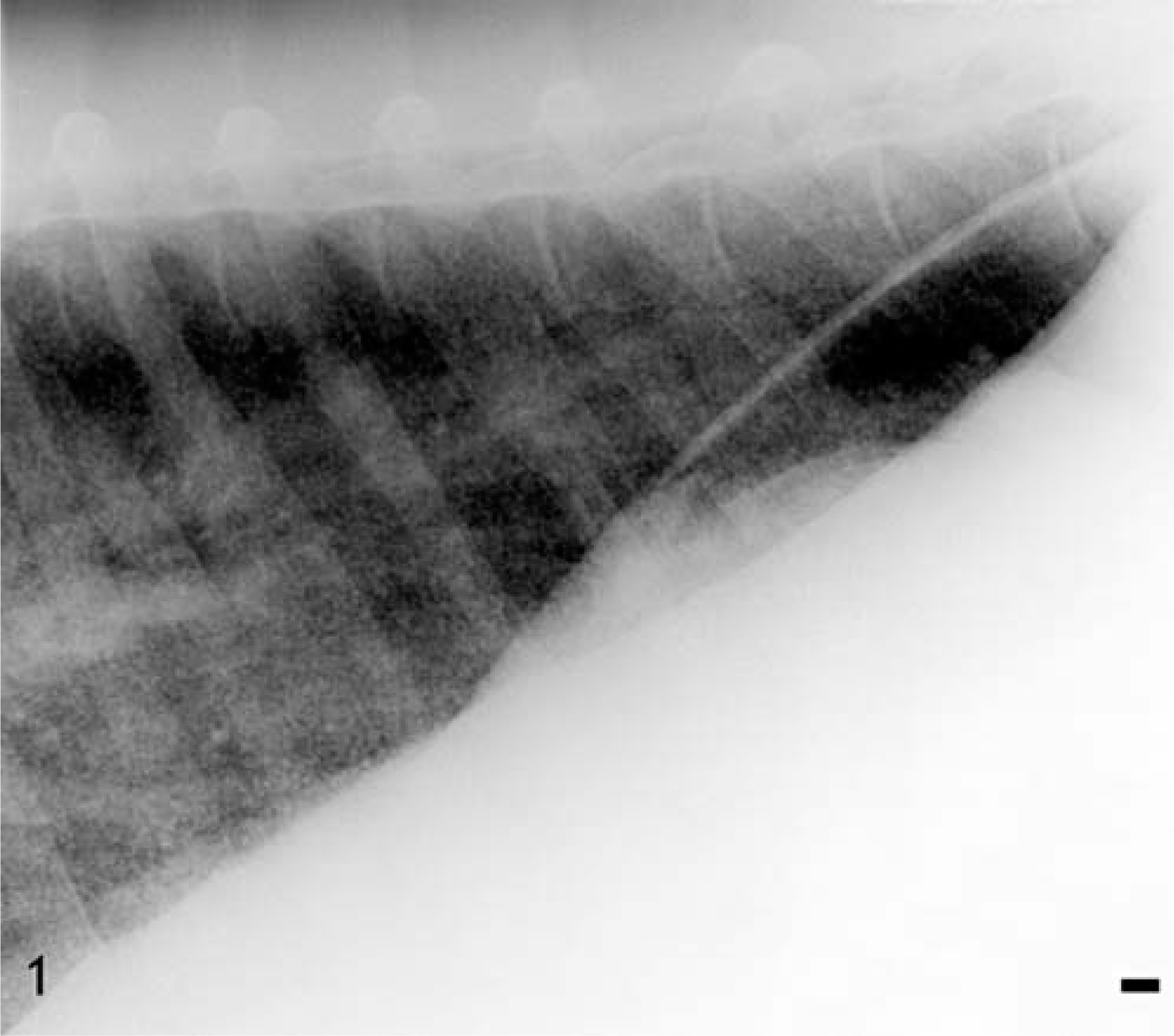
Lung; horse. Radiograph of the lung lobe illustrating the increased radiodensity of a homogeneous miliary interstitial pattern throughout the lung. Bar = 10 mm.
Mature neutrophilia (13,072/µl), hyperfibrinogenemia (500 mg/dl), and hyperglobulinemic (5.5 g/dl) hyperproteinemia (8.6 g/dl) were supportive of a diagnosis of chronic inflammatory disease. Peripheral blood eosinophil count was within normal limits (172/µl). Bronchoalveolar lavage (BAL) fluid was slightly hazy and cytologically characterized by markedly increased numbers of neutrophils, macrophages including multinucleated cells, and mildly increased numbers of eosinophils, with an abundant background of thick eosinophilic mucus. Lung biopsy revealed a significant chronic peribronchiolar fibrosis, with fibrin, neutrophils, and focal areas of clustered eosinophils. Multiple rod-shaped bacteria were also observed, but these were considered contaminants, because haired skin and hair shaft elements were also present. Transtracheal wash culture was negative. Fungal elements were not identified cytologically in BAL fluid or in the lung biopsy specimen.
Considering the disseminated nature of the pulmonary disease and undetermined etiology, the owner elected euthanasia. The mare was subsequently presented for necropsy evaluation. At necropsy, all lung lobes were diffusely pale pink, poorly collapsed, palpably nodular, and markedly heavy. Visceral pleura contained disseminated, pale white, spherical, variably-sized (approximately 0.1- to 0.3-cm diameter), hemispherically raised nodules, giving the lung a nodular appearance. On cut surface, the parenchyma had a similar nodular appearance (Fig. 2). Nodules could be easily removed from the parenchyma and were firm, spherical, and difficult to cut. Renal lymph nodes were markedly enlarged and contained similar cortical and medullary spherical nodules. A few randomly scattered small nodules were also present in the liver capsule and parenchyma. No gross lesions were detected in the skin.
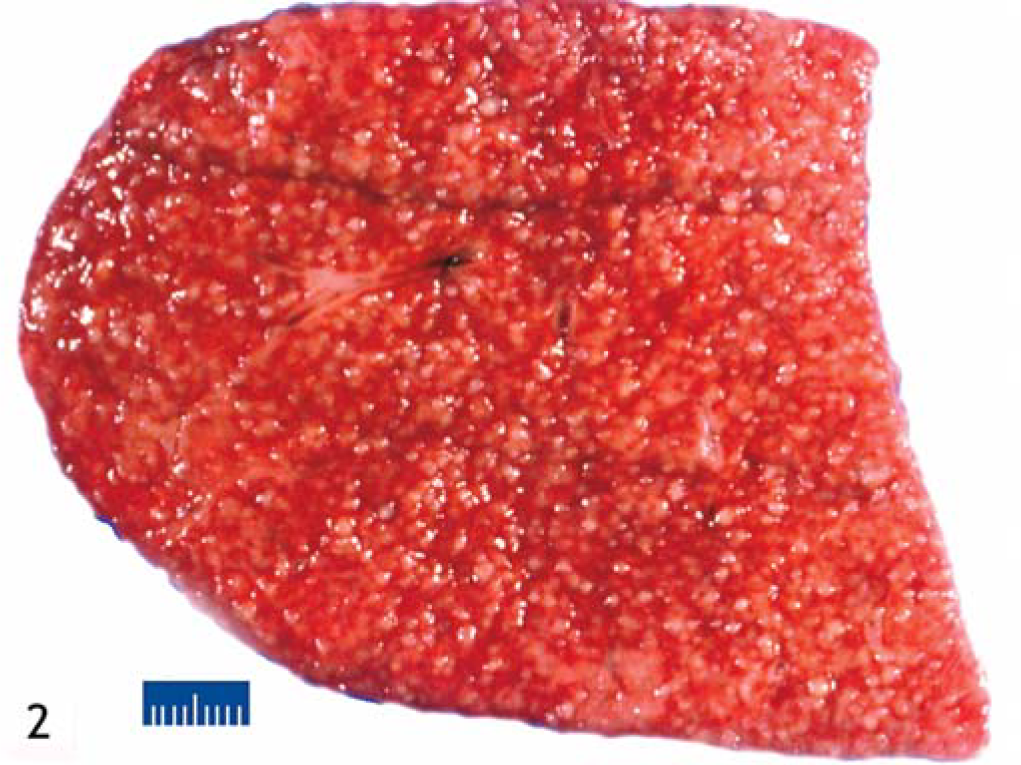
Lung; horse. Macrophotograph of the cut surface of the lung. There are multiple pale white, hemispherically raised nodules distributed throughout the cut surface. Bar = 1 cm.
Representative tissues, collected during necropsy, were fixed in 10% neutral buffered formalin, routinely processed, and embedded in paraffin. Tissue sections of 4–5 µm were stained with hematoxylin and eosin (HE), periodic acid–Schiff (PAS), Grocott's methenamine silver (GMS), toluidine blue, Ziehl-Neelsen acid-fast, and Fite's acid-fast stains and examined microscopically. Immunohistochemistry to ascertain the type of cellular infiltrate was done using anti-CD3 and anti-79a antibody for T and B cells, respectively. The sections were deparaffinized and rehydrated through a series of graded alcohols. Antigen retrieval was done by pressure cooking slides for 9 minutes in a 0.01-M sodium citrate buffer, pH 6.0. Endogenous peroxidase was blocked using a 3% aqueous solution of hydrogen peroxide. Background staining was blocked using a 10% normal goat serum for the CD3 antibody and a 10% normal rabbit serum for the CD79a antibody. Polyclonal rabbit antisera A0452 (Anti-CD3, DakoCytomation, 6392 Via Real, Carpenteria, CA 93013) and mouse antisera M7051 (anti-CD79a, DakoCytomation) were applied at 1 : 600 and 1 : 160 for 30 minutes, respectively. The sections were then rinsed in PBS-Tween and the biotinylated secondary antibodies (Zymed Laboratories, 561 Eccles Avenue, South San Francisco, CA, 94080) were applied for 15 minutes at room temperature. The sections were then rinsed again with PBS-Tween and a horseradish-streptavidin conjugate (Zymed Laboratories) was applied for 15 minutes at room temperature. The sections were rinsed and then incubated with a color reagent (DAB, Research Genetics, 2130 Memorial Parkway, Huntsville, AL 35801) for 5 minutes. All sections were rinsed with buffer and then counterstained with Mayer's hematoxylin. The sections were dehydrated with graded alcohols, cleared in toluene, and mounted in Permount. Negative controls were incubated with a nonimmune fraction of either mouse or rabbit IgG instead of the primary antibody.
Histologically, the lesions varied from focal aggregation of eosinophils to the formation of large, fibrosing eosinophilic granulomas in the lung, the liver, and the renal lymph nodes. The mature granulomas contained abundant collagen admixed with fibroblasts, macrophages, and eosinophilic debris in the center. Intact eosinophils, macrophages, fibroblasts, CD 3+ T cells, a few plasma cells, and, rarely, multinucleate giant cells were seen on the periphery of the granulomas. Often the centers of large granulomas were mineralized. Mast cells were rarely seen. The lung parenchyma was multifocally expanded by discrete, closely placed, well-circumscribed, fibrosing eosinophilic granulomas often associated with bronchiolar epithelium (Figs. 3, 4). In some regions, bronchiolar lumens were obliterated by fibrous tissue and inflammatory cells (bronchiolitis fibrosa obliterans). The surrounding smooth muscle was often hypertrophied. Bronchiolar epithelium was often severely hyperplastic and occasionally nonciliated and cuboidal. Multiple newly formed tubules of proliferating bronchiolar epithelium were frequently associated with granulomas. Alveolar septa surrounding the granulomas were often thickened by fibrous connective tissue and occasionally lined by hyperplastic type II pneumocytes. Distant alveolar septa were normal.
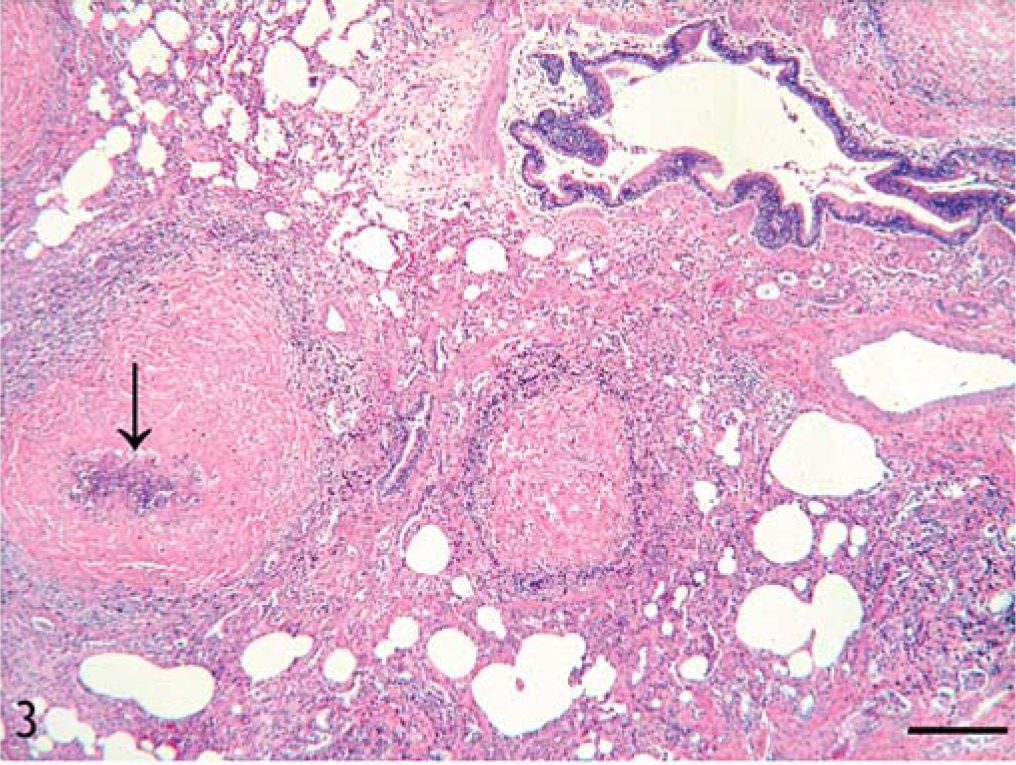
Lung; horse. Photomicrograph of the lung parenchyma expanded by multiple, closely placed, fibrosing granulomas, often with mineralized center (arrow). HE. Bar = 120 µm.
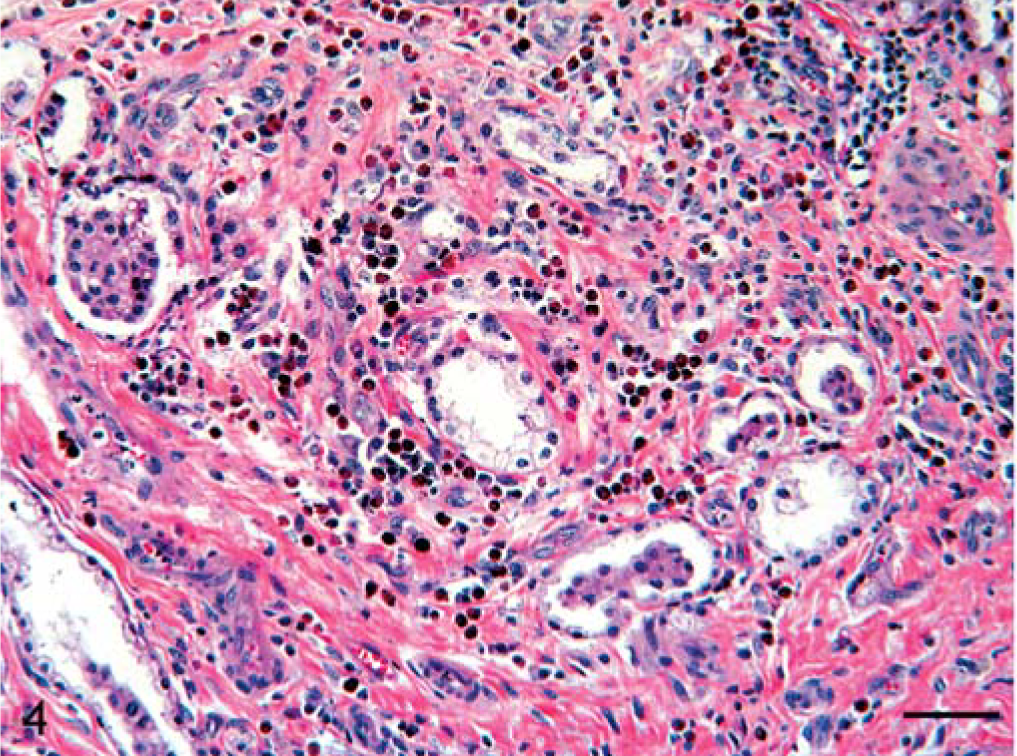
Lung; horse. Photomicrograph showing the region of a fibrosing eosinophilic granuloma. Aggregation of numerous eosinophils and macrophages scattered in fibrocollagenous tissue involving parabronchial region. Note displacement of multiple parabronchial glands. HE. Bar = 60 µm.
In the liver, portal triads were expanded by variably-sized foci of eosinophils admixed with few lymphocytes, plasma cells, and macrophages and, rarely, the formation of mature fibrosing granulomas. These inflammatory cell foci were closely associated with biliary epithelium (Fig. 5). The biliary epithelium was occasionally hyperplastic and disorganized. In some regions, the cellular infiltrate extended into the adjacent periportal regions. Often eosinophils in portal triads were accompanied by fibrosis and multiple small arterioles. The small and large intestine appeared grossly normal, but histologically the lamina propria diffusely contained small numbers of scattered eosinophils admixed with few lymphocytes and plasma cells. Bone marrow appeared active and contained moderate numbers of mature and immature eosinophils along with normal erythrogenic and myelogenic cell lines. There was a focal region in the pancreas with a mild periductal fibrosis and loss of adjacent exocrine tissue.
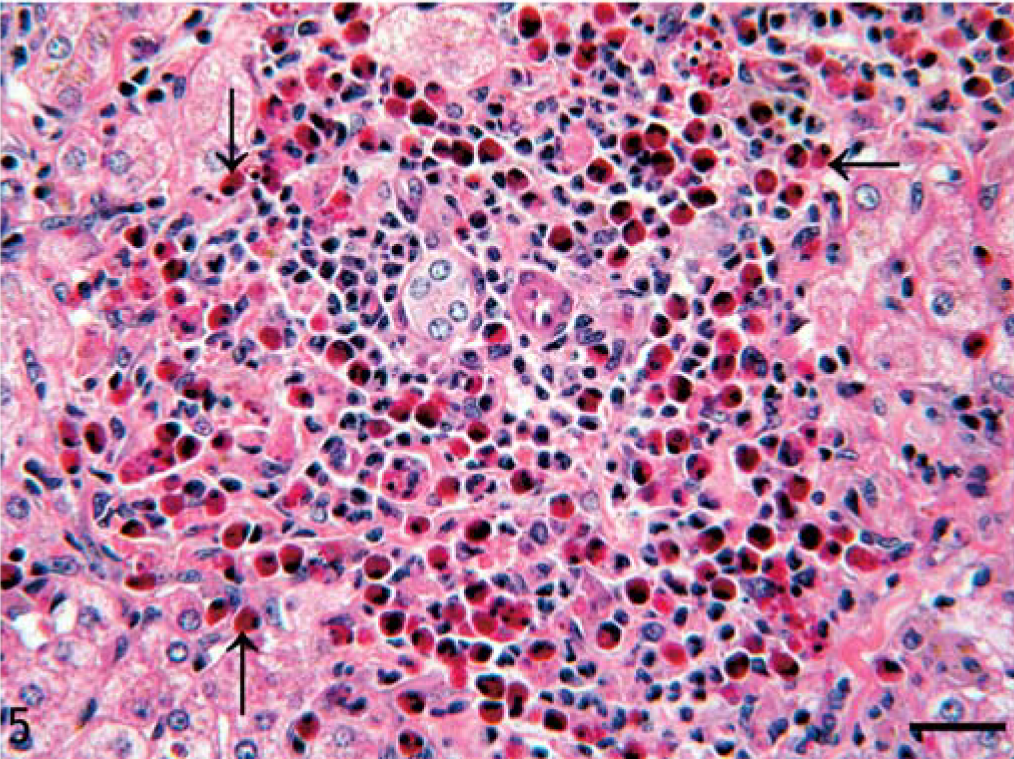
Liver; horse. Aggregation of eosinophils (arrows) and few lymphocytes, plasma cells, and macrophages in the portal triad. HE. Bar = 40 µm.
Histochemical stains for fungi (PAS and GMS) and acid-fast bacteria (Ziehl-Neelsen and Fite's) were negative. There was no histologic evidence of a parasite or inorganic material (asbestos, silica, etc.) within the lung. Based on histologic findings and the absence of an etiologic agent, a diagnosis of MEED was made.
Moderate numbers of eosinophils admixed with few lymphocytes and plasma cells were diffusely scattered in the lamina propria of the small and large intestine. Typical eosinophilic granulomas were not observed; this pattern of eosinophilic infiltration may also be observed in MEED. 12 However, it also suggests a response to foreign body, parasites or an allergy. Neither parasites nor a foreign body were detected grossly or histologically, and fecal samples submitted for parasitic evaluation were negative. In a retrospective study, grossly and histologically similar fibrosing granulomas were reported predominantly in the liver and occasionally in the small intestine, lung, and diaphragm of 11 horses from Florida. In only one horse there were poorly stained broken egg shells or a ghostlike larva that suggested schistosomal infection. 4 Based on this occasional finding, authors circumstantially concluded that the fibrosing granulomas were the result of chronic schistosomiasis of undeterminable origin. In the absence of clinical, gross, or histologic evidence of a parasite, it seems highly unlikely that our horse had a parasitic infection.
Granulomas usually described in MEED are formed by aggregation of eosinophils and other mononuclear cells without deposition of abundant collagen. In humans, immunocytochemical studies have shown that eosinophil cationic protein may play a central part in the development of granulomas. 18 In this case, the presence of abundant collagen in the granulomas is presumably attributable to eosinophilic granular enzymes such as eosinophilic cationic protein. Eosinophil-derived TGF-α and TGF-β are responsible for stimulating enhanced fibroblastic proliferation 9 and extracellular matrix deposition. 2
The majority of horses with MEED have cutaneous lesions. A generalized chronic, progressive, exfoliative dermatitis occurs that is histologically characterized by eosinophilic, lymphocytic, and plasmacytic dermatitis, accompanied by marked acanthosis and hyperkeratosis. 12 Marks and Shuster suggested that skin lesions could be a sequel to the lesions in the intestine and pancreas resulting in malabsorption especially of fat-soluble vitamins such as vitamin A. 10 In our horse, the absence of gross lesions in the skin and the absence of marked intestinal and pancreatic pathology suggest that skin lesions may be secondary to the involvement of intestine and pancreas. However, without experimental investigation, this proposal is only a hypothetical assumption. Levels of fat-soluble vitamins, in particular vitamin A, in our horse and in horses with MEED are not known.
The clinical and pathologic findings in our horse bear a close resemblance to idiopathic granulomatous disease. 15 Equine generalized granulomatous disease is a multisystemic noncaseating granulomatous disease that closely resembles sarcoidosis in humans. 16 However, the granulomas in the equine generalized granulomatous disease are mostly formed in the skin and lung and do not involve infiltration of eosinophils.
In animals, multisystemic eosinophilic infiltration may be caused by the abnormal proliferation of T lymphocytes, as has been demonstrated in humans. 3 It has been demonstrated that a significant proportion of patients with HES present with a clonal expansion of type 2 helper T lymphocytes overproducing eosinophilopoietic cytokine IL-5. 17 Other eosinophilopoietic cytokines are IL-3 and GM-CSF, which also act on other cell lineages of bone marrow–derived cells, whereas IL-5 in humans is restricted to stimulating eosinophil production. Treatment with anti-IL-5 antibodies in human patients with HES and dermatologic manifestation resulted in a decrease in the peripheral blood eosinophil count and in the serum level of eosinophilic cationic protein. 14 Although we did not quantify IL-5, presumably the presence of large numbers of CD3+ T cells in the granulomas support that involvement of IL-5 and/or abnormal proliferation of T cells may be an underlying cause. 8 Studies focusing on identification of the eosinophilic attractant proteins responsible for localization in particular organs and role of cytokine IL-5 in MEED may help in understanding the pathogenesis and in designing possible therapeutic agents.
Footnotes
Acknowledgements
We thank Ms Betty Handlin for manuscript preparation.
